Abstract
The protease-resistant infectious prion protein, PrPres, that causes transmissible spongiform encephalopathies, is remarkably resistant to conventional physical and chemical sterilization methods, including heat. It was hypothesized that thermal-dependent PrPres degradation has been underestimated, and the effect of prolonged incubation at 37°C, 55°C, and 80°C on PrPres detection was examined using brain homogenates from chronic wasting disease-affected elk and mule deer (PrPCWD). Immunoblotting demonstrated progressive loss of PrPCWD immunoreactivity with time in all incubated samples as temperature increased, and PrPCWD was virtually undetectable after 90 days of incubation at 55°C and 80°C. These results indicate that decontamination methods and tissue disposal systems maintaining elevated temperatures for long periods of time could interfere with immunodetection, and the reliability of assays for PrPres detection could be compromised when applied to tissues exposed to heat with time. Although these results may suggest that such prolonged heat treatment could destroy prions, the observed loss of immunoreactivity does not necessarily correlate with a concurrent loss of infectivity. Bioassay is needed to determine if samples that have been incubated under these conditions retain infectivity.
A large proportion of samples evaluated by transmissible spongiform encephalopathy (TSE) surveillance programs are affected with some degree of postmortem or postcollection tissue decomposition. Given the economic and public health implications of misclassifying prion disease status, concern has arisen in regard to the reliability of TSE tests when applied to deteriorated tissue. Severely decomposed nervous tissue cannot be evaluated by immunohistochemistry (IHC) or histology since tissue architecture is lost. However, it has largely been concluded that autolysis does not compromise the accuracy of assays that detect the proteinase-resistant, pathogenic isoform of the prion protein (PrPres). 2,5,7,8,12,16 It was hypothesized that over a relatively long period of time, exposure to heat may have a more substantial effect on PrPres immunodection than previously considered. The objective of the present study was to characterize PrPCWD detectability with time in brain samples subjected to heat in vitro as determined by Western blot.
Brain from 2 elk and 1 mule deer testing positive for chronic wasting disease (CWD) by IHC were studied. The brain tissues were not collected from the animals aseptically; however, a sterile technique was used in the laboratory preparations to avoid introduction of additional microorganisms. Brain tissue from each animal was homogenized separately using a homogenizer a . Homogenates were incubated without dilution or at a concentration of 20% (weight/volume) in sterile protease-free water. Homogenates were incubated at 37°C, 55°C, or 80°C for 0−198 days. Samples of incubating brain were removed at various time points and stored at −70°C until tested for PrPCWD. Detection of PrPCWD was achieved by testing equal amounts (mg weight) of brain tissue with a commercially available Western blot (WB) for TSE diagnosis b . Samples were digested with proteinase K (PK) before immunoblotting (as directed by the manufacturer). Equal volumes (μg weight) of brain tissue were PK treated and tested so that results between samples incubated with or without dilution could be directly compared. Samples used for this study were strongly positive by IHC; unincubated samples produced banding patterns in the WB that obscured neighboring lanes and were above the linear detection limits of the densitometer. Therefore, after incubation and PK digestion, samples were diluted to more accurately produce treatment response correlations. Dilutions depended on prion load in each stock homogenate, as reflected by signal strength in WB. Specifically, samples originating from elk homogenates 1 or 2 were always diluted 1:2 or 1:4, respectively, before mixing with equal volumes of sample buffer for testing. Samples from the mule deer homogenate required no dilution before mixing with sample buffer. PrPCWD signal intensity was compared between samples using densitometric analysis of all 3 PrPCWD bands. c
Results demonstrated a consistent loss of PrPCWD signal with time. When brain tissue was incubated at 37°C, 55°C, and 80°C, the rate of PrPCWD signal loss increased with temperature (Figs. 1 and 2). When CWD-affected brain tissues were incubated at 55°C as 20% suspensions in water, mean PrPCWD signal loss was 56% (±2.5) by day 16, and PrPCWD was almost below detection after 98 days of incubation (mean signal loss of 94.2% ± 4.1). Chronic wasting disease-affected brain homogenates incubated without dilution at 55°C under the same conditions showed less signal reduction. However, after 198 days of incubation, the PrPCWD signal was at least 95% reduced in all samples, regardless of homogenate concentration during incubation (Fig. 3).
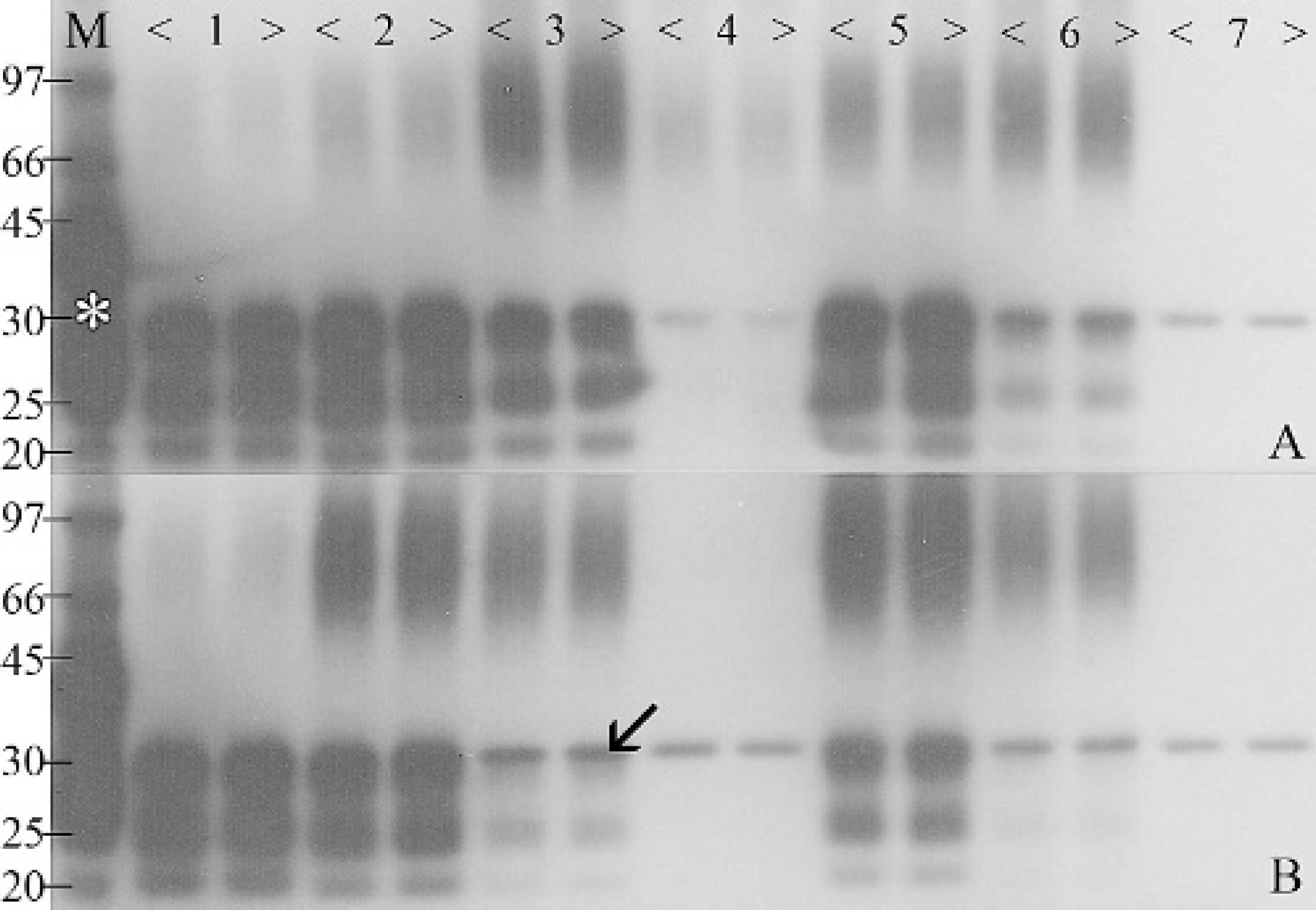
Western blot and corresponding percent PrPCWD signal intensity of homogenized brain tissue from a chronic wasting disease-affected elk incubated at 37°C, 55°C, and 80°C for up to 90 days. M denotes protein markers of molecular weight and positive control (asterisk indicates non-protease-truncated PrP). Prion proteins produce a signal consisting of 3 bands ranging from 25 kD to 35 kD, depending on the glycosylation. The single 31-kD band (arrow) is produced because of nonspecific reactions between residual proteinase K and secondary antibody.
There are a number of possible explanations for the time- and temperature-dependent reduction in PrPCWD immunoreactivity demonstrated in these studies. One possibility is that the effect is due to an increase in susceptibility to PK, which is used routinely in the WB method. However, this is not the case, since signals were similarly reduced in samples tested without PK treatment (data not shown). Dilution of samples with water enhanced the signal loss. The mechanism for this is unclear, but could involve hydrolysis or reduced PrPCWD structural stability. It is possible that incubated brain tissue could have been contaminated during necropsy tissue collection with microorganisms that may contribute to PrPCWD destruction. However, no microorganisms were isolated from samples of brain homogenates before and after 16 days of incubation at 55°C. Furthermore, because this phenomenon was observed with brain tissue from 3 different animals collected at various times, it is unlikely that similar microbiological conditions could have occurred in all 3 samples. It is possible that the reduction of immunoreactivity relates to the formation of aggregates that cannot enter the gel. Whereas high molecular weight smears that may indicate oligomeric PrPCWD aggregate formation are evident in many of the samples, especially those incubated for longer periods of time at 37°C, they are not present in samples that exhibit markedly reduced immunoreactivity. When monoclonal antibody-based immunoassays are used, immunodetection could be limited by partial PrPCWD degradation and selective epitope loss or cross-linking. Tissue examination using assays based on other monoclonal antibodies and other formats (e.g., enzyme-linked immunosorbent assays) and could reveal such selective epitope loss. Although modifications to PrPRes have the potential to diminish prion titer, 6 infectivity has been established with scrapie samples that produce no signal on WB. 13 Ultimately, in order to determine if temperature- and time-dependent reduction of PrPCWD immunodetection correlates with prion degradation and reduced infectivity, kinetic bioassay studies are required.
Signal intensity with time. “Undiluted” brain samples correspond to those portrayed by the Western blot. “Diluted” brain samples were diluted to a 20% solution in sterile protease-free water before incubation. Signal intensity of 100% represents initial signal strength for each sample at Day 0; signal intensities below 100% indicate the proportionate signal strength relative to intitial signal.
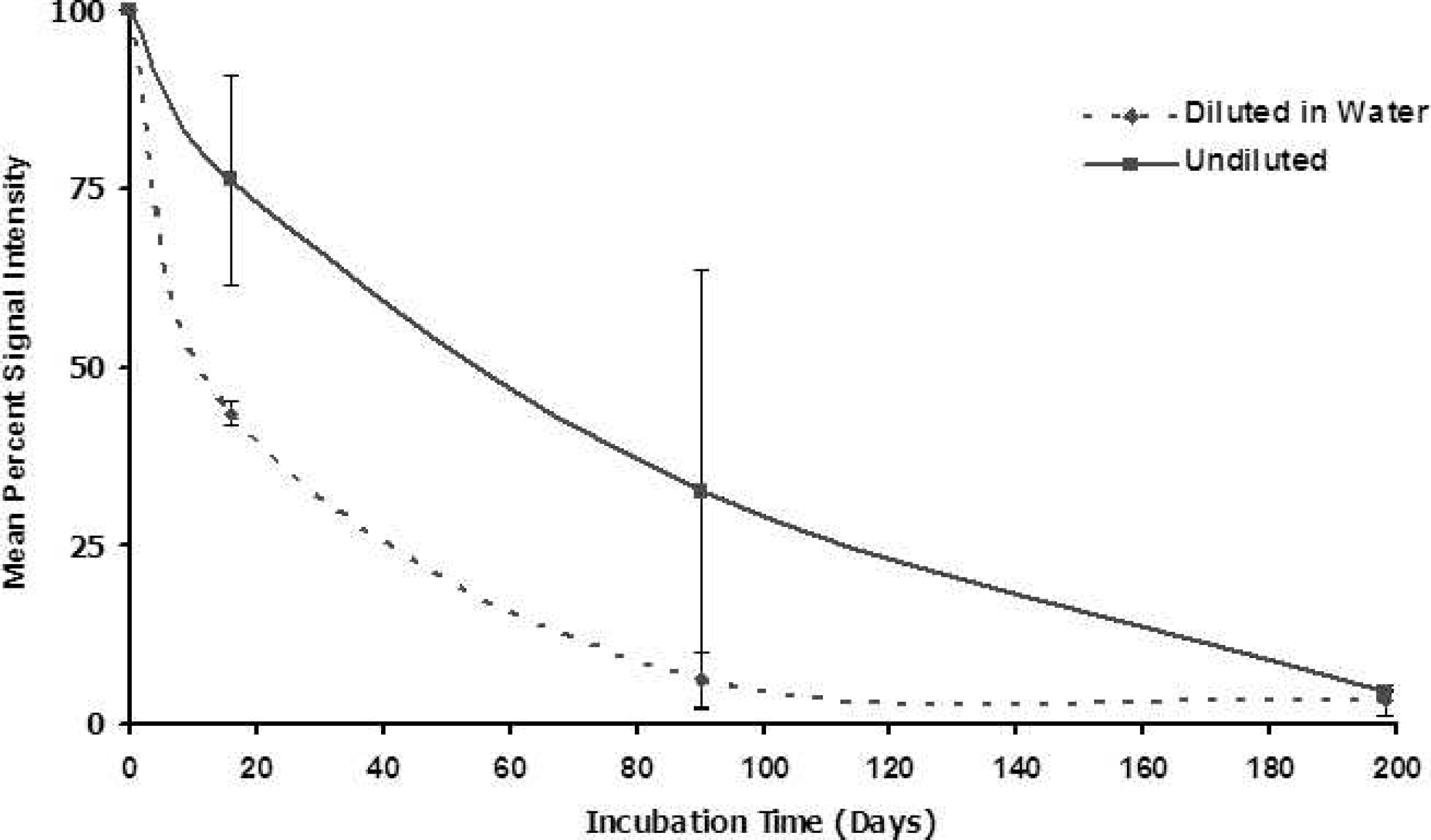
Mean percent PrPCWD signal intensity with time in homogenized brain tissue from 2 chronic wasting disease (CWD)-affected elk and 1 CWD-affected mule deer incubated at 55°C. “Diluted” brain samples signify 20% homogenized brain solution in sterile protease-free water. A signal intensity of 100% represents full strength of signal at Day 0. Signal intensities below 100% indicate the proportionate signal strength relative to the starting signal. For diluted samples, mean values at each time point were derived from 3–5 trials on tissues from each animal. For undiluted samples, mean values at each time point were derived from 3–4 trials on tissues from elk No. 1, in combination with 2 trials (day 14) and 1 trial (days 90 and 198) on tissues from elk No. 2.
Prions are resistant to inactivation by several conventional physical or chemical sterilization treatments including heat, irradiation, detergents, acids and bases, alkylating agents, organic solvents, oxidizing agents, salts, and proteolytic enzymes. 3,4,9,14 Since techniques that destroy prions (i.e., alkaline hydrolysis, treatment with the phenolic disinfectant LpH) may not be available or practical for disposal of infected animals or tissues, 3,14,15 alternative, practical methods for decontamination of TSE-contaminated biomaterial are needed. Because the reduction of PK-resistant PrP can be correlated to infectivity loss, 1,8 we speculate that the time- and temperature-dependent reduction in PrPCWD immunoreactivity could reflect its destruction. Temperatures in the range of 55°C are similar to average temperatures occurring in decomposing or composting livestock carcasses. 10,11 If the disappearance of PrPCWD immunoreactivity after exposure to elevated temperatures for extended periods of time correlates with infectivity loss, then it is possible that composting conditions might result in prion degradation. Furthermore, the results presented here raise a concern regarding the validity of PrPres immunoassays applied to decomposed tissues that have been subjected to high levels of heat for long periods of time. Investigation of the described effect using immunoassays that target different epitopes and bioassay are necessary to establish correlations between the loss of PrPCWD immunodetection and infectivity.
Acknowledgements. This work was supported by the US Environmental Protection Agency (EPA) Cooperative Agreement Number CP978015-01 and the Animal Population Health Institute at Colorado State University. The authors thank Drs. Michael Miller (Colorado Division of Wildlife) and Margaret Wild (National Park Service) for providing cervid tissues essential for this project and Prionics AG for providing western blot kits for these studies.
Footnotes
a.
Prionics FastPrep Homogenizer, Prionics AG, Zurich, Switzerland.
b.
Prionics-Check WESTERN, Prionics AG, Zurich, Switzerland.
c.
ChemiImager IS-5500, AlphaInnotech Corporation, San Leandro, CA.
