Abstract
Poisoning with amanitin-containing hepatotoxic mushrooms demands extensive efforts from clinicians, toxicologists, and pathologists. Presumptive diagnoses are established by positive identification of the suspect mushroom along with the occurrence of consistent clinical signs. If the animal dies, hepatic lesions may suggest exposure to amanitin-containing mushrooms; however, lesions are nonspecific. A 15-week-old female Dachshund was presented to the referring veterinarian for acute onset of lethargy that quickly progressed to sternal recumbency. Despite supportive care, the dog remained lethargic and died approximately 12 hours after initial presentation. A pale tan liver was noted at necropsy. Microscopically, the liver showed panlobular coagulative necrosis of hepatocytes. A presumptive diagnosis of amanitin poisoning was based on suspect history of exposure to mushrooms, clinical signs, and pathologic findings. Exposure to amanitin was confirmed through detection of α-amanitin in the liver by liquid chromatography/mass spectrometry. The objective of this case report is to illustrate the essential components to a successful diagnostic work-up of a suspect case of hepatotoxic mushroom poisoning. Although hepatotoxic mushroom poisoning has been documented in dogs before, confirmatory techniques for biologic specimens have not been used previously in diagnostic investigations.
The apparent frequency of mushroom poisoning in animals is likely low because of the lack of methods to confirm exposure to toxic mushrooms. Most cases are diagnosed by positive identification of the suspect mushroom along with the occurrence of consistent clinical signs. Amanita phalloides, the ubiquitous death cap or death angel, is found throughout North America, commonly in association with oaks and birch, and is the species most frequently resulting in fatalities in humans. 2 , 15 Additionally, 2 other genera, Galerina and Lepiota, are known to contain hepatotoxic cyclopeptide toxins. 13 There are 3 groups of cyclopeptides, including the amatoxins, phallotoxins, and virotoxins. Amatoxins are bicyclic octapeptides and include the amanitins (α-, β-, γ-, and ε-amanitins), amanin, amanullin, and proamanullin. 17 Severe poisonings and lethality are mainly attributable to the amanitins. The bicyclic heptapeptide phallotoxins were once thought to be the cause for gastrointestinal clinical signs; however, they are no longer believed to exert any acute toxicity. Although the amount of research is limited, bicyclic heptapeptide virotoxins are not considered to have toxic effects after oral exposure. Therefore, diagnostic approaches to confirm hepatotoxic Amanita mushroom poisoning need to be based on the identification of amanitins in biologic specimens.
In October 2005 a 15-week-old female Dachshund was presented to the referring veterinarian for lethargy. The puppy was current on vaccinations. The owner noted that the puppy had consumed mushrooms approximately 12 hours before the onset of clinical signs. At the time of clinical examination the dog was in sternal recumbency and demonstrated generalized twitching. Serum chemistry results revealed marked hypoglycemia (19 mg/dl; normal: 62–108 mg/dl) and elevated alanine aminotransferase (above the upper limit of 75 U/liter of an in-house analyzer; normal: 8–57 U/liter). A complete blood count (CBC) demonstrated a moderate leukocytosis (28.9 × 109/μl; normal: 6.0–16.9 × 109/μl) that was composed primarily of granulocytes (24.8 × 109/μl; normal: 3.3–12.0 × 109/μl). A fecal floatation test and parvovirus ELISA test were negative. Supportive care, including an oral glucose supplement, was implemented; some diarrhea was noted throughout the day. Although still lethargic, the dog was discharged that afternoon. The dog died approximately 24 hours after ingestion of mushrooms and 12 hours after onset of clinical signs.
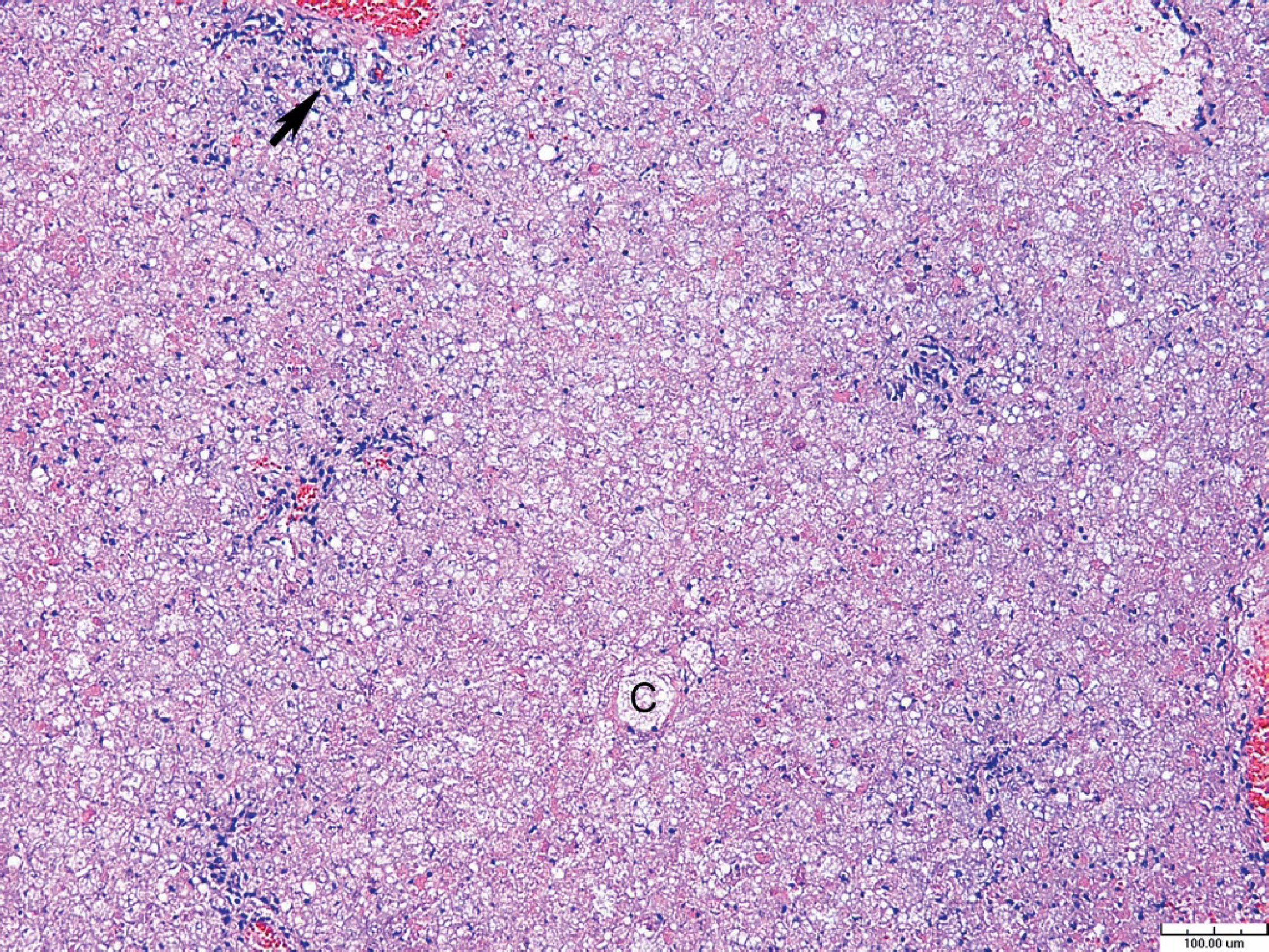
Liver, dog. Amanitin toxicosis. Massive hepatocellular necrosis with collapse of hepatic cords. Preserved bile duct (arrow) in portal area. “C” denotes central vein. Hematoxylin and eosin staining, 10X. Bar = 100 μm.
A complete necropsy was performed on the dog with subsequent histologic evaluation of tissues. Gross examination revealed that the dog was moderately thin with decreased body fat. The lungs appeared wet and exuded clear fluid from cut surfaces. The caudodorsal aspect of the left lung lobe was dark red; the lungs contained multiple 1–3-mm dark red foci, identified as petechiae. The stomach contained a small amount of brown to black ingesta. The gastric mucosa contained a focal 20-mm stellate ulcer. The small intestines contained a small amount of brown, liquid ingesta. The liver was pale tan to yellow, slightly swollen, and extended approximately 4 cm beyond the costal arch. Portions of brain, heart, lung, liver, kidney, urinary bladder, spleen, lymph node, thyroid gland, adrenal gland, pancreas, tongue, stomach, small intestine, and colon were fixed in 10% neutral buffered formalin. Specimens were routinely processed, paraffin embedded, cut to approximately 5-μm thickness, stained with hematoxylin and eosin, and examined by light microscopy. Microscopic examination of multiple sections of liver revealed a panlobular, uniform coagulative necrosis of hepatocytes extending from central veins to portal areas (Fig. 1). Hepatic plates were collapsed, and sinusoids were obliterated. Rarely, small clusters of periportal hepatocytes were spared. The majority of hepatocytes contained multiple small cytoplasmic vacuolations. A smaller number of hepatocytes contained dark eosinophilic, finely granular cytoplasm and pyknotic or karyorrhectic nuclei (Fig. 2). In multiple sections of lung alveolar septa were expanded by eosinophilic, proteinaceous fluid, and multiple foci of hemorrhage were frequent in alveoli. The alveolar septa and alveolar lumina contained mild mixed infiltrates of alveolar macrophages and fewer lymphocytes, plasma cells, and neutrophils. A moderate amount of fibrillar, eosinophilic material (fibrin) was observed in occasional alveoli. Few macrophages and small amounts of fibrin were also within a moderate number of bronchioles. Culture of the lungs was negative. No significant lesions were noted in sections of brain, heart, kidney, urinary bladder, spleen, lymph node, thyroid gland, adrenal gland, pancreas, tongue, stomach, small intestine, and colon. The lesions in the liver suggested exposure to a hepatotoxin.
As a consequence of the finding of acute hepatic necrosis and a history of exposure to mushrooms, fresh liver was submitted to the California Animal Health and Food Safety Laboratory for evaluation of the presence of amanitins. Liver was analyzed for α-amanitin using liquid chromatographymass spectrometry (LC/MS). In brief, the amanitin was extracted from liver tissue with acetonitrile in 0.1 M phosphate buffer. The acetonitrile was removed from the aqueous phase by extraction with methylene chloride, and the resulting extracts were purified by solid-phase extraction using a modified version of a previously reported purification procedure. 6 High-pressure liquid chromatography (HPLC) separation was performed using an 1100 HPLC system a equipped with a 150 × 4.6-mm Polar-RP column. b The HPLC system was interfaced with an LTQ linear ion trap mass spectrometer, c and α-amanitin was detected in MS/MS/MS (MS3) mode. A method detection limit study of 7 replicate analyses of liver fortified with 1 μg/liter (1 part per billion [ppb]) of α-amanitin gave an average recovery of 98% and a calculated method detection limit of 0.50 μg/liter (0.5 ppb). Analysis of unfortified negative control liver samples showed no interferences at the α-amanitin retention time. Alpha-amanitin was positively identified in the liver analysis based on comparison of the retention time and MS 3 spectrum obtained from analysis of purified α-amanitin standard material (Fig. 3). d Beta-amanitin can also be detected using this technique; however, the detection limit for β-amanitin d is approximately 5 times higher than that for α-amanitin. The liver sample in question was not analyzed for β-amanitin as the detection of α-amanitin was considered to be adequate for confirmation of exposure to hepatotoxic mushrooms.
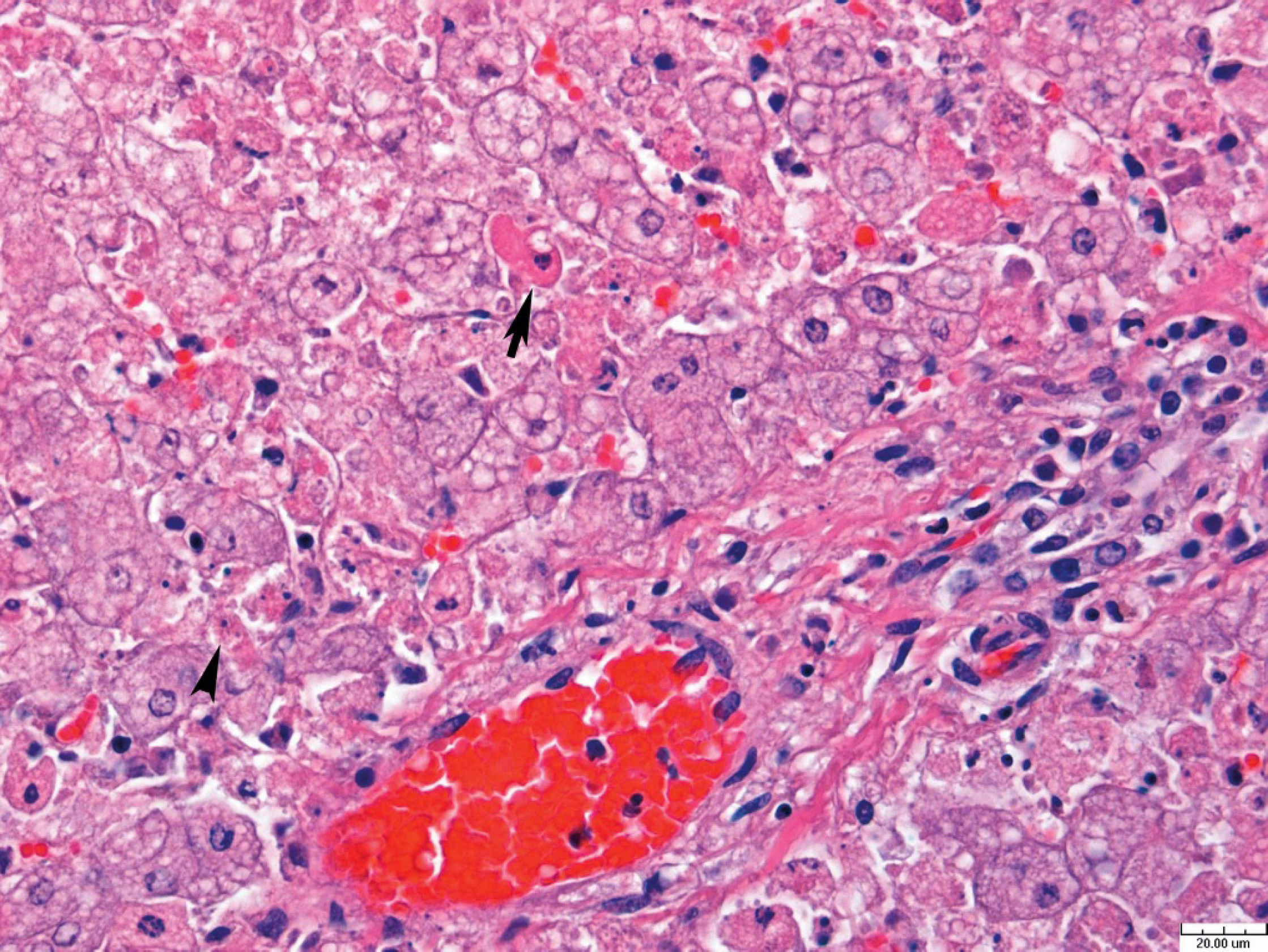
Liver, dog. Amanitin toxicosis. Diffuse centrilobular to midzonal hepatic necrosis is seen in the upper left of the photomicrograph. Periportal hepatocellular necrosis with pyknotic nuclei (arrow) and karyorrhexis (arrowhead). Hematoxylin and eosin staining, 60X. Bar = 20 μm.
On the basis of pathologic and toxicologic findings, a diagnosis of amanitin toxicosis was established. Suspected mushrooms were not submitted for identification and evaluation of their toxicity potential. This is commonly the case in suspect exposures in animals and emphasizes the need for accurate and sensitive confirmatory techniques. To the authors' knowledge, this case of fatal amanitin toxicosis in a dog is the first report that was confirmed by the presence of amanitins in liver tissue. Although dogs are highly susceptible to amanitin poisoning, only a few documented cases of incidental poisoning exist. 11 , 16 In most cases, diagnosis of amanitin toxicosis was aided by identification of amanitin-containing mushrooms in the environment of the animal, along with clinical and pathologic findings. In some cases, mushroom pieces may be found in gastric contents and can confirm exposure. In all cases, accurate mushroom identification requires consultation with an experienced mycologist.
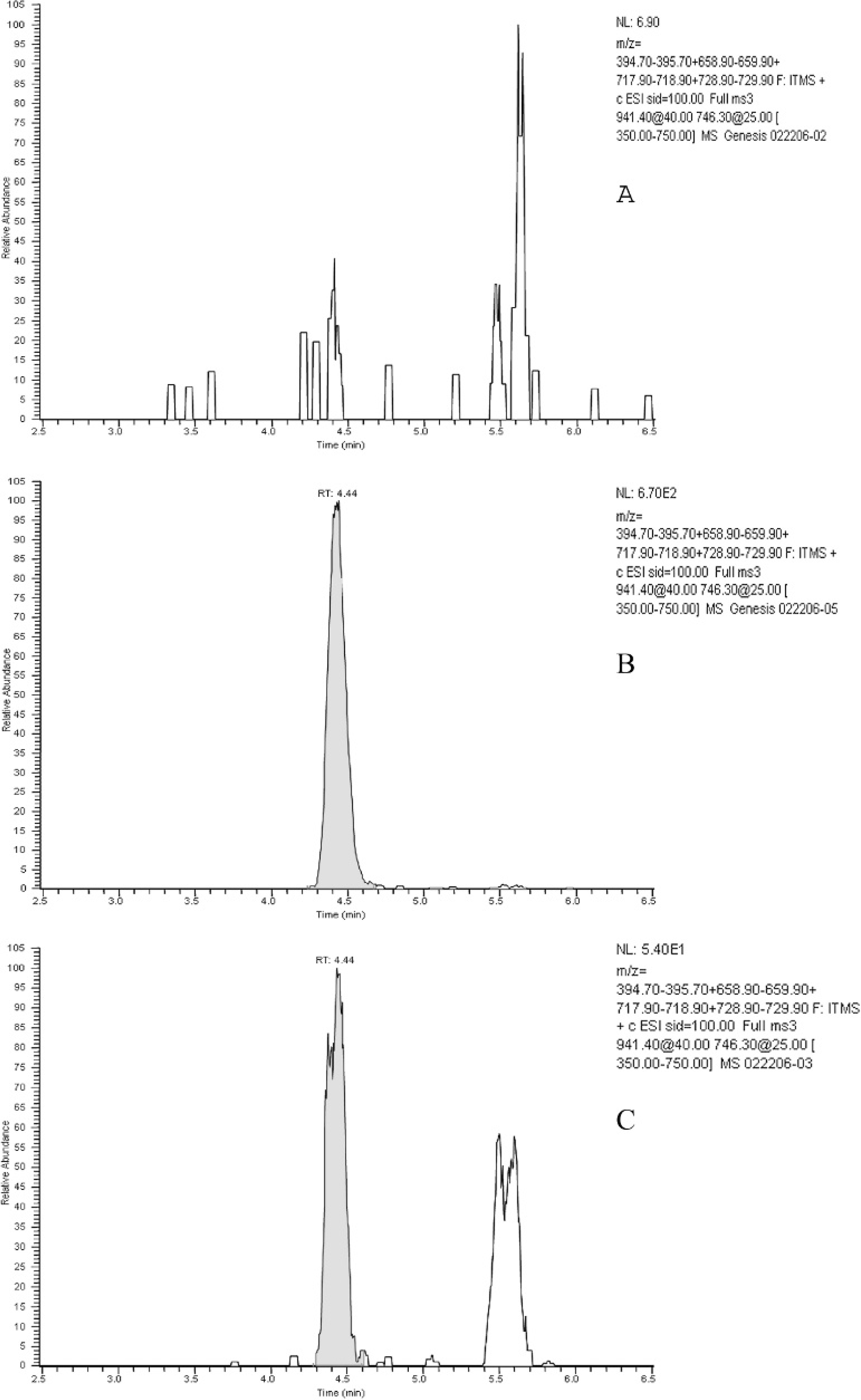
Selected product ion chromatograms (summed ions of m/z 395, 659, 718, and 729) from analysis of negative control matrix fortified with α-amanitin compared with analysis of liver from affected dog.
Amanitin exposure was confirmed through LC/MS detection of α-amanitin in liver. Currently the most commonly used method for diagnostic analysis of amanitin is a commercially available ELISA e test. This test is validated for urine specimens of human origin and has not been used for the diagnostic work-up of veterinary cases. In addition, this ELISA does not detect β-amanitin, 3 although it is known that some Amanita spp. contain β-amanitin only. A competitive ELISA was constructed recently that can detect β-amanitin in human serum and urine, but this assay is not yet available in clinical settings and is not validated for use in specimens of animal origin. 1 The newly developed LC/MS method applied in the presented case to analyze liver tissue has also successfully been used to detect α-amanitin in serum and urine of dogs (Filigenzi, personal communication). While detection of amanitins postmortem provides invaluable information to establish a cause of death, rapid confirmation of amanitins in serum and urine in suspect exposures assists in the early recognition and treatment decisions. It is known in humans that amanitins can be detected in urine well before any clinical signs are apparent, whereas routine laboratory tests such as CBC and serum chemistry profiles are unremarkable until liver or kidney damage has occurred. 10 Early recognition of exposure is critical since survival rates are greatly improved with timely therapeutic intervention. Interestingly, amanitin concentrations in kidneys and livers in people ingesting A. phalloides have been detected up to 22 days post-ingestion. 10 Detection of the parent compound is considered adequate because there is no known metabolism of α-amanitin. In dogs after IV administration of amanitins, it was shown that the plasma half-life is 25 to 50 minutes and that amanitins are not detectable in plasma after 4–6 hours. 8 Between 80% and 90% of the administered dose of amanitins is eliminated in urine and up to 7% is eliminated in bile. Toxicokinetics data after oral ingestion of A. phalloides are only available for humans. 10 In plasma, α-and β-amanitins were detected up to 36 hours after oral exposure, while toxins could be detected up to 72 hours later in urine. Plasma and urine amanitin concentrations do not seem to correlate with clinical severity or outcome.
The clinical course of amanitin toxicosis is usually divided into 4 phases, with the initial phase being the latency period. In dogs the onset of clinical signs occurs generally 6 to 24 hours after mushroom ingestion. 18 This is followed during the second phase of poisoning by severe gastrointestinal signs such as nausea, vomiting, bloody diarrhea, and severe abdominal pain. While the gastroenteric phase can have a large number of etiologies, serum and urine collected during this phase is considered most useful in detecting amanitins by chemical analysis. Gastrointestinal signs are often followed by a period of several hours to a few days during which the animal will appear to have recovered. During this third phase close monitoring of liver and kidney function can aid in the diagnostic approach and is essential to prevent misdiagnosis. The last phase of toxicosis begins approximately 36 to 84 hours after exposure to amanitins and is characterized by fulminant liver, renal, and multiorgan failure. Significant elevations in serum of aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase, and bilirubin are commonly observed in dogs. 11 , 18 As a result of liver failure, coagulopathy, encephalopathy, and coma may develop. Prothrombin (PT) and partial thromboplastin (PTT) times are important liver function parameters in dogs and can provide critical information in the assessment of a case. 16 The prothrombin index along with serum creatinine concentrations evaluated between 3 and 10 days after ingestion are useful predictors of death in humans, 9 but such data does not exist for dogs. If the animal survives the hepatopathy, renal failure may occur as a consequence of proximal and distal tubular necrosis. Severe hypoglycemia has been observed in dogs with amanitin toxicosis and is associated with the breakdown of liver glycogen. 7 Fifty percent of dogs administered amanitin toxins or pieces of A. phalloides in lethal doses died from hypoglycemia 1 to 2 days after exposure. The presented puppy had severe hypoglycemia at approximately 12 hours after exposure to amanitins, which is consistent with amanitin poisoning. While the classic 4 stages of amanitin poisoning have been observed in dogs, not all cases present that way. In cases of large exposure to amanitins, or ingestion by puppies, the animal may die within 24 hours. 4 In the presented case, the puppy died approximately 24 hours after exposure to amanitins.
Amanitins are extremely toxic. In dogs the reported IV LD50 of α-amanitin is 0.1 mg/kg body weight, while an oral LD50 has only been established for methyl-γ-amanitin and is estimated to be 0.5 mg/kg body weight. 7 Considering the average concentration of amanitins per mushroom of 4 mg/g, 17 the ingestion of 2 A. phalloides has the potential to be lethal to an adult dog while a smaller amount may kill a puppy. Amanitins inhibit nuclear RNA polymerase II, leading to decreased concentrations of mRNA and decreased protein synthesis and ultimately resulting in cell death. 14 , 19 Hepatocytes, crypt cells, and proximal convoluted tubules of the kidneys are highly susceptible to the toxic effects because of their high metabolic rates. Recently studies have shown that other cellular effects, such as apoptosis, 12 a dose-dependent insulin-releasing and β-cytotoxic effect, 5 may play a role in the toxicity of amanitins.
The diagnostic approach described here provides information crucial in the diagnosis of amanitin poisoning in a dog. It is imperative to consider amanitin toxicosis when evaluating a dog that is presented with acute onset of liver disease, even if there is no history of exposure or no grossly identifiable mushroom material in the stomach content. The time course and severity of clinical signs may be dependent on the dose and the age of the dog. Clinical signs and pathologic findings may be inconclusive, and toxicologic analysis should be performed. Based on preliminary data, applying the LC/MS methodology in suspect cases of amanitin poisoning, serum and urine samples should be collected at various time points beginning as early after exposure as possible. In postmortem presentations liver and kidney samples are suitable for testing to confirm exposure. The suspect mushroom or vomited gastrointestinal contents should also be saved for further analysis. The procedure is especially suited to veterinary diagnostic laboratory situations for which rapid diagnosis of exposure to amanitins is necessary.
Acknowledgements. We thank Dr. Pat McCoy for valuable input on the histopathologic diagnosis and the article. We also thank Dr. Stephen King for his help and timely assistance in providing the diagnostic specimens.
Footnotes
a.
Agilent, Palo Alto, CA.
b.
Phenomenex, Torrance, CA.
c.
Thermo Electron Corp., San Jose, CA.
d.
Axxora LLC, San Diego, CA.
e.
Bühlmann Laboratories AG, Schönenbuch/Basel, Switzerland.
