Abstract
A 5-day-old Thoroughbred foal was submitted to the necropsy service at the University of Kentucky Livestock Disease Diagnostic Center. The foal had a clinical history of seizure activity and severe icterus. A complete blood count and serum chemistry analysis indicated that the foal was anemic (hematocrit, 16%), hyperbilirubinemic (45 mg/dl), and hypoglycemic. At necropsy, all tissues were discolored various shades of yellow. Microscopically, there was degeneration and necrosis of cerebral neurons and cerebellar Purkinje cells; severe hepatocellular degeneration and necrosis; and deposition of amorphous golden-yellow material in the cerebellar granular cell layer, pulmonary alveoli, renal tubular epithelium, splenic trabecula, and the lamina propria of the small and large intestine. The golden-yellow material in the brain, lung, spleen, and small intestine was identified as bilirubin by histochemistry. Based on the macroscopic and microscopic findings, a diagnosis of kernicterus (bilirubin encephalopathy) was made. This report describes a rare case of equine neonatal kernicterus.
Kernicterus (“nuclear jaundice”) is a potentially fatal neurologic disorder induced by the neurotoxic effects of unconjugated bilirubin (UB) on the central nervous system. 1,20,21 Classically, kernicterus is defined as the combined clinical neurologic manifestations and characteristic yellow staining of the basal nuclei induced by high levels of circulating UB. 21 Varying degrees of hyperbilirubinemia are commonly found in neonatal infants, but the high levels associated with kernicterus and permanent neurologic damage are infrequently attained. 12 Infant hyperbilirubinemia is most commonly associated with Rh and ABO isoimmune disorders and less frequently with glucuronyl transferase deficiencies, e.g., Crigler-Najjar syndrome. 1,5,19,20
Clinical manifestations of kernicterus in humans can vary from acute to chronic. In acute cases, infants may present with jaundice, hyperthermia, hypotonia, hypertonia, high pitched crying, retrocollis, opisthotonus, impaired upward gazes, seizures, or acute death. 20 The classical tetrad of chronic kernicterus in humans include the following: athetoid cerebral palsy, hearing impairment, impairment of upward gaze, and enamel dysplasia. 20 Clinical symptoms are consistent with lesions involving the cerebellum; the globus pallidus; and the subthalamic, auditory, vestibular, and oculomotor nuclei. 20,21,25
Macroscopic pathologic changes of kernicterus consist of brain edema, severe jaundice, yellow staining of the basal nuclei, and frequently hepatomegaly and splenomegaly. 1,19,21 Histologic lesions include necrosis of cerebral neurons, Purkinje cell necrosis and loss, yellow pigment in the cerebellar granular cell layer, and deposition of variable amounts of yellow pigment in other organ systems. 1,4,11,21
Kernicterus is most frequently diagnosed in human infants, but reports have been described in Gunn rats, rhesus monkeys, a kitten, and a foal. 4,12,14,15,20,23 To the authors' knowledge, this is the second report of kernicterus in a foal.
A recumbent 5-day-old female Thoroughbred foal with marked seizure activity presented to a veterinary referral center. The attending clinician observed severe icterus; the foal was given a blood transfusion, started on fluid therapy, and treated with phenobarbital and broad spectrum antibiotics. A complete blood count and serum chemistry analysis revealed anemia (hematocrit, 16%), hyperbilirubinemia (45 mg/dl), and hypoglycemia. Despite treatment, the foal died and was submitted to the necropsy service at the University of Kentucky Livestock Disease Diagnostic Center, Lexington, Kentucky.
The foal was thin at necropsy. Regions of the cerebrum, cerebellum, brain stem, lungs, skin, subcutaneous tissues, mesentery, and intestines were discolored various shades of yellow. The spleen was moderately enlarged, and the lungs were red, heavy, and wet. The kidneys were dark red and moderately enlarged. Dark red urine was in the urinary bladder. Tissue samples were taken for routine bacterial culture and for Leptospira sp. florescent antibody (FA) testing. Appropriate tissues were placed in phosphate buffered 10% formalin, allowed to fix for 24 hours, routinely processed, and stained with Harris hematoxylin and eosin for microscopic evaluation.
Pathogenic bacteria were not isolated from any tissue samples, and FA tests were negative for Leptospira sp. Histopathologic changes were noted in the cerebellum, cerebrum, lungs, liver, spleen, kidneys, small intestine, and large intestine. Microscopic alterations in the cerebellum were primarily localized to the Purkinje and granular cell layers. There was Purkinje cell loss, and multiple Purkinje cells were necrotic and characterized by variably angular borders, intense acidophilia, and nuclear pyknosis (Fig. 1). The cerebellar granular cells contained varying amounts of intracytoplasmic globular golden-yellow pigment (Fig. 1). There was severe neuronal degeneration and necrosis within the cerebral cortex (Fig. 2). Many cerebral astrocytes were swollen and had vesicular nuclei, with peripherally displaced chromatin, consistent with Alzheimer type 2 cells. Within the lung, the alveolar spaces contained and the alveolar epithelium was lined by amorphous strands of golden-yellow material (Fig. 3). In addition, the alveoli contained low numbers of macrophages and keratinized squamous epithelial cells. Portions of the alveolar septa were mildly thickened, with small amounts of acidophilic proteinaceous material. Hepatic lesions were suggestive of hypoxia and characterized by severe centrilobular to midzonal hepatocellular degeneration and necrosis with infiltrates of low numbers of neutrophils and macrophages. Many hepatocytes and Kupffer cells contained intracytoplasmic yellow pigment. In addition, varying amounts of golden-yellow pigmented material were evident in the splenic fibrous connective tissue, the renal tubular epithelium, the intestinal lumen, and within the lamina propria of the small and large intestines (Fig. 4). The golden-yellow pigmented material in the cerebellar granule cell layer, alveoli, hepatocytes, splenic connective tissue, small intestinal lumen, and small intestinal lamina propria was confirmed as bilirubin with Hall bilirubin stain. A Perl stain demonstrated iron in alveolar macrophages, splenic macrophages, and Kupffer cells. Significant lesions were not evident in the heart, thymus, or stomach. Based on the clinical, gross, and microscopic findings, a diagnosis of kernicterus was made.
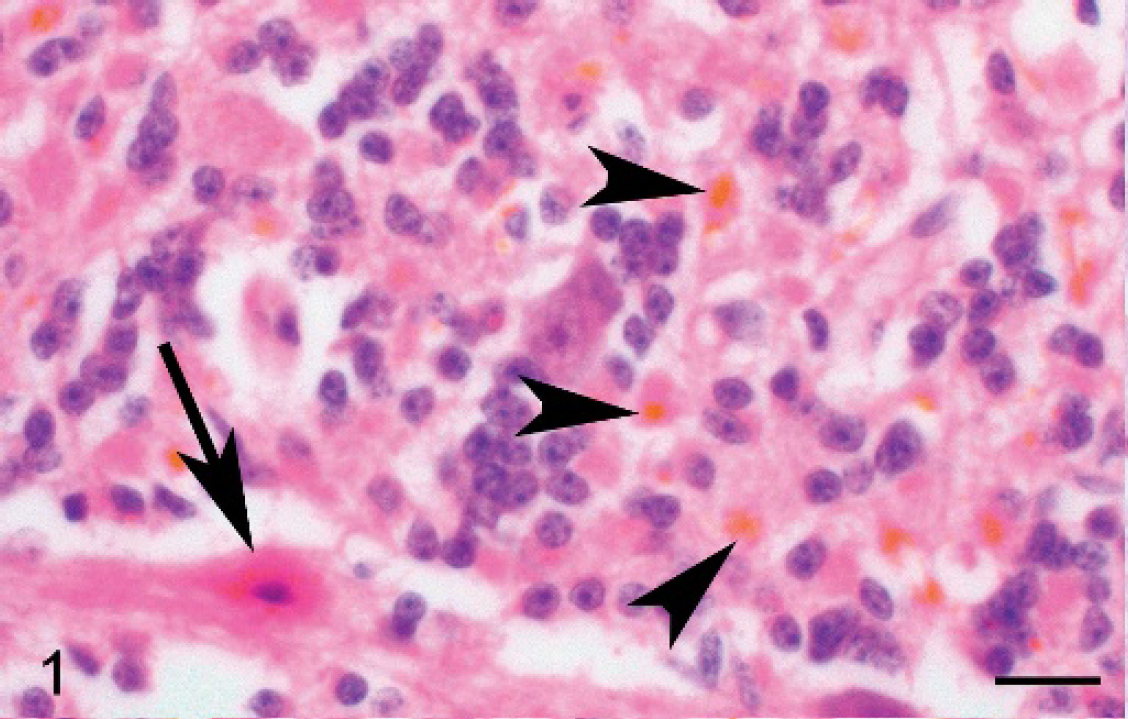
Cerebellum, Purkinje, and granular cell layers, Thoroughbred foal. There is Purkinje cell necrosis (arrow). The granular cell layer contains multifocally distributed golden-yellow globular pigment (arrowheads). Hematoxylin and eosin. Bar = 20 μm.

Cerebrum, Thoroughbred foal. Moderate numbers of neurons are necrotic. Hematoxylin and eosin. Bar = 40 μm.

Lung, Thoroughbred foal. Amorphous strands of golden-yellow pigmented material are within the alveolar spaces, often lining alveolar septa. Hematoxylin and eosin. Bar = 40 μm.

Small intestine, Thoroughbred foal. Globular golden-yellow pigment is distributed in the extracellular lamina propria and within occasional macrophages (arrow). Hematoxylin and eosin. Bar = 40 μm.
Kernicterus is most commonly described in neonatal humans. Newborns are more susceptible to hyperbilirubinemia and bilirubin encephalopathy because of increased bilirubin production, decreased hepatic bilirubin uptake and conjugation, and underdeveloped blood-brain barriers. 5,25 Prematurity, ischemia, sepsis, acidosis, length of exposure, and anemia have been shown to enhance the toxic effects of UB and may increase the likelihood of inducing bilirubin encephalopathy. 1,4,5,15,25
The pathogenesis of kernicterus has been thoroughly studied. Bilirubin is produced through erythrocyte catabolization. 3 As erythrocytes are broken down, carbon monoxide and biliverdin are produced. Macrophages and hepatocytes further catabolize biliverdin to UB, which is nonpolar and water insoluble. Unconjugated bilirubin is then bound to plasma proteins, primarily albumin, and released into the circulation. Within the liver, UB is conjugated with glucuronic acid to form a nontoxic molecule that is excreted in the bile. 5,10 Saturation of plasma protein receptors can result in increased levels of circulating free UB. 1,5,10,25 Water-insoluble UB can enter different cell types and discolor them yellow. In infants, UB can easily cross the underdeveloped blood-brain barrier; enter neurons; and induce degeneration, necrosis, and/or apoptosis. 25 Within the neuron, UB disrupts mitochondrial functions, uncouples oxidative phosphorylation, and inhibits calmodulin-dependent protein kinase II. 5,20,21 Neuronal death occurs because of the disruption of calcium homeostasis and glutamate excitotoxicity. 5,9,20,21 Excitotoxic death occurs because of the negative effect that UB has on the cellular uptake of glutamate, which ultimately allows extracellular glutamate accumulation. Increased levels of extracellular glutamate bind to glutamate receptors and cause increased neuronal excitement, which leads to degeneration, necrosis, and/or apoptosis. 7,9,13,16 The extensive neuronal necrosis, in the neonate reported here, was most likely manifested as a combinatory result of UB toxicity on neurons, hypoxia because of anemia and seizure activity, and hypoglycemia.
The presence of yellow pigmented material within this foal's alveolar spaces is unique. Meconium aspiration is a common microscopic finding in stillborn and neonatal foals that suffer from asphyxiation or periparturient stress and should be considered as the primary differential diagnosis in foals whose alveolar spaces contain yellow pigmented material. Although meconium aspiration, which cannot be definitively excluded, may be, in part, responsible for the alveolar material, an alternative theory of direct bilirubin deposition because of hyperbilirubinemia must be addressed. Yellow discoloration of the pulmonary parenchyma and alveoli, from UB, is rarely reported in the human literature and has been referred to as pulmonary yellow hyaline membranes. 6,11,24 The macro- and microscopic alterations of this rare manifestation are documented by Doshi et al., 6 who reported its incidence in kernicteric infants to be only 33.3%. The significance, occurrence, and pathogenesis of yellow hyaline membranes in humans and animals is not definitively known. 6,11,24 The definitive differentiation of bilirubin from meconium by histochemical methods is difficult. Subtle differences in routine light microscopy and Hall and Perl histochemical stains were evident in this foal, compared with those diagnosed with meconium aspiration (unpublished observations), which suggests that this case may represent a rare case of pulmonary yellow hyaline membranes.
In horses, increased levels of UB have been documented in cases of neonatal liver disease, isoerythrolysis, glucose-6-phosphate dehydrogenase deficiency, and, in other cases, of intravascular hemolysis. 2,3,8,18,22 In this foal, the cause of hyperbilirubinemia was not determined because of the short clinical course and the lack of antemortem ancillary tests on the mare and the foal. Although unproven, the age of the foal, the clinical history, and the severe anemia were consistent with neonatal isoerythrolysis (NI). Pathologic findings of foals with NI can consist of variable degrees of anemia, icterus, splenomegaly, centrilobular hepatocellular necrosis, and pigmentary nephrosis. 17 Based on the authors' experience with NI, the macroscopic findings, microscopic findings, and bilirubin deposition can occur in similar organs (excluding brain) but to lesser degrees when compared with kernicteric animals. Additional differences in this kernicteric neonate compared with foals with NI include deposition of UB within the cerebellar granular cell layer, the extensive neuronal and Purkinje cell necrosis, and the presence of yellow pigmented material in the alveoli.
Kernicterus is an extremely rare condition in animals and has only been reported in Gunn rats, rhesus monkeys, a kitten, and a foal with neonatal isoerythrolysis. 4,14,15,23 A diagnosis of kernicterus, bilirubin encephalopathy, was made in this foal based on the clinical signs, gross and microscopic lesions, and identification of bilirubin in multiple organ systems. Foals with a history or clinical signs suggestive of neonatal isoerythrolysis and seizure activity should be carefully examined for kernicterus at necropsy.
Acknowledgements. This report was published (06-01-034) with the permission of the dean and director of the Kentucky Agriculture Experiment Station and College of Agriculture, University of Kentucky, Lexington.
