Abstract
Abomasal coccidiosis was diagnosed in an 11-month-old female sheep that died after a 2-week period of anorexia and diarrhea. The abomasal mucosa was grossly thickened with a nodular surface and focal areas of hemorrhage. Microscopically, the middle to deep abomasal mucosa contained many intact and ruptured giant protozoal schizonts associated with hyperplasia of mucous neck cells, parietal cell atrophy, moderate lymphocytic-plasmacytic inflammation, and fibrosis centered on mineralized remnants of degenerate schizont walls. Sexual tissue stages and oocysts were not present. Microscopic features of schizonts and ultrastuctural features of merozoites were comparable to previous descriptions of Eimeria (Globidium) gilruthi, a coccidian of uncertain taxonomic status historically associated with incidental infections of the abomasum in sheep and goats. The distinctive lesions suggest that, similar to ostertagiasis, heavy coccidial infection of the abomasum should be considered as a cause of anorexia, diarrhea, and proliferative abomasitis in sheep.
Abomasal coccidiosis attributed to Eimeria (Globidium) gilruthi infection is sporadically reported in sheep 5,6,10,11,14,16 and goats 12,16 from different parts of the world. Infections are generally believed to be incidental and are characterized by the presence of giant schizonts within the mucosa of the abomasum and, less commonly, the duodenum. 1,6,11,14 This article reports a case of abomasal coccidiosis in a sheep associated with proliferative abomasitis, parietal cell atrophy, and mucosal fibrosis.
An 11-month-old, female, cross-breed sheep from north-central Indiana was submitted dead to the Animal Disease Diagnostic Laboratory at Purdue University for complete necropsy examination. The animal died after a 2-week period of diarrhea and anorexia. Treatments included 2 doses of oxytetracycline administered 5 days apart. The animal had been recently shown at county and state fairs.
At necropsy, the abomasum was distended with approximately 1 liter of turbid reddish-brown, watery fluid admixed with a small amount of plant material and sand. The abomasal wall was diffusely thickened by edema, and the mucosal surface was faintly nodular and mottled dark red-purple with patchy areas of hemorrhage (Fig. 1). The terminal colon contained unformed, dark green fecal material. Adult nematodes were not detected in abomasal or intestinal contents.
Approximately 40% of the cranioventral lung lobes was firm and dark red with multifocal hemorrhage and edematous distension of interlobular septa. The overlying pleural surface was roughened with loosely adherent strands of fibrin.
Selected tissues were fixed in 10% neutral buffered formalin, routinely processed, paraffin-embedded, sectioned, and stained with hematoxylin and eosin (HE) and Brown and Brenn Gram stain. Formalin-fixed specimens of the abomasum were postfixed in glutaraldehyde followed by osmium-ferrocyanide, stained with uranyl acetate and lead citrate, and examined with a transmission electron microscope. Fresh samples of lung, abomasum, colon, and mesenteric lymph node were routinely cultured for aerobic, anaerobic, and Salmonella bacteria. A fecal sample was submitted to the parasitology laboratory for qualitative flotation.
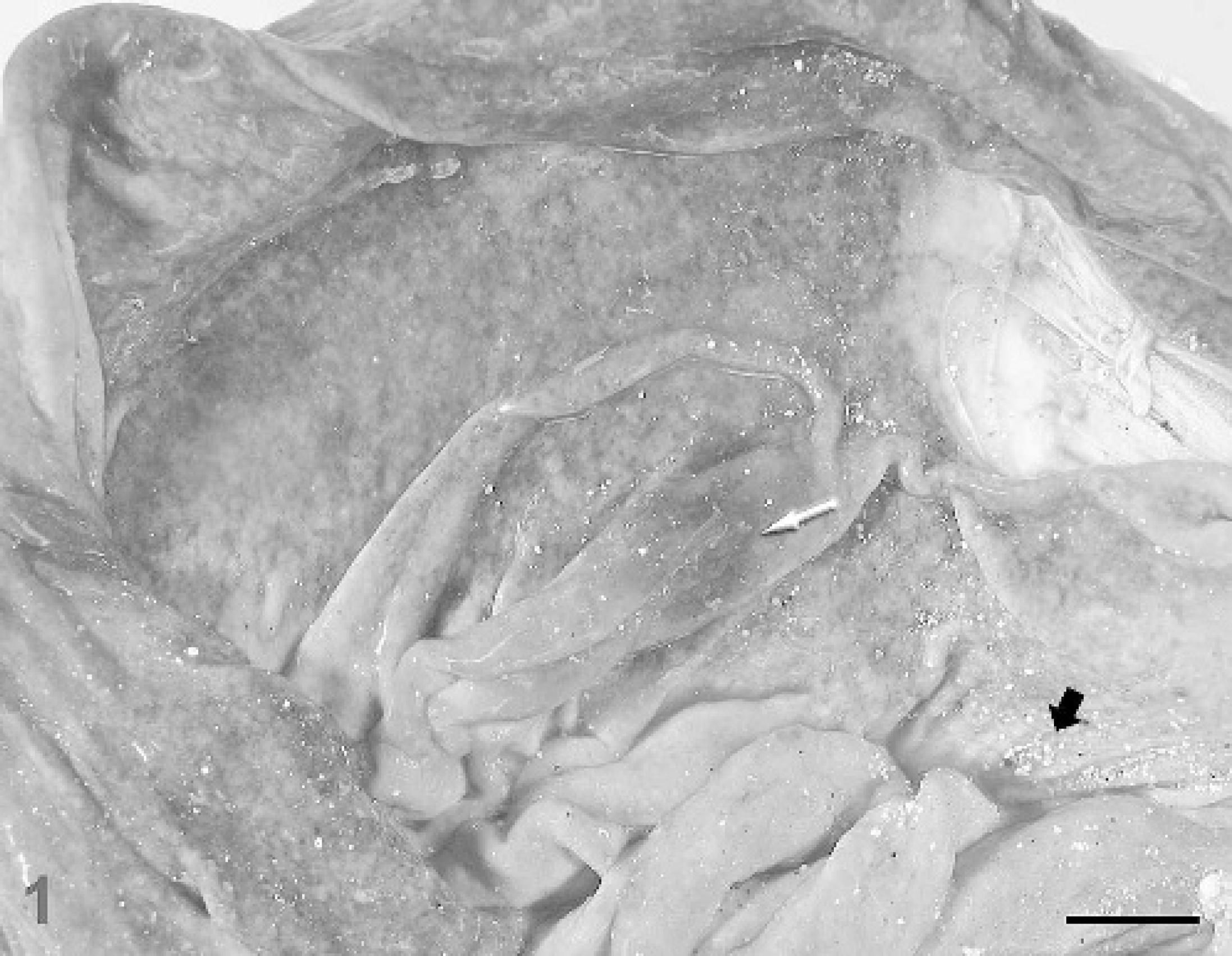
Abomasum; sheep. The mucosal surface is diffusely nodular and congested with focal areas of hemorrhage (white arrow). Note the presence of sand (black arrow) on the mucosal surface. Bar = 1.5 cm.
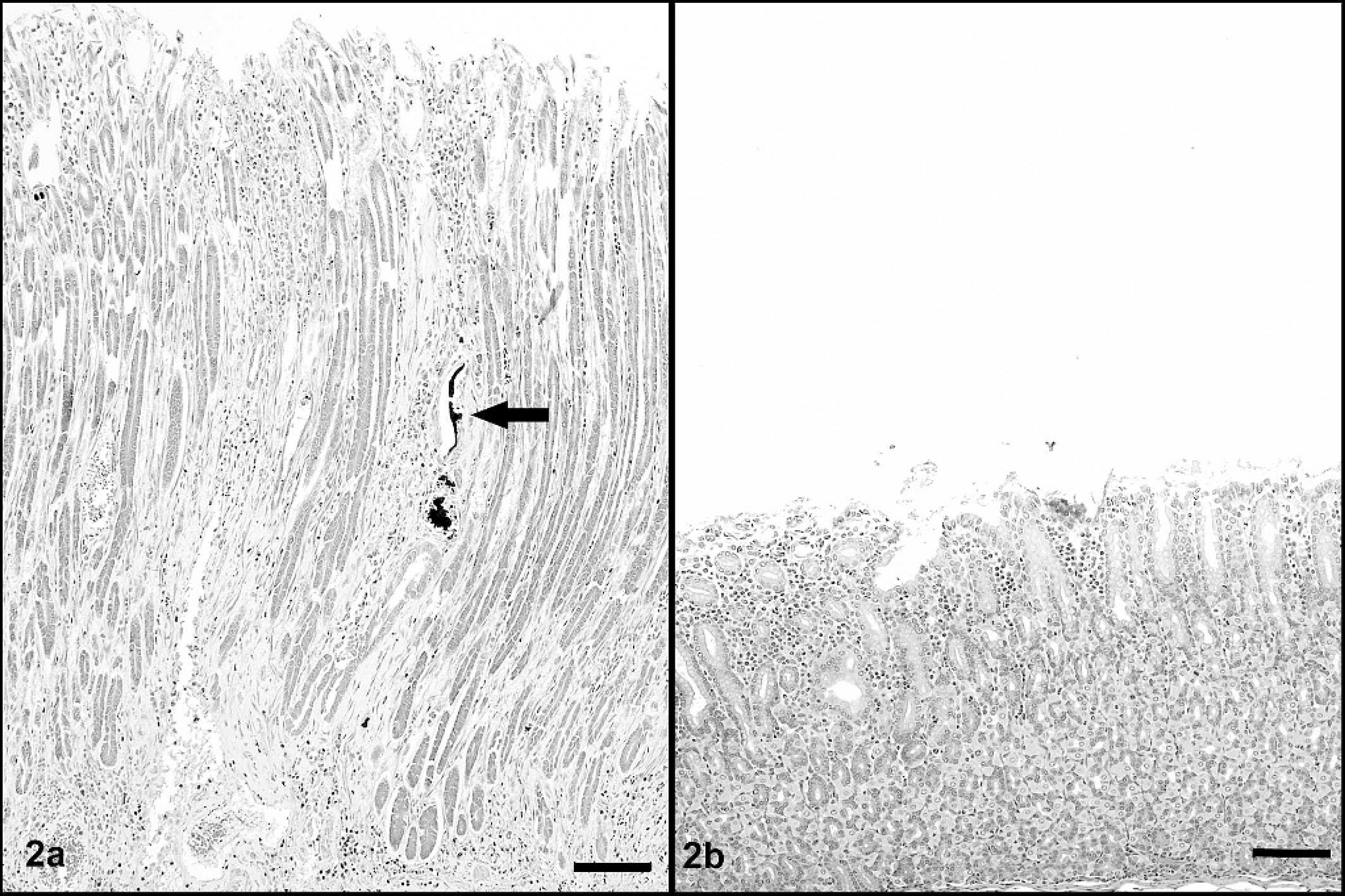
Abomasum; sheep.
Microscopically, the abomasal mucosa was diffusely thickened by elongated fundic glands lined by hyperplastic mucous neck cells and contained markedly decreased numbers of parietal cells (Fig. 2). Scattered fundic glands had loss or attenuation of the epithelial lining and contained necrotic cell debris; mitotic figures were occasionally observed in the isthmus and neck regions. Many intact or degenerate protozoal schizonts measuring 108–400 μm × 299–826 μm were observed in the middle to deep mucosa and rarely extended into the underlying submucosa. Schizonts were round to ovoid, thick-walled, and contained thousands of merozoites and variable numbers of circular blastophores within a large parasitophorous vacuole (Fig. 3). Merozoites were elongate, measured approximately 1.5 μm × 5 μm and had a nucleus at one end. The schizont wall was homogenous, palely eosinophilic, 8–15 μm thick, and occasionally contained a single hypertrophied host cell nucleus that measured up to 12 μm × 74 μm. Many schizonts were ruptured, infiltrated by neutrophils and macrophages, and sometimes surrounded by hemorrhage and fibrin (Fig. 4). Many free and intrahistiocytic merozoites were observed within and around ruptured schizonts. The mucosa was multifocally effaced by areas of fibrosis centered on mineralized fragments of schizont walls. The lamina propria and submucosa were diffusely expanded by edema and moderately infiltrated with lymphocytes and plasma cells, including a few microscopic lymphoid nodules, with fewer neutrophils, eosinophils, and macrophages. In one area, the superficial mucosa was eroded and covered by exudate composed of neutrophils, fibrin, and necrotic cell debris admixed with large dense aggregates of gram-positive rods and cocci and gram-negative rods.
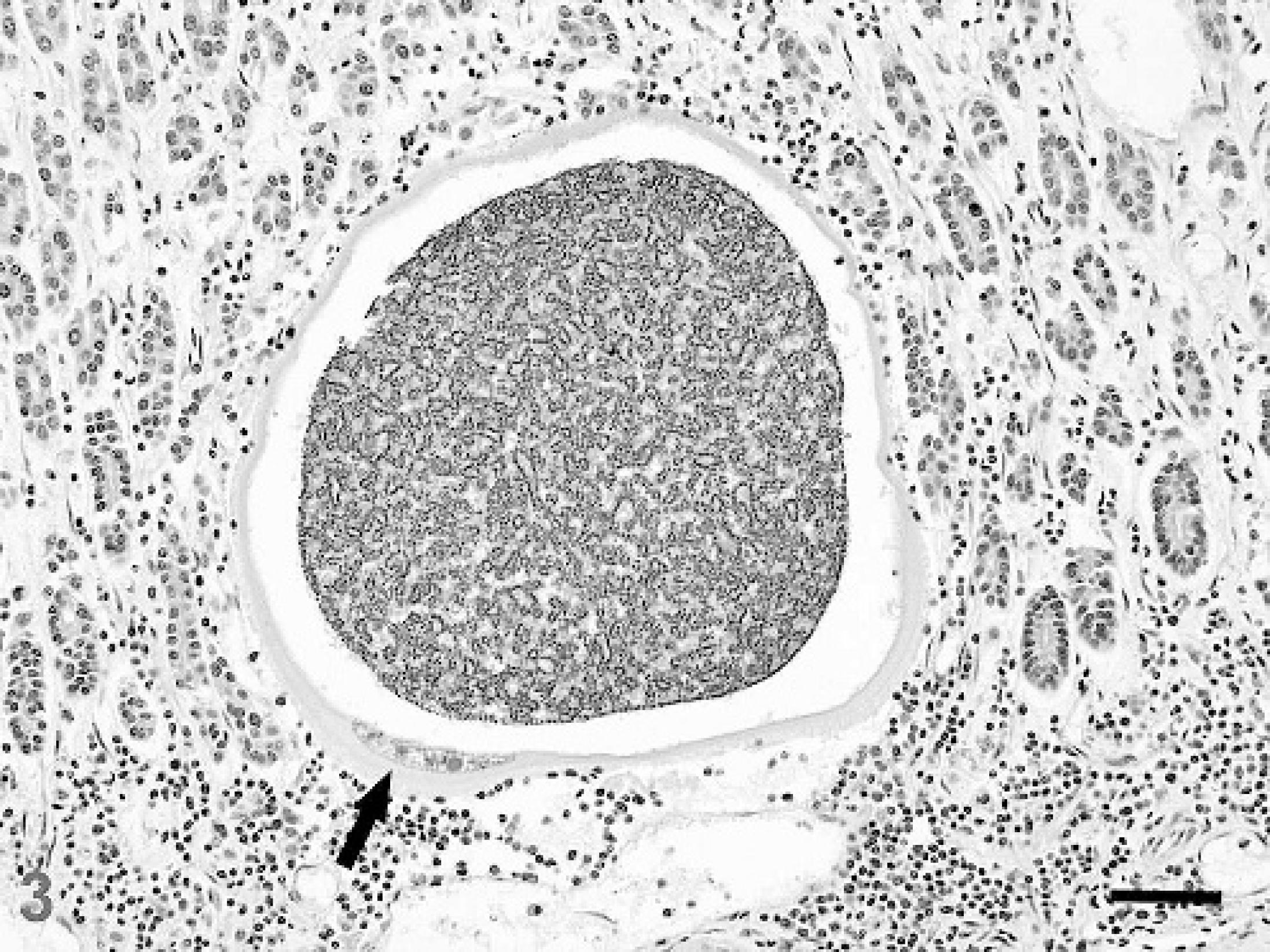
Abomasum; sheep. A 299 μm × 317 μm protozoal schizont in the abomasal mucosa is surrounded by mild lymphocytic-plasmacytic inflammation. The schizont has a thick eosinophilic wall containing a remarkably hypertrophied host cell nucleus (arrow) and is filled with thousands of merozoites and blastophores within a large parasitophorous vacuole. HE stain. Bar = 50 μm.
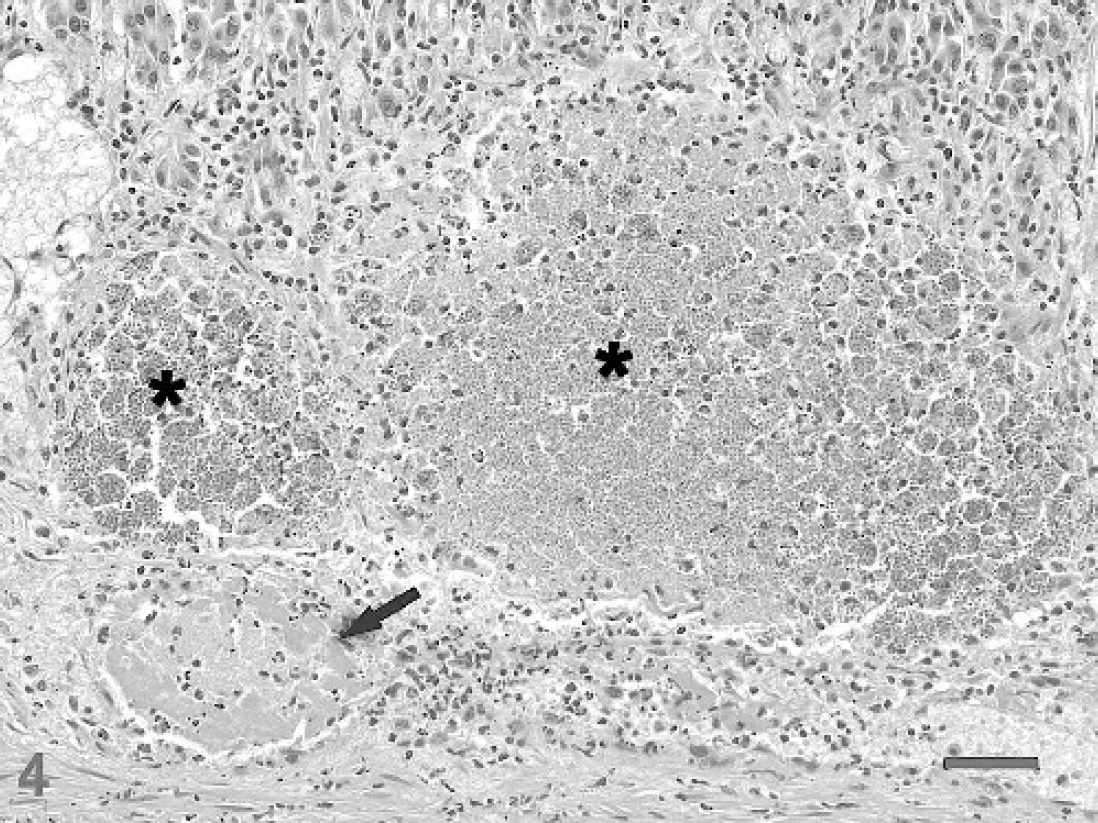
Abomasum; sheep. Two degenerate schizonts (*) are infiltrated by neutrophils and surrounded by inflammation, hemorrhage, and necrosis. Note the thrombosed blood vessel (arrow) adjacent to the ruptured schizonts. HE stain. Bar = 50 μm.
Ultrastructurally, merozoites were ovoid to elongate with tapered anterior ends and rounded posterior ends, measured 3.72-6.56 μm long × 1.36-2.56 μm wide, and possessed typical apicomplexan features, 2,4,9,13 including a pellicle, polar ring, conoid, paired rhoptries, and numerous micronemes in the anterior portion of the cell (Fig. 5). In cross-sectional views, 22 subpellicular microtubules were observed in the apical complex region (Fig. 6). The nucleus was located in the posterior half of the cell and contained distinct peripheral plaques of electron-dense heterochromatin. Virtually all merozoites contained one to four 0.64-1.52 mm diameter, single membrane-bound ovoidal bodies situated anterior to the nucleus. Each ovoidal body contained abundant granular material and a single electron-dense endosome.
Schizonts were restricted to the abomasum. Neither sexual stages nor oocysts were observed in the abomasum, intestines, or extraintestinal tissues. Qualitative flotation of feces yielded a moderate number of Nematodirus sp. eggs, but no coccidia. Whereas Nematodirus spp. can cause enteritis with villus atrophy in the proximal small intestine of ruminants, 1 small intestinal lesions with nematode larvae were not observed microscopically. Adult nematodes were not identified in the small intestinal lumen of the sheep, though it is possible that a small number of worms may have been present and overlooked. Escherichia coli, Streptococcus sp., Corynebacterim sp., and Clostridium perfringens were cultured from the abomasum. The isolates were morphologically consistent with the bacteria observed in histologic sections. The referring veterinarian declined polyerase chain reaction (PCR) typing of the C. perfringens isolate.
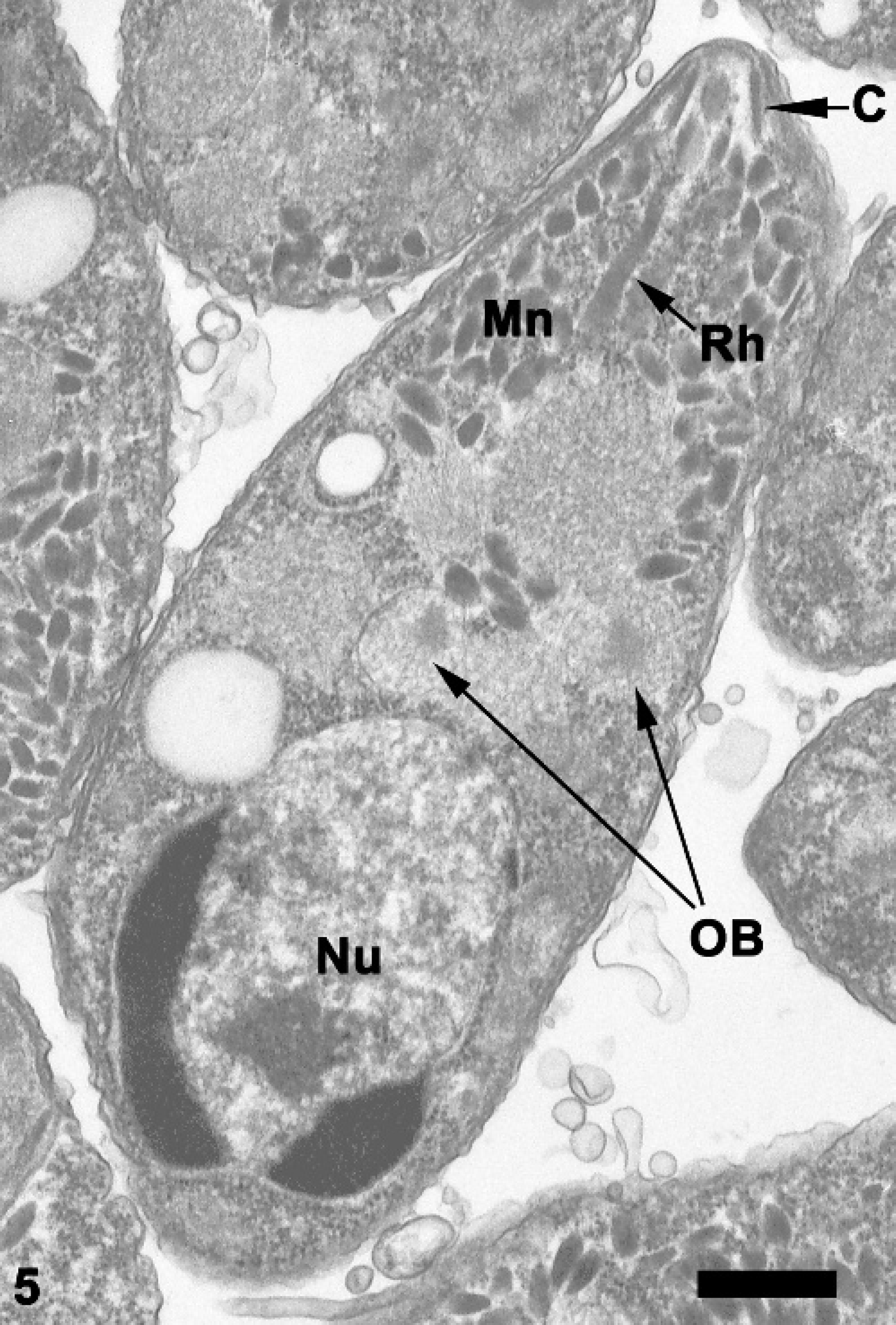
Abomasum; sheep. Longitudinal section through a merozoite with typical apicomplexan features including conoid (C), rhoptries (Rh), and micronemes (MN). A posteriorly placed nucleus (Nu) and several ovoidal bodies (OB) are present. Transmission electron micrograph. Bar = 0.2 μm.
Microscopically, lesions in the cranioventral lung lobes consisted of acute fibrinosuppurative bronchopneumonia with necrosis, capillary thrombosis and moderate fibrinous pleuritis, and colonies of small bacterial rods. Mannheimia hemolytica was aerobically cultured from lung tissue.
Light microscopic features of schizonts and ultrastructural features of merozoites from the abomasum of this sheep are consistent with previous descriptions of E. gilruthi. 5,6,10,11,13,14 Because only the schizont stage of E. gilruthi is known, the taxonomic status of the parasite is uncertain. Definitive identification of coccidian genera and species is based on morphologic characteristics of sporulated oocysts, including the number of sporocysts, distribution of sporozoites, size, shape, and appearance of the oocyst wall. 7 Ultrastructurally, the presence of 2 rhoptries and numerous micronemes in the apical complex of merozoites is consistent with Eimeria spp., 2 and the distinctive ovoidal bodies are apparently unique to E. gilruthi. 13 Eimeria spp. are homoxenous, completing all endogenous stages of the life cycle within 1 host. 4 The absence of oocysts and sexual tissue stages suggests that sheep may be abnormal hosts for E. gilruthi, with infections resulting in 1 or more generations of schizogony in the abomasum, but without progression to gametogony. Members of the genus Eimeria are normally highly host-specific, and cross-transmission between different host genera, including sheep and goats, is uncommon. 8
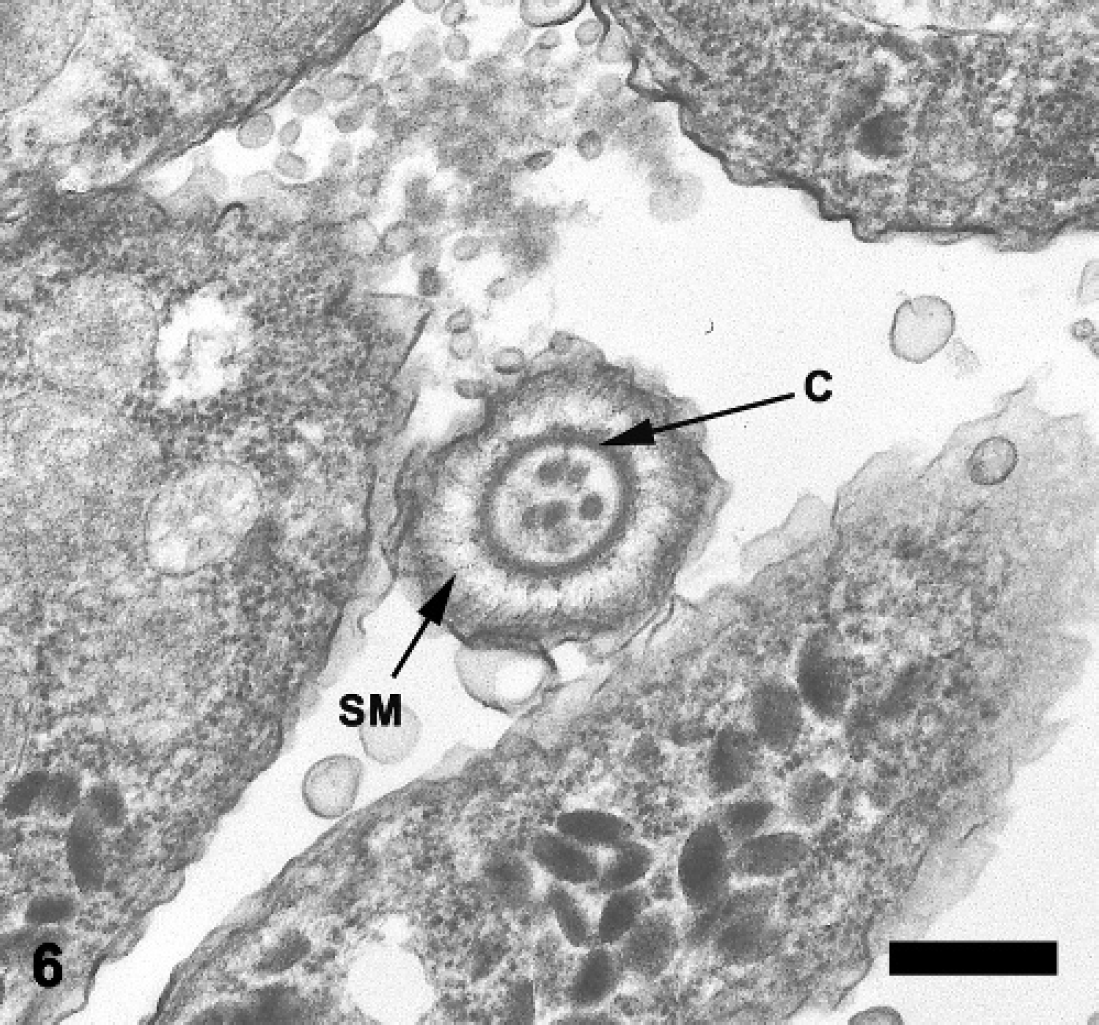
Abomasum; sheep. Cross section through the apical complex of a merozoite demonstrating the subpellicular microtubules (SM) and conoid (C). Transmission electron micrograph. Bar = 0.4 μm.
The marked proliferative abomasitis, fibrosis, and parietal cell atrophy in this sheep are distinct from the incidental white pinpoint foci in the abomasum described in most previous reports of abomasal coccidiosis. 1,6,10,16 Gross and microscopic changes in the abomasal mucosa were similar to those of ostertagiasis, in which progressive mucous neck cell hyperplasia and loss of parietal cells leads to hypochlorhydria and elevation of abomasal pH, which may approach or exceed neutrality. 1,15 Alkalinization of the abomasal contents is associated with development of diarrhea through uncertain mechanisms 1 and can cause overgrowth of rumen microbes when the abomasal pH rises to 4 or higher. 15 Abomasal pH was not, however, evaluated in this sheep. Acute bronchopneumonia caused by Mannheimia hemolytica presumably also contributed to the death of the animal.
To the authors' knowledge, this is the first report of abomasal coccidiosis in a sheep associated with diffuse mucous neck cell hyperplasia and parietal cell atrophy. The intralesional schizonts and merozoites were morphologically consistent with previous descriptions of E. gilruthi; however, definitive species identification was not possible. Abomasal lesions were similar to those of ostertagiasis in ruminants and suggest that abomasal coccidiosis should be considered as a potential cause of anorexia, diarrhea, and proliferative abomasitis in sheep.
Acknowledgements. The authors thank Dr. Joe Camp for reviewing the manuscript, Phyllis Lockard for assistance with transmission electron microscopy, Steve Vollmer for help with digital imaging, and Dr. Cheryl Miller for submission of this interesting case.
