Abstract
Background:
Acute musculoskeletal pain is one of the common causes of emergency admissions. Topical analgesics are an easily tolerated option in the treatment of these pains.
Objective:
This study aimed to compare the efficacy of topical capsaicin and topical non-steroidal anti-inflammatory drugs in acute musculoskeletal injuries.
Methods:
In this prospective, randomized, controlled, double-blinded study, patients were allocated to two groups—the topical non-steroidal anti-inflammatory drug (n = 60) and topical capsaicin (n = 59). For both groups, the first dose treatment was applied as a thin layer in the emergency department to a maximum area of 5 × 5 cm. The remaining doses were administered at home by the patients for 72 h. The initial visual analog scale scores were compared with the 60th and 120th minute as well as the 24th and 72nd hour values. Differences between the visual analog scale scores, clinical response to the treatment, and side effects were evaluated.
Results:
The proportional reduction in visual analog scale scores for topical capsaicin was significantly higher, especially at 36 and 72 h. These rates were 0.75 (95% confidence interval: 0.77–1.42; p = 0.029) and 9.08 (95% confidence interval: 1.02–17.14; p = 0.028) at 36 h, respectively. At 72 h, they were 1.27 (95% confidence interval: 0.48–2.05; p = 0.002) and 17.07 (95% confidence interval: 8.39–25.76; p < 0.001), respectively. In the topical capsaicin group, the clinical response to treatment was significantly higher (p = 0.001). Treatment-related systemic side effects were experienced only in the topical non-steroidal anti-inflammatory drug group (1.7%).
Conclusion:
Topical capsaicin can be used safely in patients presenting to the emergency department with acute pain with its high analgesic efficacy and absence of systemic side effects.
Introduction
Pain is discussed under two main categories as nociceptive (somatic and visceral) and neuropathic pain according to its formation mechanism. 1 Somatic pain is usually seen after acute musculoskeletal trauma in emergency departments. Musculoskeletal injuries constitute about 10% of emergency department admissions. 2 Many medications can be used as analgesics after musculoskeletal trauma. Non-steroidal anti-inflammatory drugs (NSAIDs) are among the most essential ones. NSAIDs can be administered intravenously, intramuscularly, as well as topically.
Topical analgesics are often prescribed and easily tolerated in pain originating from local trauma. The primary purpose of topical administration is to reduce the side effects of the systemic intake of the same drug. The use of topical NSAIDs instead of oral NSAIDs is recommended for mild to moderate pain associated with joint involvement in diseases such as osteoarthritis and rheumatoid arthritis. These treatment recommendations of the European and American Rheumatology Associations include topical use of capsaicin in addition to NSAIDs. 3
Capsaicin {6-nonenamide, N-((4-hydroxy-3-methoxy-phenyl) methyl)-8-methyl-(6E)} is a natural alkaloid that gives the red pepper (
It is known that capsaicin creams, whose concentration ranges between 0.025% and 0.075%, have been used for many years in the treatment of temporary joint and muscle pain, often without requiring a prescription. 7 However, the traditional treatments with NSAIDs, strong competitors in pain management, have always been a matter of concern. In past studies, the efficacy of these two treatments in the presence of chronic pain was frequently discussed. This study aimed to investigate the effectiveness of capsaicin cream in the treatment of acute musculoskeletal pain. For this purpose, we compared the analgesic efficacy of topical capsaicin with topical NSAID (ibuprofen), a frequently preferred treatment method.
Methods
Study design and setting
The study was designed as a prospective parallel randomized controlled trial using restricted randomization via the Random Allocation Software (RAS) 8 with random permuted blocks of 4 and an allocation ratio of 1:1 according to the Consolidated Standards of Reporting Trials (CONSORT) guidelines. 9
All patients were informed in advance about the study procedures and written consent was obtained from the volunteers, who agreed to participate. Our study was conducted in the emergency department of a university hospital between 1 June 2019 and 31 July 2019. The study was approved by the Ethics Committee at Ataturk University Faculty of Medicine.
Patients with isolated upper extremity trauma (UET) who had only blunt trauma and had no fracture and/or dislocation were included in the study. Eligible patients who met the inclusion and exclusion criteria were randomized to receive one of the two treatments. The patients were allocated to two groups: (1) topical NSAID and (2) topical capsaicin groups.
Sample size and patient selection
A literature study evaluating the efficacy of capsaicin in the treatment of pain associated with diabetic neuropathy was used for sample size calculation. 10 We calculated that at least 36 patients were required for each patient group to determine an average 10% reduction in pain scores between the two treatment groups (type I error 0.05 and test strength 90%). 11 We found that the difference in the decrease in pain intensity between the two groups was 17%.
Patients aged 18 years or older and having a visual analog scale (VAS) score of 5 or more, who were admitted to the emergency department with acute UET complaints, were assessed for their suitability for the study. Those who did not want to participate in the research, a time lapse of more than 6 h after trauma, who were pregnant or breastfeeding, patients with chronic diseases and chronic pain conditions, who had a history of allergic disease and drug allergy, history of gastrointestinal system bleeding, consumptions of regular medication (such as antihypertensive, anticoagulant, antidiabetic, and NSAIDs), cases with a fracture and/or discoloration, additional trauma findings, inability to give written consent (non-Turkish speaking, having an altered mental state), and those who had received any analgesic treatment before applying to the emergency room were excluded from the study. Patients who received an additional analgesic drug while the experiment was in progress and patients who did not continue the treatment according to the methodology were excluded as well.
Interventions
While cream containing 5% ibuprofen was administered to patients in the topical NSAID group (Dolgit Cream Adeka Pharmaceutical Industry and Trade, İstanbul, Turkey), patients in the topical capsaicin group received a cream containing 0.05% capsaicin (Sanli Pharmaceutical Pharmaceutical Co., İstanbul, Turkey). To conceal the visual difference between the two topical drugs, food color was added to the creams. Both drugs were numbered by placing them in the same opaque plastic containers. The first dose was administered within 30 min after the patient was examined in the emergency room. The clinician was blinded regarding the medication used. A thin layer of cream was applied to the trauma site where the pain was described in the upper extremity with a maximum application area of 5 × 5 cm and rubbed for 1 min. All topical applications were performed by the same clinician, and the patients were observed for 120 min. Discharged patients were instructed to apply the medication as a thin layer and rub it onto the painful area three times a day for 72 h. Patients included in the study were called to the emergency department for a control visit after 72 h. The amount of medication remaining in the container given to the patients was recorded.
Outcome measurements
Demographic data, vital parameters, VAS values at the time of presentation, as well as mechanism and localization of the trauma were recorded. A VAS was used by patients to rate their pain on a numeric scale from 0 (
Side effects that were thought to be related to topical drug administration were recorded during the treatment process. They were classified as local and systemic effects.
Statistical analysis
All statistical analyses were performed using the Statistical Package for the Social Sciences, version 20.0 (SPSS, Chicago, IL, USA). Percentages and frequencies for categorical variables and mean (±standard deviation (SD)) values for continuous variables were determined. The suitability of the numerical variables to normal distribution was evaluated with the Shapiro–Wilk test. Student’s t-test or the Mann–Whitney U tests were used for pairwise comparisons of continuous variables, depending on the data distribution. General estimating equation (GEE) modeling was used to discover any significant difference in repeated measurements of patients’ VAS scores.
Results
A total of 18,654 patients were admitted to the emergency department during the study period. Among these, 972 patients applied for UET were evaluated for suitability; 812 patients were excluded from the study. The remaining 160 patients were randomized into two groups receiving topical NSAID or topical capsaicin. During the study, 19 patients in the NSAID group and 18 patients in the topical capsaicin group could not be reached by telephone. Besides, four patients were excluded because they used additional medications. As a result, 119 patients completed the study, of which 60 were in the topical NSAID group and 59 were in the topical capsaicin group (Figure 1; CONSORT).
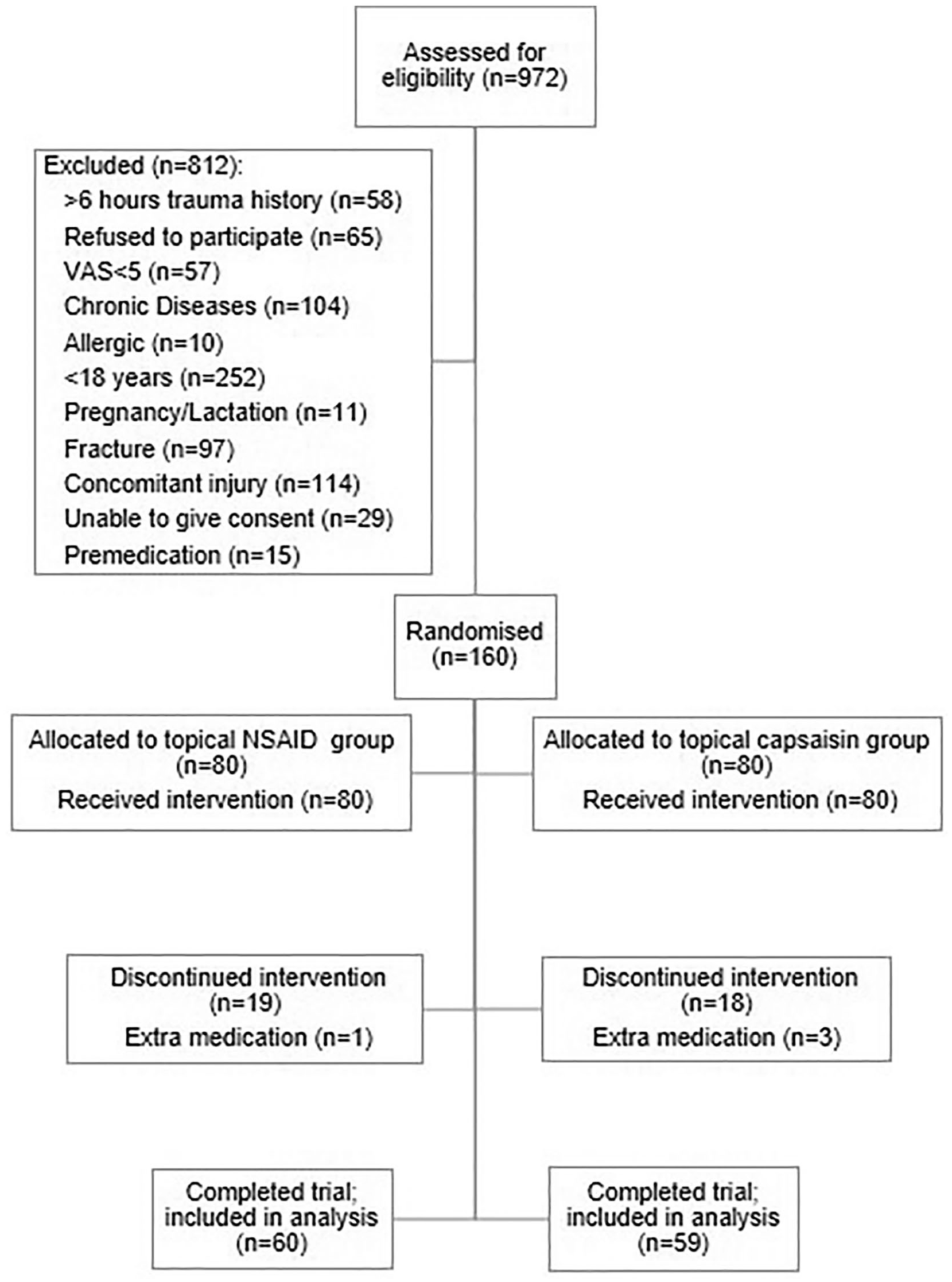
CONSORT (Consolidated Standards of Reporting Trials) diagram.
Fifty-five patients (46.2%) were female and 64 (53.8%) were male. The mean age was 43 years (SD: 12.4, range: 18–63). The most common mechanisms of injury were falls (30.3%), sports injuries (22.7%), motor vehicle accidents (14.3%), strain/sprain (11.8%), and others (21%) (Table 1). There was no significant difference between the treatment groups regarding age, sex, and trauma distribution (p values were 0.325, 0.641, and 0.905, respectively).
Demographic and baseline characteristics.
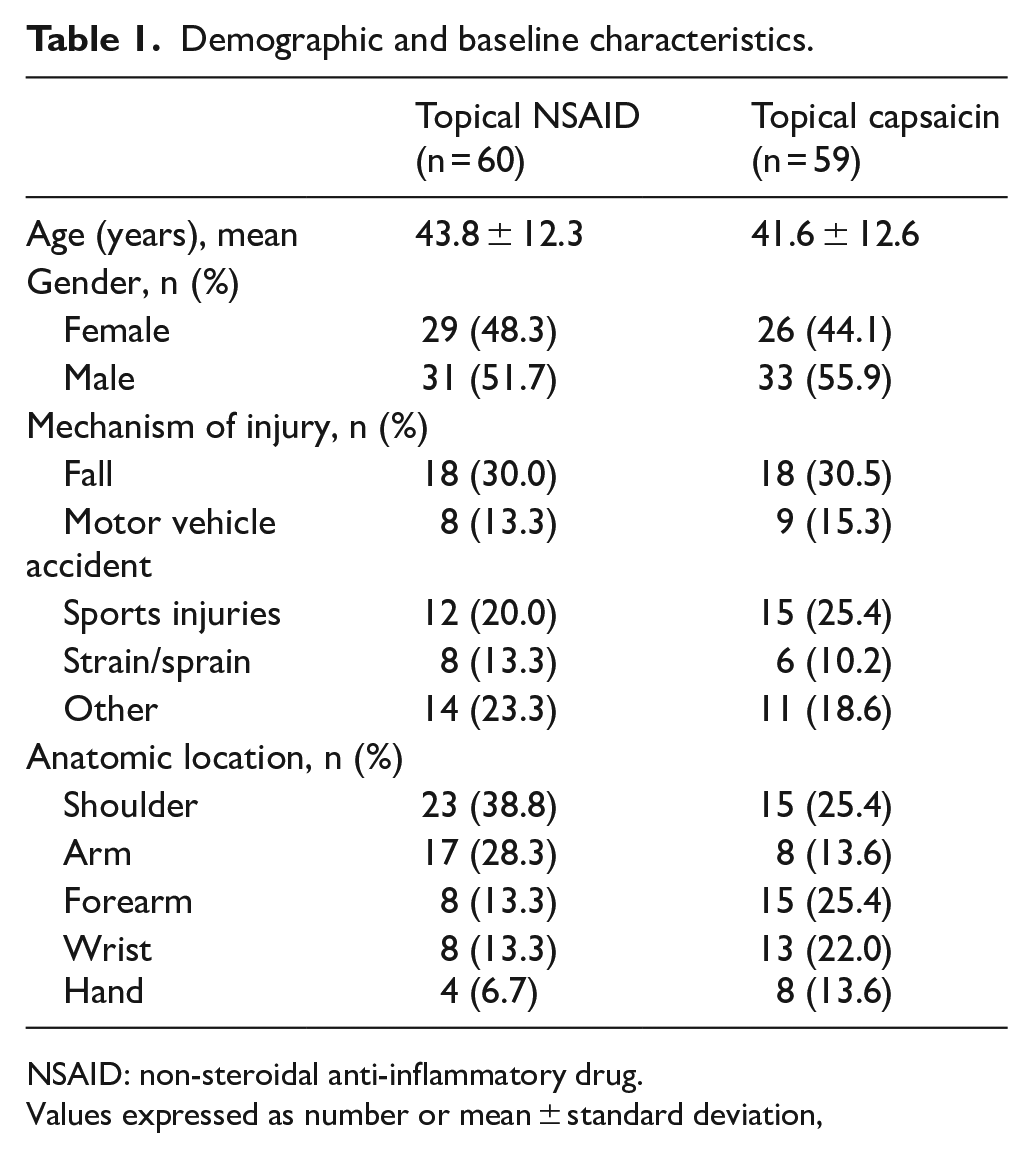
NSAID: non-steroidal anti-inflammatory drug.
Values expressed as number or mean ± standard deviation,
The mean V0 values of the groups were 7.47 (capsaicin) and 7.15 (ibuprofen), and this difference was not statistically significant (p = 0.335). GEE modeling showed a statistically significant difference between the mean VAS values at each measurement time (p = 0.000). These mean VAS scores for all measurement times are shown in Figure 2. When the mean delta VAS values were examined, the V0 – V1, V0 – V2, V0 – V3, and V0 – V4 differences were higher in the topical capsaicin group. However, this difference was only significant for V0 – V3 (mean difference: 0.75, 95% confidence interval (CI): 0.77–1.42; p = 0.029) and V0 – V4 (mean difference: 1.27, 95% CI: 0.48–2.05; p = 0.002) (Table 2). When the percentages of decrease in the VAS scores compared to the baseline were examined, the mean values for V0 – V1, V0 – V2, V0 – V3, and V0 – V4 were higher in the topical capsaicin group. Likewise, the difference between the ratios was only significant in between V0 – V3 (mean difference: 9.08, 95% CI: 1.02–17.14; p = 0.028) and V0 – V4 (mean difference: 17.07, 95% CI: 8.39–25.76; p < 0.001).
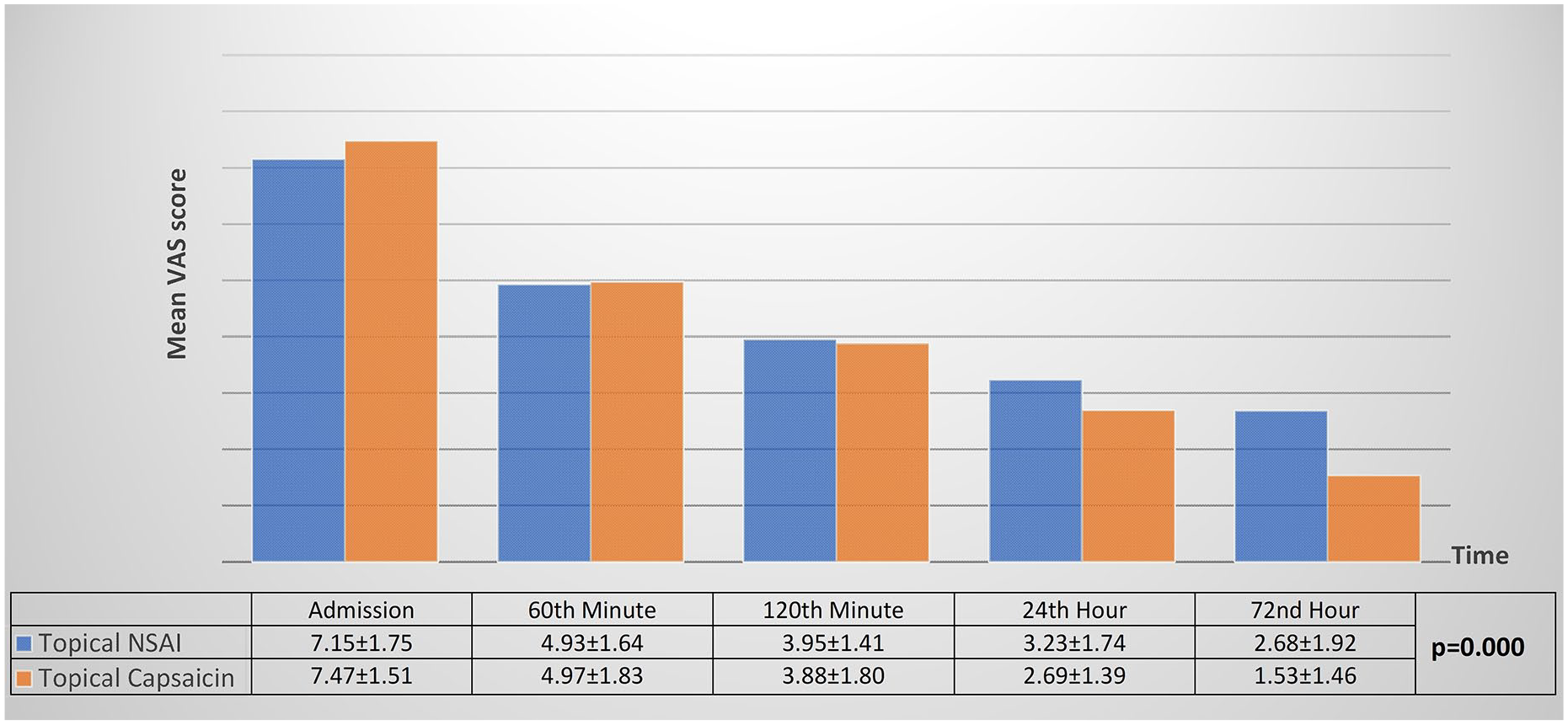
Mean VAS scores of patients at each time point. General estimating equation modeling showed a statistically significant difference between the mean VAS values at each measurement time (p = 0.000).
Delta VAS values of patient groups.
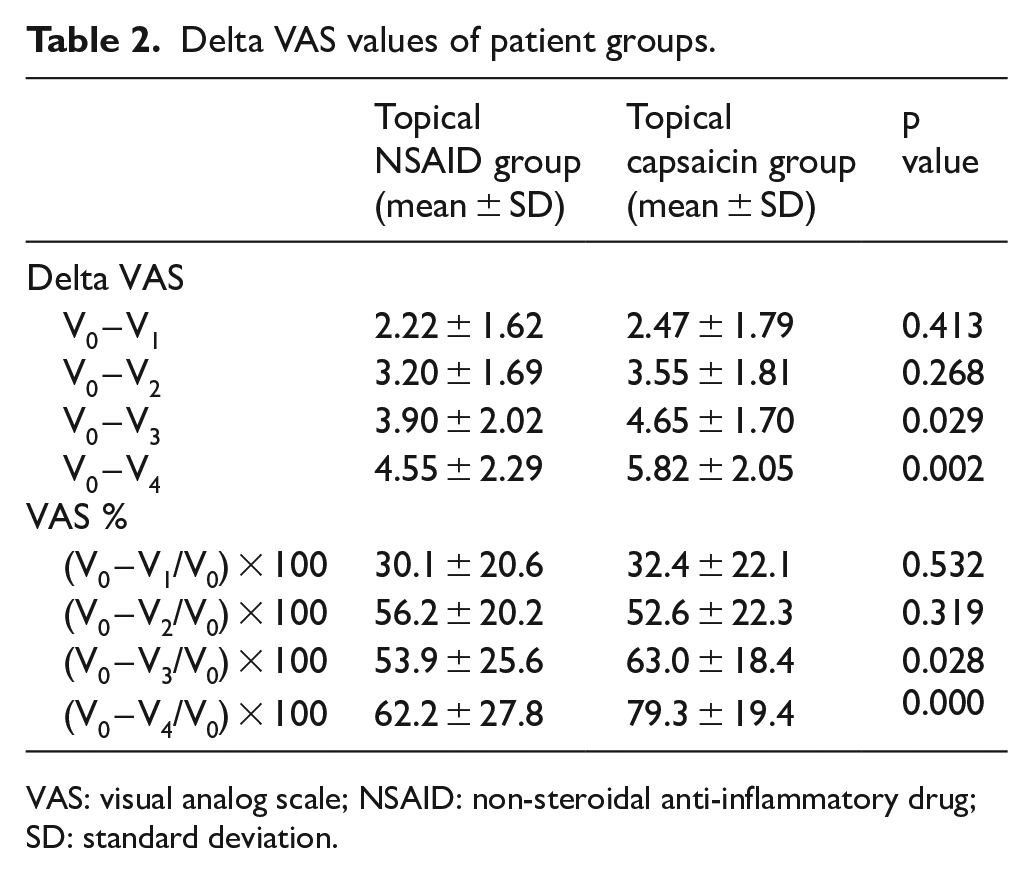
VAS: visual analog scale; NSAID: non-steroidal anti-inflammatory drug; SD: standard deviation.
At the end of the study, patients with VAS scores decreased by more than 50% were 36/60 (60.0%) in the topical NSAID group and 50/59 (84.7%) in the topical capsaicin group (p = 0.003). The proportion of patients with a VAS value ⩽4 at the end of the study was 37/60 (61.7%) in the topical NSAID group and 52/59 (88.1%) in the topical capsaicin group (p = 0.001). When these two results were evaluated, the clinical response to treatment was significantly higher in the topical capsaicin group.
The amount of medication remaining in the containers after use was 4.8 ± 4.6 g for capsaicin and 6.5 ± 5.8 g for NSAID, which was a non-significant difference (p = 0.152).
The incidence of total side effects was 10% and 18.6% in the topical NSAID and topical capsaicin groups, respectively (p = 0.178). The side effects observed in two topical treatment groups are listed in Table 3. Systemic side effects were experienced only in the topical NSAID group. There was not any significant difference between the two groups regarding the incidence of transient skin reactions (p > 0.05).
Treatment-Related Side Effects.
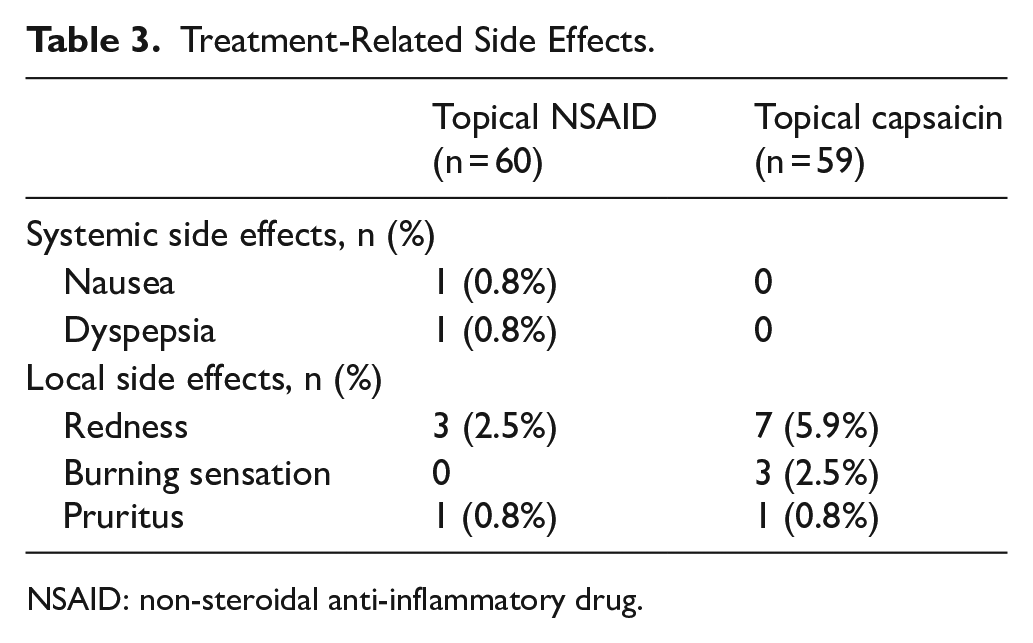
NSAID: non-steroidal anti-inflammatory drug.
Discussion
In this study, we aimed to investigate whether topical capsaicin application is an effective method in the treatment of acute musculoskeletal pain. We compared the efficacy of topical NSAID (ibuprofen) with topical capsaicin in a randomized controlled study. The primary outcome of the study was to evaluate the clinical significance of the response to the treatments. For this reason, we have determined two different criteria: a reduction in VAS values of more than 50% and a final VAS value of ⩽4. Based on these two outcomes, we found that the response to topical capsaicin treatment in patients with acute musculoskeletal trauma was clinically significant. The present study indicates that topical capsaicin applied three times a day for 72 h in the treatment of acute musculoskeletal pain was significantly better than NSAID.
Capsaicin has been used for many years in the treatment of chronic or neuropathic pain. As studies in different areas increase, topical capsaicin is suggested as a new method in the treatment of musculoskeletal pain. 14 There have been studies on the use of capsaicin in diseases requiring long-term analgesics such as osteoarthritis and rheumatoid arthritis. Furthermore, capsaicin has been included in the guidelines as a treatment method that can be used after topical NSAIDs in these diseases. 7
There are many studies in the literature investigating the efficacy of capsaicin on chronic pain.7,15 –19 Mathias et al. 17 demonstrated the effectiveness of topical capsaicin in patients with chronic neck pain. Also, Chrubasik et al. 18 reported positive results after topical capsaicin treatment in patients with chronic soft tissue pain. However, in the literature, the effectiveness of capsaicin administration was frequently compared with a placebo. In our study, we studied the efficacy of capsaicin in pain due to acute trauma to a much stronger competitor. We found that VAS scores decreased more in the topical capsaicin group for all measurement times. However, this difference between the two groups was statistically significant only at 24 and 72 h. We think that this may be related to the delayed peak plasma values after the topical application of capsaicin. 20
In the literature, oral and topical use of NSAIDs were compared, and it has been shown that the frequency of systemic adverse effects decreases in the topical application. 21 However, the possibility of severe systemic effects such as hypersensitivity reactions, renal, and gastrointestinal complications cannot be excluded even in the use of topical NSAIDs. Although topical NSAID administration is considered to be quite innocent, drug blood levels may increase in elderly patients, in cases of decreased drug clearance, and when used in large body areas.22,23 The frequency of systemic adverse effects will also increase due to elevated drug blood levels. When capsaicin is applied topically, its systemic absorption is very low and so is the frequency of systemic adverse events. Local and transient skin reactions (burning, redness, pain, and pruritus) have been reported to be the most common side effects. 24 No studies could be retrieved evaluating the rates of adverse effects in topical administration of capsaicin and NSAIDs. In our study, we did not encounter any severe systemic adverse events in both treatment groups. Two patients in topical NSAID group had nausea and dyspepsia. There was not any significant difference between the two groups regarding the incidence of transient skin reactions. We think that capsaicin cream, which is superior concerning its pain-reducing effect, may be safer to use because of the less likely systemic side effects.
Limitations
Our study has some limitations. Since the blood levels of the medications given by topical applications could not be clearly determined, no precise information could be provided about the daily dose. To standardize the amount of drug used in both groups, the amounts remaining in the containers after treatment were measured. This limitation of the study was tried to be compensated in this way. Also, similar containers were used to achieve health provider blinding. However, odor similarity could not be obtained between the topical forms. We think that this might have caused a breach of blinding.
Conclusion
Topical capsaicin, which does not pose a risk of systemic side effects in many clinical conditions causing chronic pain, has been routinely used as a treatment option. Acute pain due to musculoskeletal trauma is a new and unexplored indication for the topical use of capsaicin. With its high analgesic efficacy and absence of systemic side effects, topical capsaicin can be used safely in patients presenting to the emergency department with acute pain.
Footnotes
Author contributions
S.T.A.G., S.D., A.O.K., T.S.M., I.A., M.B.K., and Z.C. contributed in concept; S.D., A.O.K., and T.S.M. helped in design; A.O.K., T.S.M., I.A., and M.B.K. helped in supervision; A.O.K., T.S.M., I.A., M.B.K., and Z.C. helped in resources; S.T.A.G., S.D., A.O.K., T.S.M., and I.A. contributed to materials; S.T.A.G., S.D., A.O.K., M.B.K., and Z.C. contributed to data collection and/or processing; S.T.A.G., S.D., A.O.K., T.S.M., and I.A. helped in literature search; S.T.A.G., S.D., A.O.K., T.S.M., and I.A. helped in writing the manuscript; and S.T.A.G., S.D., A.O.K., T.S.M., I.A., M.B.K., and Z.C. helped in critical review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Data obtained from the patients who attend Ataturk University Medical Faculty, Emergency Department.
Informed consent
An informed consent form is obtained from each patient who wants to join the study.
Ethical approval
Ethics committee approval was received for this study from the Ethics Committee of Ataturk University Medical Faculty (2019-season number:5, decision number:2).
Human rights
There is no violation of human rights for the article.
