Abstract
Background:
Subarachnoid hemorrhages is an important emergency condition due to its high morbidity and mortality. Early diagnosis and rapid intervention are very important to prevent poor clinical outcome of this disease.
Objective:
The aim of this study was to determine the value of glial fibrillary acidic protein in the diagnosis and prognosis of subarachnoid hemorrhage.
Methods:
Patients presenting to the emergency department and undergoing computerized tomography and/or lumbar puncture due to suspicion of subarachnoid hemorrhage were included in this prospective study. Based on the computerized tomography–lumbar puncture results, cases were divided into subarachnoid hemorrhage group and non–subarachnoid hemorrhage control group. Subarachnoid hemorrhage patients were classified on the basis of severity of subarachnoid hemorrhage and were classified as good or poor clinical outcome groups based on Glasgow Outcome Scores. Glial fibrillary acidic protein levels were then compared.
Results:
A total of 111 patients were evaluated due to suspicion of subarachnoid hemorrhage and diagnosed with subarachnoid hemorrhage (n = 73) or without subarachnoid hemorrhage (n = 38). Cerebrospinal fluid glial fibrillary acidic protein levels were significantly higher in the subarachnoid hemorrhage group than in the non–subarachnoid hemorrhage group (p < 0.001) (median (25%–75%): 11.62 (2.64–68.04) and 2.26 (1.5–4.83), respectively). Serum glial fibrillary acidic protein levels of the subarachnoid hemorrhage patients with poor clinical outcomes were higher than those of subarachnoid hemorrhage patients with good clinical outcomes (p = 0.003) and cerebrospinal fluid glial fibrillary acidic protein levels were similar (p = 0.379). Glial fibrillary acidic protein levels at the time of presentation exhibited a low level of correlation with Glasgow coma score, World Federation of Neurological Surgeons scale, Hunter–Hess Scale, Ogilvy–Carter Scale, Glasgow Outcome Score, and modified Rankin score.
Conclusion:
Cerebrospinal fluid glial fibrillary acidic protein levels may be a valuable diagnostic parameter at the time of presentation for diagnosis of subarachnoid hemorrhage. And also serum glial fibrillary acidic protein levels may be useful in predicting subarachnoid hemorrhage patients with poor clinical outcomes.
Introduction
Subarachnoid hemorrhages (SAHs) most commonly occur due to rupture of intracranial aneurysms. The estimated incidence is 9 out of 100,000 inhabitants/year, with a relative frequency of approximately 5% of all strokes. 1 SAH is an important emergency condition due to its high morbidity and mortality. Outcome can be dramatically impacted by early, aggressive, expert care. 2 Early confirmation of diagnosis in the presence of suspected SAH is, therefore, of crucial importance. The first imaging technique required in cases of suspected SAH is non-contrast computerized tomography (CT) of the brain. If clinical suspicion persists although CT is normal, lumbar puncture (LP) must be performed for diagnosis, unless contraindicated. 3
Biomarkers are currently used in the diagnosis of many diseases and in predicting prognosis. However, there is currently no biomarker for use in diagnosing SAH or evaluating prognosis. A biomarker capable of helping the diagnosis and prognosis of SAH and of predicting a poor clinical course in patients diagnosed with SAH may be of considerable benefit in SAH patient management. Our research was based on the hypothesis that glial fibrillary acidic protein (GFAP) levels in serum and cerebrospinal fluid (CSF) may be useful in these patients.
GFAP, an intermediate filament protein not routinely present in the peripheral blood circulation, is released almost entirely by astrocytes following breakdown of the astrocyte skeleton after glial injury. GFAP is known to reach measurable levels in serum and CSF as a result of breakdown of the blood–brain barrier (BBB) following acute brain damage.4–8 In addition, passage into the blood circulation by means of CSF flow following release into the interstitial spaces after existing injury has been proposed as another means whereby GFAP levels can become detectable. 7 Previous studies have revealed increased serum and CSF GFAP levels in SAH patients. However, none has demonstrated the diagnostic and prognostic value of serum and CSF GFAP levels in cases of suspected SAH patients or that these can be used in clinical practice in the emergency department.4,7,9–11
The purpose of this study was to investigate the potential use of GFAP levels measured in serum and CSF specimens obtained under clinical conditions from patients with suspected SAH in terms of diagnosis and predicting prognosis.
Materials and methods
Study design
This research was planned as a prospective, time-restricted, cohort study. Following receipt of approval from the ethical committee (2016/115), patients presenting to the Karadeniz Technical University Faculty of Medicine, Department of Emergency Medicine, a tertiary institution, and undergoing CT and/or LP due to suspected SAH (headache including exertional onset, vomiting, neck pain, seizures, neurologic deficit, and/or syncope) between October 2016 and October 2017, were included in the study.
Study setting and population
The center where the study was carried out is the emergency department of one of the most advanced institutions in Turkey in terms of SAH patient care, including interventional radiology and surgery. It serves a population of approximately million and receives 100,000 adult patient presentations a year.
Non-traumatic patients aged 18 years or above, presenting to the emergency department, and undergoing CT and/or LP on suspicion of SAH were enrolled in the study. Patients presenting to our hospital with SAH determined at CT performed on suspicion of SAH at another center or referred to us due to suspicion of SAH although SAH was not diagnosed at another center were also included in the study.
Patients with exposure to trauma, with known intracranial space-occupying lesion, contraindicated for CT or LP, refusing to take part, dropping out or transferred elsewhere during the study, or with missing data were excluded. Time of onset of symptoms was recorded for patients intended for inclusion in the study, and we planned not to include subjects presenting to the emergency department more than 24 h after onset.
Data collection
Informed consent forms were obtained from all patients presenting to the emergency department and scheduled for CT or LP on suspicion of SAH, or from their families. These patients’ sociodemographic characteristics, symptoms, physical examination findings, and medical histories were then recorded. CSF pressure and characteristics for patients undergoing LP and CT angiography, magnetic resonance imaging (MRI) angiography, and angiography results if applicable were recorded.
The Glasgow coma score (GCS), Hunt–Hess Scale (HHS), modified Rankin scale (MRS), and the World Federation of Neurological Surgeons (WFNS) SAH scale combined with the GCS were used in the evaluation of the clinical severity of SAH patients, while the Fisher Grading Scale (FS) and Ogilvy–Carter Scale (OCS) system were calculated based on CT findings and then recorded. HHS, WFNS, FS, MRS, and OCS were calculated by a brain surgeon blinded to the patients’ data and biochemical results.12–15 We anticipated determining patients’ clinical outcomes criteria on the basis of the Glasgow Outcome Score (GOS) investigated within a 6-month time frame.
In order to determine the diagnostic value of serum and CSF GFAP levels in patients with suspected SAH, subjects were divided into SAH and non-SAH groups based on CT and/or LP findings. In addition to initial CT and LP, patients in the non-SAH group also underwent additional diagnostic procedures depending on their diseases, and definite diagnosis was made following these. Since serum GFAP elevation has been shown in previous studies in different neurological diseases other than SAH, such as bacterial meningitis, status epilepticus, and Alzheimer’s disease, and since the characteristics of meningitis patients occupy an important place in the differential diagnosis of SAH patients presenting with severe headache, nausea, and vomiting, the non-SAH group was further divided into subgroups based on severe central nervous system (CNS) infections (meningitis and encephalitis) and other causes, and additional comparisons were carried out.
Patients were assessed using the GOS in terms of prognosis in the 6-month period after SAH. Patients with GOS 1–2 were classified as the poor clinical outcome group and those with GOS 3–5 as the good clinical outcome group. The prognostic value of serum and CSF GFAP levels was determined by comparing serum and CSF GFAP values measured at the time of presentation in these groups.
Biochemical analysis
Following receipt of consent forms from the individuals, serum specimens were placed into biochemistry tubes containing gel separator. These were then allowed to coagulate for 20 min at room temperature, after which the tubes were centrifuged at 1800 × g for 10 min. At the end of this period, the serum parts above the gel separator were carefully transferred to small tubes and stored at −80°C until analysis. Time of collection of serum specimens was recorded for all patients.
GFAP level measurement
GFAP levels in serum and CSF were determined using an enzyme-linked immunosorbent assay (ELISA) kit (Catalog No. RD192072200R; BioVendor, Brno, Czech Republic) following the manufacturer’s instructions. GFAP levels were expressed as ng/mL. Serum, plasma, and CSF are among the validated and recommended specimens in the commercial sandwich enzyme immunoassay (ELISA) kit used in the study.
Comparison of serum and CSF GFAP levels in the SAH and non-SAH groups represented the primary outcome of the study. Secondary outcomes were the comparison of serum and CSF GFAP levels in the poor and good clinical outcome groups established on the basis of the GOS, a marker of survival and neurological status in the 6-month period following SAH, and the determination of correlation between serum and C GFAP levels and clinical and radiological severity scores.
Statistical analysis
The data obtained in the study were recorded onto Excel software, and statistical calculations were performed on SPSS 23.0 and MedCalc software. Compatibility with normal distribution normality was assessed using the Kolmogorov–Smirnov test during statistical analysis. Normally distributed numerical data were expressed as mean and 95% confidence interval (CI), and non-normally distributed numerical data were expressed as median and 95% CI, while categorical variables were expressed as n and %. During the statistical analysis, WFNS and FS, scores showing severity of hemorrhage, were dichotomized (WFNS 1–2 and FS 1–2 mild–moderate; WFNS 3–5 and FS 3–4, severe). GOS scores, a marker of survival and neurological status, were also dichotomized (GOS 1–2 representing poor, and GOS 3–5 good clinical outcomes). Student’s t-test was used to compare normally distributed numerical data at two-way comparisons, and the Mann–Whitney test was used for non-normally distributed data. The chi-square test was used to compare categorical variables. Correlation between serum or CSF GFAP levels and patients’ clinical and radiological severity scores was measured using Spearman’s correlation analysis. Values of p less than 0.05 were regarded as significant. Receiver operating characteristic (ROC) curve analysis was performed, and area under the curve (AUC) and optimal cut-off were determined in order to determine the diagnostic value of serum and CSF GFAP levels in SAH and in predicting a good or poor course.
Results
A total of 126 patients with suspected SAH based on clinical symptoms and findings were evaluated between the study dates. The initial diagnostic approach adopted in all 126 patients with suspected SAH was CT, as a result of which SAH was identified in 79 patients, but not in the other 47. Six of the 79 patients in whom SAH was determined at CT were excluded for various reasons (transferred (n = 2), lost follow-up (n = 1), or refusal to participate (n = 3)). The remaining 73 patients constituted the study group. Forty-one individuals from whom consent was received underwent LP for the measurement of CSF pressure and CSF GFAP levels. Nine of the 47 patients in whom SAH was not detected using CT were excluded from the study for various reasons (transferred (n = 2), lost follow-up (n = 2), refusal to participate (n = 3), or data form deficiency (n = 2)), and all the remaining 38 patients underwent LP to exclude SAH. No SAH was determined in any patient following LP and CSF GFAP level measurement, and these 38 patients constituted the control group. The study flow chart is shown in Figure 1, and the clinical and demographic characteristics of the study and control groups are shown in Table 1.
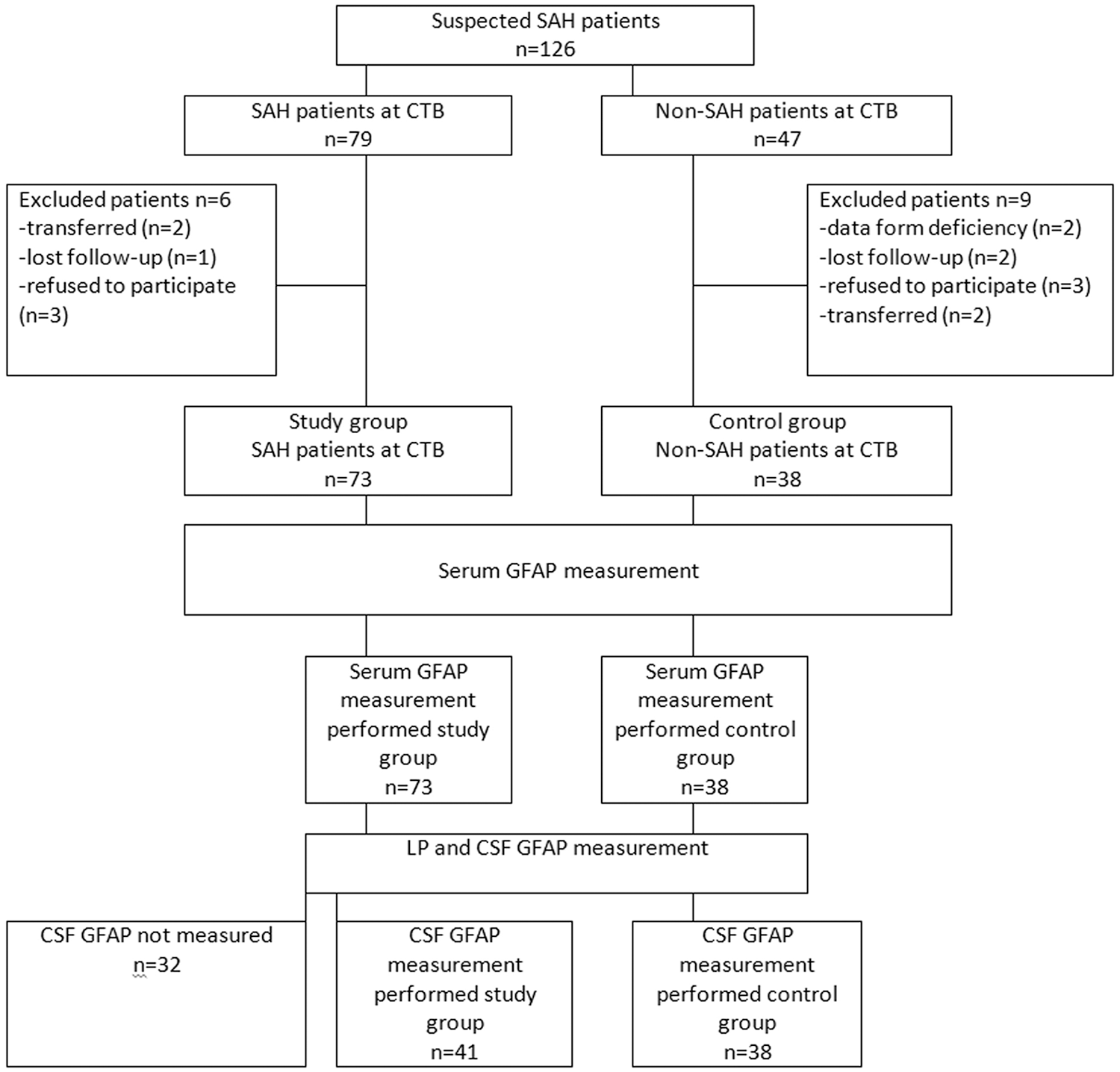
Patient flow chart.
General characteristics of the patients in the study.
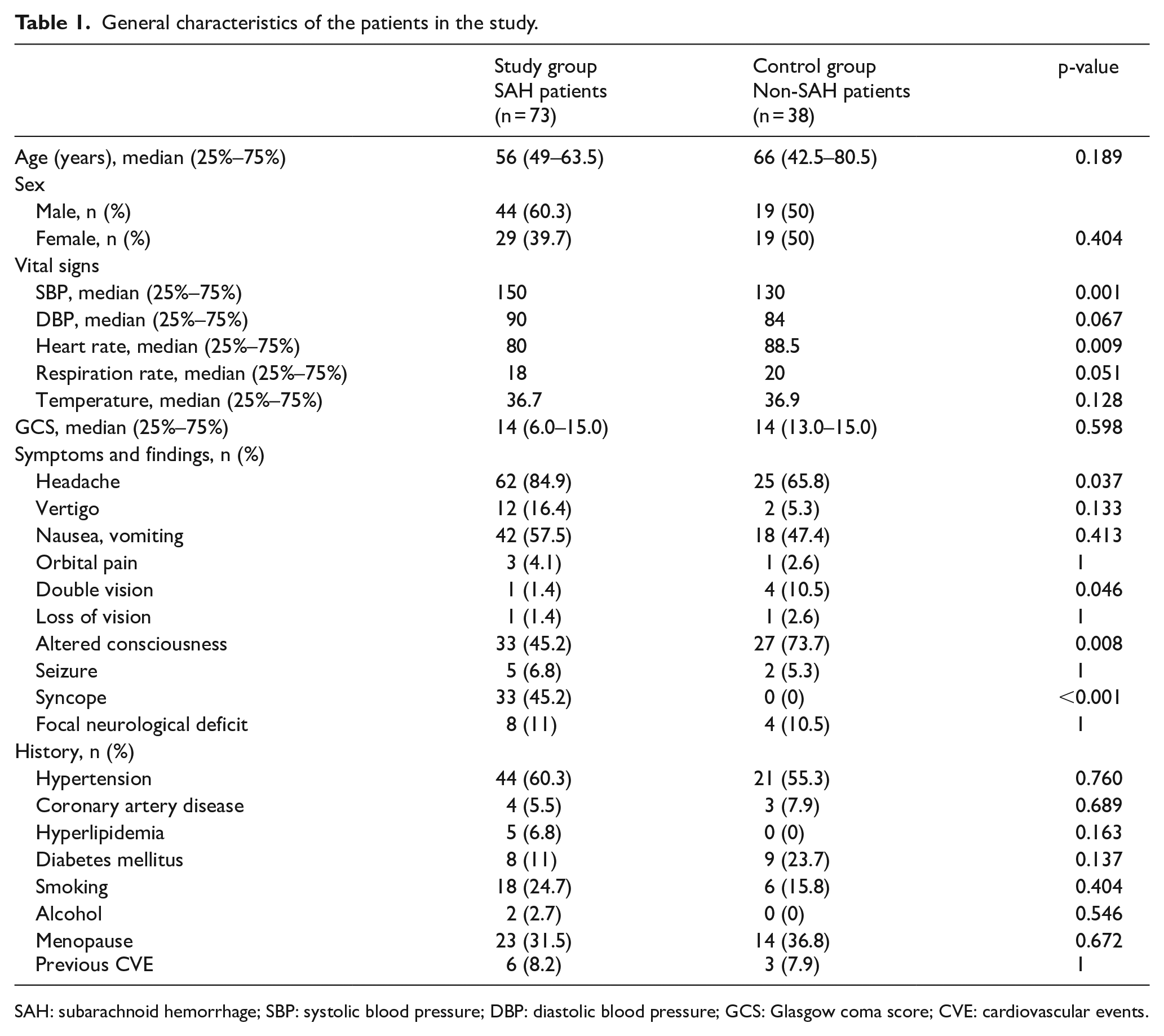
SAH: subarachnoid hemorrhage; SBP: systolic blood pressure; DBP: diastolic blood pressure; GCS: Glasgow coma score; CVE: cardiovascular events.
Mean time from onset of symptoms in SAH patients to collection of GFAP specimens was 3.1 ± 1.9 h (min: 1 h, max: 12 h), and mean time to collection of CSF GFAP specimens was 4.1 ± 2.3 h (min: 2 h, max: 13 h). No significant correlation was determined between time from onset of symptoms to specimen collection and serum or CSF GFAP levels (r = −0.04, p = 0.74 for serum GFAP and r = −0.06, p = 0.69 for CSF GFAP).
A comparison of serum and CSF GFAP levels in the study group diagnosed with SAH based on CT and LP findings and in the non-SAH control group is shown in Table 2. Based on the results, only CSF GFAP levels at the time of presentation were higher in the SAH patients than in the non-SAH controls (p < 0.001). Serum and CSF GFAP levels were evaluated whether there is a correlation between these parameters. No correlation was observed between serum and CSF GFAP levels in the SAH and non-SAH groups (r = 0.216, p = 0.18; r = 0.012, p = 0.94, respectively).
Distribution of serum and CSF GFAP levels by groups.
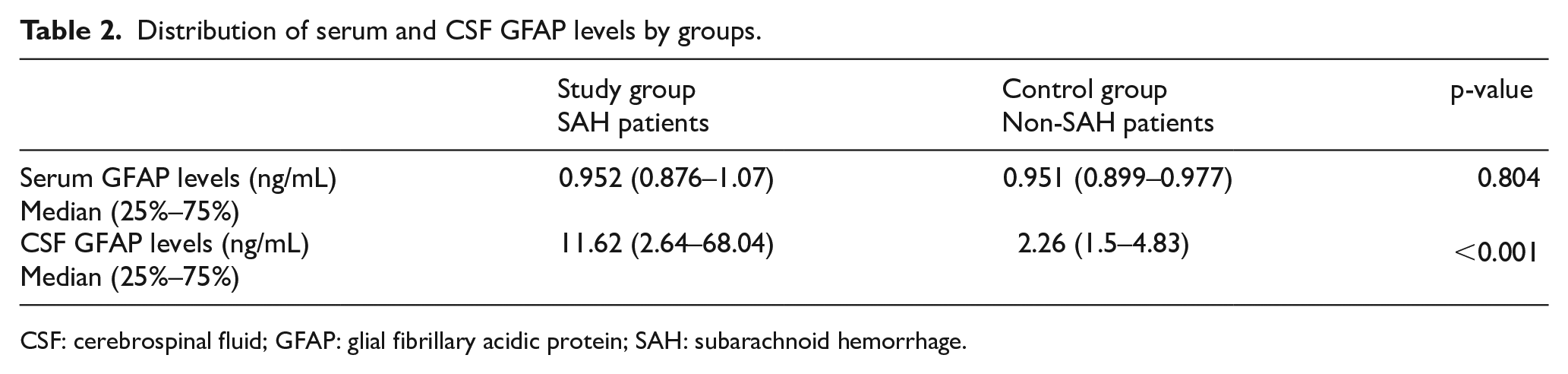
CSF: cerebrospinal fluid; GFAP: glial fibrillary acidic protein; SAH: subarachnoid hemorrhage.
According to the ROC curve for CSF GFAP levels in the diagnosis of SAH, AUC was 0.761 (95% CI = 0.652–0.850; p < 0.001), and the best cut-off value of 8.5 ng/mL exhibited sensitivity of 58.5% (95% CI = 42.1–73.7), specificity 92.1% (95% CI = 78.6–98.2), +PV 88.9% (95% CI = 70.4–97.6), −PV 67.3% (95% CI = 52.9–79.9), +LR 7.4 (95% CI = 5.6–9.8), and −LR 0.45 (95% CI = 0.1–1.4) (Figure 2). According to the ROC curve drawn for serum GFAP levels for diagnosis of SAH, AUC was 0.486 (95% CI = 0.389–0.583), and p = 0.804.
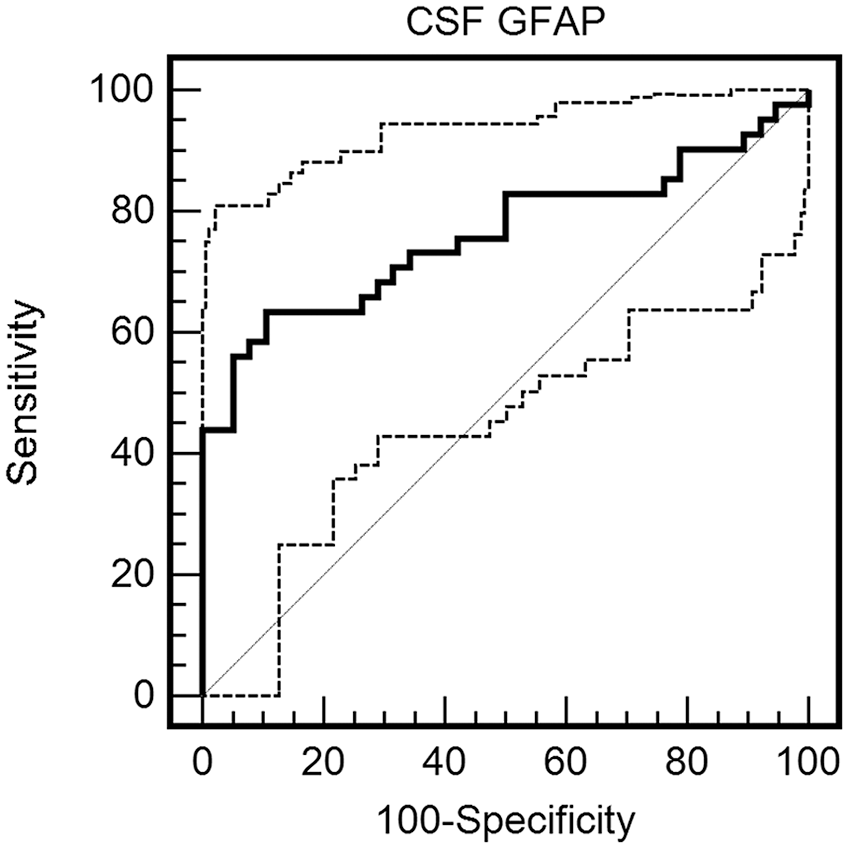
ROC curve for CSF GFAP levels in the diagnosis of SAH.
The 38 patients enrolled in the control group were subdivided into severe CNS (n = 16; encephalitis (n = 6) and meningitis (n = 10)) and other diagnoses subgroups (n = 22; metabolic encephalopathy (n = 10), hypertensive encephalopathy (n = 1), unruptured intracranial aneurysm (n = 1), subdural effusion (n = 1), and no-pathology (n = 9)) based on final diagnosis. A comparison of the results based on these subgroups is shown in Table 3. Accordingly, CSF GFAP levels were highest in the SAH patients and were also higher in the patients with CNS infection than in those with other diagnoses.
Distribution of serum and CSF GFAP levels by subgroups.
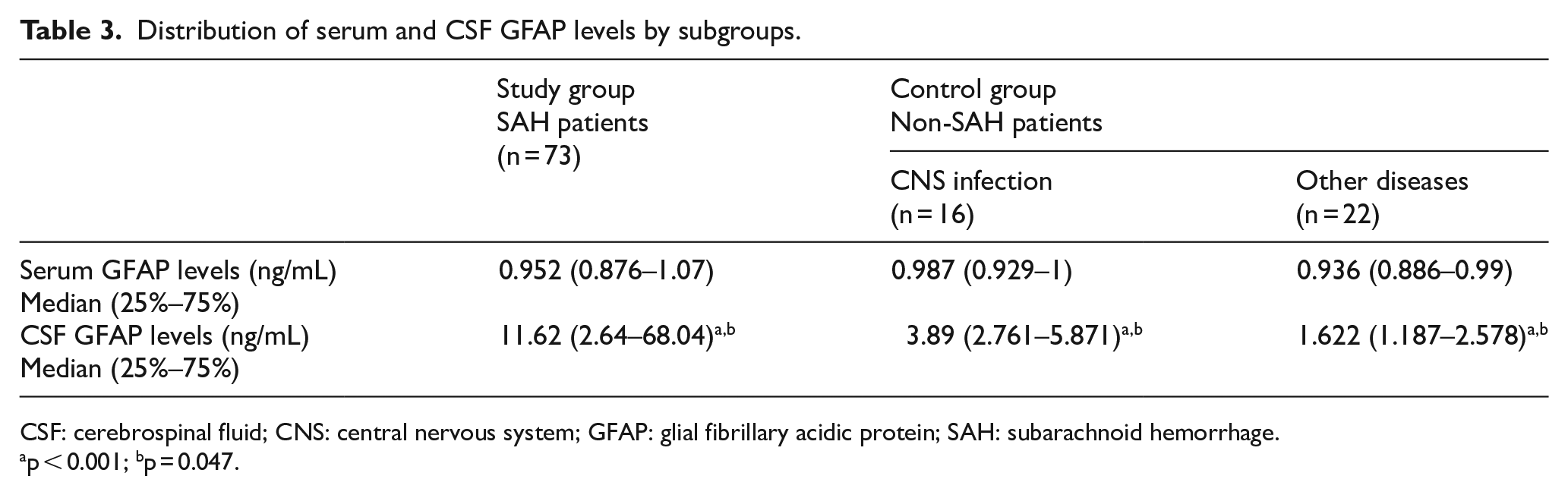
CSF: cerebrospinal fluid; CNS: central nervous system; GFAP: glial fibrillary acidic protein; SAH: subarachnoid hemorrhage.
ap < 0.001; bp = 0.047.
Serum and CSF GFAP levels in the poor and good clinical outcome groups established based on the GOS, a measure of survival and neurological status, in the 6-month period after SAH, are shown and compared in Table 4. These findings revealed higher serum GFAP levels in the SAH patients with poor clinical outcomes than in those with good clinical outcomes (p < 0.001). According to the ROC drawn for serum GFAP levels in predicting the best clinical outcome in the course of SAH, AUC was 0.704 (95% CI = 0.584–0.806; p < 0.001), and the best cut-off point value of 0.99 ng/mL exhibited sensitivity of 56.7% (95% CI = 39.5–72.9), specificity 88.2% (95% CI = 72.5–96.6), +PV 84.0% (95% CI = 63.9–95.4), −PV 65.2% (95% CI = 49.8–78.6), +LR 4.8 (95% CI = 3.5–6.6), and −LR 0.49 (95% CI = 0.2–1.3). AUC for the ROC curve drawn for CSF GFAP levels was 0.585 (95% CI = 0.420–0.736), p = 0.362.
Relations between SAH patients’ serum and CSF levels and GOS scores.
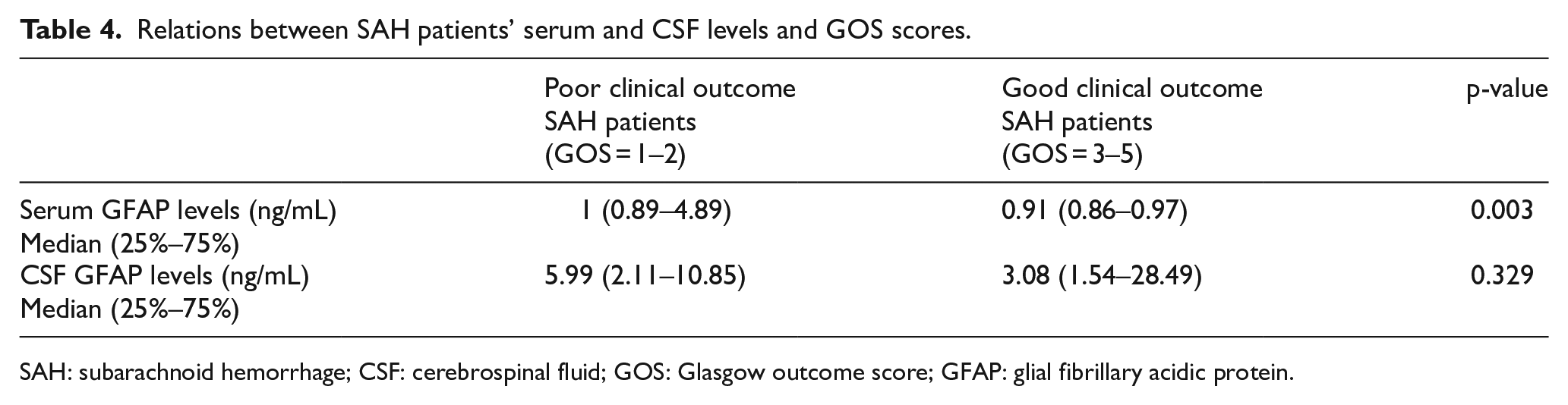
SAH: subarachnoid hemorrhage; CSF: cerebrospinal fluid; GOS: Glasgow outcome score; GFAP: glial fibrillary acidic protein.
Correlation results between serum and CSF GFAP levels and patients clinical and radiological severity scores in the patients diagnosed with SAH are shown in Table 5. Accordingly, the serum GFAP parameter exhibited weak negative correlation with GCS and also weak correlation with the WFNS, Hunt–Hess, and Ogilvy–Carter scoring systems.
Correlation results between serum and CSF GFAP levels and patients clinical and radiological severity scores.
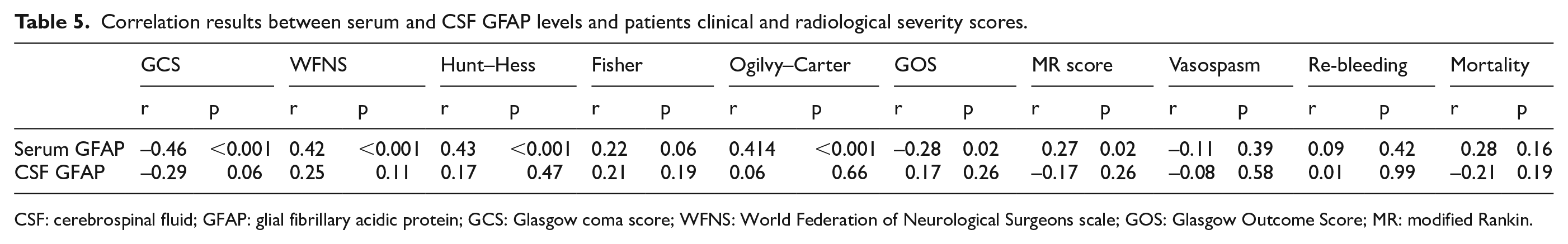
CSF: cerebrospinal fluid; GFAP: glial fibrillary acidic protein; GCS: Glasgow coma score; WFNS: World Federation of Neurological Surgeons scale; GOS: Glasgow Outcome Score; MR: modified Rankin.
Discussion
Our study evaluated the diagnostic and prognostic performance of GFAP levels measured in serum and CSF in patients presenting to the emergency department with suspected spontaneous SAH after clinical assessment. The data obtained suggested that CSF GFAP levels measured within 12 h post-event may be a valuable parameter in diagnosing SAH.
The structural integrity of neuronal and glial cells is impaired by SAH. Specific proteins, such as neuron-specific enolase (NSE), S100B, and GFAP, are thought to enter the circulation and CSF as a result of that impairment. The serum or CSF concentrations of some of these proteins may be correlated with degree of cerebral damage, and these proteins may have diagnostic and prognostic applications. 9 Various studies have examined serum GFAP levels in SAH patients. In their precursor study, Vos et al. investigated NSE, S100B, and GFAP serum levels in peripheral blood as potential biomarkers of cerebral damage in SAH patients. They reported that the glial proteins S100B and GFAP were above normal levels, while the neuronal protein NSE was within normal limits. 4 Despite being of great value as a precursor study, the absence of any information concerning the time between onset of symptoms and collection of blood specimens from patients with SAH represented a significant problem in terms of being able to compare the findings. Nylen et al. evaluated serum GFAP levels in SAH patients admitted to the neurological care unit as early as possible after admission and at subsequent checks on Days 1, 2, 3, 4, 6, 8, 10, and 15. High serum GFAP levels were determined in 81 out of 116 patients, and these high levels were correlated with clinical prognostic markers in SAH patients such as WFNS, re-bleeding, or vasospasm-related ischemia. 7 Despite revealing high serum GFAP levels in SAH patients and this elevation may be prognostic, the different aim of that study means that it is not possible to draw reliable conclusions regarding the diagnostic value of serum GFAP levels in patients with suspected SAH. Another important and noteworthy study of serum GFAP levels was performed by Mayer et al. They considered a broad spectrum of neurological diseases in 331 patients and examined serum GFAP levels. 10 They reported serum GFAP level elevation in patients with intracerebral hemorrhage, bacterial meningitis, status epilepticus, and Alzheimer’s disease. High serum GFAP levels were particularly significant in patients with meningitis, which occupies an important place in the differential diagnosis of SAH patients due to causing severe headache, nausea, vomiting, and nuchal rigidity.
The number of studies evaluating the relation between CSF GFAP levels and SAH is extremely limited. In their study of a very restricted number of subjects, Petzold et al. 11 reported high CSF GFAP levels in 93% of SAH patients, and that this elevation was most pronounced on the first day, before decreasing by the third day and reaching very low levels on Day 10. In particular, they emphasized that a secondary increase in CSF GFAP levels might have been related to delayed cerebral ischemia associated with vasospasm, and that this might be an indicator of mortality and morbidity. Petzold et al.’s study differs from ours in terms of design, in that they included only SAH patients requiring ventriculostomy for CSF drainage, and also in terms of timing of CSF GFAP measurement.
With its aims and design, our study differs significantly from other research into serum and CSF GFAP levels. Our primary aim was to determine whether serum and CSF GFAP levels can be used in the diagnosis of SAH, while our secondary aim was to determine whether these levels are of any value in predicting prognosis in SAH patients. Since the target population consisted of patients with suspected SAH presenting to the emergency department, the mean time between onset of symptoms and collection of serum and CSF GFAP specimens was only 3 h, with a minimum of 1 h and maximum of 12 h. These values were much shorter than those between onset of the event and serum or CSF GFAP measurement in previous studies. The fact that serum GFAP levels had not yet risen even though CSF GFAP levels were high enough to be used in the diagnosis of SAH may derive from serum and CSF specimens being collected very soon after the SAH event. GFAP in CSF increases rapidly with glial cell damage occurring with SAH, and that elevation reaches levels capable of use in diagnosing SAH patients. In the light of other studies determining high serum GFAP levels in SAH patients, an increase in serum GFAP very probably occurs after BBB impairment, and at a late stage. However, high GFAP levels in CSF in the early period are of low prognostic value, and serum GFAP levels are even of high prognostic value. From that perspective, high serum GFAP levels in the early period after SAH may indicate a patient with more severe hemorrhage and poorer prognosis.
LP is required in the presence of strong clinical suspicion of SAH together with negative or ambiguous CT or if CT is unavailable. It is important for the emergency physician to distinguish SAH from other pathologies and also from traumatic LP. Some studies have reported that a decrease exceeding 30% in RBC values between the first and the last tubes may be diagnostic of traumatic LP, although this has yet to be confirmed. 16 In designing our study, we planned to use the latest tubes, rather than excluding traumatic LPs. However, no traumatic LPs occurred during the study period.
The fact that the majority of patients without SAH in our study had another CNS pathology, and the possibility that this might increase GFAP levels in the control group, may also have led to our observing statistically insignificant serum GFAP levels. Mayer et al.’s 10 study clearly shows that several CNS pathologies, and particularly CNS infections, cause a rise in serum GFAP levels. Comparison of serum GFAP levels of patients in groups based on SAH and CNS infections, the most important clinical outcomes in patients presenting with headache, and on less significant causes, revealed similar serum GFAP levels in all three groups. The CSF GFAP examined in our study provided more contributory data in terms of differentiation between these subgroups. Mean CSF GFAP levels were higher in the SAH patients than in the CNS infection subgroup, but the difference was not statistically significant. From that perspective, CSF GFAP levels do not appear capable of differentiating between SAH and CNS infections. However, CSF GFAP levels were significantly higher in both the SAH patients and the CNS infection patients compared to the “other diseases” subgroup consisting of diseases with less severe clinical outcomes. This may be due to rising GFAP levels resulting from acute injury in brain cells in SAH and cell damage resulting from CNS infection. Indeed, one experimental study on this subject reported GFAP elevation in CSF specimens from bulldogs in a manifestation of necrotizing meningoencephalitis. 17 In addition, studies have also reported that GFAP elevation in the CSF of children with tuberculous meningitis may be a prognostic marker for that disease. 18 On that basis, we may infer that CSF GFAP levels may be of assistance, not only in differentiating patients with SAH but also in differentiating conditions capable of producing severe and adverse clinical outcome unless diagnosed early and accurately and unless treated promptly and accurately, such as SAH or CNS infections, from other causes of relatively less severe clinical outcomes.
Serum GFAP level elevation in our SAH patients was related with poor clinical outcomes. This may be of assistance in predicting the clinical course of SAH patients. Clinical evidence suggests that neuronal and astrocytic cell death may play a role in the poor prognosis of patients with SAH. In their experimental study, Sabri et al. 17 revealed that neuronal and astrocytic damage following SAH is a probable cause of poor prognostic course in these patients. Nylen et al. 7 reported that in addition to serum GFAP level elevation in SAH patients, this elevation was closely associated with focal brain damage and clinical outcomes in SAH patients. Vos et al. 4 revealed that GFAP levels were associated with severity of SAH. Serum GFAP levels have also been reported to be of prognostic value in stroke and other neurological diseases, and particularly in traumatic brain damage, other than SAH.19–23
Limitations
There are some limitations to this study. The first limitation is that the timing of serum collection could not be standardized since our study was performed in a tertiary hospital receiving referrals and due to the differing periods elapsing between onset of symptom and patients arriving at our hospital. Mean time to measurement of serum GFAP from the onset of the event was 3 h (min: 1 h, max: 12 h), and mean time to measurement of CSF GFAP levels was 4 h (min: 2 h, max: 13 h). These time intervals should, therefore, be considered in evaluating the diagnostic efficacy of serum of CSF GFAP levels. Serum and CSF specimens were collected at a single time-point (time of presentation), and repeat specimens were not collected subsequently during follow-up.
As this study was performed in an emergency department, patients were assessed by an emergency physician at the time of presentation, and since follow-up was performed together with other departments, the diagnostic and prognostic values of GFAP levels at the time of presentation are of greater importance for the emergency department.
The number of individuals identified as being in a healthy condition following clinical evaluations among the patients with suspected SAH was very low. The principal reason for this is that the majority of patients with suspected SAH at the time of presentation but with no SAH determined at imagine refused to provide serum, and particularly CSF specimens. Moreover, our emergency department being in a tertiary hospital receiving referrals from external centers and more complicated cases with uncertain diagnosis being assessed by us may also have contributed to this.
Conclusion
Based on our study findings, CSF GFAP levels may serve as a diagnostic parameter for SAH patients. In addition, serum GFAP levels may be useful in predicting SAH patients with poor clinical outcomes.
Footnotes
Acknowledgements
The authors gratefully thank the Karadeniz Technical University Scientific Research Projects Unit for their support.
Author contributions
Ozgur Tatli, Metin Yadigaroglu, Suleyman Turedi, Selim Demir, and Ahmet Mentese substantially contributed to conception and design, acquisition of data, analysis and interpretation of data, drafting the article or revising it critically for important intellectual content, and provided final approval of the version to be published; Mustafa Cicek, Melih Imamoglu, Seniz Dogramaci, Senol Ardic, and Sinan Pasli contributed to acquisition of data, interpretation of data, drafting the article or revising it critically for important intellectual content, and provided final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Karadeniz Technical University Scientific Research Projects Unit (Grant No. TTU-2017-6948). Karadeniz Technical University Scientific Research Projects Unit funded our research on providing the biochemical assay kits.
Availability of data and materials
The datasets generated and analyzed during the current study are available from Metin Yadigaroglu and Sinan Pasli
Informed consent
Informed consent forms are obtained from all patients included in the study.
Ethical approval
The ethical approval is obtained from Karadeniz Technical University Faculty of Medicine Ethics Committee (2016/115).
Human rights
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
