Abstract
Background:
The confirmation of prothrombin time international normalized ratio by a central laboratory often delays intravenous thrombolysis in patients with acute ischemic stroke.
Objectives:
We investigated the feasibility, reliability, and usefulness of point-of-care determination of prothrombin time international normalized ratio for stroke thrombolysis.
Methods:
Among 312 patients with ischemic stroke, 202 who arrived at the emergency room within 4.5 h of stroke onset were enrolled in the study. Patients with lost orders for point-of-care testing for the prothrombin time international normalized ratio or central laboratory testing for the prothrombin time international normalized ratio (n = 47) were excluded. We compared international normalized ratio values and the time interval from arrival to the report of test results (door-to-international normalized ratio time) between point-of-care testing for the prothrombin time international normalized ratio and central laboratory testing for the prothrombin time international normalized ratio. In patients who underwent thrombolysis, we compared the time interval from arrival to thrombolysis (door-to-needle time) between the current study population and historic cohort at our center.
Results:
In the 155 patients included in the study, the median door-to-international normalized ratio time was 9.0 min (interquartile range, 5.0–12.0 min) for point-of-care testing for the prothrombin time international normalized ratio and 46.0 min (interquartile range, 38.0–55.0 min) for central laboratory testing for the prothrombin time international normalized ratio (p < 0.001). The intraclass correlation coefficient between point-of-care testing for the prothrombin time international normalized ratio and central laboratory testing for the prothrombin time international normalized ratio was 0.975 (95% confidence interval: 0.966–0.982). Forty-nine of the 155 patients underwent intravenous thrombolysis. The door-to-needle time was significantly decreased after implementation of point-of-care testing for the prothrombin time international normalized ratio (median, 23.0 min; interquartile range, 16.0–29.8 vs median, 46.0 min; interquartile range, 33.5–50.5 min).
Conclusion:
Utilization of point-of-care testing for the prothrombin time international normalized ratio was feasible in the management of patients with acute ischemic stroke. Point-of-care testing for the prothrombin time international normalized ratio was quick and reliable and had a pivotal role in expediting thrombolysis.
Keywords
Introduction
Approximately 1.9 million neurons are lost during every minute when a large cerebral artery occlusion is left untreated. 1 Emergent reperfusion therapy may recanalize occluded cerebral arteries and save ischemic neurons. 2 The only approved thrombolytic agent to date is recombinant tissue plasminogen activator (tPA), provided that it is administered within 4.5 h following stroke onset. As previous studies have shown that earlier administration of tPA is associated with better clinical outcomes, earlier infusion of tPA is considered the most important proven therapy in patients with acute ischemic stroke.3,4 Accordingly, the current guidelines state that in-hospital delay for infusion of tPA, that is, door-to-tPA time, should be within 60 min of hospital arrival. 4 However, in-hospital delay in thrombolysis is still common and a substantial issue in routine practice.5–9 Patient selection based on laboratory tests and multistep in-hospital processes significantly contribute to such delays, even in patients arriving in the emergency room (ER) within 4.5 h of stroke onset. 10
The prolongation of prothrombin time international normalized ratio (INR) is a major exclusion criterion for administration of tPA. 4 However, confirmation of INR in a central laboratory (central laboratory testing for the prothrombin time international normalized ratio (CL-INR)) requires at least 60 min. 10 In contrast, point-of-care testing for the prothrombin time international normalized ratio (POC-INR) requires at most 1 min after placing a blood drop on the test strip. Previous studies on POC-INR in patients with stroke have focused on reducing the time interval between POC-INR and CL-INR11–17 or on the feasibility of POC-INR in outpatient clinics. 18 One study found a mean effective time gain of 28 min when POC-INR was used instead of CL-INR in patients with acute stroke. 13 Some studies have shown time gains only in patients with acute stroke treated with warfarin.13,19 Most point-of-care devices can decrease testing time, although previous studies have not proven the usefulness of POC-INR in stroke thrombolysis.11,14
To our knowledge, however, reports on expedited stroke thrombolysis through the integration of POC-INR into routine practice are lacking. Recently, we reduced door-to-tPA time to 20 min through the implementation of a hospital quality improvement activity wherein incorporation of POC-INR had a pivotal role. 20 Here, we aimed to investigate the feasibility, reliability, and usefulness of POC-INR in decreasing door-to-tPA time in the routine practice of stroke thrombolysis.
Methods
Patients
This retrospective study was performed at Asan Medical Center, Seoul, Korea, between 1 June 2016 and 31 December 2016. Data were prospectively collected for all adult (age ⩾ 18 years) patients who presented with symptoms suggesting acute stroke to our ER. In this study, we included consecutive patients (1) for whom stroke code was activated, (2) who were diagnosed with acute ischemic stroke, (3) whose onset-to-door time was <4.5 h, and (4) in whom CL-INR and POC-INR were tested. This study was approved by the Institutional Review Board of Asan Medical Center, and the need for written informed consent was waived.
Stroke Alert Team
This study was performed as a hospital quality improvement project called Stroke Alert Team (SAT). 20 Beginning on 2 May 2016, the SAT introduced ambulance pre-notification; moved patients directly to a resuscitation room in the ER; obtained venous blood samples for POC-INR and CL-INR in the resuscitation room; automatically transferred the results of POC-INR, CL-INR, and other laboratory tests to the hospital electronic medical record (EMR) system; transported patients to the computed tomography (CT) room within 5 min; and administered tPA in the CT room. The decision to infuse intravenous tPA was guided by patients’ history, neurological examination findings, POC-INR reports on the EMR, finger-stick blood sugar test, and CT scan results. INR values were routinely measured when SAT was activated. Patients with INR values above 1.5 were excluded from tPA thrombolytic therapies. Confirmation of CL-INR and other laboratory tests on the EMR were deferred until after tPA administration.
Measurement of prothrombin time
We measured POC-INR using the CoaguChek® XS Pro (Roche Ltd, Switzerland). 21 Venous blood samples for both POC-INR and CL-INR were obtained simultaneously in the resuscitation room. Tests for POC-INR were performed in the resuscitation room and samples for CL-INR with conspicuous SAT labels and audible alarms were delivered to the laboratory using a pneumatic carrier tube. The transmission of the results of POC-INR to the EMR system was established on 1 June 2016. The door-to-INR time was defined as the time interval from ER arrival to the transmission of POC-INR results to the EMR system or the time interval from ER arrival to the transmission of CL-INR results to the EMR system, whichever occurred first. We conducted quality assessments of POC-INR using the CoaguChek XS Control International Reagent. Such assessments were performed daily until July 2016. Beginning on 1 August 2016, quality assessments were performed weekly according to the manufacturer’s instructions. 22 Laboratory technicians educated ER nurses in the measurement of POC-INR and the conduct of quality assessments. Nurses who passed a test after the educational course were allowed to handle the device. The POC-INR results were displayed on the device screen within 1 min (Supplementary Figure 1). These results were then automatically transmitted to the EMR system within minutes.
Statistical analyses
Data are presented as medians with interquartile ranges (IQRs) for continuous variables and as absolute numbers or relative frequencies for categorical variables. Pearson’s chi-square tests were used for categorical variables and Wilcoxon’s signed-rank tests were used for continuous variables, as appropriate. We compared POC-INR values with CL-INR values using an intraclass correlation for patients admitted from 1 June 2016 to 31 December 2016. The agreement between POC-INR and CL-INR was also assessed using Bland–Altman analysis and Bland–Altman bias plots. The door-to-INR time, door-to-CT time, and door-to-tPA time after implementation of the SAT (from 1 June 2016 to 31 December 2016) were compared with those before the implementation (from 1 January 2014 to 1 May 2016). 20 Statistical significance was defined as two-tailed p < 0.05. All statistical analyses were performed using SPSS Version 21 (IBM Corp., Armonk, NY, USA).
Results
The SAT was activated in 413 patients from 1 June 2016 to 31 December 2016. Patients with hemorrhagic stroke, transient ischemic attack, onset-to-door times ⩾ 4.5 h, and loss of orders for POC-INR or CL-INR were excluded (Supplementary Figure 2). The 155 remaining patients were included in the study (Table 1).
Patient characteristics.
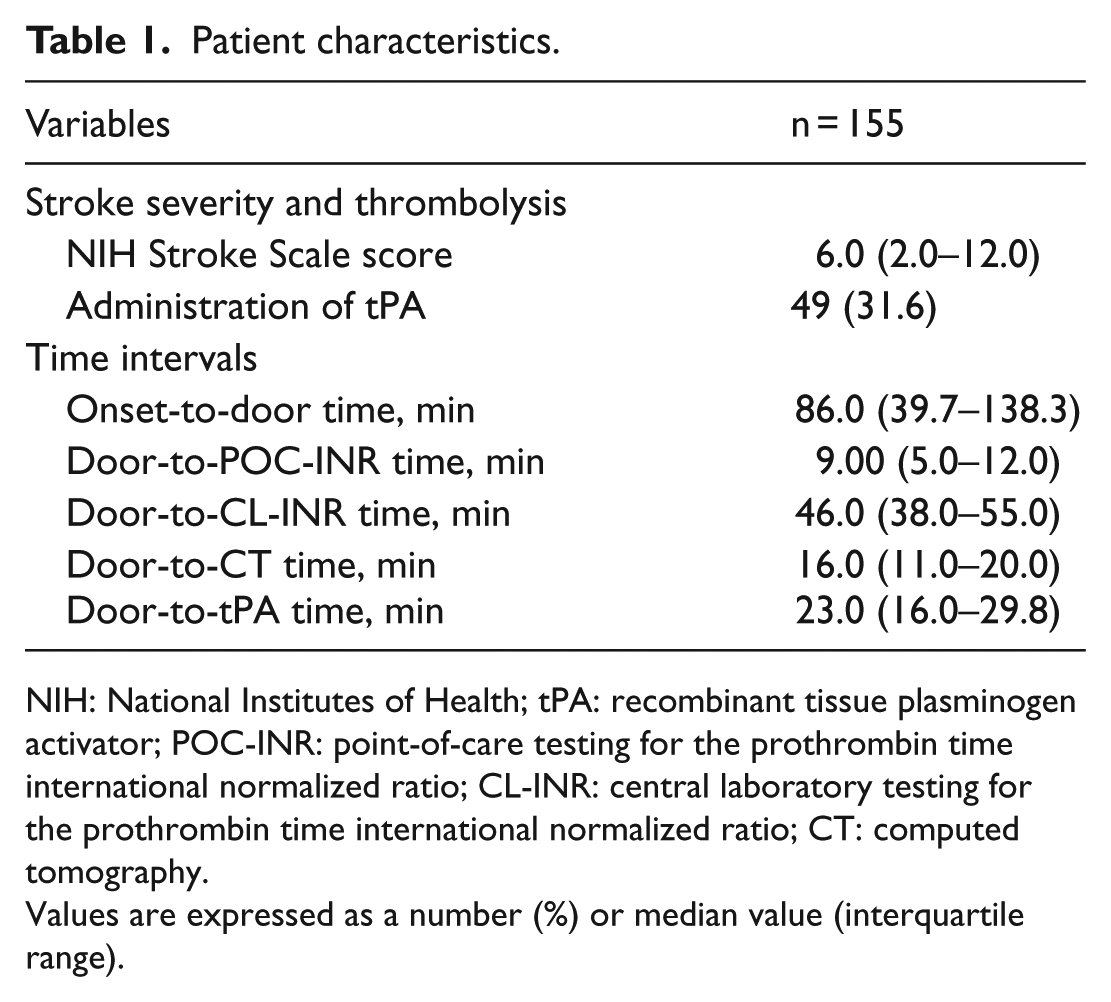
NIH: National Institutes of Health; tPA: recombinant tissue plasminogen activator; POC-INR: point-of-care testing for the prothrombin time international normalized ratio; CL-INR: central laboratory testing for the prothrombin time international normalized ratio; CT: computed tomography.
Values are expressed as a number (%) or median value (interquartile range).
Feasibility
The POC-INR test was available 24 h/day. Among the 202 patients with acute ischemic stroke who presented to the ER within 4.5 h after the onset of stroke, 47 (23.3%) did not undergo POC-INR simply because of lost orders for POC-INR. Among 155 patients who underwent POC-INR, none had technical reasons to make the POC-INR test unfeasible.
Reliability
The median POC-INR was 1.02 (IQR, 1.0–1.1), and the median CL-INR was 1.00 (IQR, 1.0–1.1). The intraclass correlation coefficient between POC-INR and CL-INR was 0.975 (95% confidence interval, 0.966–0.982; Figure 1). Bland–Altman analysis indicated that the mean and standard deviation of the pair-wise differences were 0.01 ± 0.10. POC-INR had a 0.01 positive bias compared with CL-INR. The agreement range of the differences between POC-INR and CL-INR was from less than 0.19 to more than 0.21. Six pairs of values (3.87%) were located outside the agreement range (Figure 2). Of six patients having CL-INR >1.5, the intraclass correlation coefficient between POC-INR and CL-INR was 0.991 (95% confidence interval, 0.932–0.999; Table 2). In two patients, POC-INR values were higher than 1.5, but CL-INR values were less than 1.5. On the other hand, no patients had POC-INR values less than 1.5 but CL-INR values higher than 1.5. A total of 74 patients were taking antithrombotic agents (antiplatelet agents, warfarin, or direct oral anticoagulants (DOACs)) at the time of presentation. In these patients, the intraclass correlation coefficient between POC-INR and CL-INR was 0.975 (95% confidence interval, 0.959–0.984; Table 3).
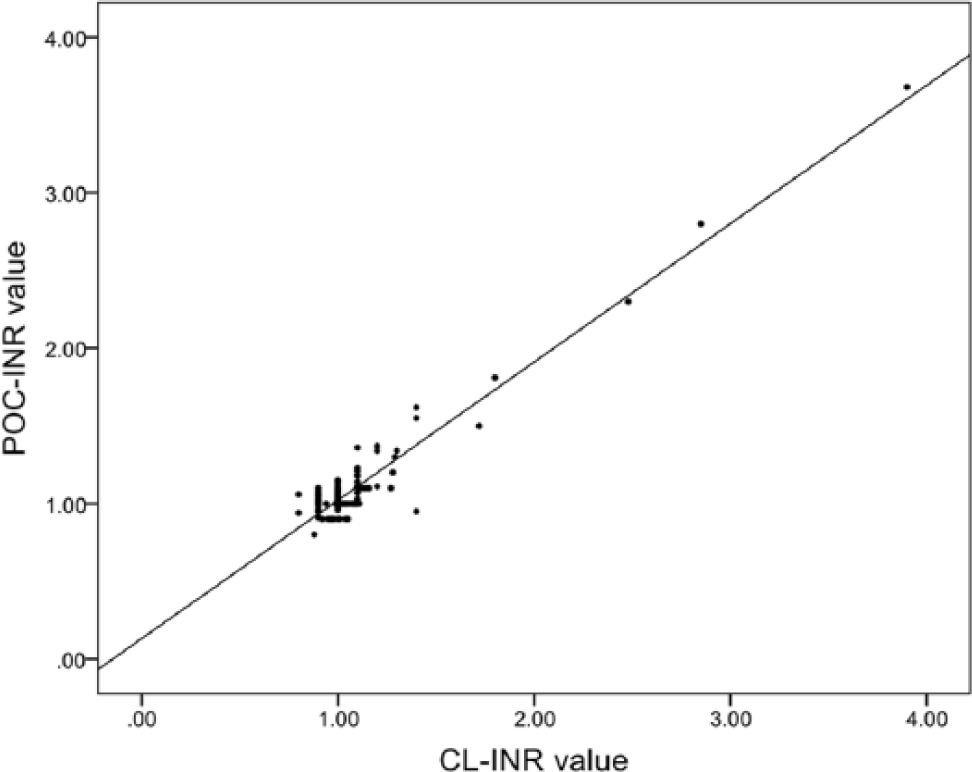
Scatter plot of the pairs of POC-INR and CL-INR.
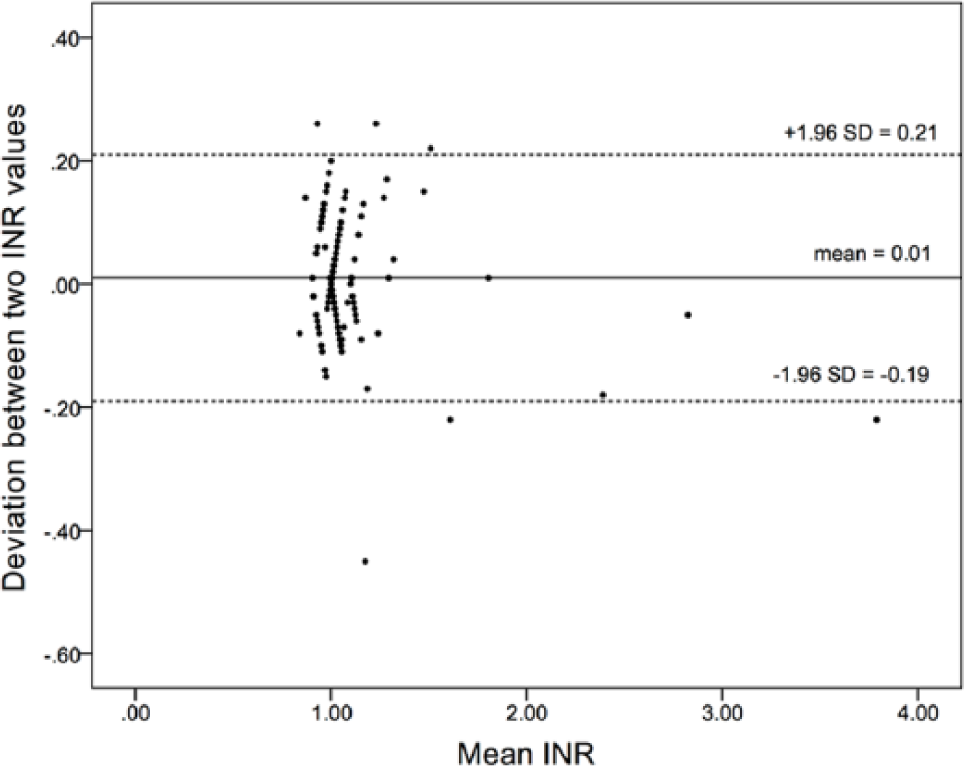
Bland–Altman bias plot for comparison of POC-INR to CL-INR.
Comparison of POC-INR values and CL-INR values in six patients whose POC-INR values were over 1.5.

POC-INR: point-of-care testing for prothrombin time international normalized ratio; CL-INR: central laboratory testing for prothrombin time international normalized ratio; ICC: intraclass correlation coefficient; CI: confidence interval.
Values are expressed as median (interquartile range).
Comparison of POC-INR values and CL-INR values in patients who were on antithrombotic agents at the time of presentation.
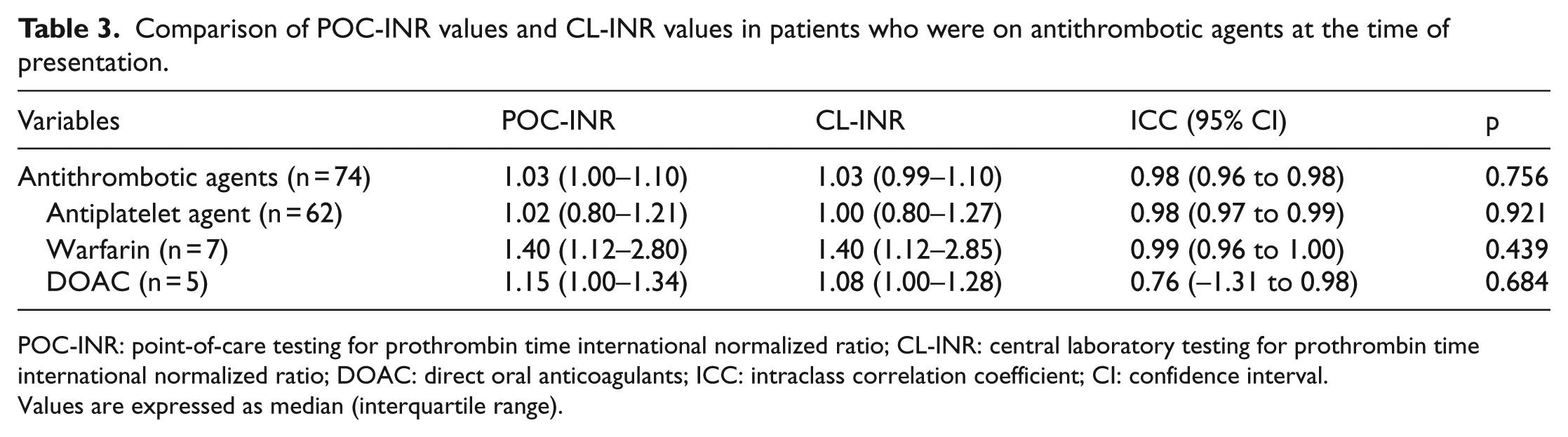
POC-INR: point-of-care testing for prothrombin time international normalized ratio; CL-INR: central laboratory testing for prothrombin time international normalized ratio; DOAC: direct oral anticoagulants; ICC: intraclass correlation coefficient; CI: confidence interval.
Values are expressed as median (interquartile range).
Usefulness
Among the 49 patients who received intravenous tPA, the median door-to-POC-INR time was 8.0 (IQR, 4.0–13.5) min, and the median door-to-CL-INR was 40.0 (IQR, 33.5–50.5) min. Thus, after implementation of the SAT, the door-to-INR time was significantly decreased by 32 min in patients who received thrombolysis treatment (p < 0.001). The door-to-tPA time was also decreased from 46.0 (IQR, 36.0–57.0) min to 23.0 (IQR, 16.0–29.8) min (Figure 3).
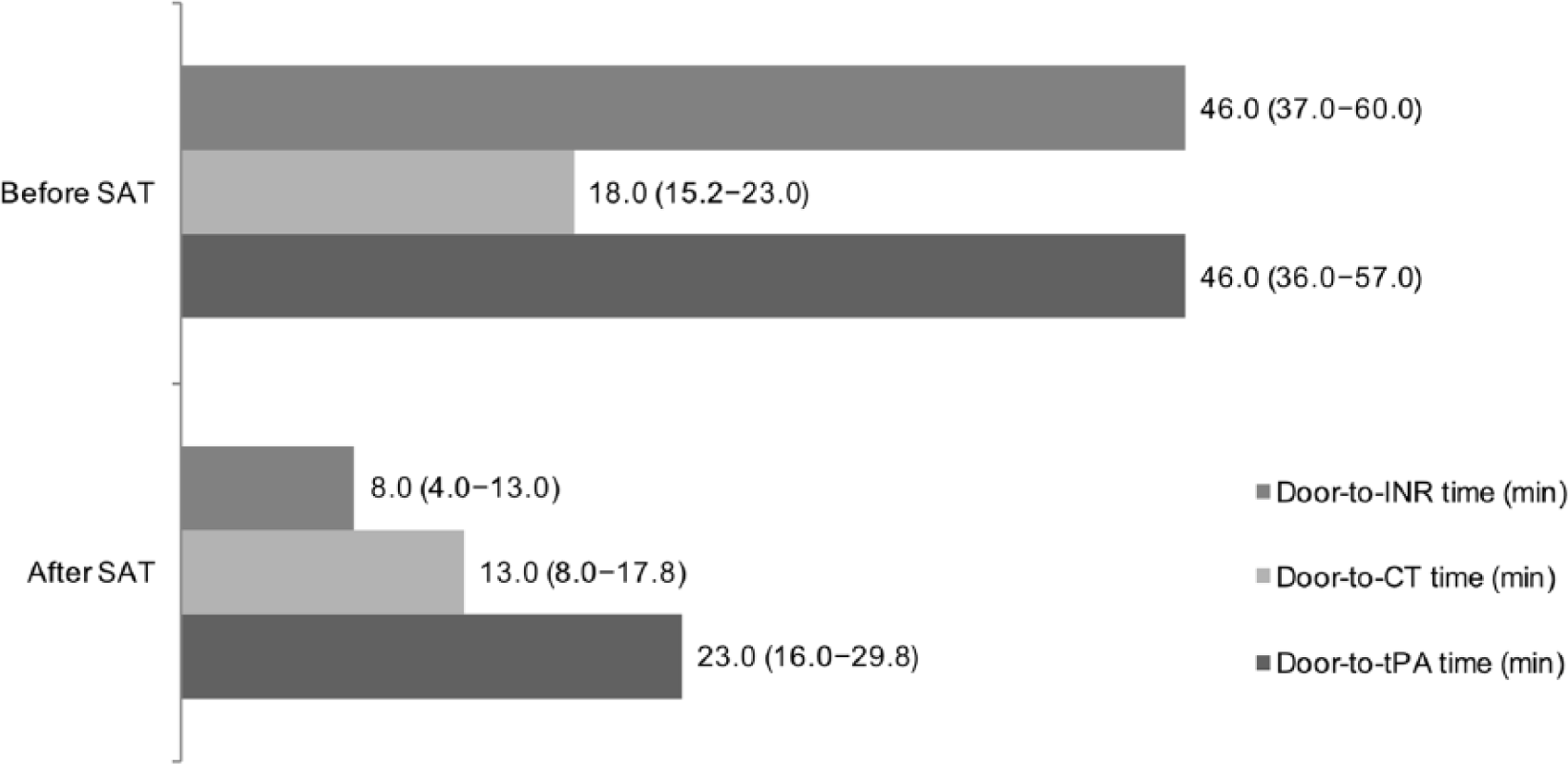
Comparison of time intervals between the two time periods before and after implementation of the Stroke Alert Team (SAT). Data are shown as median (interquartile range).
Discussion
The main finding of this study was that POC-INR was reliable compared with CL-INR and contributed to decreases in door-to-INR and door-to-tPA time when managing candidates for stroke thrombolysis. The intraclass correlation of the INR values between point-of-care testing and central laboratory testing was 0.975, and the deviation of means was only 0.01. We could decrease door-to-INR time to 8 min in patients receiving thrombolysis. Earlier confirmation of INR using point-of-care testing had a pivotal role in decreasing the door-to-tPA time by 23 min in our hospital-based quality improvement project. Thus, measurements of POC-INR in the ER were feasible, reliable, and useful for stroke thrombolysis.
Earlier infusion of tPA is associated with better patient outcomes. 2 However, there are multiple steps before the infusion of tPA. Obtaining CL-INR results is probably the most time-consuming process in thrombolysis.23,24 In this study, we showed that utilization of POC-INR reduced the door-to-INR and door-to-tPA times.
Previous studies on POC-INR for acute stroke have only been reported in patients who were taking anticoagulants.13,19 Patients with stroke, however, may have coagulopathies, including hepatic diseases and hematological diseases, or incorrect histories regarding anticoagulants. Thus, confirmation of INR values is important for the avoidance of the possible risk of post-thrombolysis hemorrhagic complications.
There is some controversy regarding the accuracy of POC-INR in patients taking anticoagulants. Some studies have suggested a poor correlation between POC-INR and CL-INR in patients with warfarin-induced INR elevation.25,26 In addition, there is controversy regarding the accuracy of POC-INR compared with that of CL-INR in patients taking DOACs.27–33 In this study, the two methods used to measure INR had a good correlation in 74 patients using antithrombotic agents (antiplatelet agents, warfarin, or DOACs). Nevertheless, our results should be interpreted with caution because we had a small number of study subjects. Inconsistencies between previous studies and our study may be due to the use of different blood sampling methods. Previous studies have used finger-stick blood for POC-INR and venous blood for CL-INR.25,34 However, we measured INR using venous blood drawn once and divided into two samples for both POC-INR and CL-INR. Venous sampling may be more accurate than the use of finger-stick blood. 34 Further studies are required to determine whether the sampling method affects the results of POC-INR and the correlation between POC-INR and CL-INR.
In addition to the inherent limitations of the retrospective design and selection bias, our study has other noteworthy limitations. First, although we implemented a point-of-care testing for prothrombin time INR, we did not use other point-of-care tests, such as activated partial thromboplastin time or platelet counts. The study population comprised patients with out-of-hospital stroke. Thus, there were no patients who met contraindications for tPA because of prolongation of activated partial thromboplastin time. In our patients, there were no candidates for thrombolysis among patients whose platelet counts were less than 100,000/µL. Thus, a lack of other point-of-care tests than POC-INR is likely to confound our findings. Second, POC-INR was used as a part of a quality improvement project in a single hospital. Thus, the decrease in door-to-tPA time was not solely a result of POC-INR. However, according to our experience, confirmation of CL-INR values was the most time-consuming step in thrombolysis before we implemented the SAT. Thus, we used POC-INR testing as a part of the SAT. The use of the POC-INR device was the only procedure that led to a higher cost in our SAT project. Third, the timing for the confirmation of POC-INR in this study was based not on the monitor of the POC-INR device but on the EMR. The POC-INR results were transferred from the device to the EMR system in a few minutes. However, for the thrombolysis practice, we could confirm POC-INR on the screen of the device. Fourth, the number of patients who were taking anticoagulants (warfarin, n = 7; DOAC, n = 5) or had INR values above 1.5 (n = 6) was too small to draw the conclusion that POC-INR is reliable when the INR results are expected to change the decision making of tPA administration, although this study showed a good correlation of POC-INR and CL-INR.
Conclusion
In conclusion, utilization of POC-INR was feasible in the management of candidates for stroke thrombolysis. POC-INR was quick and reliable and had a pivotal role in expediting thrombolysis.
Supplemental Material
a_POCT_suppl_data_final – Supplemental material for Point-of-care coagulation testing for reducing in-hospital delay in thrombolysis
Supplemental material, a_POCT_suppl_data_final for Point-of-care coagulation testing for reducing in-hospital delay in thrombolysis by Jung Hee Han, Seongsoo Jang, Mi-Ok Choi, Mi-Jeong Yoon, Seung-Bok Lim, Jeong-Ran Kook, Dong-Wha Kang, Sun U Kwon, Jong S Kim and Sang-Beom Jeon in Hong Kong Journal of Emergency Medicine
Footnotes
Acknowledgements
J.H.H., S.J., M.-O.C., M.-J.Y., S.-B.L., J.-R.K., D.-W.K., S.U.K., J.S.K., and S.-B.J. collected data. J.H.H. and S.-B.J. performed literature search and wrote the report. J.H.H. and S.-B.J. prepared figures. J.H.H. and S.-B.J. analyzed the data. J.H.H., D.-W.K., S.U.K., J.S.K., and S.-B.J. interpreted the data. J.H.H., M.-J.Y., D.-W.K., S.U.K., J.S.K., and S.-B.J. recruited all participants. J.H.H. and S.-B.J. designed the study. All authors appraised and edited the report and approved the manuscript before submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
All data submitted comply with Institutional Review Board requirements and applicable government regulations.
Informed consent
The need for written informed consent was waived by the Institutional Review Board of Asan Medical Center, because of the retrospective nature of this study and anonymization of patient data for the study.
Ethical approval
This study was approved by the Institutional Review Board of Asan Medical Center on 28 September 28 2016 (IRB No. 2016–1050).
Human rights
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
