Abstract
Objectives:
This study aimed to assess the accuracy of ocular ultrasound performed by emergency physicians for diagnosis of retinal detachment.
Methods:
This was a prospective single-center observational study conducted between September 2013 and February 2015. All adult patients presenting to the emergency department with acute onset of visual changes were eligible to participate. Patients who met one of the following criteria were excluded: a diagnosis of retinal detachment had already been made by an ophthalmologist prior to attendance; pre-existing retinal detachment currently under treatment; hemodynamically unstable; suspected rupture eyeball; and unable to give consent. Six emergency physicians performed emergency ocular ultrasound using a linear 10-MHz ultrasound probe. An abnormal lifting of the retina or the presence of a retinal flap was considered diagnostic of retinal detachment. The final diagnosis of the ophthalmologist who was blinded to the study was used as the gold standard.
Results:
Among the 139 patients evaluated, 16 (12%) had retinal detachments and 14 of them were correctly identified (true positives). Of the 123 patients (88%) without retinal detachment, 107 patients were correctly identified (true negatives). The sensitivity and specificity of emergency ocular ultrasound for retinal detachment were 88% (95% confidence interval = 60 to 98) and 87% (95% confidence interval = 79 to 92), respectively.
Conclusion:
Emergency ocular ultrasound is a useful adjunct for the diagnosis of retinal detachment.
Introduction
Retinal detachment (RD) is one of the most time-critical eye emergencies encountered in the emergency department (ED). RDs were uniformly blinding until the 1920s when Jules Gonin, MD, pioneered the first repair of RDs in Lausanne, Switzerland. With the advent of new techniques nowadays, rapid ED diagnosis and treatment of RD can be vision-saving.
In most cases, RD is caused by retinal breaks in the retina which allow fluid to enter beneath the retinal space and results in separation of the retina from the back of the eyeball. Since the detached retina is partially deprived of blood and nutrition supply, it will degenerate and will not function properly. The vision will be impaired and may progress to blindness if left untreated. Therefore, patients with RD should be diagnosed and managed promptly to save vision.
RD itself is painless. Early symptoms of rhegmatogenous detachment may include irregular or dark vitreous floaters (particularly a sudden increase), light flashes (photopsias), and blurred vision. As detachment progresses, the patient often notices a curtain or veil in the visual field. If the macula is involved, central vision becomes poor.
Diagnosis of RD in the ED setting is mainly by direct fundoscopy. Direct fundoscopy may detect vitreous hemorrhage (VH) and large detachment of the posterior pole, but it is inadequate for complete examination because of the relatively low magnification and illumination, lack of stereopsis, and limited view of the peripheral retina. Indirect ophthalmoscopy by ophthalmologist remains the definitive means of diagnosing RD. However, urgent assessment by ophthalmologist may not always be immediately available.
Emergency ocular ultrasound (EOUS) has been identified as a useful diagnostic tool for RD in ED in recent years.1–3 EOUS was advocated as early as the 1960s and 1970s4,5 and has been gaining popularity in the ED for the past decade. Since 2002, a number of studies have demonstrated that emergency physicians (EPs) can accurately identify a variety of ocular pathologies using EOUS, including RD.2,6,7 It is rapid, safe, and non-invasive. However, EOUS is not widely performed in EDs in Hong Kong.
The primary objective of this study was to determine the accuracy of EOUS for the detection of RD by EPs in the local ED setting.
Subjects and methods
This was a prospective single-center observational study of a convenience sample of patients between September 2013 and February 2015. The study was approved by the Cluster Research Ethics Committee of our hospital.
Study setting and population
This study was conducted at a local ED with 146,000 annual visits. All adult patients (>18 years) presenting to the ED with acute onset (defined as less than 48 h in duration) visual changes (decreased visual acuity, floaters or flashes, or visual field loss) were eligible for participation if one of the participating EPs was available to perform EOUS for RD. Patients who met one of the following criteria were excluded: a diagnosis of RD had already been made by an ophthalmologist prior to attendance; pre-existing RD currently under treatment; hemodynamically unstable; suspected rupture eyeball; and unable to give consent.
Study protocol
Six EPs volunteered to participate (one consultant, four associated consultants, and one higher trainee in emergency medicine) in the study. All of the participating EPs had prior experience with EOUS, and a standard protocol was agreed upon 1 month prior to start of the study. Informed consent was obtained prior to examination by EOUS. Patient was asked to close his or her eyes while ultrasound gel was applied to the closed eye covered with Tegaderm (a transparent dressing; Figure 1). To perform ocular ultrasound, a linear 10-MHz high-frequency ultrasound probe was used. A standard preset for small part was used. The scan was done in both sagittal and transverse planes. The examination was done with special care to avoid putting excessive pressure on the globe using a copious amount of ultrasound gel. A standard form was used to record patient’s presenting visual symptom, sonographic findings, preliminary diagnosis by EPs, and final diagnosis by ophthalmologists. Sonographic images with the relevant pathologies were also printed out for record. All patients were referred to ophthalmologists for evaluation.
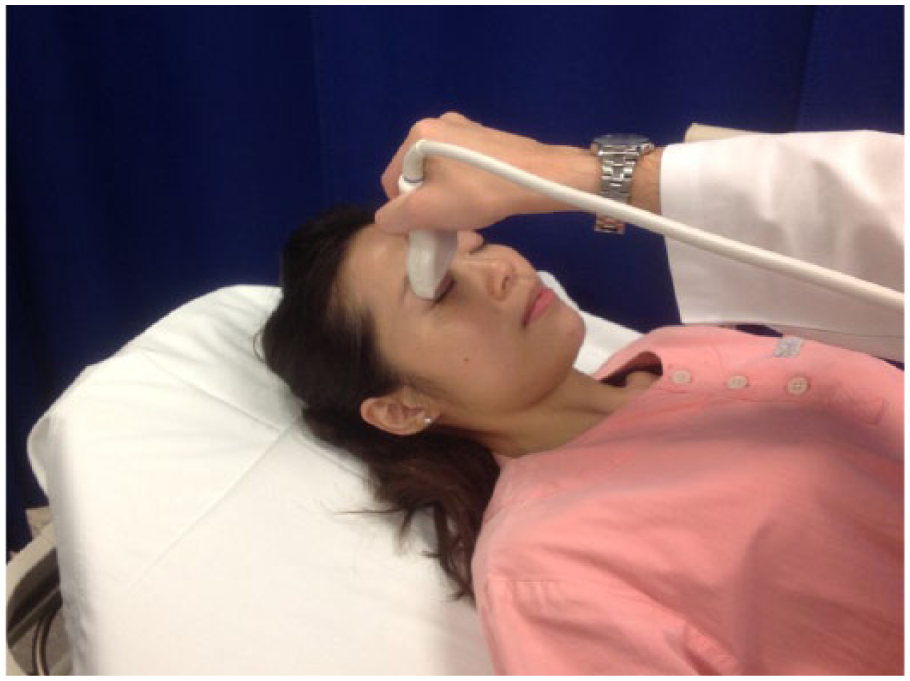
A high-resolution linear probe is applied in transverse plane to the closed eyelid.
Outcome measures
The primary outcome measure was RD as defined by an abnormal lifting of the retina or the presence of a retinal flap (Figure 2). The ophthalmologists’ final diagnoses were used as the gold standard. The ophthalmologists were blinded to our EOUS findings. The sensitivity, specificity, and likelihood ratios and their 95% confidence intervals (CIs) were calculated.
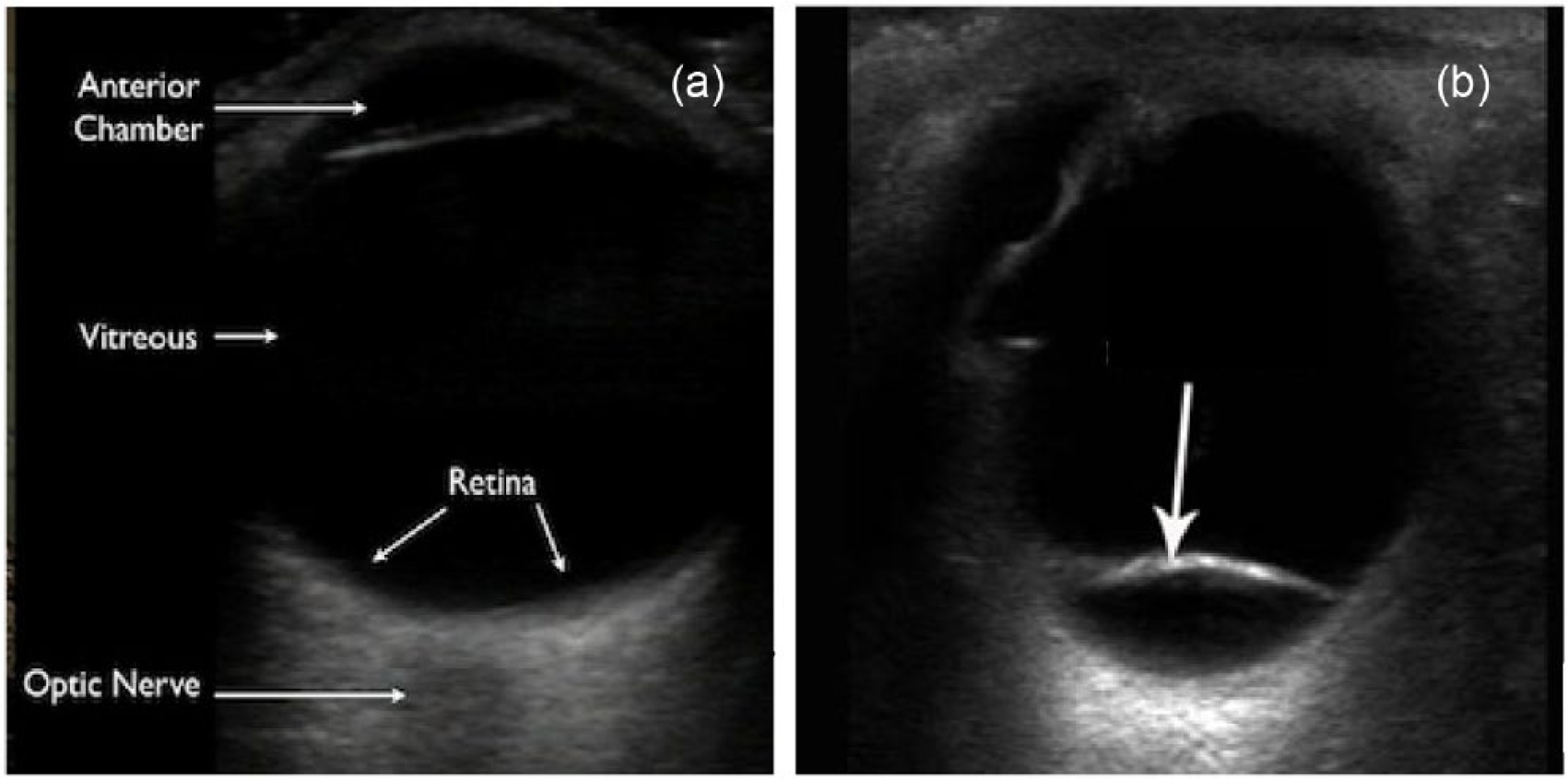
(a) Ocular ultrasound of a normal eye is shown. (b) A retinal flap (arrow) is seen in retinal detachment.
Results
During the period from September 2013 to February 2015, 148 patients were recruited into the study. Two patients were subsequently found not eligible because they were just below 18 years at the time of their recruitment and two were excluded because they did not attend subsequent ophthalmology appointment. Another five patients were excluded since no ophthalmology consultation records could be traced. Among the 139 patients recruited, 92 were female (66%) and the median age was 58 years (range: 18–92 years). The number and percentage of RD as diagnosed in each symptom group are listed in Table 1. The final diagnoses by ophthalmologists are listed in Table 2.
Retinal detachment diagnosed in each symptom group.

Final diagnoses by ophthalmologists.
In total, 16 (12%) patients had RD and 14 of them were correctly identified (true positives). Of the 123 patients (88%) without RD, 107 patients were correctly identified (true negatives). Therefore, the sensitivity and specificity of EOUS for RD were 88% (95% CI = 60 to 98) and 87% (95% CI = 79 to 92), respectively; the positive predictive value of EOUS was 0.47 (95% CI = 0.29 to 0.65) and the negative predictive value was 0.98 (95% CI = 0.93 to 1.0), with a positive likelihood ratio of 6.7 (95% CI = 4.1 to 11.0) and a negative likelihood ratio of 0.14 (95% CI = 0.04 to 0.55).
Discussion
Siegel et al. 8 found a 38% miss rate of retinal pathologies that required intervention when non-dilated direct fundoscopy was used alone. Thus, EOUS can potentially be used as an adjunct to improve the clinical assessment of RD given the high sensitivity (88%) and negative predictive value (98%) found in this study.
The negative predictive value (98%) of our study was comparable to previous studies. However, we performed less well in terms of sensitivity and specificity. In a study by Shinar et al. 9 published in 2011, the sensitivity was 97% (95% CI = 82 to 100) and specificity was 92% (95% CI = 82 to 97). The positive predictive value was 85% (95% CI = 68 to 95) and the negative predictive value was 98% (95% CI = 91 to 100). Another study by Yoonessi et al. 1 found a sensitivity of 100% (95% CI = 78 to 100) and specificity of 83% (95% CI = 65 to 94) with positive predictive value being 78% (95% CI = 56 to 92) and negative predictive value 100% (95% CI = 83 to 100).
There were two cases of false negatives. The first patient presented to the ED for 1-day history of floaters. The attending EP found no retinal flap on EOUS and referred the patient to the ophthalmology department. She was subsequently diagnosed to have macular hole RD. B-scan ultrasonography is generally not sensitive enough to distinguish macular holes from masquerading lesions. Optical coherence tomography has shown great potential in the diagnosis of macular disease, including macular hole.10,11 The second patient presented with flashes for 1 day. EOUS showed no retinal flap. She was later diagnosed to suffer from left inferior RD. It is postulated that the fanning of the probe from cephalad to caudad may not be thorough enough to pick up peripheral lesions. Views of the peripheral retina are rather difficult using the axial section. Further refinement of the scanning protocol may help to prevent missing peripheral lesions. For example, peripheral lesion is better appreciated with the transverse section when the probe is shifted from limbus to fornix to examine the posterior pole and peripheral retina.
Concerning the false positives, most of the misidentified patients (11 out of 16 false positives) had posterior vitreous detachment (PVD). PVD is a common mimic of RD on ocular sonography. 12 In general, RD appears as a prominent, continuous linear density arising from the fundus. Depending on the timing and severity of the detachment, the retinal separation may be visible only as a small peripheral convexity or, with an extensive detachment, as a complex array of bright, intersecting lines (Figure 2). Because the retina is firmly adhered to the optic disc posteriorly and the ora serata anteriorly, complete RD appears tethered to the optic disc giving a “funnel” appearance. However, PVD may also appear as a hyperechoic linear density that has been lifted off the posterior globe; however, it typically appears as a thinner and smoother structure compared to RD (Figure 3). Moreover, PVD takes on a “swaying seaweed” appearance on dynamic scanning.8,13 The linear flap of PVD will float back and forth upon ocular movement, cross the optic nerve, and will not be tethered to the back wall. 14 PVD often fades when the ultrasound gain is reduced because the posterior hyaloid surface is less dense than the retina. 8 This is useful to differentiate from a retinal flap which is highly reflective. 15 Thus, we can reduce misdiagnosing PVD for RD if we add dynamic scanning and reducing the gain in our scanning protocol.
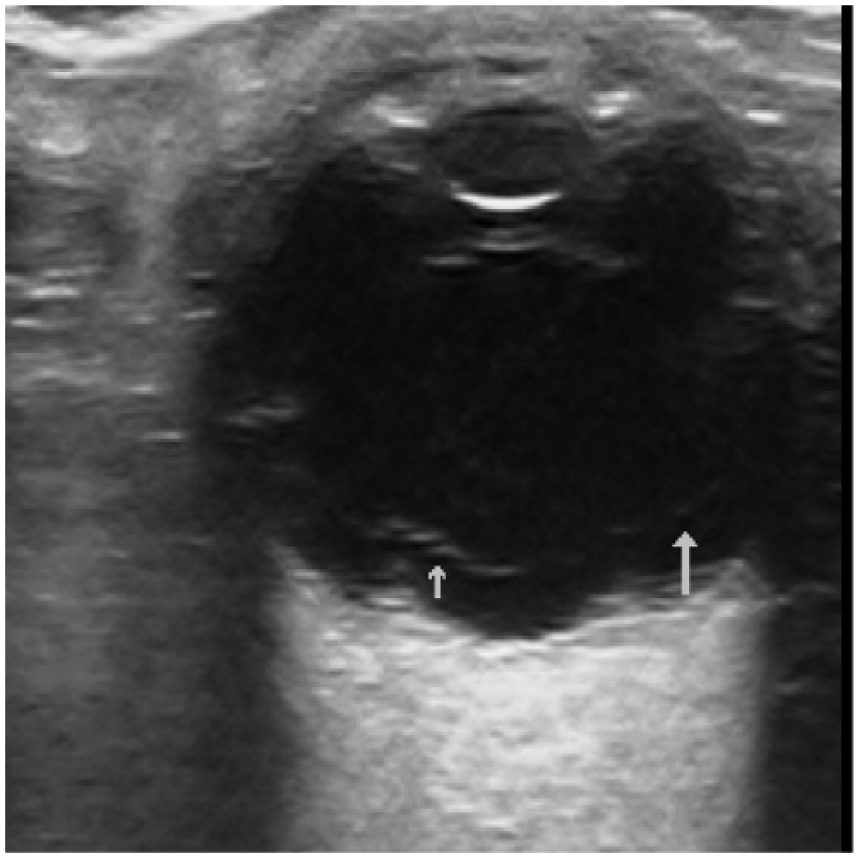
Low reflective vitreous opacities in posterior vitreous detachment.
Two other false positive cases belong to VH (Figure 4). To differentiate VH from RD, dynamic ocular sonography can be used. RD will be tethered to the globe producing an undulating flap movement, 16 while VH will swirl around without apparent attachment to the posterior globe. Moreover, RD can be seen at low gain levels, while VH requires high gain to identify. VH will fade when the ultrasound gain is reduced. 16 The remaining three false positive cases had ophthalmological diagnoses of floaters, branch retinal vein occlusion, and visual field defect, respectively.
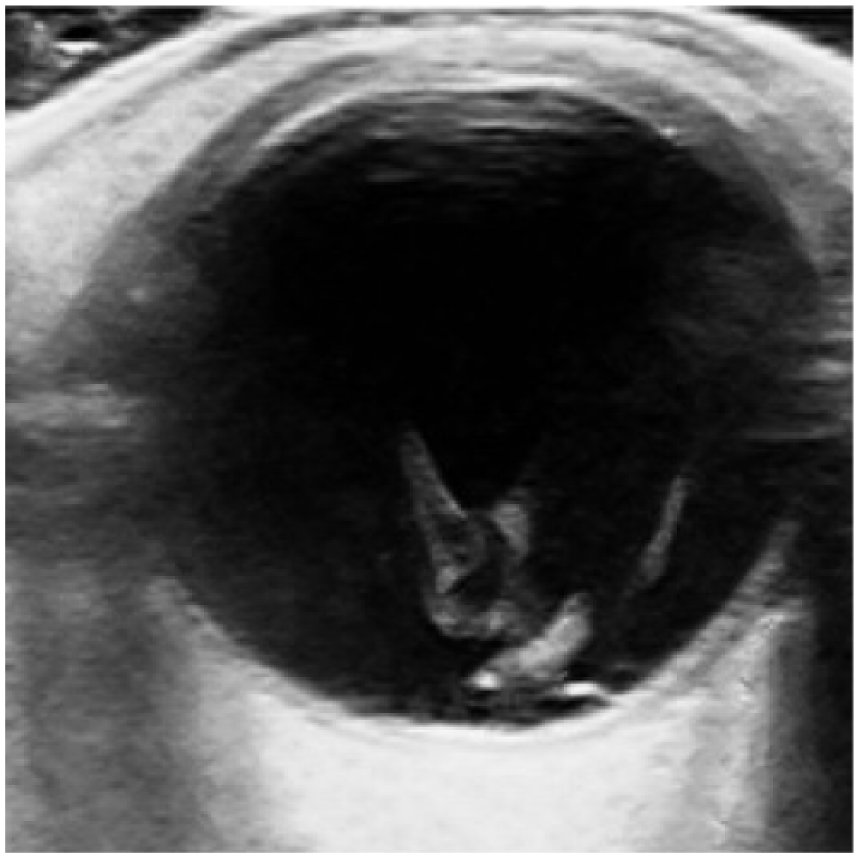
Vitreous hemorrhage. Note the diffuse mobile opacities in the posterior chamber.
Limitations
There are a few limitations in this study. First, the sample size was small which accounted for the wide CI of sensitivity and specificity. There were 139 patients recruited, and the number of RD was only 16. This was due to limited availability of participating physicians and convenience sampling. Second, this was a single-center study and ultrasound diagnosis was operator-dependent; therefore, generalizability to other settings may be difficult.
Conclusion
EOUS is a useful adjunct in the ED for the diagnosis of RD.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
