Abstract
Ultrasonic perfusion imaging predicts size and localization of acute stroke. It is unclear whether irreversibly damaged tissue can be differentiated from tissue at risk. Thirty-four patients (ischemic stroke <12 h) were included (Phase Inversion Harmonic Perfusion Imaging; bolus kinetic; fitted model function). Three patterns of perfusion were defined in 14 prespecified regions of interest (ROI): ‘normal', ‘hypoperfusion', and ‘no perfusion'. Clinical status was assessed using the National Institutes of Health Stroke Scale (NIHSS) (at baseline and at days 2 to 4). Cranial Computed Tomography (CCT) (days 2 to 4) displayed final infarction. The pattern ‘hypoperfusion’ (ROIs presumably representing tissue at risk) was tested twofold: (i) Functional impairment by correlating their number with baseline NIHSS. (ii) Viability by correlating their recruitment rate to infarction with clinical course (ΔNIHSS days 2 to 4). In addition, various predictive values were assessed. Twenty-seven patients were eligible for analysis. The sum of ROIs with ‘no perfusion’ and ‘hypoperfusion’ correlated highest with baseline NIHSS (ρ = 0.78, P < 0.001). Recruitment of hypoperfused ROIs to infarction highly correlated with clinical course (ρ = 0.79, P < 0.001). Clinical course dichotomized the patients into subgroups A ('stable', ΔNIHSS ≥ −3) and B ('improved', ΔNIHSS ≤ −4). In A, sensitivity and specificity for hypo- and nonperfused tissue being eventually infarcted were 96% and 88% positive predictive value, PPV 89%, negative predictive value, NPV 96%). In B, sensitivity and specificity for nonperfused tissue eventually being infarcted were 81% and 99% (PPV 99%, NPV 84%). Different perfusion patterns (hypoperfusion, no perfusion) and dysfunctional but viable tissue at risk can be reliably detected by ultrasonic perfusion imaging. This method may give Supplementary information in cases illegible for perfusion-weighted magnetic resonance imaging (PW-MRI).
Introduction
In acute stroke, sophisticated computed tomography (CT)- and magnetic resonance imaging (MRI)-based protocols supply information on the status of vessels and the brain (Schellinger et al, 2003; Shih et al, 2003). In a nonacademic setting, native CCT scan and color-coded sonography are probably the most commonly used diagnostics to decide on the appropriate therapy. Ultrasonic perfusion imaging may supply supplementary information, for example, once the risks and benefits of thrombolysis are considered. A bilateral examination employing the bolus kinetic approach with a model-based evaluation of time-intensity curves (TIC) takes approximately 10 mins (data acquisition, region-wise assessment of perfusion conditions). The resulting parameters can be displayed in parametric images so that the affected hemisphere can be visually compared with the unaffected hemisphere. The parameters are robust in a region-wise evaluation throughout both hemispheres (Eyding et al, 2003; Krogias et al, 2005). First results suggest that the core of an infarction can be discriminated from hypoperfused and potentially salvageable tissue (Eyding et al, 2004).
The aim of this study was to correlate bilateral parametric Phase Inversion Harmonic Perfusion Imaging (PIHI) to follow-up CCT scans with respect to clinical course in acute middle cerebral artery (MCA) stroke. It was hypothesized that a significantly delayed perfusion kinetic in a specified region of interest describes hypoperfused and dysfunctional ‘tissue at risk', whereas the absence of a perfusion kinetic depicts ischemic tissue destined to undergo infarction. With respect to the individual clinical course, it was further hypothesized that hypoperfused tissue should not eventually be infarcted in follow-up CCT once the patient experienced definite early clinical improvement. However, once the clinical course stayed stable, these regions should be infarcted in CCT.
Materials and methods
Patients
Thirty-four consecutive patients with a hemispheric syndrome were included (acute onset of a clinical stable sensory–motor hemiparesis, neglect, or aphasia <12h). Exclusion criteria were rapid clinical improvement or worsening before admission, hemorrhage on initial CCT, pregnancy, severe pulmonary, cardiac, or renal dysfunction. Initial color-coded duplexsonography ultrasound examinations followed guidelines as described earlier (Hennerici and Meairs, 1999). Clinical status was examined using the National Institutes of Health Stroke Scale (NIHSS; Lyden et al, 1994) on admission and 2 to 4 days later. Two native CCT scans were performed: on admission and a routine follow-up examination to confirm size and localization of the infarction 2 to 4 days later.
Informed consent was obtained before entering the study. The local ethics committee approved to the present study. The procedures followed in this study were in accordance with institutional guidelines.
CCT Evaluation
CCT scans were performed on a Siemens Somatom™ Plus 4 Scanner. All cranial scans were obtained in the orbitomeatal projection with slice thickness of 3 to 8 mm. All scans were evaluated by a neuroradiologic experienced radiologist (MS) being masked to side of infarction and clinical severity. The plane corresponding to the ‘mid-thalamic’ ultrasound plane was chosen as defined by morphologic structures: frontal horns of side ventricles, third ventricle, basal ganglia, and posterior horns of side ventricles (compare Krogias et al, 2005). For the follow-up scan, 20 regions of interest (ROI; 10 of each side) according to the defined ROIs of the ultrasonic examination were classified as either ‘infarction’ or ‘no infarction'.
Phase Inversion Harmonic Perfusion Imaging
The protocol of the ultrasound perfusion examination using SonoVue® (Bracco International BV) as ‘contrast agent’ has been described before (Eyding et al, 2004). Experienced examiners (JE, CK, SM, and AS) were masked to the severity of clinical status and CCT results, side of supposed pathology was known. As a reference value, the median of time-to-peak intensity (TPI) in four prespecified ROIs of the nonaffected hemisphere was calculated (lentiform nucleus; anterior and posterior thalamus; head of caudate nucleus). Evaluation of 10 prespecified ROIs within the MCA territory of the affected hemisphere was performed correspondingly (ROIs 1 to 10: caudate nucleus (1), thalamus (2, 3), lentiform nucleus (4), white matter (5 to 7), and cortical structures (8 to 10); see Figure 1A; compare Krogias et al, 2005). If the ipsilateral approach had to be chosen, only seven ROIs of the affected hemisphere could be evaluated (missing the cortical regions).
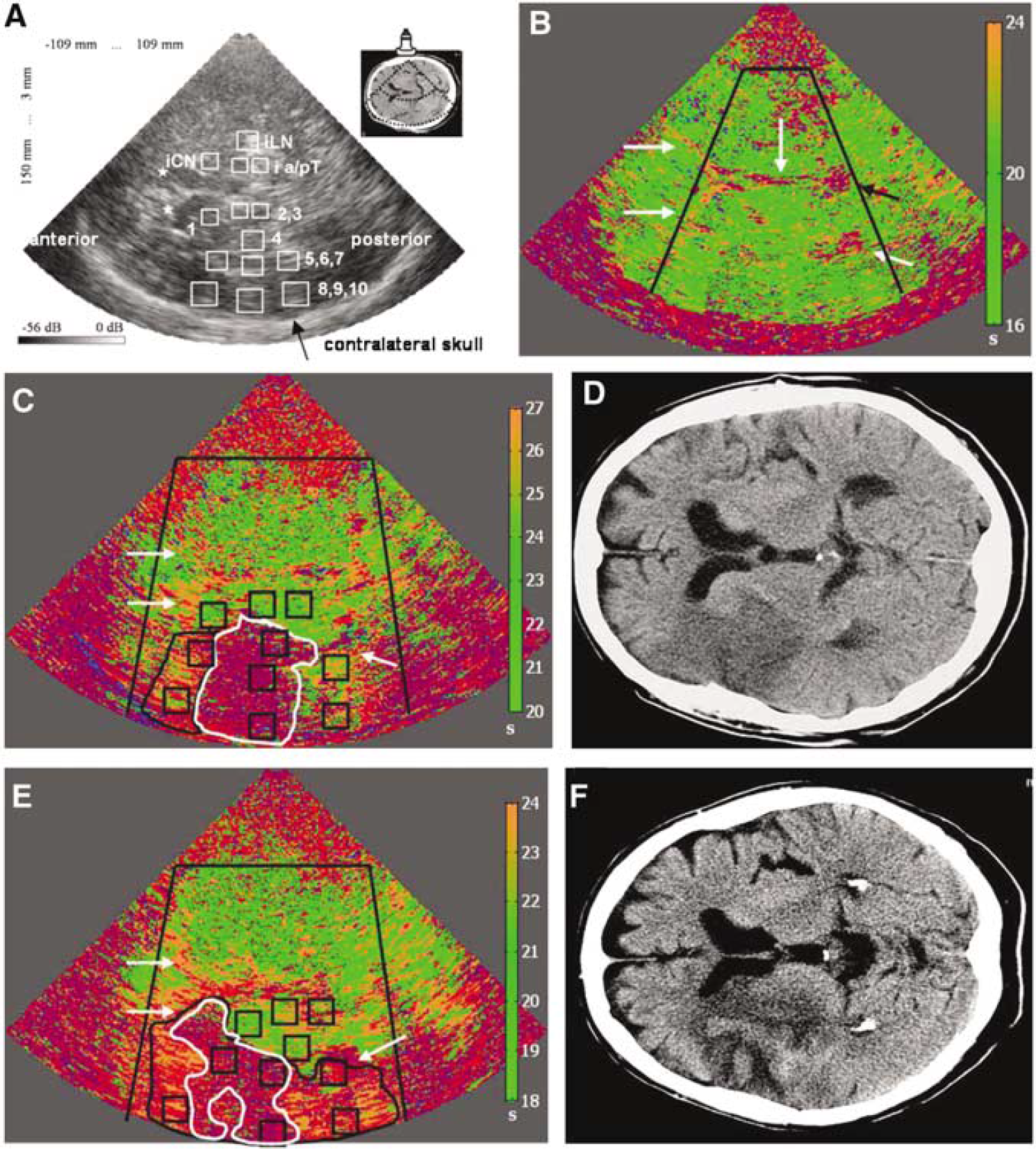
(
Data Analysis
Patients eligible for statistical evaluation were those with stable or improving clinical course defined by change in follow-up NIHSS score. Patients with complications that could possibly influence the NIHSS score (disturbing the natural course of the stroke, that is, for example, intracerebral hemorrhage) were excluded.
Region-wise evaluation of the TIC took place immediately after the examination. The respective perfusion status was allocated to each ROI. Three patterns of perfusion behavior were defined: (i) ‘normal': bolus-like progression of the TIC, TPI within ±3 secs of intraindividual median of ROIs of nonaffected contralateral hemisphere; (ii) ‘hypoperfusion', presumably ‘tissue at risk': bolus-like curve progression, TPI > 3 secs delayed; and (iii) ‘no perfusion', presumably ‘core of infarction': no bolus-like curve progression (see above; in a former study (Krogias et al, 2005), the evaluation of 280 individual TPIs in 20 healthy volunteers suggested 3 secs as a safe intraindividual cutoff).
Afterwards, a pixel-wise color-coded parametric image for TPI values was calculated, where values in the range of ±3 secs with respect to the calculated referential median (unaffected hemisphere) were coded in green, pixels with a delay of more than 3 secs in orange, pixels with undetectable perfusion kinetic in red, and pixels not evaluable because of insufficient signal enhancement of less than 1.8 dB or insufficient signal-to-noise ratio (algorithm did not converge) in pink (for a parameter image of a healthy volunteer see Figure 1B).
Statistical Evaluation
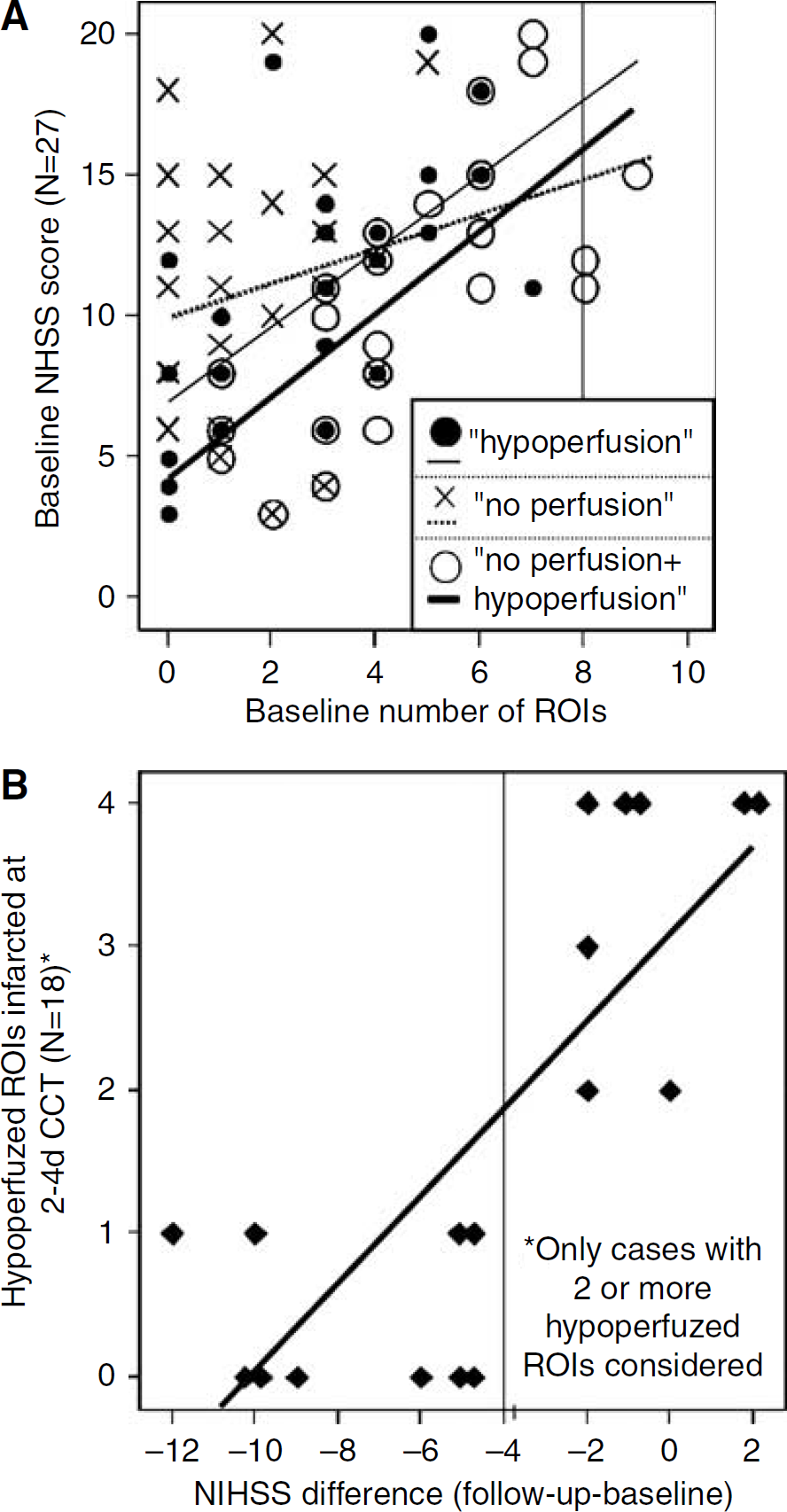
Firstly, it was analyzed whether the threshold for the definition of ‘hypoperfusion’ was appropriate to represent tissue at risk (i.e., functionally impaired but viable tissue). This was determined twofold: firstly, the correlation between the baseline NIHSS (NIHSSbaseline) and the number of differently perfused ROIs was calculated (i, hypoperfused ROIs (HP); ii, hypo- and nonperfused ROIs (HP + NP); and iii, nonperfused ROIs (NP)). The aim was to evaluate whether hypoperfused ROIs also represent functionally impaired tissue. It was hypothesized that both hypo- and nonperfused ROIs represent dysfunctional tissue in the acute state and, thus, that the correlation between (HP + NP) and NIHSSbaseline should be the strongest. Secondly, the viability of hypoperfused ROIs was shown by their recruitment rate (RR) to the final infarct as determined by follow-up CCT (days 2 to 4) in conjunction with the clinical course (ΔNIHSS score between baseline and days 2 to 4). Recruitment rate was defined as: RR = 100 (ROIinf FU)/ROIHP (%), where ROIinf FU = number of finally infarcted ROIs at follow-up CCT that were initially ROIHP at the baseline PIHI. It was hypothesized that the better the clinical course was the less hypoperfused ROIs were recruited to infarction. A correlation analysis between RR and ΔNIHSS was performed (only patients with two or more hypoperfused ROIs were included in this correlation analysis; to check for artifacts because of the rough scaling in cases with only two hypoperfused ROIs, additional correlation analyses were performed including only patients with ≥3 and ≥4). This latter correlation analysis was complemented by dichotomizing the patient sample according to their clinical course into subgroup A with a stable clinical course (ΔNIHSS > −3) and subgroup B with definite early clinical improvement (ΔNIHSS < −4). The following hypotheses were tested: if vascular compensation (namely recanalization) was sufficient, clinical course should reveal a definite improvement, and hypoperfused ROIs should not be infarcted in follow-up CCT. Else, if vascular compensation was insufficient, clinical status should not improve definitely, and these ROIs should be ‘recruited’ to infarction in follow-up CCT. A sensitivity and specificity analysis was performed to test the value of the ROI-type ‘hypoperfusion’ in the two subgroups. Therefore, sensitivity and specificity were differently defined in subgroup A (hypo- and nonperfused ROIs that were infarcted in CCT) and B (only nonperfused ROIs that were infarcted in CCT). All correlation analyses performed were nonparametric. All analyses were run with SPSS® (version 11.5). Level of significance was defined at P < 0.05 (two sided).
Results
Baseline Data
Thirty-four patients were examined. Three of these patients (11%) had to be excluded because of insufficient temporal bone window on either side. In two of the 31 remaining target patients, only an ipsilateral evaluation could be performed because of partly insufficient bone window. The median NIHSS score in these 31 patients was 12 (interquartile range (IR) 8 to 15). The score was reevaluated on days 2 to 4 (mean 66.3 h, standard deviation (s.d.) 17.8 h after initial examination). Three of these 31 patients had died because of a space occupying MCA infarction in the meantime; one patient had a symptomatic intracranial hemorrhage on day 2. The median follow-up NIHSS score of the other 27 patients (eligible for further analysis) was 6 (IR 3 to 10.75). Sixteen patients did not show significant clinical improvement or worsening in the first 2 to 4 days (ΔNIHSS ≥ −3 points); 11 patients improved clinically significantly (ΔNIHSS ≤ −4 points; same cut-point as used by, for example, Kwon et al, 2004). CCT follow-up examination on days 2 to 4 (mean 63.0 h after initial examination, s.d. 19.7 h) revealed hemorrhage in four patients (one being a symptomatic intracranial bleeding with eventual death, three being asymptomatic ‘hemorrhagic transformations'). Further data are displayed in Tables 1 and 2.
Characteristics of target patients and subgroups A (stable clinical course) and B (definite clinical improvement)
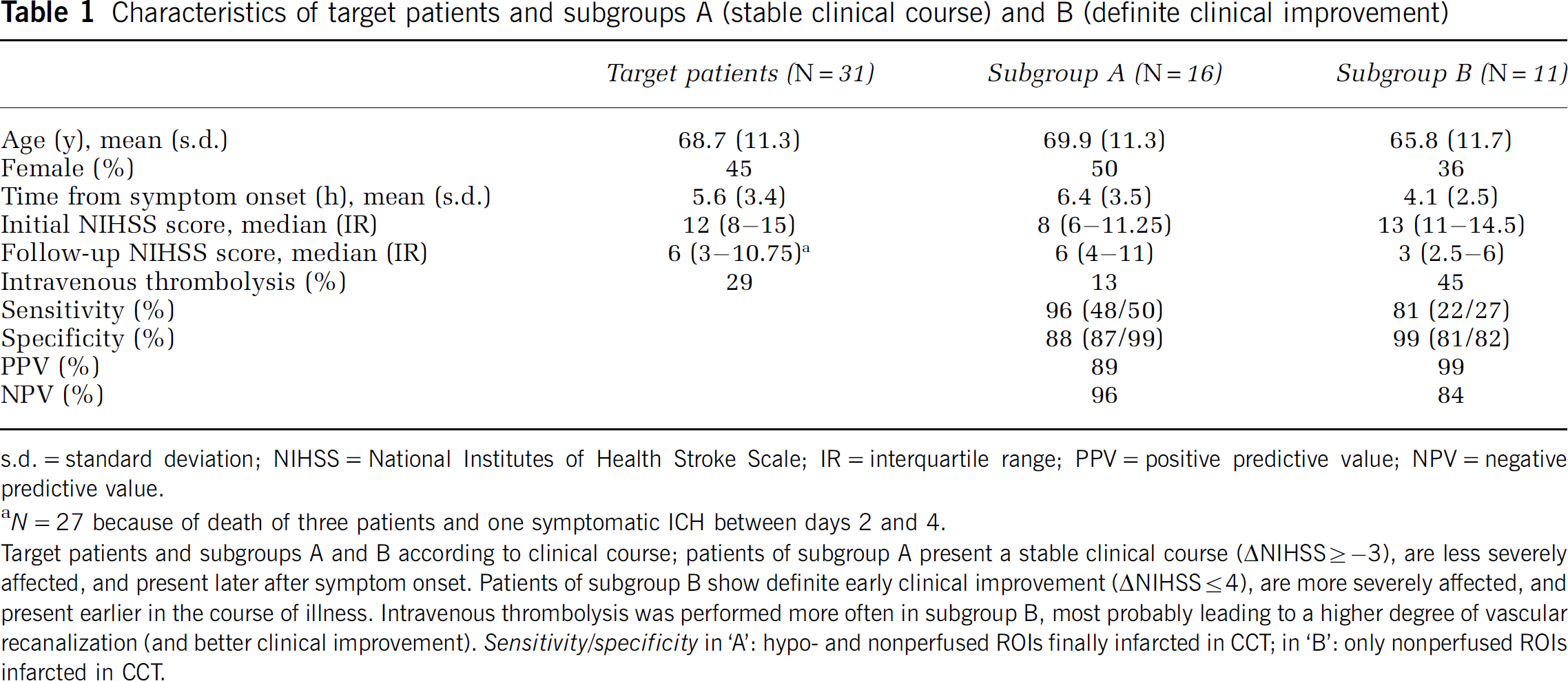
s.d. = standard deviation; NIHSS = National Institutes of Health Stroke Scale; IR = interquartile range; PPV = positive predictive value; NPV = negative predictive value.
N = 27 because of death of three patients and one symptomatic ICH between days 2 and 4.
Target patients and subgroups A and B according to clinical course; patients of subgroup A present a stable clinical course (ΔLNIHSS ≥ −3), are less severely affected, and present later after symptom onset. Patients of subgroup B show definite early clinical improvement (ΔLNIHSS ≤ 4), are more severely affected, and present earlier in the course of illness. Intravenous thrombolysis was performed more often in subgroup B, most probably leading to a higher degree of vascular recanalization (and better clinical improvement). Sensitivity/specificity in ‘A': hypo- and nonperfused ROIs finally infarcted in CCT; in ‘B': only nonperfused ROIs infarcted in CCT.
Phase Inversion Harmonic Perfusion Imaging (PIHI) in 27 patients: characteristics of study patients
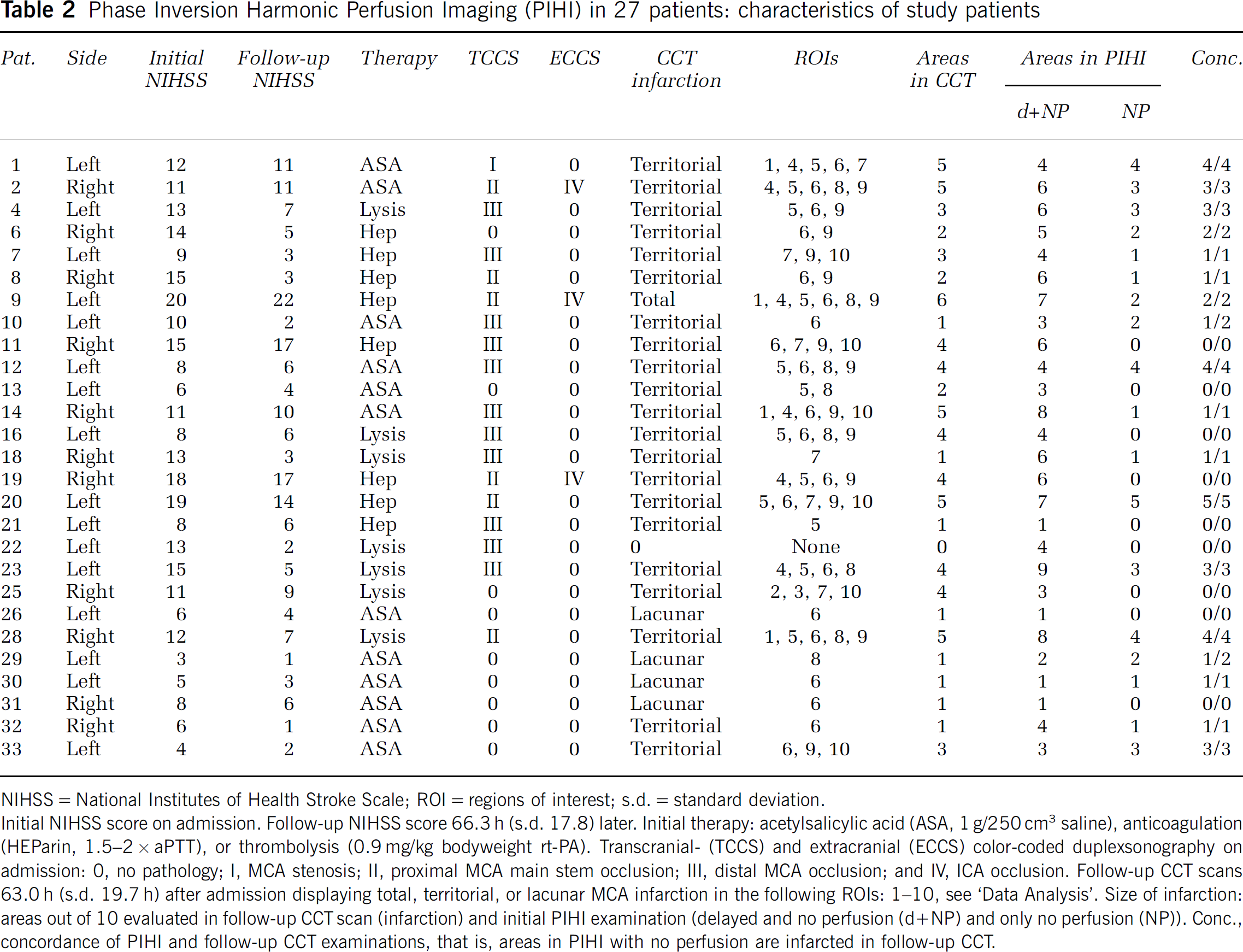
NIHSS = National Institutes of Health Stroke Scale; ROI = regions of interest; s.d. = standard deviation.
Initial NIHSS score on admission. Follow-up NIHSS score 66.3 h (s.d. 17.8) later. Initial therapy: acetylsalicylic acid (ASA, 1 g/250 cm3 saline), anticoagulation (HEParin, 1.5–2 × aPTT), or thrombolysis (0.9 mg/kg bodyweight rt-PA). Transcranial- (TCCS) and extracranial (ECCS) color-coded duplexsonography on admission: 0, no pathology; I, MCA stenosis; II, proximal MCA main stem occlusion; III, distal MCA occlusion; and IV, ICA occlusion. Follow-up CCT scans 63.0 h (s.d. 19.7 h) after admission displaying total, territorial, or lacunar MCA infarction in the following ROIs: 1–10, see ‘Data Analysis'. Size of infarction: areas out of 10 evaluated in follow-up CCT scan (infarction) and initial PIHI examination (delayed and no perfusion (d + NP) and only no perfusion (NP)). Conc., concordance of PIHI and follow-up CCT examinations, that is, areas in PIHI with no perfusion are infarcted in follow-up CCT.
Phase Inversion Harmonic Perfusion Imaging
Application of the contrast agent was tolerated well by all patients, no side effects occurred. Region-wise evaluation of perfusion-related parameters in the referential hemisphere was successful in 106/108 (98%) possible ROIs; in two patients, the caudate nucleus was not evaluable because of partial insufficient temporal bone window. The median TPI in these 106 ROIs was 20 secs (s.d. 7.4 secs); however, the mean quartile deviation in the 27 patients was 0.45 secs (s.d. 0.28 secs). In the 27 patients eligible for statistical evaluation (26 bilateral examinations), 9/267 (3%) possible ROIs of affected sides were not evaluable because of partial insufficient temporal bone window (ROI 5 in three patients, ROI 1 in three patients, ROI 8 in two patients, and ROI 10 in one patient).
The predictive value of the number of nonperfused ROIs alone on the clinical status was low and not significant (Spearman-ρ = 0.18). The number of hypoperfused ROIs significantly correlated with the clinical status (Spearman-ρ = 0.67, P < 0.001). The correlation was highest, however, if all impaired ROIs (i.e., hypo- and nonperfused) were analyzed against the clinical status (Spearman-ρ = 0.78, P < 0.001; Figure 2A). The RR of hypoperfused ROIs to the final infarction strongly and robustly correlated with the clinical course (Spearman-ρ = 0.79, P < 0.001; patients with two or more hypoperfused ROIs, see also Figure 2B; the control analyses (see Data Analysis) with patients considered with three (4) or more hypoperfused ROIs resulted in Spearman-ρs of 0.81 (0.84) with P = 0.002 (P < 0.01)), that is, the higher the ratio of hypoperfused ROIs that became infarcted at days 2 to 4 (on CCT), the worse the clinical course. The same robust correlation resulted if the patient sample was dichotomized according to its clinical course. A cut-point for differentiating whether hypoperfused ROIs would be recruited either to infarction or not depending on clinical course was ΔNIHSS score of four points (see Figure 2B; the selected cut-point corresponded to an RR of the hypoperfused ROIs to infarction of <50 and ≥50%, respectively). A subgroup analysis was made with special respect to the characterization of hypoperfused areas. In patients with a stable clinical course (subgroup A), it was tested if they were infarcted in follow-up CCT. Sensitivity and specificity, respectively, were 96% and 88% with PPV 89% and NPV 96%. In patients with a definite early clinical improvement (subgroup B), it was tested if they were not infarcted in CCT. Sensitivity and specificity, respectively, were 81% and 99% with PPV 99% and NPV 84%. The results are displayed in Table 1.
(
Discussion
Ultrasonic cerebral perfusion imaging has been introduced in the late 1990s (Postert et al, 1998; Seidel et al, 1999). First studies on acute stroke patients indicated the ability to predict localization and size of final infarction (Federlein et al, 2000; Seidel et al, 2003). However, the studies were purely qualitative. Some effort was made to introduce (semi) quantitative parameters, employing different ultrasonic techniques and data acquisition and processing techniques (Eyding et al, 2002). The aim, of course, is to differentiate tissue at risk and core of infarction in acute stroke. Analyzing the Bolus Kinetic (which was used in this study), it is possible to describe a normal variation of parameters in healthy volunteers, and, theoretically, define cutoff scores of impaired perfusion. The deviation of individual TPI values, for example, is distinctly small, so that an intraindividual cut-point describing a significant delay was hypothesized with 3 secs (Krogias et al, 2005). Lately, there have been reports on ultrasonic parametric perfusion imaging (Wiesmann et al, 2004), the ability of predicting outcome (Seidel et al, 2004), as well as on a ‘destructive’ mode and its ability to display cerebral infarction (Kern et al, 2004). However, none of these studies aimed to differentiate ‘tissue at risk’ and ‘core’ of infarction.
The present study was designed to evaluate the potential of bilateral PIHI using the Bolus Kinetic for differentiating infarcted from hypoperfused and dysfunctional tissue ('tissue at risk') in acute stroke. To cover only acute changes of perfusion (rather than, for example, compensatory hyperperfusion), patients with a stable or improving clinical course were included with a time window of 12 h after symptom onset. Clinical follow-up examinations were performed on days 2 to 4 to cover only effects of immediate vascular compensation (recanalization) rather than effects of, for example, rehabilitation.
The functional status of our patients correlated most strongly to the sum of ROIs both with a delayed and without perfusion kinetic. This validates the hypothesis that our method detects hypoperfused tissue that also is dysfunctional. Hypoperfused ROIs were differently recruited to eventual infarction. In patients with a stable clinical course, it was tested if they were infarcted in follow-up CCT. In patients with a definite early clinical improvement, it was tested if they were not infarcted in CCT. Sensitivity and specificity ranged from 81% to 99% with PPV of 89% and 99% and NPV of 96% and 84%. Our conclusion is that regions with a delayed perfusion kinetic implicate dysfunctional ‘tissue at risk', which is not salvaged in patients with a stable clinical course (no recanalization). In patients with a definite improvement (recanalization), these regions are salvaged and not infarcted in follow-up CCT. Kwon et al (2004) have reported on stroke patients undergoing intravenous thrombolysis after MR-angiography (MRA) proven MCA or internal carotid artery (ICA) occlusion, where follow-up MRA and NIHSS score dichotomized patients with and without recanalization. NIHSS score increased (12 to 13) in those without recanalization, whereas it dropped (12 to 5) in patients with recanalization.
The limitations of the method are obvious: there is only one plane of insonation. Patients need to have a temporal bone window. So far, only infarctions in the MCA territory are assessable. However, with the bilateral approach, all of the affected MCA territory is displayed in one examination (cortical structures are missed by the unilateral approach). The main potential indication for the method could be the depiction of larger perfusion deficits in the MCA territory. Since approximately over 95% of larger MCA infarctions should be located in the chosen plane, the method should be strong enough to provide satisfying diagnostic impact. However, this should be tested in future studies with correlation to perfusion- and diffusion-weighted MRI or perfusion CCT.
With the improvement of parametric imaging, a single image may help to identify perfusion deficits at a glance. Once MRI- or CT-based perfusion imaging is not applicable, this may be essential for therapeutical decision finding (e.g., acute stroke between 3 and 6 h; Schellinger et al, 2003). Our method is potentially widely distributable and also easily applicable for follow-up examinations as a bed-side test, since the study was performed on a standard machine with standard settings. Once technological development results in transportable high-end ultrasound machines, the examination can be performed in the ambulance with the potential of preclinical decision finding on the adequate therapy of acute stroke.
