Abstract
Purpose
This study aimed to evaluate the diagnostic success of percutaneous biopsies performed for suspected musculoskeletal tumors and to identify clinical, anatomical, and procedural factors associated with the need for repeat biopsy (re-biopsy). The secondary objective was to assess the impact of multidisciplinary team (MDT) evaluation on reducing unnecessary re-biopsy procedures.
Methods
A total of 565 patients (331 bone, 234 soft tissue lesions) who underwent percutaneous biopsy for suspected musculoskeletal tumors between 2020 and 2024 were retrospectively analyzed. Diagnostic success was defined as a biopsy yielding a definitive histopathological diagnosis sufficient to guide treatment planning without additional tissue sampling. Predictive factors including lesion size, depth, location, and performing specialty were evaluated using univariate and multivariate logistic regression analyses.
Results
The initial diagnostic yield was 65.5%. Multivariate analysis revealed that lesion width <2 cm (OR = 2.01; 95% CI, 1.19–3.39; p = 0.009) and biopsy performed by an interventional radiologist (compared to an orthopedic surgeon) (OR = 5.25; 95% CI, 2.83–9.74; p < 0.001) were independent predictors of re-biopsy recommendation. Among 72 patients recommended for re-biopsy, MDT evaluation averted the need for a second procedure in 38 cases (52.8%).
Conclusion
Smaller lesion size (<2 cm) and the medical specialty performing the biopsy were independent predictors of re-biopsy recommendation, with the latter likely reflecting systematic differences in case complexity and procedural environment rather than operator skill. Structured MDT evaluation significantly reduced unnecessary repeat procedures.
Keywords
Introduction
Musculoskeletal tumors encompass a broad spectrum of benign and malignant lesions originating from bone and soft tissue. 1 Accurate histopathological diagnosis is critical for effective treatment planning and optimal clinical outcomes. 2 The success of biopsy procedures, which remain the gold standard for tissue diagnosis, depends on a complex interplay of clinical, anatomical, and procedural factors. 3
In some cases, an initial biopsy may not provide a definitive diagnosis due to non-representative sampling or interpretive difficulties, necessitating a repeat biopsy (re-biopsy). Such situations delay treatment, increase patient anxiety, and place an additional burden on healthcare resources.4,5 Although several studies have reported that lesion size, depth, and anatomical site influence diagnostic yield, the independent contribution of each factor remains inadequately characterized by robust multivariate models. 6
Both interventional radiologists and orthopedic surgeons play active roles in performing biopsy procedures. For bone biopsies, the Jamshidi trephine needle (commonly 8–14 gauge) remains the standard instrument for obtaining core specimens, while for soft tissue lesions, Tru-cut-type cutting needles (commonly 12–18 gauge) are preferred.2,3,7,8 Institutional workflows often influence which specialty performs the biopsy, with differences in procedural setting, sedation protocols, imaging guidance modality, and the number of cores obtained potentially contributing to variation in diagnostic yield.9–11
Despite these recognized procedural differences, few studies have used multivariate analysis to determine whether the performing specialty is an independent predictor of diagnostic failure after controlling for lesion-related confounders. Furthermore, the role of multidisciplinary team (MDT) evaluation in managing non-diagnostic biopsies has received limited attention in the orthopedic oncology literature.12–14
Therefore, this study was designed to determine the rates of diagnostic failure and re-biopsy recommendations in a large single-center cohort, to use multivariate analysis to identify independent clinical and procedural predictors for re-biopsy, and to measure the impact of a multidisciplinary tumor board on the final management of non-diagnostic cases.
Methods
Study design and ethical approval
This study was designed as a single-center retrospective cohort and approved by the local ethics committee (Approval No: 2024-126). Due to the retrospective nature of the analysis, the requirement to obtain informed consent from patients was waived. This study was conducted in accordance with the Declaration of Helsinki and reported following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Patient population
A total of 741 patients who underwent their first percutaneous biopsy for suspected musculoskeletal tumors at our tertiary referral hospital between June 2020 and January 2024 were evaluated. After applying the exclusion criteria, 565 patients were included in the final analysis. Of these, 266 (47.1%) had biopsies performed by interventional radiology (IR) and 299 (52.9%) by orthopedic oncology (OO). The study cohort comprised 331 bone and 234 soft tissue lesions (Figure 1). Biopsy outcomes and current practice flowchart. The flowchart represents current practice showing actual study results (n = 565 patients). Initial diagnostic yield was 370/565 (65.5%), with 123 non-specific results and 72 re-biopsy recommendations. Interventional radiology biopsies had a significantly higher re-biopsy recommendation rate (21.8%) compared to orthopedic oncology (4.7%) (p < 0.001). MDT evaluation averted re-biopsy in 38/72 (52.8%) cases.
Patients were eligible if they had a clinically or radiologically suspected bone or soft tissue tumor, if this was their first percutaneous biopsy, and if their clinical and radiologic data were complete. Patients were excluded if they had incomplete demographic, clinical, or radiologic information, if their biopsy had been performed at an outside institution, or if the biopsy did not involve imaging guidance.
Of the 176 excluded patients: 15 had incomplete clinical or radiologic data, 6 had biopsies performed at an outside institution, 64 had superficial lesions biopsied without imaging guidance, 53 had axial skeletal involvement outside the sacrum, and 38 had soft tissue biopsies surrounding the chest or abdominal wall.
Data collection and variable definitions
Independent variables included patient demographics (age, sex), lesion characteristics (type [bone vs soft tissue], anatomical region [upper extremity, lower extremity, pelvis], size [length and width measured in mm on pre-biopsy MRI/CT], and depth [suprafascial/subfascial for soft tissue; cortical/medullary for bone]), and procedural factors (performing specialty [IR vs OO], needle type and gauge, number of cores obtained, imaging guidance modality, and sedation protocol). All data were extracted from institutional electronic medical records and the Picture Archiving and Communication System (PACS) by two investigators independently, with discrepancies resolved by consensus.
Biopsy technique
Biopsy procedural protocols by performing specialty.

CT, computed tomography; IR, interventional radiology; IV, intravenous; OO, orthopedic oncology.
For soft tissue lesions, both specialties used Tru-cut-type cutting needles: IR used 11–16 gauge needles under ultrasound guidance with local anesthesia only, while OO used 11–14 gauge needles under ultrasound guidance with sedoanalgesia. Tru-cut needles are the standard instrument for soft tissue core biopsy, with 18 gauge generally considered sufficient for most musculoskeletal soft tissue lesions, while 11–16 gauge needles may provide improved specimen quality in heterogeneous or partially calcified tumors.3 Notably, IR typically obtained a single core specimen per bone biopsy, whereas OO routinely obtained 3–6 cores. For soft tissue biopsies, IR obtained 3–5 cores while OO obtained 3–6 cores; additionally, OO consistently used larger gauge needles (11–13G vs 11–16G). For bone biopsies, IR used local anesthesia supplemented by intravenous sedation (50 µg fentanyl), whereas for soft tissue biopsies, IR used local anesthesia only (10 mL 2% prilocaine).
Outcome measures
The primary outcome was whether the initial percutaneous biopsy was diagnostically successful, defined as yielding a definitive histopathological diagnosis that enabled treatment planning without additional tissue sampling. This determination was made by the treating orthopedic oncologist after reviewing the pathology report in the clinical context, and it represented the initial clinical assessment prior to and independent of subsequent MDT review. The primary outcome measure was the result of the initial percutaneous biopsy, classified into three categories: (1) Diagnostic (n = 370, 65.5%): a definitive histopathological diagnosis was established enabling treatment planning without additional tissue sampling; (2) Non-specific (n = 123, 21.8%): pathology revealed non-specific reactive, inflammatory, or necrotic tissue without a definitive diagnosis; and (3) Re-biopsy recommended (n = 72, 12.7%): the treating orthopedic oncologist determined that the available pathological material was insufficient for reliable treatment planning, and repeat tissue sampling was formally recommended. The secondary outcome was whether MDT evaluation successfully resolved the diagnostic uncertainty and averted the need for a second invasive procedure.
Follow-up and multidisciplinary team evaluation
Median follow-up duration was 14 months (interquartile range, 8–24 months). All cases with non-diagnostic or inconsistent biopsy results were evaluated by a weekly orthopedic oncology council consisting of an orthopedic oncologist, radiologist, pathologist, and medical oncologist. The final decision regarding the need for re-biopsy was determined through MDT consensus rather than by any single clinician.
Statistical analysis
Continuous variables were summarized as mean ± standard deviation (SD) or median and interquartile range (IQR) after assessing distributions using the Shapiro-Wilk test. Categorical variables were reported as numbers and percentages. For univariate analysis, chi-square or Fisher’s exact tests were used for categorical variables, and independent samples t-tests or Mann-Whitney U tests were used for continuous variables, as appropriate. All statistical analyses were performed using IBM SPSS Statistics, version 26.0 (IBM Corp., Armonk, NY, USA).
The following variables were included in the univariate analysis: age, sex, lesion type (bone vs soft tissue), anatomical region (upper extremity, lower extremity, pelvis), lesion length (mm), lesion width (mm), lesion depth (suprafascial/cortical vs subfascial/medullary), and performing specialty (IR vs OO). The complete univariate analysis results are provided in Supplemental Table S1. A multivariate binary logistic regression model was constructed to determine independent predictors of re-biopsy recommendation. Variables with p < 0.20 in univariate analysis were entered into the model using forward stepwise (likelihood ratio) selection with entry criterion p < 0.05 and removal criterion p > 0.10. The final model included lesion width (dichotomized as < 2 cm vs >= 2 cm), lesion type (bone vs soft tissue), lesion depth, and performing specialty (IR vs OO). Model fit was assessed using the Hosmer-Lemeshow test, and discriminative ability was evaluated by the area under the receiver operating characteristic (ROC) curve. Procedural variables such as number of cores obtained, needle gauge, and sedation protocol were not included in the multivariate model because these parameters were inherently determined by the performing specialty rather than varying independently across individual cases, and their inclusion would have introduced severe multicollinearity with the specialty variable. Results were expressed as adjusted odds ratios (OR) with 95% confidence intervals (CI).
This study is a retrospective analysis of all eligible cases during the defined period. No formal a priori power analysis was performed. However, post hoc sensitivity analysis demonstrated that with a sample size of 565 and a re-biopsy recommendation rate of 12.7%, the study had over 80% power to detect an odds ratio of 2.0 or greater at alpha = 0.05. There were no missing data for the primary outcome or the independent variables included in the regression model; therefore, no imputation methods were required.
Results
Cohort demographics and lesion characteristics
Basic demographic and lesion characteristics of the study population (N = 565).

Values are presented as mean ± SD or n (%). SD, standard deviation.
Initial biopsy results and departmental comparison
Comparison of initial biopsy results according to the department performing the procedure.

Values are presented as n (%). IR, interventional radiology; OO, orthopedic oncology.
Chi-square test, p < 0.001 for overall comparison between departments.
Univariate analysis
Univariate analysis of factors associated with re-biopsy recommendation.
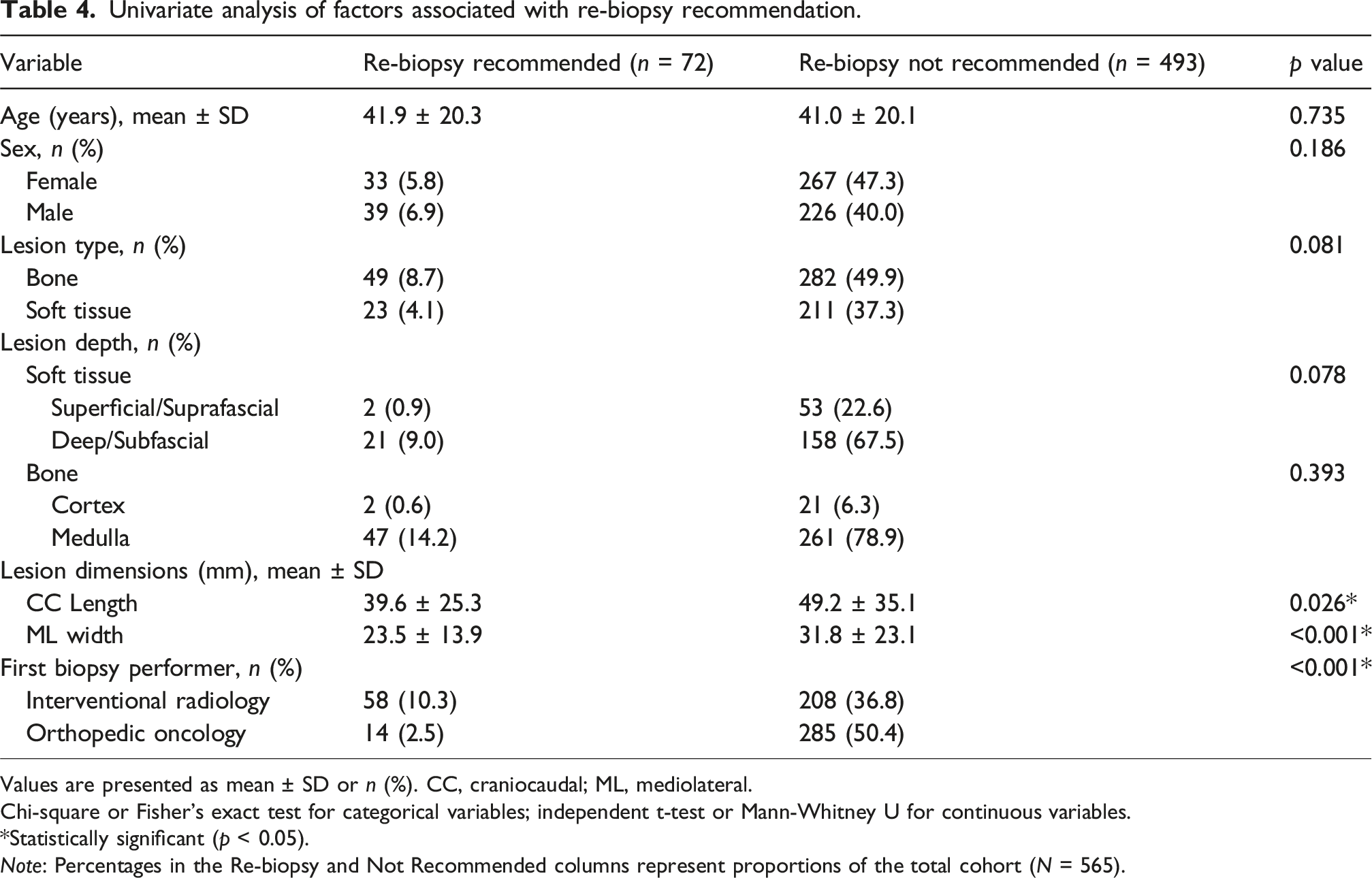
Values are presented as mean ± SD or n (%). CC, craniocaudal; ML, mediolateral.
Chi-square or Fisher’s exact test for categorical variables; independent t-test or Mann-Whitney U for continuous variables.
*Statistically significant (p < 0.05).
Note: Percentages in the Re-biopsy and Not Recommended columns represent proportions of the total cohort (N = 565).
Independent predictors of re-biopsy: Multivariate analysis
Multivariate logistic regression analysis of independent predictors for re-biopsy recommendation.

B, regression coefficient; S.E., standard error; df, degrees of freedom; OR, odds ratio; CI, confidence interval; IR, interventional radiology.
Model: forward stepwise logistic regression (likelihood ratio). Entry criterion p < 0.05, removal criterion p > 0.10.
*Statistically significant (p < 0.05).
Resolution of cases with re-biopsy recommendations
Among the 72 patients recommended for re-biopsy, 38 (52.8%) were managed successfully without a second invasive procedure following MDT review. The MDT avoided re-biopsy through three principal mechanisms: radiologic-pathologic correlation (n = 22, 57.9%), whereby integrated review of imaging findings with pathological features established a concordant working diagnosis; clinical context integration (n = 10, 26.3%), in which clinical behavior, serial imaging, or laboratory data provided sufficient diagnostic confidence; and decision for direct surgical resection (n = 6, 15.8%), where the MDT determined that definitive surgical excision served as both a diagnostic and therapeutic intervention, rendering a repeat biopsy unnecessary. The remaining 34 patients (47.2%) underwent repeat biopsy. This 52.8% re-biopsy avoidance rate represents a clinically meaningful contribution of structured MDT evaluation and highlights the value of a multidisciplinary diagnostic safety net in musculoskeletal oncology.
Discussion
This large retrospective study identified two independent predictors of initial diagnostic failure and re-biopsy recommendation in musculoskeletal tumor biopsies: lesion width less than 2 cm and biopsy performed by interventional radiology. Most importantly, we demonstrated that systematic MDT evaluation averted the need for repeat biopsy in over half of recommended cases, underscoring the role of structured multidisciplinary review as a diagnostic safety net.
Diagnostic yield and comparison with literature
The 65.5% initial diagnostic yield in our study is substantially lower than reference values in the literature, which typically report 85–98% for musculoskeletal biopsies.15–18 Our re-biopsy recommendation rate of 12.7% approaches the upper limit of the 2–13% non-diagnostic rate reported in the literature.14,19 This discrepancy is partially attributable to our inclusive study design that captured both bone and soft tissue tumors across all anatomical regions. Furthermore, our conservative definition of diagnostic success required a pathological diagnosis sufficient to enable definitive treatment planning, which is a more stringent criterion than many studies that include descriptive or suggestive pathological reports as diagnostic.15,20 Additionally, our cohort included a high proportion of small lesions (mean mediolateral width in the re-biopsy group: 23.5 mm, with smaller lesions significantly overrepresented), which are known to have lower diagnostic yield.
Lesion size as an independent predictor
Our finding that lesion width <2 cm is a strong predictor of failure (OR, 2.01; 95% CI, 1.19–3.39; p = 0.009) is consistent with previous reports.19–21 Small targets increase the risk of sampling error, with the needle potentially sampling surrounding reactive tissue or necrotic areas rather than viable diagnostic tumor.22,23 This finding has direct clinical implications: for lesions measuring less than 2 cm, pre-procedural MDT discussion should guide the optimal biopsy approach, including the consideration of operating room-based procedures with multiple sampling passes and appropriate sedation to allow precise needle placement.
Performing specialty as an independent predictor
The finding that biopsies performed by IR were independently associated with a significantly higher rate of re-biopsy recommendation (OR 5.25; 95% CI, 2.83–9.74; p < 0.001) requires nuanced interpretation. This result should not be construed as an indication that one specialty performs biopsies more competently than the other. Rather, multiple systematic and institutional factors likely contribute to this observed difference.
First, referral patterns and case selection differed between specialties at our institution. IR was typically assigned technically challenging cases, including smaller lesions, those in anatomically difficult locations, or lesions adjacent to critical neurovascular structures. Although we excluded certain anatomically complex regions (axial skeleton, chest and abdominal wall) and adjusted for measurable confounders in the multivariate model, residual selection bias inherent to the retrospective design cannot be entirely eliminated.
Second, procedural environment and sedation protocols differed substantially. OO performed biopsies in the operating room under sedoanalgesia, which provides a controlled environment with the ability to reposition the needle, take multiple passes, and obtain larger core specimens. In contrast, IR biopsies were performed in the outpatient radiology suite under local anesthesia (supplemented by 50 µg intravenous fentanyl for bone biopsies only), where patient discomfort may limit the number and quality of samples obtained.
Third, the number of cores and needle gauge differed between specialties. For bone biopsies, OO routinely obtained 3–6 cores using 11–13 gauge Jamshidi needles, whereas IR typically obtained a single core using 11–14 gauge needles. For soft tissue lesions, OO used larger gauge needles (11–13G Tru-cut) compared to IR (14–16G Tru-cut). The relationship between the number of cores, needle gauge, and diagnostic yield has been well documented; larger specimens and multiple passes increase the probability of capturing representative diagnostic tissue.3,12,20,24,25
Fourth, the choice of imaging guidance differed: OO used fluoroscopy for bone lesions, whereas IR used CT guidance. While CT provides superior cross-sectional visualization, fluoroscopy allows real-time, dynamic needle manipulation, which may facilitate more targeted sampling in certain clinical scenarios. For soft tissue biopsies, both specialties used ultrasound guidance.
Fifth, it is important to note that IR operates without an on-site pathology assessment (rapid on-site evaluation, ROSE) at our institution. Immediate specimen adequacy assessment could potentially reduce non-diagnostic sampling rates but was not available in the IR workflow during the study period.
The existing literature on specialty-related differences in biopsy yield presents mixed findings. Kubo et al. 9 performed a meta-analysis reporting comparable diagnostic accuracy between radiologists and orthopedic surgeons for soft tissue and bone sarcomas when case complexity was controlled. Cengiz et al. 10 similarly found no significant difference in diagnostic accuracy between specialties in a single-center analysis. Rozenberg et al. 11 reported that IR-performed biopsies can achieve diagnostic yields equivalent to surgical biopsies with appropriate case selection and technique. These studies, however, generally did not account for the procedural variables we have identified, including the number of cores, sedation environment, and needle gauge, which may represent modifiable factors that could improve diagnostic yield across both specialties.
We acknowledge that the retrospective nature of our study precludes definitive conclusions regarding causality. A prospective investigation controlling for case complexity, number of cores, needle gauge, sedation protocol, and imaging modality would be necessary to determine whether the observed specialty-related difference is attributable to specific modifiable procedural factors rather than confounding by indication.
Role of multidisciplinary team evaluation
Perhaps the most clinically meaningful finding of our study is the role of the MDT as a ‘diagnostic safety net.’ In our series, MDT evaluation established a reliable working diagnosis and treatment plan for 38 of 72 patients (52.8%) initially recommended for re-biopsy. This was achieved by integrating radiologic-pathologic correlation (57.9%), clinical context (26.3%), and direct surgical excision planning (15.8%). This finding aligns with the growing emphasis on multidisciplinary management in musculoskeletal oncology.19,26–29 The 52.8% re-biopsy avoidance rate supports the recommendation that all non-diagnostic musculoskeletal tumor biopsies should undergo structured MDT review before a repeat invasive procedure is pursued.
Clinical implications
Our results suggest the need for a risk-stratified approach to biopsy planning. For high-risk lesions (particularly those <2 cm in width), pre-procedural MDT discussion should guide the optimal biopsy approach, potentially directing such cases toward operating room-based procedures with multiple sampling passes and appropriate needle selection (Jamshidi for bone [8–14 gauge]; Tru-cut for soft tissue [12–18 gauge]).
30
Furthermore, the modifiable procedural differences identified between specialties, particularly the number of cores obtained and needle gauge, suggest opportunities for protocol optimization. Standardizing biopsy protocols to include multiple core specimens and larger gauge needles, regardless of the performing specialty, may improve diagnostic yield. All non-diagnostic cases should undergo structured MDT evaluation before re-biopsy is performed.
31
The proposed risk-stratified biopsy planning algorithm is presented in Figure 2. Proposed risk-stratified biopsy planning algorithm for musculoskeletal tumors. High-risk lesions (<2 cm in width) should undergo pre-procedural MDT discussion and consideration of operating room-based biopsy with extensive sampling using appropriate needle types (Jamshidi for bone [8–14 gauge]; Tru-cut for soft tissue [12–18 gauge]). All non-diagnostic cases should undergo structured MDT evaluation before re-biopsy is performed.
Strengths and limitations
The strengths of this study include its large cohort size (n = 565), inclusion of both bone and soft tissue tumors, and direct, statistically adjusted comparison between two main specialties within a single institution. The multivariate model demonstrated acceptable discriminative ability (AUC = 0.78), and all cases were reviewed through a standardized MDT process. However, important limitations must be acknowledged. The retrospective design introduces inherent selection bias, particularly regarding the non-random assignment of cases to IR and OO. Because the performing specialty was determined by institutional referral patterns rather than randomization, the observed difference in re-biopsy rates may partly reflect systematic differences in case complexity that were not fully captured by the available covariates. The single-center design may limit generalizability. Additionally, we were unable to control for individual operator experience, which may vary within each specialty. The lack of rapid on-site pathological evaluation (ROSE) in the IR setting represents a potentially modifiable factor that was not analyzed separately. The use of forward stepwise variable selection, while pragmatic, may be sensitive to the order of variable entry and can yield unstable models in smaller samples; however, the consistency of our findings with existing literature supports the robustness of the identified predictors. The lack of a formal a priori power analysis is a further methodological limitation, although the post hoc analysis demonstrated adequate power for the observed effect sizes. Future multicenter, prospective studies with standardized biopsy protocols across specialties are needed to validate these findings and to determine whether equalizing procedural parameters (number of cores, needle gauge, sedation) would eliminate the observed specialty-related difference.
Conclusion
Smaller lesion size (<2 cm) and the medical specialty performing the biopsy were independent predictors of re-biopsy recommendation in musculoskeletal tumor biopsies. The specialty-related difference likely reflects systematic variations in procedural environment, number of cores obtained, and needle gauge rather than differences in operator competence, warranting prospective investigation of modifiable biopsy parameters. Structured MDT evaluation averted unnecessary repeat procedures in over half of recommended cases, supporting its routine integration into musculoskeletal tumor management pathways.
Supplemental material
Supplemental material - Predictors of re-biopsy in percutaneous musculoskeletal tumor biopsies: A single-center retrospective cohort study
Supplemental material for Predictors of re-biopsy in percutaneous musculoskeletal tumor biopsies: A single-center retrospective cohort study by Ali Yücesan, Yavuz Arıkan, Fatih Mert Doğukan, İlhan Nahit Yılmaz, Serdar Toy, Tamer Atan, Devrim Özer in Journal of Orthopaedic Surgery
Footnotes
Ethical considerations
This study was approved by the Başakşehir Çam and Sakura City Hospital Ethics Committee (Approval No: 2024-126).
Consent to participate
The requirement for informed consent was waived due to the retrospective design.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Location of work
The investigation was performed at Başakşehir Çam and Sakura City Hospital, Istanbul, Turkey.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
