Abstract
Background
Wrist and palmar bone defects are prevalent yet challenging conditions in orthopedic clinical practice. 1 Trauma, bone tumors, chronic diseases, and surgical resection can all lead to bone defects in the wrist. 2 The mainstream approaches for reconstructing normal anatomy and restoring function are wrist arthrodesis or autogenous bone flap grafts. 3 However, these methods cannot effectively reconstruct anatomical structures, often resulting in deformities that impact postoperative function and quality of life. 3D printing customized implants appear to be a promising approach worthy of in-depth exploration. By reconstructing the same anatomical morphology, better postoperative outcomes can be achieved. The wrist bones are numerous and diverse in shape, and the absence of any part can affect wrist joint function. 4 The lack of conventional implants in the market also makes the existence of customized implants essential.
Nevertheless, there are currently few cases and literature on the application of 3D printed prostheses in the treatment of wrist bone defects. This situation restricts the selection of materials and the optimization of manufacturing processes and techniques. Orthopedic surgeons often use various materials to reconstruct wrist bone defects, each with its own unique advantages and limitations. Han et al. reported in the medical record series that the use of 3D printed Ti6Al4V alloy (TC4) addressed severe and complex wrist joint bone defects. 1 Metal implants provide unmatched biomechanical support, making them ideal for load-bearing applications. 5 Orthopedic implants should be custom-made according to varying bone defects to optimize host-implant matching. 6 Meanwhile, the material must possess excellent osteogenic potential to guarantee long-term prosthesis survival. 7 Researchers are diligently exploring and developing innovative implant materials that exhibit ideal osteogenic potential, exceptional biocompatibility and robust mechanical strength. 8 The pursuit of the ultimate bone repair material continues, aiming to optimize patient outcomes and reduce the complications associated with current treatment options. 9 Indeed, three-dimensional (3D) printing technology has revolutionized the processing and fabrication of a diverse array of biomaterials, enabling the creation of implants tailored to meet an extensive range of specific needs.10,11 Titanium (Ti) and its alloys have garnered immense approval in bone defect repair, primarily attributed to its robust mechanical characteristics.12,13 Ti alloy exhibits seamless integration with adjacent tissues, offering patients a resilient and dependable means of restoring bone functionality. 14 Numerous clinical studies supported the potential of 3D printed titanium alloys for the reconstruction of bone defects. 15 However, despite the myriad advantages of Ti alloys, those studies have also unveiled some inherent shortcomings. A notable disadvantage lies in their relatively poor bone integration capabilities, which can compromise the long-term stability of the implant,16,17 resulting in prosthesis loosening and potentially leading to revision surgery—a scenario that can be devastating in complex cases. Tantalum metal (Ta) stands out as a “biophilic” material renowned for its superior biocompatibility and osteoinductive properties.18,19 We have successfully repaired critical-size bone defects resulting from tumor resection in areas such as the pelvis and wrist joint by using the personalized 3D-printed porous Ta implants,19,20 which precisely matched the irregular shape of the defects. The porous structure enables those implants to better conform to the bone tissue, thereby preventing stress shielding. 21 The results has yielded favorable long-term clinical outcomes. Unfortunately, the scarcity of natural resources results in limited availability of Ta, thereby driving up production costs and rendering it economically unfeasible for the treatment of extensive bone defects. 22 This restriction not only curtails patients’ access to Ta implants but also poses significant hurdles for researchers and manufacturers in innovating and broadening the application of this promising material within orthopedic surgery.
To address these limitations, ongoing research endeavors are concentrated on developing alternative materials or refining surface modification techniques that can replicate tantalum’s biocompatibility and osteoinductive properties while maintaining cost-effectiveness. Our team innovatively applies magnetron sputtering (MSP) technology to prepare nano thin amorphous Ta coatings on titanium alloy (Ti - 6Al - 4V, TC4) substrate. 23 MSP is a form of Physical Vapor Deposition (PVD), which involves introducing a magnetic field on the surface of the target cathode, utilizing the magnetic field’s confinement of charged particles to enhance plasma density and thereby increase the sputtering rate. 24 MSP can be applied to various substrate materials, including metals, ceramics, glass, polymers, and more. This versatility renders the technique exceptionally adaptable across various fields, as the sputtered film boasts a dense, smooth texture and remarkable environmental stability, attributed to the energy activation during the film preparation process. 25 Additionally, MSP enables the preparation of coatings with varying thicknesses, ranging from just a few nanometers to several hundred nanometers or even thicker. 26 Shanaghi et al. deposited TaN and Ta/TaN nanostructured coatings on NiTi alloys, 27 demonstrating good osteointegration and osteogenesis. While Alias et al. also suggested that the silver-tantalum-oxide-based nanostructured thin film prepared on stainless steel 316L via magnetron sputtering exhibits excellent stability and antibacterial properties. 28 Furthermore, nano-amorphous Ta coatings produced via MSP typically exhibit excellent adhesion. 29 During the sputtering process, strong chemical bonds are formed between the target atoms and the substrate, thereby enhancing the coating’s adhesion. 30 The coatings produced by magnetron sputtering demonstrate remarkable uniformity. 31
We hypothesized that this novel TC4-based Ta-coated implant seamlessly combines the inherent strength of TC4 with the excellent properties of Ta. The application of Ta coating not only endows with better osteogenesis even than pure Ta or porous TC4 prostheses. Meanwhile, compared with pure Ta implants, TC4-based Ta-coated implant as the substrate also has the advantage of lightweight effect as the density of Ta is about four times that of Ti under the same temperature conditions. 32 TC4-based Ta-coated implant can ensure good bone integration performance, while also reducing the burden on surrounding tissues and potentially improving patient comfort and recovery rate. 33 In addition, the economic benefits of using TC4-based Ta-coating are enormous as the raw material price of Ta is approximately 10 to 15 times that of TC4.
Our previous research effectively demonstrated the safety and excellent bone integration ability of this nano-thin tantalum coating when applied to 3D printed porous TC4 through a series of in vivo and in vitro experiments.
22
Based on these basic findings, this study presents a series of clinical cases, in which TC4-based Ta-coated implant were used to treat complex wrist bone defects (See Figure 1). Schematic diagram of preparing a tantalum (Ta) coating on a TC4 substrate. (A) Schematic diagram illustrating the principle of magnetron sputtering of Ta atoms onto a 3D-printed TC4 substrate. (B) Physical images of a porous TC4 prosthesis substrate and the same substrate after being coated with Ta (taking patient 2’s lunate bone defect prosthesis as an example): (1, 3, 5) before coating; (2, 4, 6) after coating: the surface of the prosthesis appears rougher after coating, and the color of the tantalum film differs from that of the substrate.
Method
Patient selection
This study prospectively included a series of three cases of severe and complex wrist bone defects caused by different reasons. All patients underwent surgical reconstruction using 3D printed TC4-based Ta-coated prostheses.
Inclusion criteria
(1) 18 to 65 years old; (2) Clearly identified bone defects resulting from trauma, bone tumors, etc., with suitability for repair using personalized titanium-based tantalum-coated prosthesis assessed through imaging methods such as X-rays, CT scans, or MRI; (3) in good overall health and capable of enduring the surgical procedure and rehabilitation process; (4) free from severe chronic conditions like cardiovascular and cerebrovascular diseases, liver and kidney dysfunction, etc. (5) thoroughly informed about the treatment plan, associated risks, and expected outcomes, and have provided written informed consent.
Exclusion criteria
(1) presence of allergies to Ti or Ta; (2) serious immune system diseases, or systemic and local infectious diseases; (3) combining mental illnesses, psychological issues, or other conditions that may compromise treatment efficacy and patient adherence; (4)Unable to attend regular follow-up as scheduled.
From April 2023 to December 2024, two males and one females who met the inclusion criteria were enrolled in our survey. Each patient is numbered for a better elaboration.
Data acquisition and model building
All patients underwent high-resolution thin-layer computed tomography (CT) scans, performed with a layer spacing of 1.0 mm, ensuring the capture of meticulous details. The scanning area comprehensively covers the ipsilateral and contralateral limbs of the bone defect, thereby providing a comprehensive view of the relevant anatomical structures.
The raw data was meticulously collected and securely stored in the Medical Digital Imaging and Communication (DICOM) format. Subsequently, the DICOM-format data underwent sophisticated reconstruction using the Mimics Innovation Suite 19.0 software (Materialise, Leuven, Belgium). Within the Mimics software, various thresholds were meticulously adjusted to achieve precise segmentation of the STereoLithography (STL) data. The segmented STL data was then seamlessly imported into the advanced Unigraphics NX (UG) software (Siemens PLM Software, Texas, USA). Utilize the 3D modeling of UG software for preoperative surgical planning and prosthesis design. Prior to initiating the design process, designers must consult with the attending surgeon and tailor the prosthesis according to the surgical plan. During the designing, it is crucial to consider factors such as mechanical stress, the prosthesis’ shape and size, the dimensions and orientation of screw holes, and to perform mechanical testing, clinical comparisons, and any other pertinent tasks. Depending on the prosthesis’ intended usage, incorporate porous structures with varying parameters and conduct topology optimization design. Finally, print a solid model of the prosthesis to validate the design scheme and facilitate preoperative preparations.
Preparation of titanium alloy (TC4) base
Select high-quality commercial spherical TC4 (Ti-6Al-4V) powder, featuring an average diameter of 60 μm, as the foundational raw material. Utilize the advanced Electron Beam Selective Melting (EB-PBF) system (Sailong Y150 model operated at 60 kV) to print the TC4 powder into intricate 3D porous cylindrical supports (6 mm diameter × 6 mm height) and solid disks (6 mm diameter × 1 mm thickness). The stent design incorporates a dodecahedral crystal cell structure, characterized by a pore size of 600 μm and a porosity level of 80%. Upon completion of the printing process, employ high-pressure air to thoroughly remove any residual TC4 powder. Subsequently, conduct ultrasonic cleaning using anhydrous alcohol followed by ultrapure water for a duration of 5 min each, ensuring the pristine cleanliness of the substrate surface and preparing it aptly for the subsequent tantalum coating deposition.
Preparation of the nano thin tantalum coating
Deposit a tantalum coating onto the TC4 substrate utilizing a magnetron sputtering machine sourced from Shenyang Scientific Instrument Co., Ltd, model JGP-560B. Prior to deposition, initiate a pre-sputtering process lasting 1 min under an argon pressure of 1 Pa and a sputtering power of 120 W to eliminate oxides and impurities from the tantalum target surface. Next, select experimental parameters based on the equipment’s rated power and specifications, namely sputtering powers of 300 W and 250 W, along with argon pressures of 0.2 Pa, 0.4 Pa, and 0.6 Pa. To ascertain the optimal sputtering duration for achieving nanoscale tantalum coatings, measure the sputtering rate on a silicon wafer substrate using a step meter (due to the TC4 3D printed substrate’s high surface roughness). The experimental results revealed a deposition rate of approximately 0.6 nm per second for the Ta coating, leading to a final sputtering time of 30 s for depositing the nano-thin tantalum layer (Figure 1(A)). To guarantee comprehensive tantalum coating coverage within the porous structure, rotate the sample four times from various directions (up, down, front, back) during sputtering, thereby enabling a greater number of tantalum atoms to deposit within the pores (Figure 1(B)).
Surgical procedures
Anesthesia was selected according to the surgical procedure. Choose the appropriate position for the patient based on the location and surgical requirements. Select an incision with sufficient exposure and minimal damage at the site of the lesion, carefully separate the soft tissue through the incision, and expose the bone defect area. During the surgery, it is important to carefully separate and protect the surrounding blood vessels, nerves, and soft tissues to avoid unnecessary damage. Match the prosthesis with the bone defect area to ensure that the prosthesis can be stably fixed at the bone defect site. Use preoperative personalized 3D osteotomy guide plate for osteotomy to ensure correct implantation and fixation of the prosthesis. Careful hemostasis can reduce postoperative bleeding and prevent hematoma formation. After completing the implantation of the prosthesis, suture the incision layer by layer. Wrap the wound with sterile dressings to reduce the risk of infection. During the surgical process, strict adherence to the principle of aseptic operation is necessary to reduce the risk of infection.
Postoperative management
After surgery, it is necessary to closely monitor the patient’s condition and promptly detect and treat any possible complication. All patients should undergo appropriate rehabilitation training to promote bone healing and functional recovery. Regularly follow-ups were conducted to promptly identify and address potential complications. Weight-bearing and vigorous activity on the surgical limb should be banned for 12 weeks. According to the specific situation of the patient, appropriate adjuvant measures such as medication and physical therapy should be given.
During the follow-up period, regular clinical evaluations and imaging examinations were performed. These evaluations encompassed a comprehensive assessment of the patient’s pain condition using the VAS score, as well as functional assessments utilizing the HHS score for hip joints and the Cooney modification of the Green and O’Brien scores for wrist joints. Imaging examinations primarily focused on observing the prosthesis’ position and stability, the integrity of the tantalum coating, and the restoration of bone defects through X-rays or CT scans.
Result
Baseline statistical data of patients.

The follow-up period ranged from 6 to 12 months, averaging 9 months. As of the latest follow-up, all patients exhibited significant improvements in pain symptoms (assessed using VAS scores) and functional outcomes (as shown in Figure 2). Notably, no patients experienced serious complications such as infection, prosthesis loosening, or vascular and nerve damage. Comparison of VAS scores and Cooney modification of the Green and O’Brien scores of patients before and after surgery, showing significant improvement in postoperative scores compared to preoperative scores. (A) Cooney modification of the Green and O’Brien scores of 3 patients; (B) VAS scores.
Typical case - patient with left wrist scaphoid bone necrosis
A 37-year-old male patient complained of left wrist pain that had lasted for 2 years since a fall. The pain had been worsening and accompanied by mobility restriction for 1 year. Physical examination revealed slight swelling of the left wrist with no bruising, ulceration, or pigmentation. Deep tenderness was noted in the wrist’s carpal area, absent palpable bone friction. Partial restriction of left wrist joint mobility was observed: active and passive palm flexion 0–80°, dorsiflexion 0–60°; hyperextension and overflexion elicited pain. Muscle strength, tension, sensation, movement, and blood supply of the left upper limb were normal.
CT scan demonstrated an old fracture of the left wrist scaphoid bone (Figure 3). The diagnosis was old fracture of the left wrist scaphoid bone with nonunion. Given the patient’s significant symptoms affecting quality of life, surgical intervention was indicated. With informed consent from the patient and his family members, personalized 3D-printed prostheses were deemed ideal for reconstruction, which can preserve wrist joint structure and normal appearance post-scaphoid bone resection. CT plain scan and 3D reconstruction reveal an old fracture of the left wrist scaphoid bone. (a/b/c) CT scans from different planes demonstrate significant bone defects and non-union; (d) the scaphoid bone (marked in red) reconstructed using mimics software clearly shows obvious bone non-union.
Mimics 17.0 software was used to store and analyze CT data in DICOM format. Following 3D remodeling of bilateral palms and wrists, we imported the data into UG software for prosthesis design. Using the healthy right limb as a reference, we simulated and designed a TC4 prosthesis base for the left wrist scaphoid bone (Figure 4(a)). The prosthesis included suture fixation holes (1.0 mm) to enhance stability and functionality. The prosthesis base was fabricated by using TC4 powder according to the preoperative design, incorporating porous structure weight reduction (Figure 4(b)). Then MSP technology was employed to apply a nano-thin amorphous Ta coating to the TC4 base. The surface was polished to achieve a roughness of 0.2–0.4 μm. (Figure 4(c)) Design and manufacturing process of 3D printed titanium-based tantalum-coated implants. (A): (1) Three-dimensional reconstruction of CT scans of the right hand and wrist on the healthy side was performed using Mimics software. (2) The right wrist scaphoid bone was selected separately, and (3) the shape of the prosthesis was designed in UG software. (B): (1) Setting of porous structure parameters for the TC4 substrate, (2) Design of sutured fixation channels, (3)/(4) Final morphology of the prosthesis. (C): Finished product of the wrist scaphoid bone titanium-based tantalum-coated prosthesis. (1) (2) (3): TC4 substrate that has undergone surface polishing treatment without Ta coating; (4) (5) (6): Finished product after coating.
After conducted satisfactory brachial plexus block anesthesia, the patient was positioned supine with a tourniquet applied to the proximal end of the left arm. The surgical site was disinfected, draped, and the tourniquet inflated to 30 kPa. The left upper limb was abducted and pronated on the operating table. Guided by 3D electromagnetic navigation, a curved incision of approximately 4.0 cm was made on the dorsal radial side of the left wrist, centered on the scaphoid bone. The skin, subcutaneous tissue, deep fascia, and retinaculum were incised. The radial nerve branch was carefully protected, and the extensor tendons were retracted to expose the scaphoid bone. Exploration revealed an old fracture in the wrist region of the scaphoid bone with obvious non union, peripheral osteophyte proliferation, and enlarged, sclerotic fracture ends. A protruding styloid process of the radius was noted. A bone saw was used to resect approximately 0.4 cm of the radial styloid process, which was then repaired and shaped using bone forceps. The necrotic scaphoid bone was completely removed from the dorsal, scaphoid, and palmar ligaments under C-arm electromagnetic navigation. A titanium-based tantalum-coated prosthesis was implanted, and its size and position were confirmed to be satisfactory via C-arm fluoroscopy. Preoperatively designed suture fixation holes were used to suture and reconstruct the scaphoid, palmar, and dorsal ligaments, establishing preliminary scaphoid stability. The wrist joint exhibited radial deviation without restriction or compression during movement. The surgical site was irrigated with saline, and the instruments and gauze were counted. The incision was closed in layers, dressed sterilely, compressed with elastic bandages, and the tourniquet was deflated. The intraoperative blood loss was 5 mL.
After the surgery, the patient experienced a swift recovery, with their forearm and wrist immobilized in plaster for a duration of 3 weeks. Antibiotics were administered to ward off infections, while oral and topical painkillers effectively managed any pain symptom. The wound healed remarkably well, prompting the successful removal of stitches after 2 weeks. A postoperative X-ray, conducted 6 months after the surgery, revealed that the prosthesis placed in the left wrist scaphoid bone was in a satisfactory position, with no signs of displacement or loosening (Figure 5). During the most recent follow-up, the patient reported a satisfactory restoration of function in their left wrist, without any notable discomfort. The range of motion for the radial wrist joint and forearm was as follows: dorsiflexion at 40°, palmar flexion at 45°, radial deviation at 25°, and ulnar deviation at 30°. Occasionally, the patient experiences mild pain in their left wrist. Pre or postoperative X-ray images of three patients. (a/b) A postoperative X-ray of patient 3, conducted 6 months after the surgery, revealed that the prosthesis placed in the left wrist scaphoid bone was in a satisfactory position, with no signs of displacement or loosening. (a) AP view; (b) lateral view; (c/d) pre (c) or postoperative (d) X-ray images of patient 2; (e/f) pre (e) or postoperative (f) X-ray images of patient 1.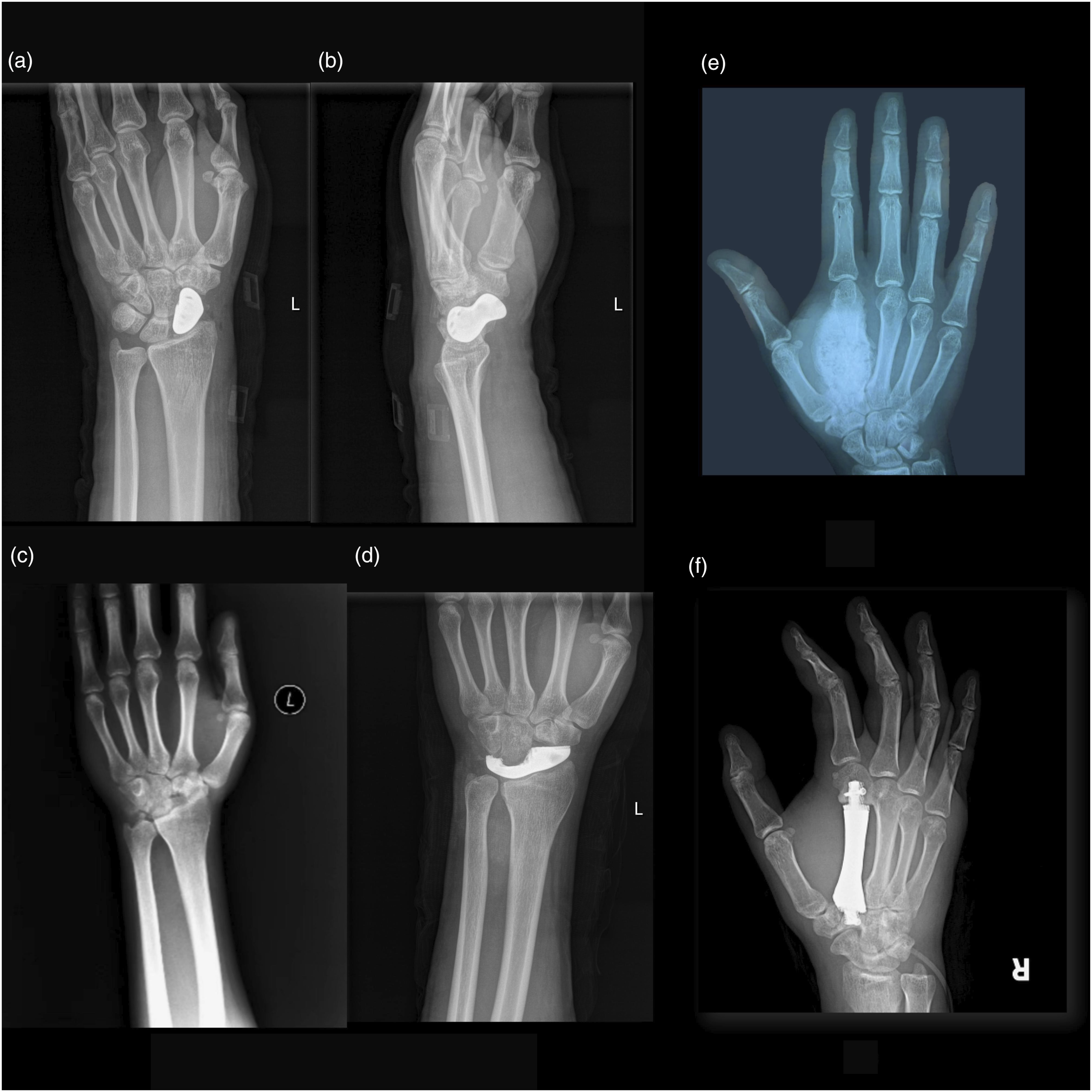
Discussion
Here we presented a sole clinical research on the application of titanium-based nanoscale tantalum-coated prostheses in patients with critical-size bone defects, offering valuable references for future research on additive manufacturing (AM) technology and the clinical applications of Ta coatings. Our outcomes reveal that these customized TC4-based Ta-coated implants, prepared through 3D printing and surface-modified with MSP, exhibit significant improvements in bone integration and clinical outcomes. In the clinical context, this innovation holds significant importance as it offers a viable solution to overcome the limitations of traditional Ta alloys and Ta metal implants, while seamlessly integrating and harnessing the strengths of these two widely used metals. If the manufacturing process can be further optimized, it will bring immense benefits to the vast number of patients suffering from various bone defects.
The main outcomes, including pain symptoms assessed using VAS scores and functional outcomes, demonstrated substantial improvements in all patients. Notably, no serious complications such as infection, prosthesis loosening, or vascular and nerve damage were observed during the follow-up period. These results suggest that the TC4-based Ta-coated prostheses not only enhance bone integration but also ensure patient safety and satisfaction. The adverse reaction rate remained within an acceptable range, further validating the safety and efficacy of this novel approach.
The advantages of nano thin amorphous Ta-coating
We deposited a nano-thin amorphous Ta coating of approximately 20 nm on a 3D printed TC4 substrate using MSP, achieving excellent interface bonding, high coverage, and remarkable biological properties. 22 3D printed TC4 and Ta prostheses have been widely reported for clinical applications in treating different bone defects, although most of them were isolated cases. Zheng et al. reported the use of 3D printed customized TC4 prostheses for treating metacarpal bone defects, 33 which demonstrated more significant improvements in pain and function compared to autologous bone transplantation. Similarly, Cheng et al. employed 3D printed customized titanium alloy prostheses to treat traumatic fibular defects, and the reconstructed distal fibula and lateral malleolus had structures and functions close to normal. 34 Han et al. applied 3D printed customized semi-pelvic titanium alloy prostheses to treat iliac bone tumors and achieved satisfactory clinical efficacy. 35 Zhao et al. used 3D printing-assisted shaping of titanium cages to treat osteomyelitis of the calcaneus with bone and soft tissue defects, attaining good clinical efficacy. 36 As for Ta implants, Ao et al.’s 3D printed customized porous tantalum prosthesis was utilized to treat bone defects caused by total knee replacement revision surgery. 37 During an average follow-up period of 26.3 months, these patients showed significant improvement in pain and function. Li et al. also achieved satisfactory therapeutic effects in the treatment of ilium chondrosarcoma by using 3D printed customized porous tantalum prostheses. 38 However, limitations of those metals have been identified, such as the poor bone integration performance of Ti and the high production cost of Ta. To further enhance the bone integration property of personalized implants, reduce production costs, and expand their clinical indications, our research team deposited a nano-thin amorphous tantalum coating on a 3D printed porous TC4 substrate. In previous in vitro and in vivo studies, we found that Ta coatings exhibited higher cell proliferation and bone growth rates in the early stage of implantation compared to pure Ta and TC4. Amorphous Ta coating may demonstrate higher hardness, superior corrosion resistance, and excellent wear resistance. 39 Furthermore, nano-thin coatings can offer a larger surface area, thereby enhancing the adhesion with the substrate and influencing cellular responses. 40 In vivo experiments, 22 micro-CT analysis revealed that the new bone in the Ta-coated group grew to the center of the scaffold, while only a portion of the TC4 group reached the edge of the scaffold, indicating that the Ta-coated scaffold had a faster bone growth rate in the initial stage than the TC4 group. In addition, the amount of bone tissue in the central region of the nano-Ta-coated scaffold is notably greater than that of pure Ta. This difference may be attributed to the surface oxidation composition, surface morphology, and porous structure of the tantalum coating, which enable proteins or cells to directly interact with three distinct oxide layers. 41 The results of this study showed that patients treated with TC4-based Ta-coated prostheses had significant improvements in pain symptoms and functional outcomes, while no serious complications were reported during follow-up. These findings suggested that our novel prosthesis has the potential to overcome the limitations of previous studies and provide more effective and cost-efficient treatment options for different bone defects. Through process optimization, it is expected to further reduce costs and provide an economically effective solution for repairing bone defects.
The advantages of magnetron sputtering in the fabrication of nano-thin amorphous Ta-coating
Within the realm of bionanomaterials, the research on nano-thin Ta coatings stands as a pivotal direction for orthopedic implants, supported by numerous studies highlighting the extensive application potential. 42 Among various methods, MSP is presently prevalent in the preparation of Ta coatings, encompassing thin film production, surface modification, enhancement of biological activity, antibacterial properties, and other aspects. 43 For instance, Cao et al. 44 utilized thermal synthesis and MSP to deposit fluorinated hydroxyapatite and Ta composite coatings on magnesium alloys, thereby augmenting their biological activity and osteoinductive capabilities. Similarly, the antibacterial performance of Ta coatings merited attention, as evidenced by Elangovan et al., 45 who prepared TaN/Cu nanocomposite coatings demonstrating significant antibacterial activity against Pseudomonas aeruginosa while improving hardness and friction properties. Furthermore, Nasakina et al. 46 investigated the co-deposition behavior of Ta and Ti by MSP, laying a theoretical foundation for the development of novel composite materials. Inspired by those remarkable technological innovations, we tried to verify the feasibility and preeminence of MSP technique in fabricating nanoscale Ta coatings on porous TC4 surfaces.
Our studies robustly confirmed the advantages of MSP in fabricating nano-thick Ta coatings. 22 Conducted in a vacuum environment, 26 MSP eliminates the contamination from harmful elements and impurities, thereby guaranteeing the safety of materials. Furthermore, the Ta coating deposited by MSP, in conjunction with the porous TC4 interface, exhibits a uniform distribution and robust adhesion, enhancing the coating’s durability and long-term survival. Additionally, we have achieved precise control over MSP deposition parameters, including power, gas pressure, and sputtering time, paving the way for larger-scale manufacturing endeavors in the future.
Clinical significance of TC4-based Ta-coated implants on treating different bone defects
The evaluation of the value of a new technology in medicine mainly depends on its ability to be continuously applied and promoted in clinical practice. 47 3D printed prostheses have been widely used in various orthopedic clinical environments, which demonstrates the potential and value of 3D printing. 48 Researchers are also making efforts to further promote this technology in clinical settings. However, a major obstacle for orthopedic 3D printed implants is the high cost of raw materials associated with high-performance orthopedic implant materials such as Ta, which subsequently increases the overall cost of treatment. Combining nanostructure technology and surface modification has become a promising solution. 49 Qin et al. demonstrated this by combining 3D printing with electrochemical nanostructures and hydroxyapatite (HA) modification to produce titanium implants that enhance bone integration, thereby improving efficiency and reducing costs. 50 Similarly, Gulati et al. used a combination of 3D printing and anodizing processes to create a titanium alloy (Ti6Al4V) bone implant, resulting in a scalable, simple, and cost-effective dual form that includes micrometer sized spherical particles and vertically aligned titanium dioxide nanotubes. 51 Similar to these studies, our TC4-based Ta-coated implants also modified the surface of TC4 with nano thin Ta coating at a lower price, and confirmed that it significantly reduced manufacturing costs by about 10 times while achieving better osteogenic ability.
In addition, based on the fact that traditional prostheses can basically meet the treatment of common bone defects, 3D printed customized prostheses are of great significance for the treatment of diseases with complex structures but without specialized implants. This study took bone defects in the wrist scaphoid bone as an example to demonstrate the application and value of 3D printed prostheses. Traditional reconstruction methods, such as autologous or allogeneic bone transplantation, often fail to fully adapt to the anatomical structure of the wrist joint, resulting in unsatisfactory long-term outcomes for diseases such as bone necrosis. 52 Internal fixation with iliac bone transplantation not only causes serious surgical trauma, but may also lead to complications such as pain, infection, and donor site bleeding. 53 Although arthroscopic bone transplantation combined with percutaneous fixation is minimally invasive, the operation time is longer compared to open surgery.53,54 The clinical study by Yi et al. showed that even with robot assistance, the average surgical time was 167.5 min (range: 105–255 min). 55 Our research has successfully shortened the surgical time to approximately 1 h by customizing prostheses, thereby improving existing treatment methods and increasing surgical efficiency. Accurate prosthesis design for irregular bone defect patterns, while possessing excellent osteogenic performance, sufficient mechanical strength, and lightweight characteristic, makes TC4-based Ta-coated implants more conducive to clinical application and promotion. In addition, the failure of prostheses after complex bone defect reconstruction may have catastrophic consequences for patients. Sevelda et al.’s study showed that the most common periprosthetic infection caused by Staphylococcus aureus is the main cause of proximal tibial tumor type knee prosthesis failure. 56 The antibacterial performance of tantalum coated prostheses has been widely validated,55,57 and there have been no reports of periprosthetic infections in current research on Ta implants. This highlighted the clinical significance of our research on TC4-based Ta-coated implants.
The limitations of this study are readily apparent. Firstly, the sample size is relatively small, which restricts the generalizability of the findings. Despite preliminary results indicating the promising efficacy of personalized titanium-based tantalum-coated prostheses in treating bone defects, further validation through larger-scale studies is necessary to confirm their effectiveness and safety. Secondly, the follow-up period is limited, necessitating extended observation and research to assess the long-term stability of the prosthesis, coating durability and potential complications such as prosthesis wear and coating detachment. Since the focus of this study is to introduce the innovative aspects of nano tantalum coating, we will continue to follow up with all patients to further evaluate long-term indicators that have not yet been included. Lastly, the absence of a control group prevents a direct comparison with traditional treatment methods, thereby undermining the persuasiveness of the research outcomes to some extent.
Conclusion
In conclusion, our clinical study introduces a novel and promising approach to treating patients with critical-size bone defects of wrist and palmar through the use of personalized titanium-based tantalum-coated prostheses. These prostheses were meticulously designed using advanced 3D printing technology and surface-modified with a nano-thin amorphous tantalum coating, imparting exceptional biocompatibility and osteoinductive properties. Results from our study demonstrate successful surgeries in all three patients, with the prostheses exhibiting favorable bone integration and clinical outcomes. This innovation offers a significant advancement in addressing limitations associated with traditional bone replacement materials, particularly in terms of bone ingrowth, mechanical performance, and economic viability. As we look toward the future, continued research and long-term follow-up are essential to further validate the effectiveness and safety of these customized tantalum-coated titanium prostheses in the realm of orthopedic reconstruction.
Footnotes
Author contributions
C.C and Z.L completed the manuscript writing, H.F, P.W, and G.C were responsible for literature searching, F.W and C.C collected and analyzed the data. All authors reviewed the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the funding provided by The Sichuan Science and Technology Program (2023YFQ0009) and Sichuan Chongqing Science and Technology Innovation Cooperation Program (2024YFHZ0075).
Ethical statement
Data Availability Statement
All data included in this study can be obtained from the corresponding author upon providing appropriate justification.
