Abstract
Introduction
Delayed bone union or nonunion is observed in up to 25% of patients with open long bone fracture, slowing recovery, necessitating otherwise avoidable healthcare resource utilization, and potentially resulting in skeletal malformations. 1 The main goals of bone fracture treatment are to restore biomechanical properties and normal physiological function. Thus, pharmacological interventions for accelerating fracture healing may substantially lower total medical costs and facilitate return to work and other activities. 2 Despite clear unmet clinical needs, however, no pharmacological agents are currently approved for the acceleration of fracture healing or for the treatment of delayed or nonunion fractures.
Recently, promising clinical results were reported using newly developed implants as internal fixators for osteotomy around the knee and hip joints. These rigid long plates provide high initial stability without bone grafting for early weight-bearing and can maintain optimal postoperative lower leg alignment. Despite such advances, however, most clinicians still recommend a period of protection from weight-bearing after osteotomy, which slows rehabilitation and delays return to work, sports, and various other activities of daily living. The length of time taken for the osteotomy site to unite is a rate-limiting step in patient recovery. Therefore, acceleration of bone healing at the osteotomy site can provide both social and financial benefits for patients.
Abaloparatide (Abalo) is a novel subcutaneously administered parathyroid hormone 1 receptor (PTH1R) agonist with 76% amino acid homology to native parathyroid hormone related protein (PTHrP) 1–34. Daily administration was reported to increase bone formation, bone mass, and bone strength without increasing bone resorption in both uninjured (nonfractured) rats and nonhuman primates.3–5 Abalo is currently approved by the U.S. Food and Drug Administration as a treatment for postmenopausal osteoporosis based on its ability to reduce vertebral and nonvertebral fracture risk.6,7 Similarly, bisphosphonate (BP) has been demonstrated to improve bone mineral density (BMD), normalize elevated levels of serum bone metabolism markers, and lower the risk of bone fractures by inhibiting bone resorption.8–10 Furthermore, several reports have demonstrated the efficacy of BP for increasing callus volume (CV) and dynamic strength following fracture.11,12 Zoledronic acid (or zoledronate, ZA) is a BP approved for several bone diseases. In rats, 0.1 mg/kg zoledronate (ZA) was reported to increase callus strength, bone mass: volume ratio, trabecular width, and trabecular connectivity density during bone fracture healing in an ovariectomized (OVX) rat femoral fracture model. 13 Thus, it is speculated that combination treatment with Abalo and BP may act additively or synergistically to accelerate long bone healing.
However, BP has been reported to blunt PTH action in both patients14–16 and animal models.17,18 Alternatively, Cosman and colleagues reported that a single infusion of zoledronic acid with daily teriparatide, the 1–34 N-terminal peptide of PTH, increased lumbar spine BMD more than teriparatide alone among females with postmenopausal osteoporosis during the first 13 weeks of treatment. 19 Furthermore, combined therapy led to rapid, pronounced, and persistent gains in total-hip, trochanter, and femoral neck BMD versus teriparatide alone and greater total-hip BMD gain compared to ZA alone at 13 weeks. Consistent with these findings, we reported that a combination of low-dose teriparatide and ZA promoted callus formation, increased CV, and enhanced bone union in a refractory fracture rat model. 20 Similarly, Vegger and coworkers reported an additive effect of combined PTH and ZA treatment in a rat model of disuse osteopenia, including significantly higher bone mass, trabecular bone volume, and trabecular thickness compared to monotherapy. 21 Murphy and colleagues also reported that PTH (1–34) increased bone turnover as well as ZA turnover and remobilization during fracture healing in OVX osteopenia model rats. 22 Collectively, these studies support the benefits of ZA and teriparatide combination therapy for callus development and fracture healing. Moreover, ZA plus teriparatide has been reported to be more efficacious for fracture healing than either monotherapy in OVX rats.23,24
We hypothesized that combination therapy with Abalo and ZA can promote bone healing compared to separate administration and that this effect would be more pronounced when administered preoperatively. To test this hypothesis, we established a rat femoral osteotomy model and compared preoperative combined administration to individual Abalo and ZA treatments for accelerating bone healing, increasing trabecular and cortical BMD, and improving the micro-architecture of trabecular and cortical bone at 2 and 4 weeks postosteotomy.
Materials and methods
Treatment groups
A total of 56 male Sprague–Dawley rats (9 weeks old; CLEA Japan, Inc., Tokyo, Japan) were randomly divided into four groups of 14 each: Control (administered saline), Abalo (administered 30 μg/kg s.c. abaloparatide 5 times/week for 6 weeks before osteotomy), ZA (0.1 mg/kg single dose s.c. immediately prior to osteotomy), and Abalo + ZA (administered the same doses of abaloparatide and zoledronate). Abaloparatide bulk powder was obtained from Teijin Pharma Limited (Tokyo, Japan) and zoledronate (ZOMETA®) from Novartis Pharma KK (Tokyo, Japan) (Figure 1). Study schema. Sprague–Dawley rats (n = 56, aged 9 weeks) were randomly and equally allocated to a Control group, abaloparatide (Abalo) group, zoledronate (ZA) group, and Abalo + ZA group. Unilateral (right) femoral osteotomy was performed at time 0, and rats were sacrificed for X-ray and micro-CT-based analyses of bone healing after 2 and 4 weeks of recovery. Blood samples were also taken at the indicated times pre- and postosteotomy for measurement of bone turnover markers.
Femoral osteotomy
Animal experiments were reviewed and approved by the Oita University Animal Ethics Committee (no. 1,624,003) prior to the study. Rats were anesthetized by intraperitoneal injection of 0.15 mg/kg medetomidine, 2 mg/kg midazolam, and 2.5 mg/kg butorphanol (0.3–0.4 mL total volume). The right hind limb was prepared for the operation under standard sterile conditions. With the rat in the lateral position, the right femur was located using the posterolateral approach and the midshaft exposed. A transverse osteotomy was performed, the fracture ends were contacted and stabilized, and the intramedullary spaces drawn together using a stainless-steel wire (diameter, 1.6 mm). The wire was cut on the surface of the intercondylar groove to prevent restriction of knee joint motion. The fascial and skin incisions were closed with 3–0 nylon sutures.
Harvesting of bilateral femurs
Following osteotomy, rats were housed in separate cages with food and water available ad libitum and monitored daily for health status of the rat. Animals were euthanized by injecting 0.15 mg/kg medetomidine hydrochloride +2.0 mg/kg midazolam +2.5 mg/kg butorphanol into the peritoneum, followed by cervical dislocation, 2 or 4 weeks after osteotomy, and the femoral bones carefully excised. The stainless-steel wire was removed from the osteotomized (right) femur before analysis. Both femoral bones were examined by radiography.
Radiographic analysis
Excised femoral bones were photographed using a Softex-ray apparatus (Softex CSM-2; Softex, Tokyo, Japan) and HS Fuji Softex film (Fuji Film, Tokyo, Japan) at a working distance of 45 cm, 30 kV maximum voltage, and 15 mA maximum current. Each irradiation lasted 20 s. Bone fusion was quantified based on anteroposterior (A-P) and lateral radiographs using the Radiographic Union Scale for Tibial fractures in Tibial Fracture (RUST) score, 25 which is based on the presence/absence of callus and a visible fracture line in the four cortices visible on A-P and lateral radiographs. The RUST score ranges from 4 to 12 points, with four points indicative of an unhealed fracture and 12 points a healed fracture with all cortices bridged by callus and without a fracture line on any of the four cortical bone regions. Kooistra and colleagues have verified the reliability and validity of the RUST scale for human long bone.
Measurement of serum bone turnover marker levels
Blood samples (100 µL) were also collected 6, 4, and 2 week before osteotomy, immediately before osteotomy (0 w), and both 2 and 4 weeks after osteotomy from seven animals per group. Serum levels of the osteogenesis marker type 1 amino-terminal propeptide (P1NP) and the bone resorption marker cross-linked C-telopeptides of type-1 collagen (CTX-1) were measured using specific enzyme-linked immunosorbent assay (ELISA) kits (RK03858 [CUSABIO, Houston, TX, USA] and RK03603 [Abconal, Woburn, MA,USA], respectively).
Bone histomorphometrical analysis
After extraction and X-ray imaging, the femoral bones were cleaned, fixed in 70% ethanol, embedded in glycolmenthacrylate resin, and sliced at 3 µm thick. Calcification state and cells in the fracture area were observed from five samples per group at 2- and 4-weeks postosteotomy. After staining with toluidine blue, bone morphometry was performed up to 0.43 mm proximal and distal to the fracture site using a Histometry RT camera (System Supply, Tokyo, Japan). Measurement parameters included CV, callus area, and cartilage tissue area (Ca.A).
Bone micro-CT analysis
Bone micro-CT imaging was performed according to the guidelines of Bouxsein et al. 26 Excised femoral bones were scanned using a Sky-Scan 1176 CT system (Bruker micro-CT, Kontich, Belgium) at a voxel size of 20 mm, 100 kV maximum voltage, and 100 mA maximum current. Images were reconstructed using the cone-beam algorithm. Briefly, each femoral bone was fixed in a sample holder with the vertical axis on the object stage, and scanned over 180° of rotation with an exposure time of 105 ms. A cylindrical volume of interest 20 mm in diameter and 27 mm high was selected as this size proved sufficient to capture both cortical and trabecular bones. All data were analyzed using CT Analyzer software (Bruker micro-CT). For the nonoperated (left) femur, the trabecular bone area 0.8–3.8 mm from the growth plate reference level and the cortical bone area 3.0–3.8 mm from the growth plate reference level were calculated on each scan using CT Analyzer. For the operated side, the region of interest was set at the fracture area, and the areas of trabecular bone and cortical bone ±4.2 mm from the fracture line were calculated using CT Analyzer. Measured parameters from three-dimensional analysis were bone volume/tissue volume (bone volume (BV)/TV), trabecular BV, TV, trabecular thickness (Tb.Th), trabecular number (Tb.N), trabecular separation (Tb.Sp), cortical BV, cortical bone area (Cr.Ar), and cortical bone thickness (Cr.Th).
Femoral BMD
The BMD of both trabecular and cortical bone was measured by micro-CT 3.0–3.8 mm proximal from the growth plate.
Statistical methods
Morphometric parameters and RUST scores were compared among treatment groups by one-way ANOVA with post-hoc Bonferroni tests for pair-wise comparisons. All analyses were performed using Statistical Package for the Social Sciences (SPSS V25.0; SPSS, Chicago, IL). Statistical significance was set at p < .05.
Results
Animals exhibiting improper pin placement, unduly comminuted, or poorly located fractures were euthanized by injecting 0.15 mg/kg medetomidine hydrochloride +2.0 mg/kg midazolam +2.5 mg/kg butorphanol into the peritoneum, followed by cervical dislocation; animals among the 20 spare rats underwent the fracture protocol to achieve 56 fractured animals per treatment group. Animals were allowed full weight bearing after recovery from anesthesia. Animals were weighed at least once per week as a health-monitoring measure and for weight-based treatment dosing adjustments, and were observed daily to assess general health, recovery from surgery, and reactions to treatment.
Preoperative abaloparatide plus zoledronate treatment enhanced callus bridge formation following experimental osteotomy
At 2 weeks postosteotomy, a fracture line could be clearly identified on X-rays of excised femurs from all groups, but immature callus formation was observed in femurs from Abalo and Abalo + ZA group rats, suggesting earlier initiation of repair. Moreover, callus continuity was most evident in Abalo + ZA group rats. In contrast, the fracture line was still clear, callus formation less advanced, and continuity absent in rats of the control and ZA groups. At 4 weeks, mature callus formation was observed in all groups, but the fracture line was least clear in the Abalo + ZA group and callus continuity complete only in the Abalo + ZA group (Figure 2). These group differences in callus formation were further confirmed by micro-CT (Figure 3). Mature callus and bridge formation around the fracture site were most evident in the Abalo and Abalo + ZA groups (Figure 3). X-ray images of isolated femurs showing accelerated healing by Abalo + ZA treatment. (a)–(d) Typical femur samples at 2 weeks postosteotomy from the Control group (a), Abalo group (b), ZA group (c), and Abalo + ZA group (d). (e) and (f) Typical femur samples at 4 weeks postosteotomy from the Control group (e), Abalo group (f), ZA group (g), and Abalo + ZA group (h). Examples were selected based on similarity of callus volume to the group mean. Computer tomographic images showing accelerated healing by Abalo + ZA treatment. (a)–(d) Typical femur samples at 2 weeks postosteotomy from Control (a), Abalo (b), ZA (c), and Abalo + ZA (d) groups. (e)–(f) Typical femur samples at 4 weeks postosteotomy from Control (e), Abalo (f), ZA (g), and Abalo + ZA (h) groups. Examples were selected based on similarity of callus volume to the group mean.

The RUST score results, including evaluation of cortical bone continuity and callus formation, were consistent with qualitative observations, with the Abalo and Abalo + ZA groups scoring significantly higher than the control group at both 2 weeks and 4 weeks postosteotomy (p < .05) (Figure 4). Group differences in RUST scores. The RUST score is based on the presence or absence of callus and of a clear fracture line within the four cortical views on anteroposterior and lateral radiographs. Results are presented as mean ± standard deviation of n = 7 bones per group and timepoint. *p < .05 by one-way ANOVA with post-hoc Bonferroni tests. Bars and error bars represent the mean and standard deviation, respectively.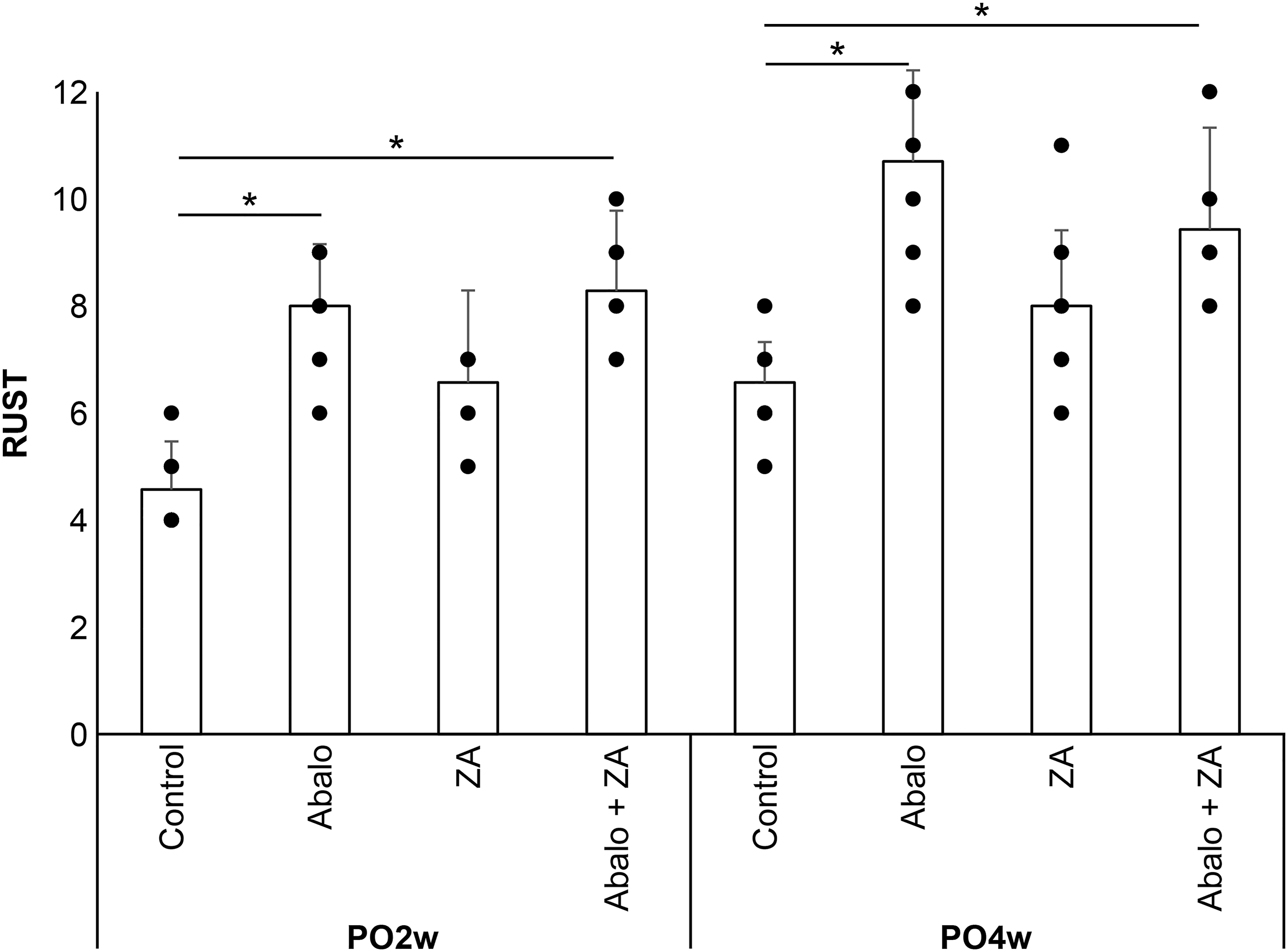
Serum bone turnover marker concentrations were comparable among treatment groups
Serum P1NP concentration rose by 60% in both abaloparatide-treated groups (Abalo and Abalo + ZA) during the first 2 weeks of treatment compared to ZA and control groups, then declined to near baseline thereafter (Figure 5, left panel). Thus, there were no significant group differences in serum P1NP at the time of osteotomy. Serum P1NP rose again in the Abalo group by 4 weeks postosteotomy but fell below baseline in the Abalo + ZA group. There were significant differences in serum P1NP levels 4 weeks postoperatively between the control and Abalo + ZA, Abalo and Abalo + ZA, and ZA and Abalo + ZA groups. In contrast, serum CTX-1 concentration (Figure 5, right panel) was reduced by 57% after 4 weeks of Abalo treatment (Abalo and Abalo + ZA groups). Serum CTX-1 concentration also fell in the ZA group and especially the control group following osteotomy and was significantly lower in the control group at 2 week postosteotomy. Nonetheless, no significant group differences were found at 4 weeks postosteotomy. In all groups, serum P1NP and CTX-1 changes varied markedly among rats within a group. Mean serum concentrations of bone turnover markers P1NP and CTX-1. Blood samples (100 µL) were collected from the tail vein 6, 4, and 2 weeks before osteotomy (Pre6w, Pre4w, and Pre2w, respectively), on the day of osteotomy (Osteotomy), and both 2 and 4 weeks after osteotomy (Post2w and Post4w) (n = 7 samples per group and timepoint). Serum concentrations of P1NP (left panels) and CTX-1 (right panels) were measured by ELISA and expressed relative to the Pre6w value. There were significant differences in serum P1NP levels 4 weeks postoperatively between the control and Abalo + ZA, Abalo and Abalo + ZA, ZA and Abalo + ZA groups. **p < .01 by one-way ANOVA with post-hoc Bonferroni tests. In contrast, the only significant difference was detected in serum CTX-1 between control and both Abalo and ZA groups at 2 weeks postosteotomy. *p < .05 by one-way ANOVA with post-hoc Bonferroni tests.
Abaloparatide plus zoledronate enhanced endochondral ossification and callus area following experimental osteotomy
Histopathological analyses using toluidine blue staining revealed no mature bridge formation or endochondral ossification at 2 weeks postosteotomy in the control and ZA groups, while callus formation was advanced in both the Abalo and Abalo + ZA groups (consistent with radiographic findings) without endochondral ossification (Figure 6). After 4 weeks, all groups exhibited mature bridge formation; however, only the Abalo and Abalo + ZA groups demonstrated endochondral ossification in the superficial layers. Moreover, callus area was numerically greatest in the Abalo + ZA group (Figure 6). Histological analysis of toluidine blue-stained nondecalcified rat femur specimens. (a)–(d) Typical femur samples at 2 weeks postosteotomy from Control (a), Abalo (b), ZA (c), and Abalo + ZA (d) groups. (e)–(h) Typical femur samples at 4 weeks postosteotomy from Control (e), Abalo (f), ZA (g), and Abalo + ZA (h) groups. Light toluidine blue staining indicates immature callus formation and endochondral ossification, while darker violet staining indicates cartilage matrix (light toluidine blue staining is indicated by a star (★) and darker violet staining is shown as a diamond shape (◆) in Figure 6(d)).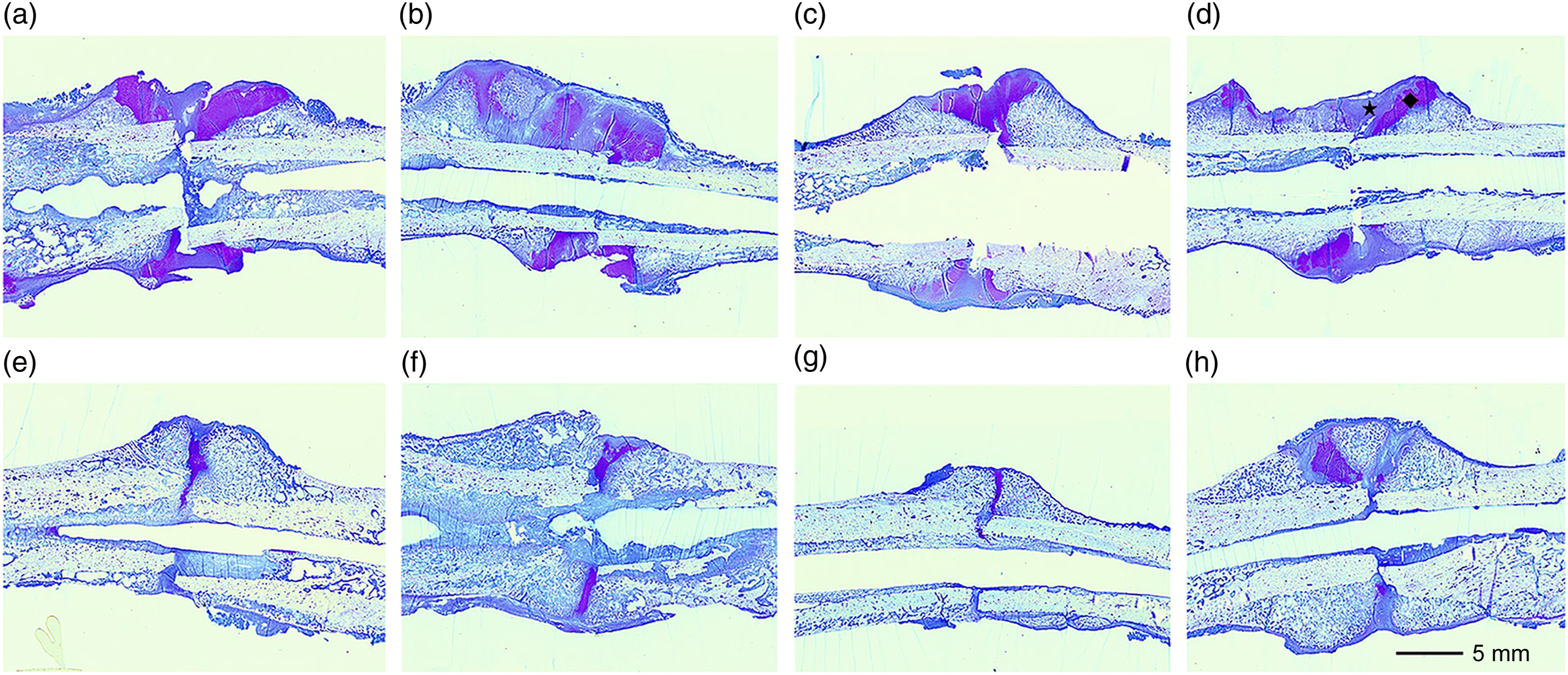
Bone micro-CT analysis of the fractured and unfractured femur
Micro-CT of the fractured femur (Figure 7) revealed significantly greater BV/TV in the Abalo + ZA group compared to the control group at 4 weeks postosteotomy. Further, BV/TV, Tb.N were numerically higher in ZA and Abalo + ZA groups than in the control group at 4 weeks postosteotomy; however, Tb, Th, and Tb.Sp were approximately the same. Treatment group differences in trabecular bone morphology on the fractured side during recovery. (a) Tissue volume (TV). (b) Trabecular bone volume (BV). (c) Trabecular bone volume to tissue volume ratio (BV/TV). (d) Trabecular thickness (Tb.Th). (e) Trabecular number (Tb.N). (f) Trabecular separation (Tb.Sp). *p < .05 and **p < .01 by one-way ANOVA with post-hoc Bonferroni tests. Bars and error bars represent the mean and standard deviation, respectively.
There were also differences in morphometric values in the noninjured femur (Figure 8). Both BV/TV and Tb-Th were higher in the Abalo + ZA group than the ZA and Abalo groups. Alternatively, Tb.Sp was significantly lower in the Abalo + ZA group compared to the control group. Treatment group differences in trabecular bone morphology on the nonoperated side during recovery. (a) Tissue volume (TV). (b) Trabecular bone volume (BV). (c) Trabecular bone volume to tissue volume ratio (BV/TV). (d) Trabecular thickness (Tb.Th). (e) Trabecular number (Tb.N). (f) Trabecular separation (Tb.Sp). *p < .05 and **p < .01 by one-way ANOVA with post-hoc Bonferroni tests. Bars and error bars represent the mean and standard deviation, respectively.
BMD of the nonoperated femur
Trabecular BMD was significantly greater in the Abalo + ZA group compared to all other groups, while cortical BMD was significantly greater in the Abalo + ZA group compared to Abalo and control groups and greater in the ZA group than the Abalo group (Figure 9). Treatment group differences in bone mineral density on the nonoperated side during recovery. Bone mineral density was calculated from micro-CT measurements within an area 3.0–3.8 mm from the growth plate. *p < .05 and **p < .01 by one-way ANOVA with post-hoc Bonferroni tests. Bars and error bars represent the mean and standard deviation, respectively.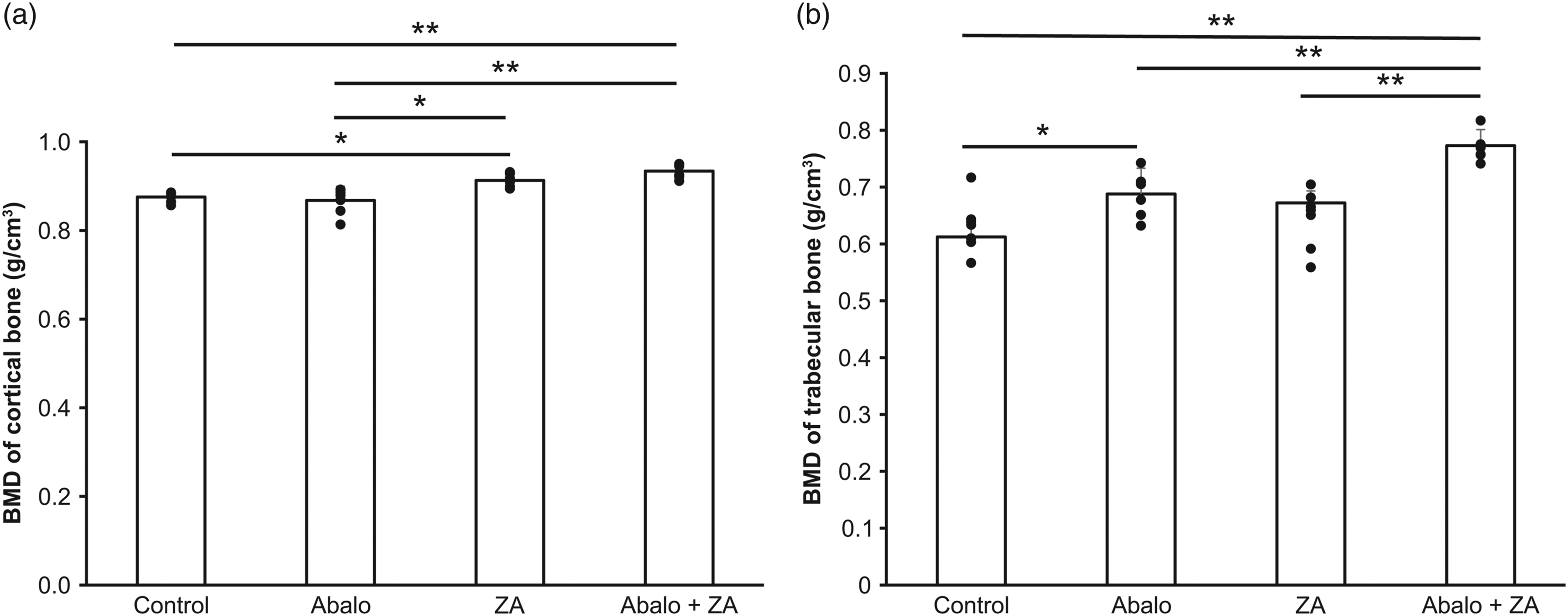
Discussion
Abaloparatide monotherapy and combination therapy with zoledronate (Abalo + ZA) prior to unilateral femoral osteotomy accelerated healing in rats within the first 4 weeks compared to ZA alone as evidenced by soft X-ray imaging, micro-CT, and histopathology. Bone morphometry of the injured and noninjured (contralateral) femur based on micro-CT also revealed increased trabecular BV relative to total BV in rats receiving combined Abalo + ZA compared to Abalo alone. Further, both trabecular and cortical BMD were higher at the post- injury site following Abalo + ZA post-treatment compared to individual Abalo and ZA post-treatments. Thus, the combination of Abalo and ZA may be an effective regimen to speed recovery and return to normal activities following femoral bone fracture.
Fracture healing occurs in three distinct stages. 27 The first stage involves various inflammatory processes for clearing injured tissue and providing an appropriate microenvironment for tissue repair. Damage to local blood vessels and bleeding leads to the formation of a hematoma and fibrin network around the fracture site. Within the first week postinjury, a number of proteinaceous growth factors and cytokines are released into this compartment to drive macrophage activity. This inflammatory stage is followed by fracture repair. At the site of the hematoma, osteoblasts are recruited by reparative cytokines to form an immature callus, 28 a process that substantially influences the pace and extent of healing. During the third remodeling phase, the calcified callus is replaced with lamellar bone that ultimately becomes incorporated into and around the fracture site. This final reparative stage determines the strengthening of fractured bone, and may take months or even years to complete. The total duration of fracture healing is highly variable, and is determined by individual reparative capacity, fracture location, and the degree of mechanical loading required for full function. It is during the second stage of fracture healing that teriparatide pretreatment may have substantial therapeutic benefits. 29
In the present study, preosteotomy administration of Abalo and a single postosteotomy dose of ZA were sufficient for accelerated bone healing. Abaloparatide may accelerate fracture healing by promoting the proliferation and differentiation of mesenchymal stem cells, chondrogenic progenitor cells, and osteoblastic progenitor cells as well as by enhancing cartilage cell maturation, bone matrix protein production, and osteoclast formation.30–32 The increase in callus formation may have resulted from enhanced proliferation and differentiation of osteoblasts and osteoblastic progenitor cells. 3 Furthermore, teriparatide was reported to increase the size of pseudogenesis via the Wnt/β-catenin signaling pathway and the expression levels of type II and type X collagen. These effects may account for the observed acceleration of bone repair as earlier callus formation was detected in the Abalo and Abalo + ZA groups compared to control and ZA groups by soft X-ray, 3D-CT, and toluidine blue staining. Furthermore, these differences were verified quantitatively by RUST scoring. Previous studies have reported accelerated fracture and osteotomy healing with increased CV and cartilage tissue during postinjury Abalo treatment. The current results thus support previous findings and further suggest that preoperative administration of Abalo can increase bone density and bone mass following osteotomy by creating an anabolic window for increased callus formation, thereby promoting more rapid bone healing.
Zoledronate inhibits bone resorption by inducing osteoclast apoptosis. It has also been reported to increase CV, bone density, and (or) mechanical strength in humans and animals without interfering with the fracture healing process. However, ZA does inhibit remodeling. 11 Nonetheless, faster bone healing was observed in the group receiving Abalo plus ZA. Murphy and colleagues reported increased metabolic turnover of bisphosphonate in a rat fracture model treated with combined PTH and bisphosphonate. 22 Thus, it is plausible that Abalo promotes earlier bone healing by remobilizing ZA.
This study has several limitations. First, we did not examine the effects of Abalo and ZA separately or together on functional outcomes such as walking ability. Femoral load differs markedly between rats and (bipedal) humans, so the degree of healing required for normal ambulation may differ. 33 Similarly, species differences in bone repair mechanisms may influence the effects of abaloparatide and ZA. For instance, the changes in bone formation markers differed from human findings. 6 In humans, bone formation markers exhibited a slow and gradual decline for 18 weeks during Abalo treatment, while in the present rat experiments, markers decreased considerably at 6 weeks after treatment initiation. Therefore, 2 weeks of preoperative administration may have been sufficient to achieve the observed improvement in bone healing. In general, the administration regimen may influence treatment efficacy but we did not examine longer or shorter Abalo treatment periods or different drug doses. Finally, we did not include a posttreatment group for comparison with our previous experiments with teriparatide and romosozumab.20,34 Nonetheless, preoperative administration of Abalo was clearly effective for promoting bone healing and callus formation.
The present results provide further preclinical evidence that abaloparatide may improve healing of traumatic fractures and fragility fractures.
From a clinical perspective, early improvement in fracture callus stability may allow earlier recovery of physical activities, including weight-bearing, potentially leading to improved fracture healing due to early regain of mechanical loading. These findings, as well as those from other experimental orthopedic models, may serve as a basis for future studies of abaloparatide as a bone-healing agent. Data are promising for patients with osteoporosis or traumatic fractures.
Conclusion
Monotherapy with abaloparatide and combination therapy with abaloparatide and zoledronate prior to osteotomy accelerated bone healing as evidenced by increased trabecular and cortical BV. Preoperative administration of Abalo and ZA may be effective for refractory fracture cases such as delayed union and pseudoarthrosis, as well as for promoting recovery following orthopedic osteotomy.
Footnotes
Acknowledgements
We are grateful to Oshima Minori and Shinohara Keiko for excellent technical assistance. We would like to thank Teijin Pharma Limited (Tokyo, Japan) for providing the abaloparatide bulk powder as a kind gift.
Author contributions
TK, YT, RT, and MK carried out the operation. MK conceived the study design. TK, YT, RT, TO, TI, MK, and NK interpreted the data and drafted the manuscript. All authors read and approved the final manuscript.
Author’s note
Corresponding author is a specialist physician and a member of the Japanese Orthopedic Association.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number 21K09302.
Ethical statement
Data availability statement
All data generated or analyzed during this study are included in this published article.
