Abstract
Background
Two injectable anti-osteoporosis medications, denosumab and zoledronic acid, have been widely used to treat patients with severe osteoporosis. The purpose of this study was to evaluate the real-world effectiveness and adherence of denosumab compared to zoledronic acid in geriatric patients after a hip fracture.
Methods
A total of 282 patients treated with osteoporotic hip fracture between March 2014 and Aug 2022 were retrospectively reviewed. The patients were asked to select the anti-osteoporosis medication after surgery. Treatment persistence was monitored by follow-up visit to the outpatient clinic at postoperative 2 years.
Results
Of 282 individuals with baseline data, 162 patients took subcutaneous denosumab and 120 patients took intravenous zoledronic acid. At postoperative 2 years, the change in bone mineral density (BMD) from baseline was greater in the denosumab group compared with the zoledronic acid group (p < 0.001). The rate of persistence to denosumab was significantly higher than that for 12-months zoledronic acid (p = 0.01). Serious adverse events were similar in the two groups.
Conclusions
Our study revealed the effectiveness and patients' persistence for two commonly used anti-osteoporosis agents after hip fracture. In this frail, elderly population, half-yearly denosumab was superior to yearly zoledronic acid in BMD and demonstrated significant higher persistence rate, indicating a potential therapeutic advantage that warrants further validation.
Introduction
Osteoporotic fractures in elderly patients often entail more devastating outcomes for the individual and higher costs for society compared to fractures in young patients. 1 As elderly population is growing, hip fracture incidence has been increasing gradually in global populations and costs for hip fracture care are slated to reach 265 billion by 2050. 2 Furthermore, patients with hip fracture are at high risk of secondary fractures. 3 Therefore, treatment for osteoporosis after hip fracture is generally recommended in the real-world clinical setting and adherence to treatment is essential to preserve bone mass for most elderly patients. 4
Zoledronic acid is the strongest bisphosphonate that can be administered intravenously at a dosage of 5 mg once yearly. It has been associated with a significant reduction of non-vertebral and vertebral fracture risks in postmenopausal women with osteoporosis. 5 Denosumab is also a widely used anti-resorptive drug that has been demonstrated to increase bone mineral density (BMD) and reduce the risk of fractures at both non-vertebral and vertebral sites. 6 Considering the more acceptable and infrequent administration, denosumab and zoledronic acid are preferable treatment options among many elderly patients. Generally, these medications require long-term management to decrease fracture risk practically. However, only a small number of patients after a hip fracture actually receive pharmacological treatment for osteoporosis after surgery, thus leading to a substantial reduction in anti-fracture effectiveness. The causes for this insufficient treatment are numerous including advanced age, increased co-morbidities, polypharmacy, and a higher level of dependence. 7 Non-adherence to therapy can increase morbidity and mortality along with significant loss of function after surgical treatment of hip fractures.5,8
To date, limited comparative effectiveness analysis exists between denosumab and zoledronic acid in real-world settings in geriatric patients following hip fracture. The aim of this study was to evaluate the effectiveness, adherence, and safety of two commonly used anti-osteoporosis medications after hip fracture.
Material and methods
Patient cohorts
A retrospective observational cohort study was conducted between March 2014 and Aug 2022, and all patients with a procedural code for management of hip fracture were identified. This single-center retrospective comparative study was approved by the institutional review board of our institution (approval number: HGH-2022-08-003).
Patients who underwent surgery for intertrochanteric or femoral neck fractures were included. The inclusion criteria were as follows: (1) patients who were treated for a hip fracture, (2) patients with osteoporosis, (3) patients with dual energy X-ray absorptiometry (DXA) scan, and (4) patients who were prescribed annual intravenous (IV) zoledronic acid or 6-months subcutaneous (SC) denosumab after surgery. The exclusion criteria were: (1) patients with a treatment switch to another drug, (2) patients who were aged less than 65 years, (3) patients with chronic kidney disease on hemodialysis, and (4) pathological fractures.
The characteristics of two agents (6-months SC or 12-months IV) were explained before injection and the patients themselves or their healthcare provider made decision. In case of denosumab, possible adverse effects of back pain, blistering or irritation of the skin, muscle or bone pain, rash, and swelling were explained. With regard to zoledronic acid, common side effects of pyrexia, myalgia, headache, influenza like symptoms and arthralgia were explained. BMD was measured by DXA (Lunar Prodigy; GE Medical Systems, Madison, WI, USA) at admission, 12 months and 24 months after therapy. BMD value of L1 through L4 (L-BMD) and total hip (H-BMD) were obtained. The changes of BMD (%) from the baseline were compared between the groups by statistical analysis. During the evaluation of BMD, fracture site and the vertebrae with an obvious deformity were avoided. Treatment persistence was monitored by discontinuation/completion rates (For the denosumab group, persistence was defined as having received two injections at intervals of 6 months). 6 Body mass index (BMI) was calculated as weight (kg)/height2 (m2). The Charlson comorbidity index (CCI) was assessed to evaluate comorbidity. 9 Smoking status, menopausal age, and the fracture history were assessed based on medical records. The average age for patients was 82 years (range, 65–98). The average follow up period was 2.5 years (range, 2.0–7.2). Any adverse effects after injections and secondary fractures occurring after hospital discharge were investigated at each follow-up. Electronic health records and radiographs were used to collect the data.
Statistical analysis
All statistical testing was conducted using SPSS 12.0 software (SPSS Inc., Chicago, IL, USA). Patient background parameters are presented as mean ± standard error of the mean. Descriptive statistics of the number of patients was performed using frequencies and percentages. Independent t-test was used to test the difference between groups for continuous variables. For categorical variables, Chi-square test was performed to determine any significant differences. A p-value less than 0.05 was considered statistically significant.
Results
The demographic data for each group (group I, denosumab group; group II, zoledronic acid group).
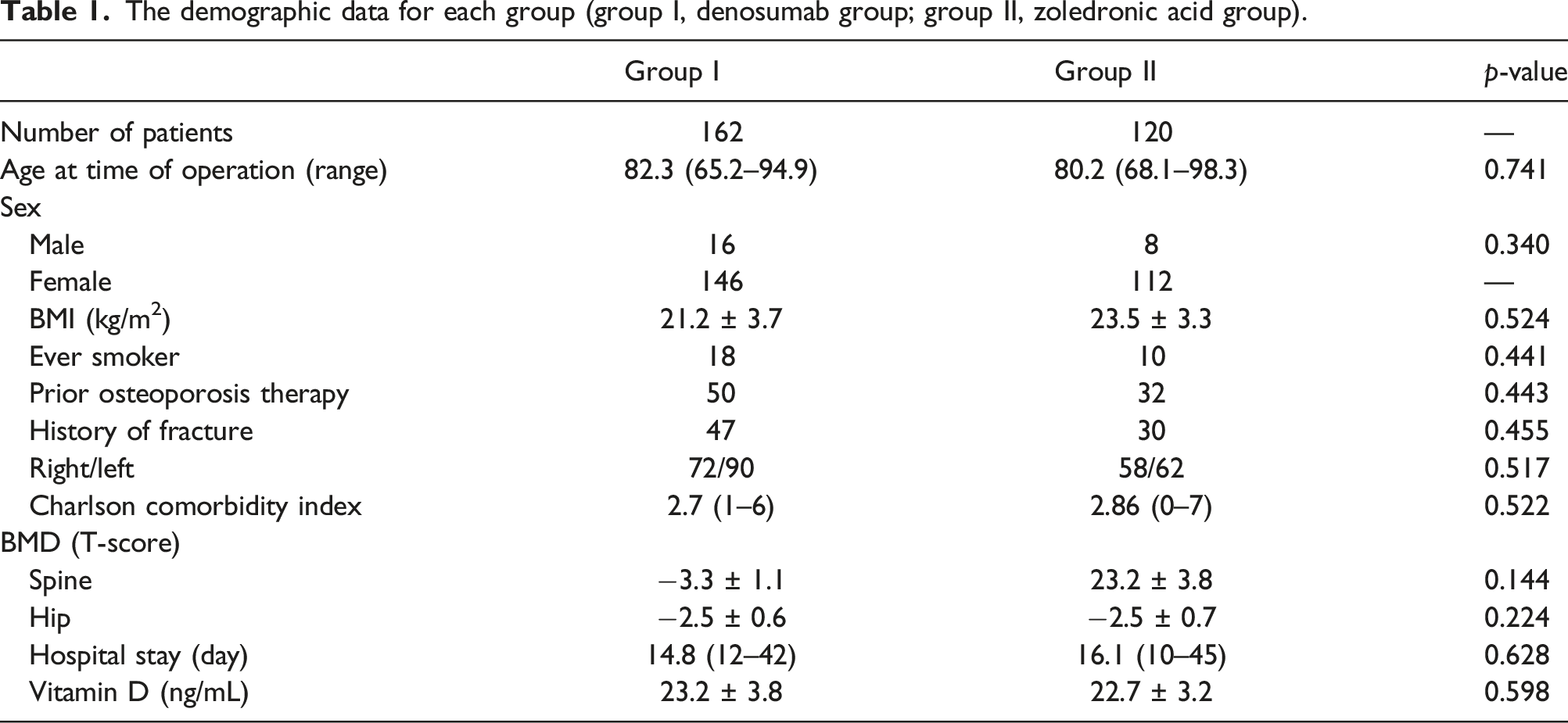
At the final follow-up, BMD increased progressively at the lumbar spine and total hip in both groups. The percentage change of L-BMD was significantly higher in the denosumab groups compared with the zoledronate group at 12 (4.5 ± 0.8 vs 3.5 ± 0.7; p = 0.01) and 24 months (8.4 ± 1.2 vs 6.1 ± 0.9; p < 0.001) after treatment (Figure 1). H-BMD change was also higher in the denosumab group compared to the zoledronate group at 12 and 24 months (p = 0.03 and p < 0.001, respectively). (a) Mean percentage change in BMD at the lumbar spine, (b) Mean percentage change in BMD at the total hip.
Discharge and follow-up medications.

Adverse effects.

Discussion
Treatment persistence is known to be higher in the patients receiving long-dose-interval medications. 6 The aim of this study was to compare the effectiveness and persistence to two commonly used long-dose-interval medications in elderly patients. In the present study, we found that half-yearly denosumab was superior to yearly intravenous injection in BMD and demonstrated higher persistence rates in this elderly population. Additionally, our study confirmed that patients’ persistence to anti-osteoporosis medications after a hip fracture is still very low.
Among the current medication options, bisphosphonates have been the mainstay therapies and widely used for postmenopausal osteoporosis treatment and prevention of new, secondary fractures.10,11 The IV zoledronic acid also has been commonly used in the treatment of osteoporosis with robust data demonstrating efficacy in fracture risk reduction. 12 Even though bisphosphonates are currently the treatment of choice for the treatment of osteoporosis, the medications faced challenges in prescribing recently due to increased risk of side effects (heart burn and abdominal pain), dental procedure, drug holidays, atypical fracture, and post-infusion symptoms. Over the past few years, it has been often reported that bisphosphonates have significant issues pertaining to inconvenience and low levels of persistence. 13 As of 2013, bisphosphonate therapy following fragility hip fracture decreased from 15 to 3%. 14
Denosumab is an anti-RANKL monoclonal antibody drug and administered subcutaneously every 6 months. Effectiveness and persistence to denosumab were already evaluated in numerous studies. 15 Compared to bisphosphonates, most patients were satisfied with its route of administration and reported greater compliance to denosumab. 16 It has been recently shown that the 2-year adherence of denosumab was almost 2 times higher than that for bisphosphonates, and that persistence rate was significantly higher for denosumab than for bisphosphonates. 17 Additionally, many articles demonstrated that denosumab was cost-saving in patients older than 75 years old with a history of fragility fractures, lower BMD values, and other multiple risk factors. 18 To evaluate the cost-effectiveness and budget impact of available treatments for osteoporosis, an accurate understanding of the costs of therapy is needed. In our institution, estimated annual costs of administration were $241 for denosumab (when administered twice annually) and $206 for zoledronic acid (both medications were covered by national health insurance).
Many clinical trials have demonstrated that medications with longer dosage interval are associated with higher compliance than are other injectable medications.15,19 Whereas patient adherence to half-yearly denosumab versus quarterly (3-monthly) bisphosphonates was not surprising in relation to the less frequent dosing schedule and the more convenient route of administration, patient preference and adherence between denosumab and zoledronic acid (yearly) could be not obvious. Many studies showed that persistence is improved with 12-monthly zoledronic acid regimen compared to other bisphosphonates. 20 However, because the medication takes quite a long time for intravenous infusion (up to 1 h) and is frequently associated with side effects of flu-like symptoms, muscle pain, and fever (even 78% of the patients) for up to 3 days after the first infusion,21,22 patients often discontinue the treatment after 12 months. 23 In our outpatient clinic, patients were recommended to take a pain reliever for 2–3 days after injection if the patient has symptoms. Recently, Lin et al. 15 demonstrated that the persistence of denosumab is superior to that of zoledronic acid at the 3rd, 4th and 5th year after treatment (20% and 16% at the 5th year). The causes of non-adherence with anti-osteoporosis medication have been shown to be multi-factorial and differ between numerous medications. 24 In our study, it seems that not many patients took zoledronic acid injection because they were afraid of post-infusion symptoms like the common side effects of COVID-19 vaccines. When prescribing anti-osteoporosis drugs for these frail patients following hip fracture, first-line therapy should be suitable to enhance patient persistence because they mostly have multiple comorbidity and high level of dependence.
While the advantages of denosumab injection have been described, potential issues regarding safety have been raised. 25 Several previous reports suggested a possible positive association between denosumab and cardiovascular disease. 26 In contrast, Choi et al. 25 reported that denosumab and zoledronic acid have similar clinical safety regarding the risk of serious infection and cardiovascular disease. Recent meta-analysis demonstrated that denosumab is as safe as zoledronic acid and may be a better option for the population with cardiovascular disease. 27 In the present study, we did not observe any differences in serious adverse events in denosumab versus zoledronic acid users in elderly patients with hip fracture.
There were several limitations in our study. First, this was a retrospective analysis with a short period of observation and the analysis of our research might have been underpowered due to a small sample size. Our study couldn’t confirm a decreased risk of secondary fracture in the denosumab group compared to the zoledronic acid group despite of a greater increase in BMD. Second, to compare the efficacy and safety of different anti-osteoporosis medications, the study of choice is a randomized clinical trial (RCT). However, because the patients usually have multiple comorbidities and the follow-up rates are quite low, there are as yet very few detailed data from a 2-years follow-up in geriatric patients following hip fracture. Even among healthier populations, there are few RCT that directly compare the effectiveness of alternative anti-osteoporosis medications, and most studies are not sufficiently powered to reveal a significant difference in hip fracture outcomes between treatment groups.28,29
In conclusion, our real-world data showed that denosumab was associated with greater BMD increases and higher adherence in very elderly people after hip fractures. Most patients who underwent surgery for a hip fracture do not take anti-osteoporosis medications, appropriately. The low compliance suggests that first-line medications are not suitable and tailor-made treatment options are required for this subset of patients. SC denosumab compared to IV zoledronic acid may provide a cost-saving treatment strategy and increase patient persistence. However, as patient compliance and persistence are complex problems, more RCTs are needed to verify this conclusion. The clinical management of osteoporosis and evidence-based prescribing will continue to be a major challenge without the information provided by such research.
Footnotes
Author contributions
S-JK had made the study design and carried out acquisition of the data. JK made the analysis and interpreted the data. D-WL had participated in conceiving the study design and revising the data and manuscript. All authors had read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
