Abstract
Keywords
Introduction
Adult spinal deformity (ASD) is characterized by abnormal spinal alignment in adulthood resulting from conditions such as de novo scoliosis, progressive adolescent idiopathic scoliosis, and degenerative disc disease.1,2 With advancements in healthcare, longer life expectancies, and an increasing number of healthy older individuals, the incidence and prevalence of ASDs are increasing.2–4 Schwab et al. 4 reported a high prevalence of ASD (68%) in adults aged >60 years. Previously, surgeons preferred conservative ASD management because of the higher operative risks, including neurological deficits. 2 However, the popularity of spinal corrective surgery has recently increased. 3 Studies have shown significant improvements in patient-reported outcomes post-surgery, including the Scoliosis Research Society-22 score, Oswestry disability index (ODI), visual analog scale score, and numeric rating scale score.5–7 A meta-analysis showed that surgical intervention improved patient-reported clinical outcomes, including pain, disability, and correction of scoliotic curves. 8
Osteotomy is an effective surgical technique for reconstruction and reshaping of rigid or severe spinal deformities. 9 The two main osteotomy techniques are posterior column osteotomies (Schwab grade II osteotomy) and three-column osteotomies (pedicle subtraction osteotomy and vertebral column resection). 9 Multiple Schwab grade II osteotomies (MOs) are relatively easy to perform; however, it is difficult to correct severe sagittal imbalance owing to limited posterior column shortening. Furthermore, multiple Schwab grade II osteotomies are associated with several complications such as excessive blood loss, pseudarthrosis, proximal junctional kyphosis, neurological deficit, rod breakage, dural tear, deep wound infection, or hematoma.9–11 Hence, minimally invasive surgery, including lateral lumbar interbody fusion (LLIF), is increasingly used to manage ASD and reduce surgery-related complications. 12 Inserting a larger interbody cage increases disc height, leading to indirect neural decompression and significant central canal decompression. 13
In severe kyphoscoliosis, the complex spatial deformation of the spinal cord is inadequately represented by plane-curve models. Moreover, information on simultaneous three-dimensional (3D) spinal cord length changes in patients with scoliosis is scarce, and reference data on cord elongation to optimize corrective surgeries without causing neurological complications are lacking. This study aimed to analyze 3D changes in the anterior vertical column length (AVCL), spinal canal length (SCL), and spinal canal volume (SCV) after ASD surgery with LLIF and posterior lumbar interbody fusion with MOs (PLIF + MOs). We hypothesized that the length and volume of the spinal canal would increase more significantly in patients undergoing LLIF compared to those undergoing PLIF + MOs.
Materials and methods
Institutional review board approval
This study was approved by the Institutional Review Board of our hospital (research approval no. 22-222) and adhered to the principles of the Declaration of Helsinki.
Study design
Data were retrospectively retrieved and reviewed from a single-centre prospectively collected database of ASD surgeries. We investigated 274 consecutive ASD patients who underwent spinal corrective fusion surgery, including LLIF and PLIF + MOs, between April 2010 and March 2021 (Figure 1). As inclusion criteria, this study included adults aged over 18 years diagnosed with ASD, who required surgical intervention for correction due to pain, neurological deficits, or functional impairment with the range of spinal fixation from the thoracic spine to the sacrum. Informed consent was obtained from all patients. As exclusion criteria, patients were excluded if they had a history of prior spinal surgery, recent spinal fractures, metabolic bone diseases, active spinal infections, spinal tumors, incomplete medical records or missing imaging data, insufficient assessment of full computed tomography imaging from T1-S1 pre- and postoperatively, or were unable or unwilling to comply with post-operative follow-up protocols. ASD was defined as the presence of at least one of the following indicators: degenerative or idiopathic scoliosis with a spinal curvature >20° in the coronal plane, a C7 sagittal vertical axis (SVA) > 50 mm, pelvic tilt (PT) > 25°, and T5-T12 thoracic kyphosis (TK) > 60°.
14
Physical examination, pre- and postoperative full spine radiography, and CT images of the whole spine of each patient were acquired for surgical planning and treatment (Figure 2). Patients were asked to complete the ODI score as an outcome measure. Complete datasets that included information regarding demographic, operative, and postoperative data were available for all patients, and sufficient radiographic data for analyses were available. Study design. Radiographs of a case managed through correction surgery with LLIF and PLIF + MOs. Case 1: A 73-year-old female with back pain and kyphoscoliosis underwent radiography in AP (a) and lateral (b) planes. The patient underwent LLIF and posterior fixation from T5 to IL. Postoperative radiographs in the plane (c) and lateral plane (d) showed good sagittal alignment, and her back pain resolved postoperatively. Case 2: A 79-year-old male with back pain and kyphoscoliosis underwent radiography in the AP (e) and lateral (f) planes. The patient underwent PLIF + MOs and posterior fixation from T7 to IL. Postoperative radiographs in the plane (g) and lateral planes (h) demonstrated good sagittal alignment, and his back pain resolved. LL, lumbar lordosis; PI, pelvic incidence; PT, pelvic tilt; SVA, sagittal vertical axis; Cobb, Cobb angle; LLIF, lateral lumbar interbody fusion; PLIF + MOs, posterior lumbar interbody fusion with multiple grade II osteotomies; AP, anterior-posterior.

Surgical methods
We used two methods for corrective surgery, LLIF and PLIF + MOs, based on the below-mentioned surgical strategy.
15
All patients were operated on by one medical team. Patients with symptoms caused by deformity, such as abnormal posture, fatigue-related low back pain, intermittent claudication, scoliotic deformity >30°, smooth kyphosis, and sagittal imbalance with SVA >50 mm, were classified as Level III. The procedures included PLIF + MOs
Radiographic parameters
Patient characteristics and surgical data were obtained from the medical records. For each patient, the radiographic parameters included the pre- and postoperative radiographic measurements of lumbar lordosis (LL), thoracic kyphosis (TK), pelvic incidence (PI), pelvic tilt (PT), sagittal vertical axis (SVA), and Cobb angle (Cobb; Figure 2).
Three-dimensional model analysis
All patients underwent CT imaging before and after the surgery, employing the following multi-phase scanning parameters: a thickness of 1 mm, power set at 120 kV, 200 mA with a 0.5-s rotation time, and matrix reconstruction of 512 × 512 pixels. To determine the AVCL from L1 to S1, we located the midpoint of the arc created by the anterior vertical column from L1 to S1 in the axial plane of each vertebral endplate segment (Figure 3(a)). We calculated the SCL from T1 to S1 by defining the central spinal canal as the midpoint of the vertebral arch. circle (on the axial plane). This was connected to each other within the surveyed spinal cord segment, from T1 through S1 (Figure 3(b)) and across the three CT scan planes (Figure 3(c)), for 3D imaging of the SCL (Figure 3(d)). To determine the SCV, circular or elliptical vertebral arch. circles (on the axial plane) were created and connected to each other within the surveyed spinal cord segment (Figure 3(e)) and across three CT scan planes (Figure 3(f)), generating 3D images of the SCV (Figure 3(g)). The delta SCL and SCV (postoperative minus preoperative) were calculated for each patient. 3D images of the length and volume were generated and processed using Synapse Vincent software (Synapse Vincent, FUJIFILM, Japan). The SCL and SCV were determined by analyzing length (mm), volume (cm3) respectively, and CT values were measured in Hounsfield units (HU). To minimize measurement errors during the analysis of the 3D model, all measurements were taken by two orthopedic surgeons with independent measurements. And utilize automated or semi-automated segmentation algorithms to minimize observer variability. These algorithms can consistently identify anatomical landmarks and structures, reducing the potential for human error. The interobserver reliability was calculated by determining the intraclass correlation coefficient (ICC) for AVCL, SCL, and SCV. ICC was interpreted as follows: below 0.50: poor, between 0.50 and 0.75: moderate between 0.75 and 0.90: good above 0.90: excellent.
18
A 3D model of the spine constructed using a CT scan image. Spine imaging overview to identify research indicators, including SCL, SCV, and AVCL (a). To determine the SCL, midpoints of the vertebral arch. circle (on the axial plane) were connected to each other within the surveyed spinal cord segment (b) and across the three CT scan planes (c), resulting in 3D rendering of the SCL (d). For SCV determination, circular or elliptical vertebral arch. circles on the axial plane were created and connected to each other within the surveyed spinal cord segment (e) and across the three CT scan planes (f), thereby generating 3D images of the SCV (g). CT, computed tomography; SCL, spinal cord length; SCV, spinal cord volume; AVCL, anterior vertical column.
Statistical analysis
Descriptive statistics, including mean and standard deviations (mean ± SD), were computed for the demographic data. To test for significant differences between groups, an independent-sample paired t test (two-tailed) was used. Statistical significance was set at p < 0.05. IBM SPSS software (version 25.0; IBM Corp., Armonk, NY, USA) was used for all statistical analyses.
Results
Preoperative characteristics and radiographic parameters between groups with LLIF and PLIF + MOs.

*The mean difference is significant at 0.05 level.
Statistical analysis was performed using independent-samples t-tests.
Body mass index (BMI), preoperative (preop), lumbar lordosis (LL), thoracic kyphosis (TK), pelvic incidence (PI), pelvic tilt (PT), sagittal vertical axis (SVA), Oswestry disability index (ODI), lateral lumbar interbody fusion (LLIF), posterior lumbar interbody fusions with multiple grade II osteotomies (PLIF + MOs).
Preoperative spinal canal dimensions between groups with LLIF and PLIF + MOs.

*The mean difference is significant at 0.05 level.
Statistical analysis was performed using independent-samples t-tests.
Anterior vertical column length (AVCL), spinal canal length (SCL), spinal canal volume (SCV), Oswestry disability index (ODI), lateral lumbar interbody fusion (LLIF), posterior lumbar interbody fusions with multiple grade II osteotomies (PLIF + MOs).
Postoperative radiographic parameters and outcomes between groups with LLIF and PLIF + MOs.
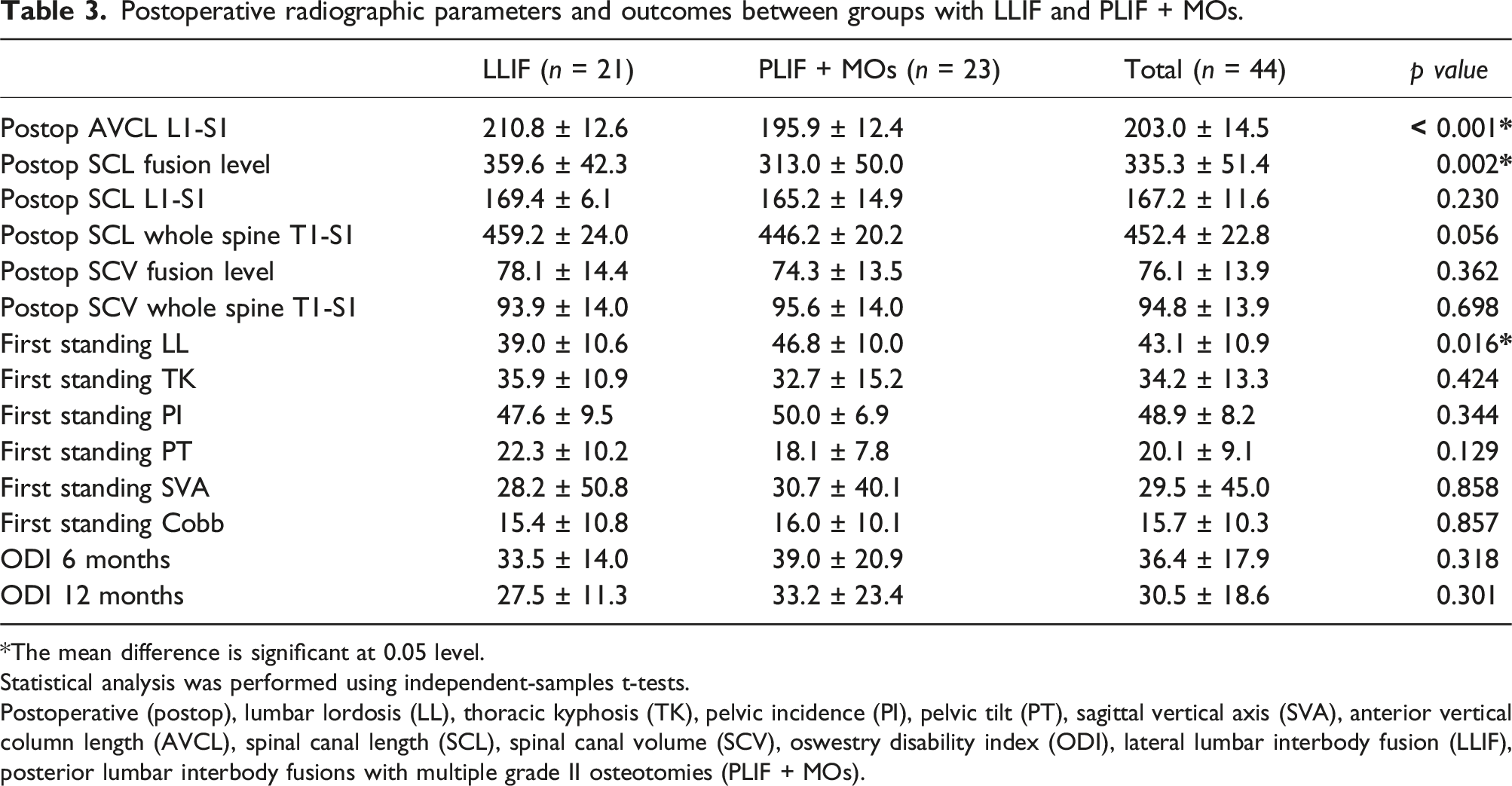
*The mean difference is significant at 0.05 level.
Statistical analysis was performed using independent-samples t-tests.
Postoperative (postop), lumbar lordosis (LL), thoracic kyphosis (TK), pelvic incidence (PI), pelvic tilt (PT), sagittal vertical axis (SVA), anterior vertical column length (AVCL), spinal canal length (SCL), spinal canal volume (SCV), oswestry disability index (ODI), lateral lumbar interbody fusion (LLIF), posterior lumbar interbody fusions with multiple grade II osteotomies (PLIF + MOs).

Variation of parameters according to each surgical method There were statistically significant differences between the two methods with regards to changes in AVCL L1-S1, SCL L1-S1, SCL T1-S1, SCV fusion level, and SCV T1-S1 (p = 0.0001, p = 0.0461, p = 0.0061, p < 0.0001, and p < 0.0001, respectively) ( ns: not significant, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001). LLIF, lateral lumbar interbody fusion; PLIF + MOs, posterior lumbar interbody fusion with multiple grade II osteotomies; AVCL, anterior vertical column length; SCL, spinal cord length; SCV, spinal cord volume.
There were significant differences in the postoperative AVCL (L1-S1), SCL fusion level, and first-standing LL between the groups (p < 0.0001, 0.002, and 0.016, respectively; Table 3). However, postoperative ODI scores did not differ between the groups. Figure 5 shows a representative case of a 73-year-old female with back pain and severe kyphoscoliosis. She underwent spinal deformity correction surgery with LLIF, in which the preoperative AVCL, SCL, and SCV significantly increased after surgery. Representative case of a 73-year-old female with back pain and severe kyphoscoliosis 3D constructed image with AVCL, SCL, and SCV based on CT scan in three planes: axial, sagittal, and coronal (a, d). She underwent spinal deformity correction surgery with LLIF, in which the preoperative SCL and SCV (b), (e) significantly increased after surgery (c), (f). AVCL, anterior vertical column length; SCL, spinal cord length; SCV, spinal cord volume; CT, computed tomography; LLIF, lateral lumbar interbody fusion.
Discussion
Previous studies attempted to measure the spinal cord or canal using 2D plane curves based on radiographs or CT images.19,20 However, patients with severe kyphoscoliosis often undergo spatial spinal cord deformations, which cannot be accurately represented by a plane curve model because of the complex geometry involved. To address this, we reconstructed 3D models of the spinal canals using high-resolution pre- and postoperative CT images using our method (3D model analysis; Figure 3). Our 3D spinal reconstruction was found to be more accurate than 2D imaging in evaluating spinal geometry due to the potential for inaccuracies caused by overlapping complicated spinal elements in 2D imaging. The primary objective of comparing canal dimensions following different correction methods in ASD surgery is to analyze the efficacy of these techniques in achieving indirect canal decompression. Indirect decompression involves reducing spinal stenosis without direct manipulation of the spinal canal, which can be particularly advantageous in minimizing surgical invasiveness and associated risks. By comparing the canal dimensions pre- and post-operatively, this study aims to assess how effectively each correction method achieves this indirect decompression and its subsequent impact on clinical outcomes. We compared the effects of LLIF and PLIF + MOs on spinal canal dimensions during ASD surgery. LLIF demonstrated an increase in AVCL (L1-S1) and SCL whole spine T1-S1 by 6.5 ± 8.0 mm and 13.8 ± 7.6 mm, respectively, compared with PLIF + MOs. Furthermore, LLIF significantly increased the SCV at the fusion level and the whole spine T1-S1 by 14.5% and 10.6%, respectively, outperforming PLIF + MOs. Our study found a significant difference in the SCL fusion levels between the LLIF and PLIF + MOs groups before the operation, but no such difference in SCV fusion levels (Table 2). Interestingly, while there was no difference in SCL whole spine T1-S1, there was a significant difference in SCV whole spine T1-S1 measurements. The difference is likely due to the indications or three-dimensional deformity patterns for LLIF and PLIF + MOs patients. Firstly, the significantly longer preoperative SCL fusion level in LLIF is naturally influenced by the greater number of fusion levels. On the other hand, there is no significant difference in SCV fusion level between the two groups, and the average value is slightly lower in the LLIF group. Results show that LLIF has an advantage over PLIF + MOs in improving spinal canal length (fusion levels) and spinal canal volume (whole spine).
Ischemic spinal cord injury, stemming from overstretching of the spinal cord during corrective surgeries, has been reported, 21 with maximum distraction of the spinal canal leading to spinal cord injury. Excessive distraction or compression of the spinal cord during these surgeries is a potential trigger of neurological deficits and spinal cord damage.22,23 Animal models have been used to quantify the maximum distraction length that can cause spinal cord injuries.24,25 Moreover, patients with scoliosis have been studied to determine the postoperative changes in SCLs associated with neurological complications.19,25 In our study, we observed an increase in SCL for the whole spine T1-S1; both LLIF and PLIF + MOs showed an increased length (13.8 ± 7.6 mm and 6.8 ± 8.4 mm, respectively) (Figure 4). This increase aligns with the data observed in previous models, such as those presented by Bridwell et al. 19 who reported an average increase of 13.5 mm using whole spine radiograph. Yahara et al. 20 found that the center of the SCL increased by an average of 12.7 mm with Lenke type 1 and 10.1 mm with Lenke type 2 between the T2 and L2 vertebrae.
PLIF + MOs enhance the flexibility of the entire spine for realignment by removing the lamina, facet joints, and ligaments between adjacent segments. 26 PLIF + MOs are simple to perform but can prove challenging in correcting severe sagittal imbalance due to limited posterior column shortening. 27 In severe cases, both the dural sac and cord can be displaced, leading to the reduction or even obliteration of anterior epidural and subarachnoid spaces, resulting in widening of the posterior space. 26 In contrast, LLIF is a modern technique that allows surgeons direct access to the intervertebral space using a lateral approach, either anterior to or through the psoas muscle. This approach provides an alternative to anterior lumbar interbody fusion with instrumentation, posterior lumbar interbody fusion, and transforaminal lumbar interbody fusion for anterior column support 28 and has gained popularity since its introduction. LLIF can open the foramen and restore the disc height by inserting substantial interbody cages, resulting in a 35% increase in the average foraminal area, a 70% increase in the posterior intervertebral height, 29 and an increase in the central canal diameter of 33.1%. Oliveira et al. reported a 42% increase in the average intervertebral disc height and a 33% increase in the central canal diameter after LLIF. 30 Furthermore, the central spinal area of the dural sac gradually expands over time. Ohtori et al. demonstrated that spinal stability can induce changes in the lumbar ligamentum flavum and cause spinal canal remodeling. 31 Additionally, Elowitz et al. observed an improvement in clinical outcomes even in patients who experience only a modest increase in the spinal canal area following LLIF. 32 Compared with data from the central spinal area, SCV can offer a more comprehensive assessment of the compressed segment of the spinal cord. Given that the spinal cord is unified from rostral to caudal, compensatory phenomena arise within the spinal cord and cerebrospinal fluid compartments. Thus, SCV can assist in better assessing the compressed spinal cord segment, especially in severe scoliokyphosis, where multiple vertebrae are compressed, leading to structural changes. As observed in our study, LLIF significantly increased the SCV at the fusion level and whole spine T1-S1 by 9.3 ± 6.2 cm3 (up 14.5%) and 9.0 ± 7.6 cm3 (up 10.6%), respectively, while PLIF + MOs only increased it by 1.9 ± 3.4 mm (up 2.6%) and 1.4 ± 3.5 mm (up 2.0%), respectively. Postoperatively, the first-standing LL of LLIF was smaller than that via PLIF + MOs (39.0° ± 10.6° vs 46.8° ± 10.0°, respectively; p = 0.016), indicating that LLIF might increase foraminal width after cage insertion. Interestingly, postoperative AVCL (L1-S1) and postoperative SCL fusion level with LLIF was also larger than that obtained with PLIF + MOs (210.8 ± 12.6 mm and 195.9 ± 12.4 mm, p < 0.0001; 359.6 ± 42.3 and 313.0 ± 50.0, p = 0.002, respectively), demonstrating that LLIF can increase both the flexibility of the lumbar spine and the length of anterior column along the longitudinal axis of the lumbar spine segment (Table 3).
Importantly, shortening the spinal cord length can affect spinal cord compression during surgery, particularly because the spinal cord relies on the three primary arteries for vascular supply. The anterior spinal artery supplies the anterior two-thirds, and the two posterolateral spinal arteries supply the posterior third of the spinal cord. 33 Our research found that the central spinal canal L1-S1 was shortened by both the LLIF and PLIF + MOs methods, by 1.8 ± 2.4 mm and 4.6 ± 5.8 mm, respectively.
Preoperative characteristics revealed significant differences in the number of fusion levels, preoperative TK, and T-score, with p-values of 0.025, 0.023, and 0.002, respectively, between the LLIF and PLIF + MOs groups. These differences may be attributable to the selection of patient populations, particularly in patients with large TK, osteoporosis, and multi-level involvement who may require LLIF surgery to achieve appropriate correction, stability and minimize surgical invasiveness to reduce the risk of complications, making LLIF a suitable option for these patients. 34 The choice of LLIF for patients with large TK, osteoporosis, and multi-level involvement is important because these factors significantly affect the surgical outcome. Large TK requires greater adjustment, which LLIF can provide more efficiently due to its approach and the ability to place cages between intervertebral. Osteoporosis and multi-level involvement require a minimally invasive approach to reduce the risk of complications, making LLIF a suitable option for these patients. In addition, LLIF can allow for better preservation of posterior components, reducing the need for extensive posterior fusion and fixation as in PLIF surgery. 35 However, despite the observed significant differences, the choice of surgery for each patient population did not significantly differ based on our surgical strategy and indication. This suggests that the selection criteria for either surgical approach were applied consistently, demonstrating a strategic adjustment in surgical practice. This adaptation may be influenced by evolving preferences or emerging evidence regarding the efficacy or advantages of LLIF in specific scenarios.
Several other studies have indicated that the most common complications of this procedure are anterior thigh pain and numbness, resulting from irritation of the psoas muscle or transient irritation of the genitofemoral nerve.28,29 Nonetheless, in our study, the ODI scores decreased at the 6- and 12-months follow-ups, although the difference between the two groups was not statistically significant (Table 3). Furthermore, no sensory or motor deficits due to ischemic spinal cord injury or spinal cord compression were during follow-up.
Our study has some limitations. First, the sample size was not sufficiently large to achieve statistically significant results, making it difficult to isolate the effect of the surgical method on spinal dimensions, a small sample size and selective bias in surgical selection, impact the generalizability of the findings. Patients with specific characteristics, such as large TK, osteoporosis, and multi-level involvement, were more likely to undergo LLIF, introducing a bias that makes it difficult to isolate the effect of the surgical method on spinal dimensions. Second, postoperative changes in the spinal cord could not be determined without postoperative magnetic resonance imaging. Therefore, it is necessary to investigate changes in the spinal canal over time after surgery. Third, owing to the use of different vertebral correction methods in our study, the fusion level was different, which may have affected the evaluation of the effectiveness of surgery. In future, a more extensive study with a larger sample size is required.
Conclusions
A comparison between LLIF and PLIF + MOs for correcting ASD revealed significant differences in their effects on various spinal canal dimensions. LLIF can significantly increase spinal canal space, lumbar lordosis, and anterior column length along the longitudinal axis of the lumbar spine segment. Our study provides valuable insights into the effect of different correction methods on spinal dimensions. In future studies, it is important for surgeons to understand how different methods can affect the spinal canal space and cord dimensions during surgery, which may help improve surgical outcomes and prevent neurological complications.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tomohiko Hasegawa and Shin Oe work at a donation-endowed laboratory and are funded by Medtronic Sofamor Danek Inc., Japan Medical Dynamic Marketing Inc., and Meitoku Medical Institution Jyuzen Memorial Hospital. For the remaining authors, none were declared. The submitted manuscript does not contain information about medical devices/drugs.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tomohiko Hasegawa and Shin Oe (Medtronic Sofamor Danek Inc., Japan Medical Dynamic Marketing Inc., and Meitoku Medical Institution Jyuzen Memorial Hospital).
