Abstract
Purpose
The purpose of the present work was to assess the specific effects and underlying mechanisms of Daprodustat (GSK1278863) on skeletal muscle injury induced by ischemia reperfusion (I/R).
Methods
C57BL/6 mice were randomized into the skeletal muscle I/R injury (I/R), Daprodustat (GSK1278863) pretreatment and I/R (I/R + GSK) and sham operation (Sham) groups. The skeletal muscle I/R injury model was established by placing an orthodontic rubber band at the left hip joint for 3 h and releasing it for 3 h. H&E staining, wet weight/dry weight ratio assessment, TUNEL assay, ELISA, qRT-PCR and immunoblot were utilized to assess the effects of Daprodustat.
Results
Daprodustat pretreatment significantly ameliorated apoptosis in skeletal muscle cells, reduced oxidative damage and suppressed inflammatory cytokines. Mechanistically, Daprodustat positively affected NF-κB signaling activation.
Conclusion
These data demonstrated that Daprodustat may provide a potential clinical approach for preventing or treating skeletal muscle injury induced by I/R.
Keywords
Background
Skeletal muscle ischemia reperfusion (I/R) injury is a common clinical challenge in many medical disciplines, including trauma surgery and vascular injury, which is observed in osteofascial compartment syndrome, limb replantation and peripheral vascular injury, and leads to limb and multiple organ dysfunction syndrome and even death.1,2 The complex pathophysiological processes of skeletal muscle I/R injury involve multiple factors, and it is generally agreed inflammation suppression ameliorates skeletal muscle IR injury.3–5 Few treatments have been proven to effectively reduce or prevent skeletal muscle IR injury, whose therapeutic interventions need further investigation
HIF-1 represents an important transcription factor comprising an oxygen sensitive subunit (HIF-1α) and an aryl hydrocarbon nuclear translocator (HIF-1β), with critical regulatory roles in controlling oxygen balance and promoting tolerance to acute ischemia. 6 In normoxia, HIF-1α undergoes hydroxylation by the prolyl hydroxylase domain-containing protein (PHD), which is the recognition site of ubiquitin ligase E3, and rapidly degrades HIF-1 through the ubiquitin protease pathway.7,8 Under hypoxic conditions, the enzymatic activity of PHD is reduced, which then allows HIF-1 to escape degradation and translocate into the nucleus, regulating multiple genes participating in hypoxic adaptation.6–8 These genes control diverse processes such as erythropoiesis, vasodilation, glucose and lipid metabolism, angiogenesis, cell proliferation and apoptosis.6,9 HIF-1αinvolvement has been demonstrated in multiple pathologies, including hypoxia and ischemia. HIF-1αSkeletal muscle preconditioning under hypoxia improved contractile force production, calcium influx and reperfusion injury compared with normal oxygen levels. 10
PHD inhibition can increase endogenous EPO production and enhance iron availability to the erythron, which could alleviate anemia in patients with chronic kidney diseases. Previously reported evidence indicates PHD inhibition reduces I/R injury. For instance, Daprodustat alleviated myocardial, renal and lung I/R injury.11–13 However, the efficacy of the PHD inhibitor Roxadustat (GSK1278863) in the treatment of skeletal muscle IR injury is not fully elucidated. Therefore, this study aimed to examine whether Roxadustat pretreatment reduces skeletal muscle I/R injury in a mouse model and to explore the underlying mechanisms.
Methods
Animals
All animal experiments were carried out as outlined by the Guide for the Care and Use of Laboratory Animals, after approval from the University Institutional Animal Care and Use Committee (IACUC number FJMU 2022-0657). The study was reported according to the ARRIVE guidelines. Eight-week-old, 20–25 g, male C57BL/6J mice were provided by Laboratory Animal Center of the University [Animal License No. SCXK (Fujian) 2020-0005] and housed with freely available food and water under a 12-h photoperiod with 50 ± 10% humidity at 21 ± 2°C.
Grouping and Daprodustat (GSK1278863) pretreatment protocols
Thirty mice were randomly divided into 3 groups: (1) skeletal muscle I/R group (I/R group, n = 10), (2) proline hydroxylase inhibitor GSK1278863 pretreated with I/R group (I/R + GSK group, n = 10), and (3) control group (Sham group, n = 10). Establishment of skeletal muscle ischemia-reperfusion injury model: First, the mice were anesthetized with a flow rate of 1 L/min with 2% isoflurane. After the mice were completely anesthetized, they were placed prone on a heating pad at 37°C to maintain a body temperature of 37°C. Sterile towels were laid on the heating pad, and the limbs of the mice were fixed with adhesive tape. After 3 h, the self-locking nylon cable ties were removed and the mice were put back into the cage for 3 h, and the mice were killed [6]. The I/R + GSK group was prophylactically given proline hydroxylase inhibitor GSK1278863 dissolved in DMSO solution, 50 mg/kg intraperitoneal injection, once a day at a fixed time for 1 week. Sham group and I/R group were given the same dose of DMSO solution in the same way and manner, and the left hip joint of mice in Sham group was not clamped after anesthesia, and the residual treatment was the same as that in I/R group.
Tissue sample collection
After euthanasia, the tibialis anterior muscle and the gastrocnemius muscle of the left hind limb were carefully dissected for biochemical and histological analyses. The proximal 1/4 gastrocnemius muscle underwent fixation with 10% formaldehyde. The remaining part of the gastrocnemius muscle was kept at −80°C for biochemical assays.
Histological examination
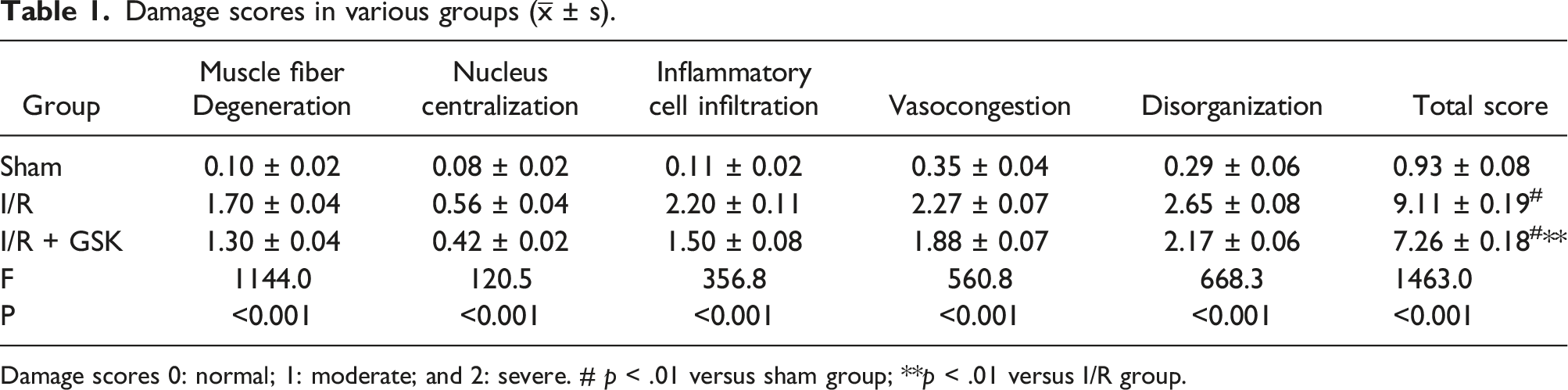
Gastrocnemius muscle fixed with 4% paraformaldehyde underwent dehydration with graded ethanol, paraffin embedding and sectioning at 5 μm. Then, staining was performed with hematoxylin and eosin (H&E), followed by imaging of 10 random high-power fields (20×) per sample under a light microscope (Leica DM2000 LED, Japan) Histological analysis was carried out by three blinded pathologists according to muscle fiber degeneration, nucleus centralization, inflammatory cell infiltration, vasocongestion and disorganization. Each parameter scored as 0 (normal findings, <10% of the specimen affected), 1 (mild findings, 10-30%), 2 (moderate findings, 30-50%) and 3 (severe findings, >50%) points. The overall scores ranged from 0 to 15, as a sum of the sub-scores 14
Wet weight/dry weight (W/D) ratios of muscle tissue samples
Tibialis anterior muscle samples from mice were weighed immediately following extraction from the left hindlimbs (wet weights) and upon dehydration in an oven at 55°C for 48 h (dry weights). The W/D ratio was determined as (wet weight/dry weight) ×100%. 15
TUNEL assay
Paraffin-embedded skeletal muscle sections were assessed with a TUNEL kit (MK1025 Boster, Wuhan, China) as directed by the manufacturer. A light microscope (Leica DM2000 LED, Japan) was utilized for imaging, followed by quantitation with Image J (NIH, USA) and ImagePro Plus 6.0 (Media Cybernetics, USA). Skeletal muscle apoptosis was quantitated by the apoptotic index (amount of apoptotic cells/total amounts of cells). Six randomly selected fields at 200× per sample.
Determination of serum TNF-α, IL-1β, MDA and KYNA levels
Blood specimens underwent a 10-min centrifugation at 3000 r/min for serum preparation. Serum tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), malondialdehyde (MDA) and kynurenic acid (KYNA) concentrations were assessed with specific mouse ELISAs (SEA133Mu, SEA563Mu, CED718Ge Cloud-Clone Corp, Wuhan, China, and E-EL-0060c, Elabscience, Wuhan, China, respectively) as directed by the manufacturers.
Western blotting
After homogenization in a lysis buffer, the BCA Protein Assay Kit (Beyotime Biotechnology, China) was utilized for protein quantitation. Equal amounts of total protein (30 μg) underwent separation by SDS-PAGE, followed by transfer onto PVDF membranes (Millipore Corporation, USA). Next, a 1-h blocking was performed with 5% (w/v) nonfat milk in Tris-buffered saline, and the samples were incubated with primary antibodies targeting β-actin (YT0099 Immunoway, USA), HIF-1α (YT0099 Immunoway, USA) and egln1 (4835 cell signaling, USA) at 4°C overnight. Upon incubation with secondary antibodies, the ChemiDocTMMP imaging system and ImageJ were utilized for gray value assessment, with β-actin as an internal reference.
Quantitative real-time PCR assay
Total RNA extraction from skeletal muscle tissue specimens utilized using TRIzol reagent (Invitrogen, USA) and cDNA was synthesized with PrimeScript RT Reagent Kit (Vazyme, China), as instructed by the manufacturers. Quantitative RT-PCR was performed to quantitate HIF-1α (F, AGATTTGGAGATGCTGGCTCC; R, CAGTGGCAGTGATGGTAGGT) and EGLN1 (F, TGGGCAACTACAGGATAAACG; R, TGTCACGCATCTTCCATCTC) with the SYBR Premix Ex Taq (RR420 A, TAKARA). The housekeeping gene β-actin (F, GGCCAACCGCGAGAAGATGAC; R, GGATAGCACAGCCTGGATAGCAAC) was utilized for normalization, and the 2−ΔΔCT method was utilized for analysis. Amplification was performed at 95°C (5 min), with subsequent 45 cycles at 95°C (5 s), 60°C (5 s) and 72°C (10 s), and final extension at 72°C for 60 s.
Statistical analysis
SPSS (IBM Corp, USA) software was employed for data analysis. The data are presented as mean ± standard deviation (SD). To account for the multiple sets of data and to perform pairwise comparisons, we initially applied one-way ANOVA to assess overall differences between groups. Subsequently, when significant differences were detected, we employed post hoc Tukey’s test for pairwise comparisons to identify specific differences between individual groups. A two-sided p-value of less than 0.05 was considered statistically significant.
Results
There were no major complications during the experiment, and behavioral changes were not observed. Basically, there were no obvious abnormalities during the experiment, and weight changes were not monitored. All experimental animals ate normally.
Effects of GSK1278863 on the pathological changes in mice with skeletal muscle I/R injury
H&E staining was used for evaluating muscle tissue injury. Figure 1 depicts histological changes in various groups. Sham mice had normal muscle morphology, with neat myofibril arrangement and clear structures. By contrast, skeletal muscle cells in the I/R group showed inflammatory cell infiltration, muscle fiber degeneration, vasocongestion, nucleus disappearance, and myoedema. The highest total damage score was obtained in the I/R group. After treatment with GSK1278863, inflammatory cell infiltration, myoedema and vasocongestion in the skeletal muscle were improved. The total damage scores for the skeletal muscle were reduced in the I/R + GSK group in comparison with the I/R group (p < .01). These scores are shown in Table 1. The above findings indicated GSK1278863 prevented I/R injury in the skeletal muscle. In addition, elevated W/D ratios were detected in the I/R group compared with sham mice (p < .01). However, after GSK1278863 pretreatment, W/D ratios for skeletal muscle tissue samples were reduced compared with those of the I/R group (p < .01) (Figure 3(a)). Skeletal muscle tissues after hematoxylin and eosin staining (H&E) staining in various groups. Sham. Normal muscle histology in sham mice. I/R. Marked tissue alterations in the I/R group. I/R + GSK. Histological improvement in the I/R + GSK group. Magnification, 200×. Damage scores in various groups ( Damage scores 0: normal; 1: moderate; and 2: severe. # p < .01 versus sham group; **p < .01 versus I/R group.
Effect of GSK1278863 on cell apoptosis in animals with skeletal muscle I/R injury
Skeletal muscle cell apoptosis is critical for the occurrence and progression of I/R injury in the mouse skeletal muscle.
16
Therefore, TUNEL staining was used to assess the effect of GSK1278863 on IR-induced skeletal muscle cell apoptosis (Figure 2). The cell nuclei in skeletal muscle samples were round or oval, with clearly delineated edges and uniform staining in sham animals; in the I/R injury group, skeletal muscle cells exhibited apoptotic bodies, indicating nuclear fragmentation, and irregular margins. As expected, the I/R injury group had starkly elevated apoptotic index (37.93 ± 2.59%) in comparison with sham mice (7.11 ± 0.66 %, p < .01; Figure 3(b)). Upon GSK1278863 pretreatment (I/R + GSK group), there were less TUNEL-positive cells (20.26 ± 2.84%) compared with the IR group (37.93 ± 2.59%, p < .01; Figure 3(b)). The above data suggested GSK1278863 inhibited skeletal muscle cell apoptosis in mice with I/R injury. Representative TUNEL images of skeletal muscle specimens. Magnification, 200×. (a) TUNEL-positive rates in skeletal muscle cells. (b) W/D ratios of skeletal muscle tissues. #p < .01 versus sham group; **p < .01 versus I/R group. Results are showed as mean ± SD.

Effect of GSK1278863 on oxidative stress in mice with skeletal muscle I/R injury
I/R injury is commonly accompanied by lipid peroxidation.
17
To assess GSK1278863’s effect on oxidative stress in the skeletal muscle in I/R injured animals, MDA amounts were determined. As depicted in Figure 4(c), elevated MDA contents were detected in the I/R group (424.50 ± 25.85 ng/mL) in comparison with sham mice (45.78 ± 7.09 ng/mL, p < .01). However, GSK1278863 administration markedly decreased MDA amounts. These findings suggested GSK1278863 relieved oxidative stress in the skeletal muscle in mice with I/R injury. (a) Serum TNF-α levels. (b) Serum IL-1β levels. (c) Serum MDA levels. (d) Serum KYNA levels. #p < .01 versus sham group; **p < .01 versus I/R group. Results are showed as mean ± SD.
Effects of GSK1278863 on inflammation in mice with skeletal muscle I/R injury
Inflammation is the top mechanism of I/R injury, and TNF-α and IL-1β are critical pro-inflammatory cytokines, whose amounts reflect the severity of inflammation and the extent of skeletal muscle injury in I/R.
18
Serum TNF-α and IL-1β amounts were notably elevated in the I/R injury group (434.34 ± 14.27 pg/mL and 365.95 ± 9.91 pg/mL, respectively) in comparison with sham mice (51.55 ± 4.65 pg/mL and 35.20 ± 4.20 pg/mL, respectively) (Figure 4(a) and (b)). However, GSK1278863 administration markedly reduced TNF-α and IL-1β amounts in comparison with the I/R group. Moreover, the levels of white blood cells in blood samples from the sham (1 ± 0.2 × 109/L) and I/R + GSK (6.67 ± 0.42 × 109/L) groups were significantly reduced compared with those of the I/R group (7.93 ± 0.35 × 109/L) (p < .01, Figure 5(b)). (a) White blood cell levels in blood samples. (b) Hemoglobin levels in blood samples. *p < .05 and #p < .01 versus sham group; **p < .01 versus I/R group. Results are showed as mean ± SD.
Effect of GSK1278863 on hemoglobin levels in mice with skeletal muscle I/R injury
PHD suppression increases erythropoietin and induces red blood cell production in the liver. 19 The I/R group had blood hemoglobin amounts of 115.00 ± 5.00 g/L. Hemoglobin levels in the Sham (126.33 ± 3.51 g/L) and I/R + GSK (138.33 ± 2.08 g/L) groups were significantly increased in comparison with those of the I/R group (p < .05 or p < .01; Figure 5(a)).
Effects of GSK1278863 on NF-κB signaling pathway effectors in mice with skeletal muscle I/R injury
In mice with skeletal muscle I/R injury, GSK1278863’s regulatory effects on NF-κB signaling were determined. 20 qRT-PCR and immunoblot were carried out to assess EGLN1 and HIF-1α mRNA and protein amounts, respectively. As depicted in Figure 5, HIF-1α and EGLN1 mRNA and protein amounts were elevated in the I/R group in comparison with sham animals. After pretreatment with GSK1278863 (I/R + GSK group), qRT-PCR showed higher HIF-1α and lower EGLN1 mRNA amounts in comparison with the I/R group. Similar findings were obtained at the protein level. It is known loss of EGLN1 results in increased levels of KYNA. 23 In this study, KYNA levels were starkly decreased in the I/R group (334.33 ± 8.02 ng/mL) compared with the sham (337.56 ± 6.41 ng/mL, p < .01, Figure 4(d)) and I/R + GSK (338.77 ± 7.55 ng/mL, p < .01, Figure 4(d)) groups. However, there was no significant difference between the sham and I/R + GSK groups (p > .05, Figure 4(d)). Previous findings have indicated HIF-1α regulation by NF-κB signaling, and EGLN1 plays an important role in the HIF-1α-mediated hypoxia signaling pathway.20–23 Taken together, GSK1278863 may reduce I/R injury partly by regulating NF-κB signaling.
Discussion
Skeletal muscle I/R injury represents a commonly problem encountered in clinic, with varying degree of I/R injury of the skeletal muscle and serious effects on prognosis and the quality of life. However, there is no widely accepted treatment at present. 4 HIF-1α, a master regulator of critical adaptive responses to hypoxia, constitutes an important transcriptional factor controlling pathophysiological events whose expression and transcriptional activity show exponential increases with decreasing amounts of cell oxygen.24,25 Ratcliffe and Kaelin proposed that HIF-1α is degraded and activated under normoxic and hypoxic conditions, respectively.26,27 Daprodustat (GSK1278863) is a small-molecule hypoxia-inducible factor (HIF)-prolyl hydroxylase (PHD), which can rapidly reduce HIF activity under normoxic conditions for the treatment of anemia in chronic kidney disease. 28
HIFα expression is also controlled via prolyl hydroxylation by α-ketoglutarate (αKG) dependent-dioxygenases termed EGLNs (also named PHDs). The EGLN family comprises three species, i.e., EGLN1, EGLN2 and EGLN3, with EGLN1 being the major regulator of HIFα. 29 EGLN1 suppression results in increased amounts of circulating αKG, which induces the synthesis and secretion of kynurenic acid (KYNA) in the liver. 21 After treatment with Daprodustat (GSK1278863), KYNA levels were markedly increased. These results showed that Daprodustat (GSK1278863) could decrease EGLN1 expression and induce HIF-1α expression. Furthermore, overexpression of HIF-1α decreased IR-induced inflammation, indicating Daprodustat (GSK1278863) reduces inflammation in IR by increasing HIF-1α expression.
Although the related mechanisms are complex, there is increasing evidence that inflammation is critical in the pathogenetic process of skeletal muscle IR injury.18,30 Following IR injury, inflammatory cells infiltrate and induce the release of multiple pro-inflammatory cytokines. The first inflammatory cells arriving at the injury site are neutrophils, which are successively replaced by monocytes and macrophages.
31
In the present study, two inflammatory cytokines, IL-1β and TNF-α, after 3 h of ischemia and 3 h of reperfusion, were highly expressed in the damaged skeletal muscle, which could be used as inflammatory markers to observe the inflammatory response. Evidence suggests that HIF-1α accumulation and activation are affected by the NF-κB pathway under inflammatory conditions.
20
Hence, we speculated that Daprodustat (GSK1278863) could affect the expression of NF-κB pathway effectors. Our data showed that after GSK1278863 pretreatment, the number of inflammatory cells, TNF-α and IL-1βlevels and white blood cell amounts were markedly decreased. These findings suggested IR induces serious systemic and local inflammatory responses, which are alleviated by Daprodustat (GSK1278863) via the NF-κB pathway. However, the mechanism by which Daprodustat (GSK1278863) affects the expression of NF-κB remains unknown (Figure 6). (a, b, c) Western blot analysis of HIF-1α and EGLN1. (d), (e) qRT-PCR analysis of EGLN1and HIF-1α. *p < .05 and #p < .01 versus sham group; **p < .01 versus I/R group. Results are showed as mean ± SD.
I/R injury is usually accompanied by lipid peroxidation, and MDA levels are widely used as an indicator of lipid peroxidation that reflects the extent of cellular oxidative injury. 17 Here, increased MDA levels in the muscle tissue after I/R and decreased MDA levels after GSK1278863 pretreatment were found, suggesting that oxidative stress might play a role in skeletal muscle I/R injury.
Increasing evidence suggests apoptosis contributes to the pathogenesis of skeletal muscle IR16,32,33; however, the associated pathways remain unknown. As assessed by TUNEL, increased apoptosis in I/R muscle was found in this work, which was significantly decreased by Daprodustat (GSK1278863). Bcl-2 represents the major anti-apoptotic molecule in human cells, which antagonizes the pro-apoptotic factor Bax, suppressing cytochrome C release by mitochondria into the cytosol and downregulating caspases, which are involved in apoptosis during skeletal muscle IR injury. 34
This study has the following limitations. First, our experimental design was built around a preclinical mouse model, and this might not fully capture all the complexities that occur in human skeletal muscle IR injury. The differences between mice and humans might mean that the way the drug behaves in the body and how it affects the body might be different, which might limit how well our findings can be used in real-world clinical situations. Second, the timing of when we gave the daprodustat in our study might not be exactly like in real-world clinical situations. We gave the drug before we caused the ischemia, so we could see if it might help protect against damage. But in real clinical situations, it is often hard to know when IR injury is going to happen and treat it before it does. So, the good effects we saw in this study might not be the same for patients who get daprodustat after they have already been injured. Even though we showed that daprodustat can help protect against damage in mice, we still need to do more studies to figure out how it works, what the best dose is, and if it is safe and effective for a long time.
Conclusions
The current results demonstrated that Daprodustat (GSK1278863) remarkably alleviates IR-induced muscle injury by reducing oxidative stress injury, inflammatory responses and apoptosis, which might be associated with the NF-κB pathway. Therefore, pharmacological preconditioning with Daprodustat (GSK1278863) may represent a viable approach for treating skeletal muscle IR injury.
Footnotes
Authors’ contributions
WQ W, YF Z and Y Z carried out the studies, participated in collecting data, and drafted the manuscript. T K, JY Z, and RB L performed the statistical analysis and participated in its design. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Fujian Province (Grant No. 2018J01241) and Startup Fund for scientific research of Fujian Medical University (Grant No. 2018QH1150).
Ethical statement
Data availability statement
The datasets generated during and/or analyzed during the current study available from the corresponding author on reasonable request
