Abstract
Objectives
Intimal hyperplasia, which is mainly caused by vascular damage during percutaneous coronary intervention, affects the prognosis of patients who undergo percutaneous coronary intervention. However, it remains unclear whether circulating microparticles, which are also affected by percutaneous coronary intervention, participate in intimal hyperplasia.
Methods
In this applied basic research (also identified as a cross-sectional study), microparticles were obtained from healthy participants (n = 20), patients with serious intimal hyperplasia (n = 33), and patients with mild intimal hyperplasia (n = 33) 1 year after percutaneous coronary intervention. After origins testing, the effects of microparticles on the proliferation and migration (crucial processes in intimal hyperplasia) of human coronary artery smooth muscle cells were determined. The expression levels of extracellular signal-related kinase (ERK), p38 mitogen-activated protein kinase (P38), and c-Jun N-terminal kinase (JNK) as well as the production of intercellular cell adhesion molecule-1 and vascular cell adhesion molecule 1 (markers related to oxidative stress, inflammation, and cell differentiation signaling pathways) were also evaluated.
Results
Although the microparticle concentration was higher in patients with mild and serious intimal hyperplasia than in healthy participants, there were no differences in the microparticle concentration between patients with mild and serious intimal hyperplasia. Flow cytometry revealed that the concentration of both endothelial-derived microparticles and platelet-derived microparticles increased in mild and serious intimal hyperplasia. Microparticles derived from patients with mild intimal hyperplasia stimulated the proliferation and migration of human coronary artery smooth muscle cells (partially blocked by PD98059); increased the phosphorylation of ERK and P38, but not JNK; and enhanced the production of intercellular cell adhesion molecule-1 and vascular cell adhesion molecule 1 (blocked by SB20358). All these effects were more pronounced in patients with serious intimal hyperplasia.
Conclusions
The effects of microparticles in patients with intimal hyperplasia may reveal a therapeutic target for intimal hyperplasia.
Keywords
Introduction
Coronary atherosclerotic heart disease (CAHD) is associated with considerably high mortality and disability rates.1,2 However, percutaneous coronary intervention (PCI) has been shown to reduce mortality in CAHD patients. In-stent restenosis (ISR), which manifests mainly as vascular intimal hyperplasia (IH), seriously affects the prognosis of CAHD patients who undergo PCI. Although intervention treatment or prevention measures have been taken for related risk factors, the incidence of IH in CAHD patients after PCI has not improved significantly as expected.3–5
As submicron particles, circulating microparticles (MPs) are mainly cell debris produced by activated or apoptotic platelets, endothelial cells, and monocytes. Our study and previous research have demonstrated that MPs participate in inflammatory reaction, coagulation activation, thrombosis, and endothelial function related to cardiovascular disease.6–10 However, the role of MPs in IH remains unclear. Thus, we conducted this study to determine the effects of MPs on the proliferation and migration of human coronary artery smooth muscle cells (HCASMCs), which play a crucial role in IH, and the potential underlying mechanisms.
Materials and methods
Study population
This study was an applied basic research (identified as a cross-sectional study). Patients with single-vessel CAHD who had serious IH (s-IH; target vessel diameter stenosis ≥50%, n = 33) or mild IH (m-IH; target vessel diameter stenosis <50%, n = 33) 1 year after the implantation of drug-eluting stents were recruited from the Shaanxi Provincial People’s Hospital in 2022. Patients with diseases that may increase the concentration of MPs, including uncontrolled diabetes mellitus, infectious disease, severe trauma, uncontrolled hypertension, multiple sclerosis, renal failure, lupus anticoagulant, or acute rheumatic diseases, were excluded. Twenty age- and sex-matched healthy volunteers were recruited as a control group. We de-identified all patient details. We conducted our study in accordance with the Helsinki Declaration of 1975, as revised in 2024. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 11 Informed consent was obtained from the patients and healthy volunteers. The research was approved by the ethics review board of the Shaanxi Provincial People’s Hospital (The Third Affiliated Hospital of Xi’an Jiaotong University) (2021-1093).
Isolation of MPs
Fasting venous blood samples of all participants were collected (3 mL of blood per participant), and MPs were subsequently obtained via centrifugation. 12 After centrifugation (11,000 ×g, 4°C, 2 min), the upper plasma sample was separated into platelet-poor plasma (50 µL of platelet-poor plasma was reserved for flow cytometry analysis). Then, the platelet-poor plasma was centrifuged (13,000 ×g, 4°C, 45 min) again to obtained MPs (precipitant at the bottom of the centrifugal tube). After the supernatant was discarded, the MPs were resuspended in RPMI1640 (100 µL; Gibco-Invitrogen, Carlsbad, CA) and used within 3 weeks (refrigerated at −80°C after subpackaging). Owing to the large number of MPs required in the follow-up experiments with limited blood samples, we pooled the MPs derived from all patients; this approach also allowed us to account for individual variations.
Flow cytometry analysis
For flow cytometry analysis, 9 50 µL of platelet-poor plasma (stored as mentioned previously) was mixed with 20 µL of fluorescein isothiocyanate-labeled anti-CD41 and phycoerythrin-labeled anti-CD31 in a 12 mm × 75 mm test tube (anticoagulant with ethylenediamine tetraacetic acid). After mixing, the mixture was incubated at room temperature in the dark for 30 min, with manual shaking every 10 min during incubation. After 30 min of incubation, 20 µL of flowmeter calibrator beads (Coulter Beckman Coulter) was added to the antibody-labeled tube before counting and analyzing (the gate size was set to <1 µm). CD31(+)/CD41(−) MPs were defined as endothelial-derived MPs (EMPs), whereas CD31(+)/CD41(+) MPs were categorized as platelet-derived MPs (PMPs).
Proliferation
HCASMCs (ScienCell Technology) were seeded into 96-well plates until the cell density reached 50%, and smooth muscle cell medium (SMCM) with or without PD98059 (20 μM; specific inhibitor of extracellular signal-related kinase (ERK)-1/2, Sigma-Aldrich) was used to preincubate the cells for 30 min before the addition of MPs (3 mg/mL) from each group. The cells were washed with phosphate-buffered saline 3 times after incubation with MPs for 24 h. Then, HCASMCs were stimulated with CCK-8 (a cell proliferation assay kit with an absorbance of 450 nm) for 2 h to test the effects of MPs on HCASMC proliferation.
Migration
HCASMCs were seeded into 24-well plates until the cell density reached 95%, and a line was drawn through the middle of the 24-well plates with a 200-µL pipette nozzle (a “road” without cells can be seen under a microscope). SMCM with or without PD98059 (20 μM; specific inhibitor of ERK1/2) was preincubated with HCASMCs for 30 min before the addition of MPs (3 mg/mL) from each group. Photos (400×) of the “road” were taken before and 12 h after MP incubation. The width of the “road” was measured before and 12 h after MP incubation to analyze the effects of MPs on migration.
Western blot analysis
HCASMCs were seeded into 6-well plates until the cell density reached 95%, after which SMCM with or without MPs (3 mg/mL) from each group was added and incubated for 1 h. Then, the cell proteins were harvested for immunoblotting. 12 Antibodies against p38 mitogen-activated protein kinase (MAPK) (P38; Cell Signaling Technology), ERK (Cell Signaling Technology), c-Jun N-terminal kinases (JNK; Cell Signaling Technology), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH, Proteintech) were used as references. 9
Enzyme-linked immunosorbent assay (ELISA) detection of intercellular cell adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1)
HCASMCs were seeded into 48-well plates until the cell density reached 95%, after which they were preincubated with or without SP600125 (a JNK inhibitor, 10 μmol/L, Sigma-Aldrich) or SB20358 (a p38 MAPK inhibitor, 2.5 μmol/L, Sigma-Aldrich) for 30 min. Subsequently, HCASMCs with or without MPs (3 mg/mL) were added and incubated for 1 h. Then, the ICAM-1 and VCAM-1 present in the supernatants were detected via ELISA. Briefly, monoclonal antibodies (mAbs) against ICAM-1 (Sigma-Aldrich) and VCAM-1 (Sigma-Aldrich) were coated on microplates, and the supernatants (100 μL) were incubated with these mAbs for 30 min. Then, the captured proteins were detected after incubation with secondary antibodies for another 30 min. ICAM-1 and VCAM-1 levels were tested using a microplate plate reader (Safire, Tecan Infinite 200, absorbance at 450 nm) after the addition of horseradish peroxidase (HRP) substrate (TMB) and HRP-linked antibody. The dry weights of aortic samples were obtained to homogenize the ICAM-1 and VCAM-1 levels (ng/mg protein).
Statistical analysis
All data were presented as mean ± SD values and were analyzed using Graph Pad Prism (version 5.0). χ2 analysis was used to compare proportions between the groups. One-way analysis of variance was used for multigroup comparison, and the Bonferroni correction for multiple comparisons was used for intergroup comparisons. Differences were considered significant when P-values were <0.05.
Results
Clinical data
With the exception of plasma lipoprotein and medications, all clinical characteristics were similar between the healthy volunteers and patients (Table 1).
Clinical characteristics of the study participants.

The values are presented as mean ± SD values.
m-IH: mild intimal hyperplasia; s-IH: serious intimal hyperplasia; TC: total cholesterol; TG: triglyceride; HDL: high-density lipoprotein; LDL: low-density lipoprotein
vs. Healthy, P < 0.05.
Plasma MP concentrations
As shown in Figure 1, compared with the control group (2.65 ± 0.52 mg/mL, n = 20), the plasma MP concentrations were slightly elevated in m-IH patients (3.72 ± 0.78 mg/mL, n = 33), whereas they were clearly increased in s-IH patients (5.16 ± 0.48 mg/mL, n = 33). Although the MP concentration clearly increased in s-IH patients, there was no linear relationship between the MP concentration and the degree of stenosis (data not shown).

MP concentrations are increased in patients with m-IH or s-IH. MP: microparticle; m-IH: mild intimal hyperplasia; s-IH: serious intimal hyperplasia.
Origin of MPs
Compared with the control group (Figure 2(a) and (b); where few EMPs and PMPs were detected), EMPs (Figure 2(c); Q2 area; Figure 2(d), 39.03% ± 3.56%, n = 33) and PMPs (Figure 2(c), Q1 area; Figure 2(d), 35.58% ± 3.13%, n = 33) accounted for the majority of the MPs in m-IH patients. Among s-IH patients, a larger proportion of EMPs (Figure 2(e), Q2 area; Figure 2(f), 44.39% ± 3.75%, n = 33) versus PMPs (Figure 2(e), Q1 area; Figure 2(f), 32.82% ± 4.24%, n = 33) were detected. These results demonstrated that both EMPs and PMPs (especially EMPs) may play the key functional role of MPs. However, there exists no method to isolate EMPs/PMPs from the blood for our subsequent experiments. Therefore, we used MPs for the following tests.
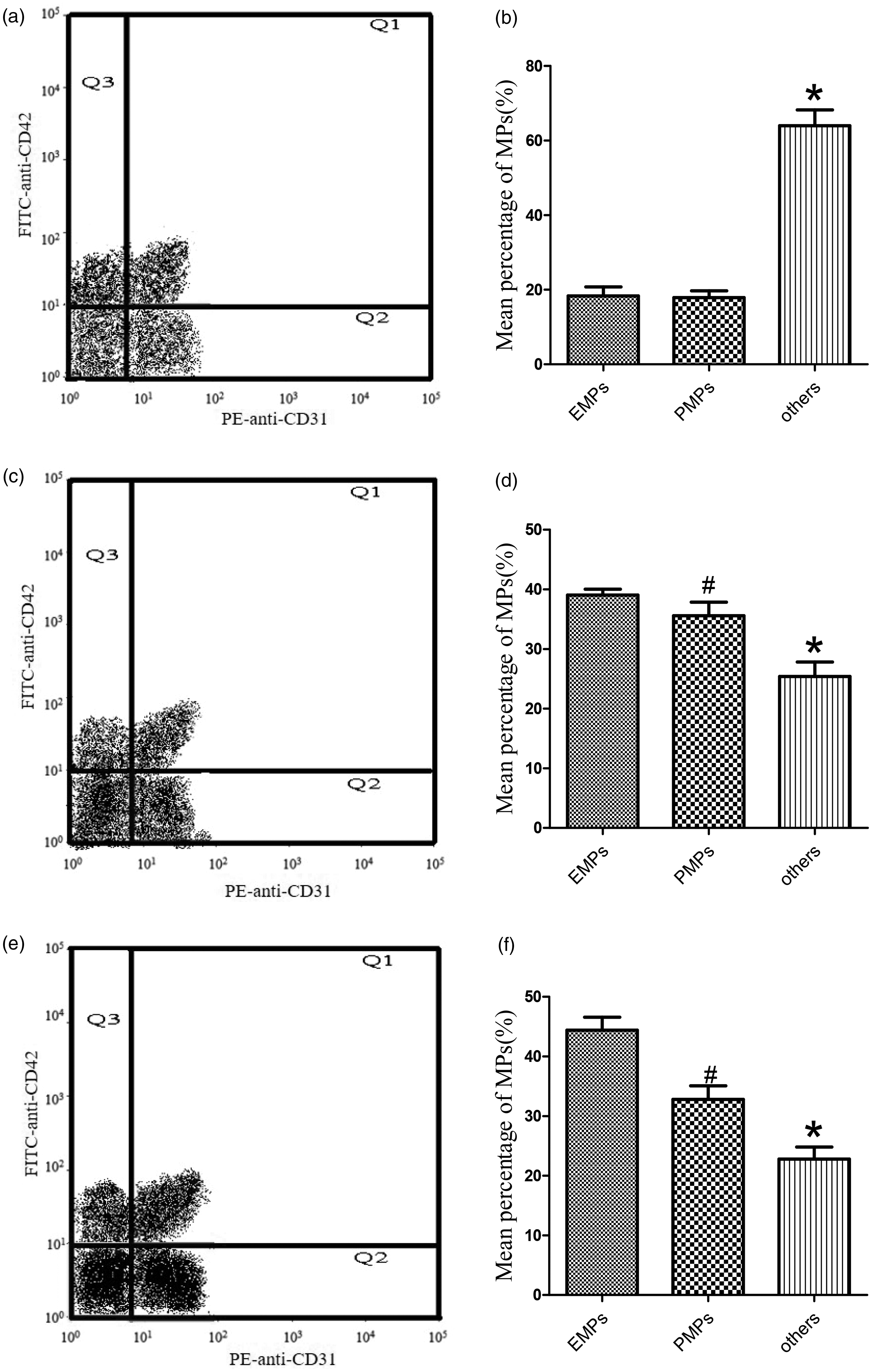
Origins of MPs in each group. (a, b) Origins of MPs from healthy participants: Few EMPs (Q2 area) and PMPs (Q1 area) were detected (n = 20). (c, d) Origins of MPs from m-IH patients: EMPs (Q2 area, (c); 39.03% ± 3.56%, (d)) and PMPs (Q1 area, (c); 35.58% ± 3.13%, (d)) constitute approximately 75% of MP component, and other MPs whose origin was not tested (others, (c); 25.39% ± 3.51%, (d)) constitute the remainder (n = 33). (e, f) Origins of MPs from s-IH patients: Compared with m-IH, EMPs increased (Q2 area, (e); 44.39% ± 3.75%, (f)), whereas PMPs (Q1 area, (e); 32.82% ± 4.24%, (f)) and other MPs whose origin was not tested (others, (e); 22.79% ± 3.32%, (f)) decreased slightly (n = 33). *P < 0.05 vs. EMPs and PMPs; #P < 0.05 vs. EMPs. MPs: microparticles.
Origins of MPs from healthy participants
Few EMPs (Q2 area) and PMPs (Q1 area) were detected (n = 20).
Origins of MPs from m-IH patients
EMPs and PMPs constituted approximately 75% of the MP component, and other MPs whose origin was not tested (others, Figure 2(c); 25.39% ± 3.51%, Figure 2(d)) constituted the remainder (n = 33).
Origins of MPs from s-IH patients
Compared with m-IH patients, the proportion of EMPs increased, whereas those of PMPs and other MPs whose origin was not tested (others, Figure 2(e); 22.79% ± 3.32%, Figure 2(f)) decreased slightly (n = 33).
MPs derived from s-IH patients can stimulate HCASMC proliferation, whereas those derived from m-IH patients can stimulate HCASMC proliferation only slightly. Preincubation with PD98059 an (inhibitor of ERK1/2) can partly block the effect of MPs (both m-IH and s-IH) on HCASMC proliferation.
Effects of MPs on the proliferation of HCASMCs
As the core process of IH, the proliferation of HCASMCs was tested after incubation with MPs. Compared with the control group, MPs from both m-IH and s-IH patients (especially s-IH patients) promoted HCASMC proliferation. The ability of MPs to stimulate HCASMC proliferation can be largely blocked by PD98059 (a specific inhibitor of ERK1/2; Figure 3), which indicated that MPs participated in HCASMC proliferation through ERK1/2.

Effects of MPs from patients with m-IH or s-IH on HCASMC proliferation. MPs: microparticles; m-IH: mild intimal hyperplasia; s-IH: serious intimal hyperplasia.
Effect of MPs on the migration of HCASMCs
Similar to proliferation, the migration of HCASMCs was tested after incubation with MPs. Compared with the control group, MPs derived from both m-IH and s-IH patients (especially s-IH patients) promoted HCASMC migration. The ability of MPs to stimulate HCASMC migration can be largely blocked by PD98059 (a specific inhibitor of ERK1/2; Figure 4), which indicated that MPs participated in HCASMC migration through ERK1/2.

Effects of MPs from patients with m-IH or s-IH on HCASMC migration. (a) Migration without stimulation (0 h). (b) Migration after incubation with MPs and PD98059 (12 h). (c) Percentage of migration conversion (12 h/0 h). MPs from s-IH patients evidently stimulated HCASMC migration, whereas MPs from m-IH patients stimulated HCASMC migration only slightly. Preincubation with PD98059 (an inhibitor of ERK1/2) can partly block the effect of MPs (both s-IH and m-IH) on HCASMC migration. The data are presented as the means ± SD values, * vs. control; # vs. m-IH; λ vs. m-IH and s-IH + PD, P < 0.05. MPs: microparticles; m-IH: mild intimal hyperplasia; s-IH: serious intimal hyperplasia; HCASMC: human coronary artery smooth muscle cell.
Effects of MPs on ERK, P38, and JNK expression
To investigate the mechanism by which MPs affect the biological functions of HCASMCs, the expression levels of ERK, P38, and JNK in HCASMCs after incubation with MPs were detected using western blot analysis. Compared with the control group, MPs derived from both m-IH and s-IH patients (especially s-IH patients) increased the phosphorylation of ERK and P38 but had no effect on the expression of JNK (Figure 5). These findings indicate that MPs affect the biological functions of HCASMCs through the ERK and P38 signaling pathways.

Effects of MPs derived from patients with m-IH or s-IH on ERK, P38, and JNK expression. (a) MPs from both m-IH and s-IH patients, especially s-IH patients, increased ERK phosphorylation. (b) MPs from both m-IH and s-IH patients, especially s-IH patients, increased P38 phosphorylation. (c) MPs from patients with m-IH or s-IH had no effect on JNK expression. *P < 0.05 vs. m-IH and control; #P < 0.05 vs. m-IH Owing to the close molecular weight of the target protein, the gels/blots from different groups were cropped. However, all samples derived from the same experiment were processed in parallel. MPs: microparticles; m-IH: mild intimal hyperplasia; s-IH: serious intimal hyperplasia; ERK: extracellular signal-related kinase; JNK: c-Jun N-terminal kinase; P38: p38 mitogen-activated protein kinase.
Effects of MPs on ICAM-1 and VCAM-1 levels
As a downstream signaling pathway of oxidative stress and inflammatory response, ICAM-1 and VCAM-1 levels in HCASMCs after incubation with MPs were detected using ELISA. Compared with the control group, MPs derived from both m-IH and s-IH patients (especially s-IH patients) increased ICAM-1 and VCAM-1 levels (Figure 6). However, these effects can be largely blocked by SB20358 (a specific inhibitor of p38) but not by SP600125 (a specific inhibitor of JNK) (Figure 6). These findings indicate that MPs activate oxidative stress and the inflammatory response through the P38 signaling pathway.

Effects of MPs derived from patients with m-IH or s-IH on ICAM-1 and VCAM-1 levels. (a) MPs from patients with both m-IH and s-IH, especially those with s-IH, increased ICAM-1 production. (b) MPs from patients with both m-IH and s-IH, especially those with s-IH, increased VCAM-1 production. These effects were largely blocked by SB20358 but not by SP600125 and (c) Hypothetical working model. *P < 0.05 vs. control; #P < 0.05 vs. m-IH and s-IH + SB; γ P < 0.05 vs. m-IH. MPs: microparticles; m-IH: mild intimal hyperplasia; s-IH: serious intimal hyperplasia; ICAM-1: intercellular cell adhesion molecule-1; VCAM-1: vascular cell adhesion molecule 1.
Discussion
This study demonstrated that the concentration of plasma MPs was higher in patients with m-IH or s-IH than in healthy individuals. A majority of the MPs derived from patients comprised EMPs and PMPs. However, in s-IH patients, EMPs, not PMPs, accounted for the majority of MPs. MPs derived from patients with m-IH or s-IH, especially s-IH patients, stimulated HCASMC proliferation and migration (which can be partially blocked by PD98059). Further mechanism testing revealed that MPs promoted the phosphorylation of ERK and P38 as well as increased the production of ICAM-1 and VCAM-1 in HCASMCs (which can be blocked by SB20358).
The concentration of MPs is increased in many diseases that are harmful to the vascular system.13–16 Our previous study demonstrated that plasma MP concentrations are increased in acute coronary syndrome patients, and these MPs can impair vascular function by inhibiting the endothelial nitric oxide synthase (eNOS) expression, uncoupling the association of eNOS with heat shock protein 90, and increasing the NO level but decreasing the O2˙− levels. 9 Our next study revealed that MPs from acute myocardial infarction patients subjected to PCI can further damage vascular function by disrupting the balance of NO and O2˙−. 17 Brodsky et al. 18 reported that EMPs impair acetylcholine (Ach)-induced vasorelaxation in a concentration-dependent manner by increasing O2˙− generation and that EMPs act as a marker of endothelial dysfunction. Similar to the findings of our study and other previous research, in the present work, we found that the concentration of MPs increased in patients with m-IH or s-IH (especially s-IH patients; Figure 1). EMPs account for the majority of MPs from s-IH patients (Figure 2). It has been reported that oxidative stress, such as an imbalance of NO and O2˙−, can result in vascular tone damage, vascular smooth muscle cell proliferation, and cell adhesion. 19 Thus, we assessed the influence of MPs derived from patients with m-IH or s-IH on HCASMC proliferation and migration. MPs from patients with m-IH or s-IH, especially s-IH patients, stimulated HCASMC proliferation and migration (which can be partially blocked by PD98059; Figure 3 and Figure 4).
Although PCI is an effective method for CAHD treatment, the occurrence of IH after PCI remains challenging both in clinical and fundamental research.20–23 Given the overlap between IH and MPs in terms of cell adhesion and inflammatory infiltration, we determined whether MPs participated in IH in the present study. MPs from patients with m-IH or s-IH, especially s-IH patients, induced the production of ICAM-1 and VCAM-1 (Figure 6). As a cell surface glycoprotein, ICAM-1 can be stimulated by various types of inflammatory and oxidative stressors. 24 Moreover, the deletion of ICAM-1 or the inhibition of its ability can prevent inflammation. 25 VCAM-1 is mainly expressed on vascular cells and is activated during the inflammatory response, which can be used as a biological indicator of poor prognosis in CAHD patients. 26 On the basis of the above research results,24–26 our study demonstrated that MPs from patients with IH could activate inflammatory and oxidative stress reactions by increasing the levels of ICAM-1 and VCAM-1.
As a MAPK, ERK plays a key role in cell proliferation, migration, invasion, and differentiation. 27 Similarly, as key proteins of the MAPK signaling pathway, P38 signaling pathways are involved in the pathophysiological process of ischemic brain injury and are key signaling pathways involved in blood–brain barrier injury. 28 Another study 29 revealed that diosgenin can regulate oxidative stress and inflammatory response by downregulating the P38 signal pathway and inhibiting its phosphorylation, thereby playing a protective role against CAHD. Our study revealed that MPs from patients with m-IH or s-IH (especially s-IH patients) increased the phosphorylation of ERK and P38, but not JNK (Figure 5). Previous studies27–29 have shown that MPs may participate in the pathological process of IH by activating oxidative stress and inflammatory response through the ERK and P38 pathways.
Limitations of the study
A mixture of PMPs, EMPs, and other materials was used in our study because there are currently no available methods for isolating single-origin MPs from human plasma. In addition, monitoring changes in the concentration and function of MPs at different postoperative time periods (such as 1, 3, 6, 9, and 12 months after PCI) may be more helpful. Finally, the small sample size and the use of stent restenosis as a marker of IH severity in the present study limit the accuracy of our findings. ISR is influenced by several other factors in addition to endometrial hyperplasia.
Conclusion
In summary, our study showed that MPs may result in or exacerbate the development of IH in patients who underwent PCI. Our findings may provide a novel therapeutic target for the prevention and treatment of IH.
Footnotes
Acknowledgements
The authors would like to thank the patients and staff at Shaanxi Provincial People’s Hospital for their assistance throughout this study.
Author contributions
F-J C made substantial contributions to study conception and design. X-L W and W-Q H implemented and conducted the experiment. W Z, Z L, and Y-J Y acquired, analyzed, and interpreted the data. X-L W, K Y, and W-Q H drafted the manuscript and revised it critically for important intellectual content. K Y and F-J C provided final approval of the version to be published.
Availability of data and materials
All data are available in the manuscript. Reprint requests can be directed to the corresponding author.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
No funding was received for this study.
Patient consent for publication
Informed consent was obtained for the publication of patient data.
