Abstract
Introduction
Balance control strategies in the human body include the ankle, hip, and step strategies, which integrate sensory information and involve different reflex circuits and cortical control of the upper and lower limbs. 1 When the posture is slightly disturbed by external or internal factors, the body adjusts its stability using an ankle strategy primarily driven by the tibialis anterior (TA) and gastrocnemius medial (GM) muscles.2,3 Surface electromyography (sEMG) is a noninvasive, objective, and real-time evaluation method for analyzing muscle activation. Observing the activation characteristics and influencing factors of the ankle plantar flexion and dorsiflexion muscles under different instability conditions can enhance our understanding of ankle strategies for application in balance recovery training and postural control rehabilitation.4–6
Several studies have investigated factors related to the activation of ankle plantar flexion and dorsiflexion muscles. Vestibular information is an important factor that influences the ankle strategy. Horak performed research on individuals with vestibular loss while walking on tilting surfaces and found that the onset of ankle dorsiflexion was delayed and ankle plantarflexion inhibition was delayed or absent. 7 Aging is another critical factor that influences the posture control response. Tsai found that older adults experiencing an unexpected external perturbation on a six-degree-of-freedom platform showed increased EMG activity in their lower limbs as a compensation strategy for their decreased postural control ability. 8 In addition, vision, proprioception, load lifting, sudden lower limb inversion, and upper limb movement can affect ankle muscle activation.9–13
Among numerous influencing factors, unstable standing surfaces and single-leg standing have become outstanding research topics. Studies have shown that standing on unstable surfaces and single-leg standing (SLS) induce increased activation of ankle plantar flexion and dorsiflexion muscles to maintain standing stability.14,15 During rehabilitation, an unstable surface or single-leg standing position is often used to increase ankle muscle activation, thereby improving ankle stability. 16 Munoz-Martel observed the activity of lower limb muscles during both-leg standing (BLS) or single-leg standing on different standing stability surfaces and found that unstable situation and single-leg standing increased muscle synergy. 17 Borreani described the activation of ankle flexor extensor muscles by employing unstable platforms and resistance and found that single-leg standing on a soft surface with resistance induced the greatest electromyographic activity. 9 However, Harput suggests that ankle muscle activity is not related to standing platforms. 18 Di performed a study to assess the co-contraction of ankle muscles and provided a normal reference range for CI. 19
From this, it can be seen that previous studies have explored ankle muscle activity on suddenly unstable surfaces, normal gait, and when standing with both legs or a single leg. However, inconsistent results from different studies still exist, and reference frames for GL/TA co-contractions are still needed. Furthermore, unstable surfaces and the type of standing position are important for balance rehabilitation; however, the activation strategy of ankle muscles when standing on different persistent unstable surfaces during different types of standing positions is still unclear. This study aimed to evaluate the electromyographic activity of ankle plantar flexion and dorsiflexion muscles in healthy young adults during single-leg standing and both-leg standing positions on flat ground (FG), soft mat (SM), and BOSU ball (BB) surfaces, and to provide insights into the neuromuscular control mechanisms of ankle strategy.
Methods
Participants
Thirty healthy young adults, 15 males and 15 females, participated in this study, with age: 21.40 ± 0.93 years, height: 169.57 ± 6.29 cm, and body mass: 59.50 ± 6.77 kg. The exclusion criteria were pre-existing neuromuscular disorders or leg surgery, and the study protocol was approved by the Ethics Committee of Sir Run Run Shaw Hospital, affiliated with Zhejiang University School of Medicine (Approval No. 2022-75). Written informed consent was obtained from all participants before they participated in the study.
Tasks
The tasks included maximum voluntary isometric contractions (MVIC) and postural control tasks. The MVIC task was based on the study by Hirono et al. For the both-leg standing (BLS) postural control task (see Figure 1), participants stood barefoot on FG, SM, and BB surfaces in a natural position with their heels separated at a comfortable interval distance.
15
Their arms hanging on both sides of the body, and they are required to gaze at a mark placed at eye level. For the single-leg standing postural control task, we asked the participants to lift their left ankle joint toward the left knee joint level. Participants perform the postural control tasks during two standing postures on three surfaces. Six postural control tasks, include (a) BLS on FG, (b) SLS on FG, (c) BLS on SM, (d) SLS on SM, (e) BLS on BB, and (f) SLS on BB. BLS: Both-leg standing; SLS: Single-leg standing; FG: Flat Ground; SM: Soft Mat; BB: BOSU Ball.
Procedure
Participants performed MVIC by exerting maximal effort to contract the TA and GM muscles while maintaining a stationary position. EMG signals were recorded for 3 s during each MVIC task, which was repeated twice for reliability.
The standing task required participants to maintain a standing posture on various surfaces, including both-leg standing and single-leg standing. Both-leg standing involved standing with both legs, while single-leg standing involved standing on one leg. These tasks were performed with the right leg on surfaces such as flat ground, a soft mat, and a BOSU ball. Each standing posture was held for 10 s, during which EMG signals from the TA and GM muscles were recorded. To minimize bias, the order of surfaces and standing postures was randomized for each participant.
Electromyography measurements
Surface EMG of the right leg TA and GM muscles were measured (ME6000 T8, Mega Electronics, Finland) during the MVIC and standing tasks. The EMG electrodes were placed in the middle of the muscle belly. The parameter settings for surface EMG were as follows: sampling at 1000 Hz, bandwidth between 20 and 500 Hz, Amplifier with input impedance of 2 MV, and CMRR of 130 dB.
The MVIC of the TA and GM muscles was conducted twice with an interval of 60 s, and the average electromyography (AEMG) was calculated. In the standing tasks, each task was conducted twice for 10 s with an interval of 60 s. Muscle activation during standing tasks was calculated as the ratio of AEMG during the standing task to AEMG during MVIC and represented as MVIC%. Muscle co-contraction was calculated as the ratio of the AEMG of the TA to that of the GM and represented by the co-contraction index (CI).
20
The assessment strategy is illustrated in Figure 2. Strategy for assessment. Strategies for assessing the activation of ankle plantarflexor and dorsiflexor muscles during single-leg stance and both-leg stance postures on flat, cushioned, and BOSU ball surfaces by surface electromyography observations.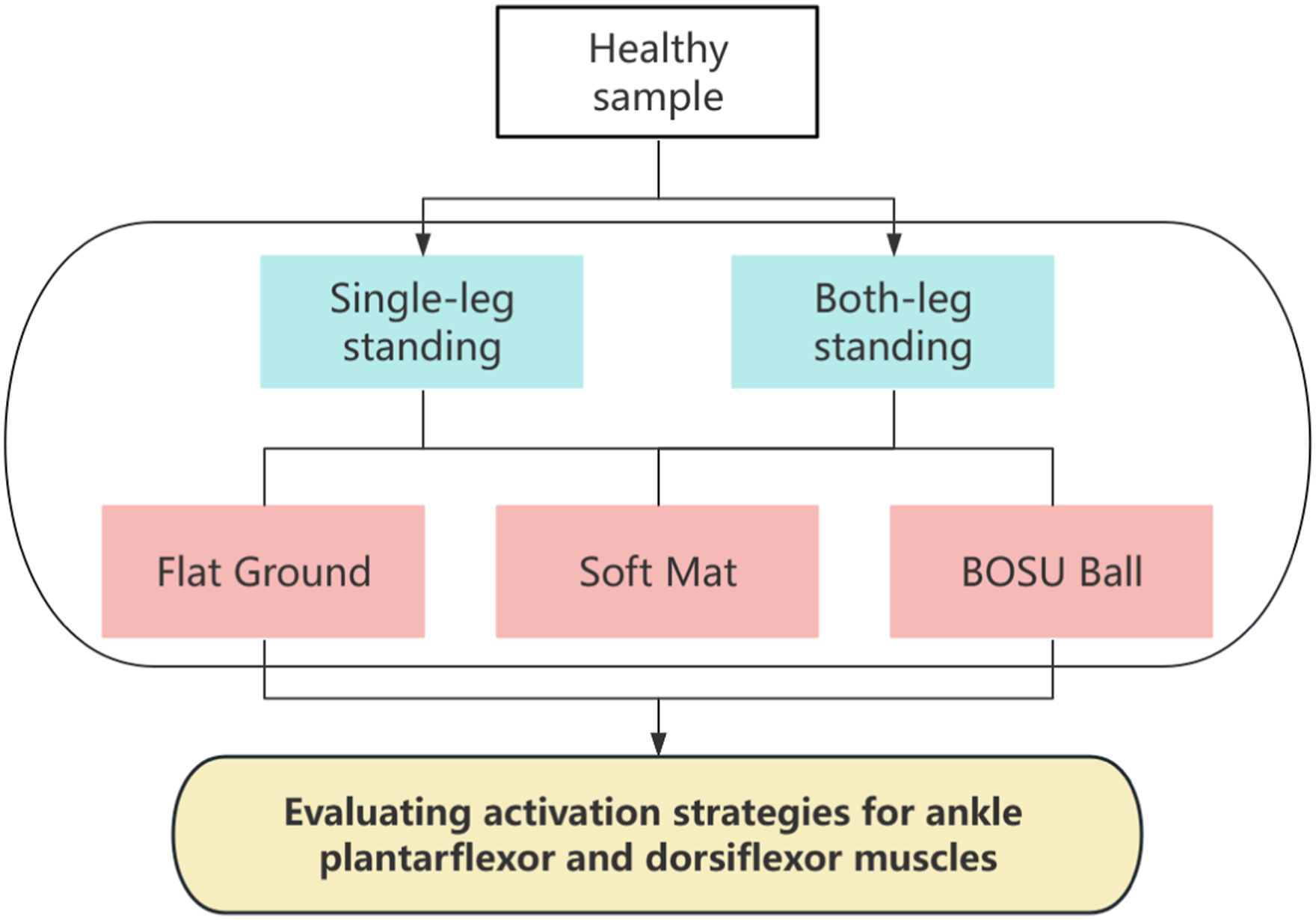
Statistical analyses
All Data are presented as mean ± SD (standard deviation). Differences between the two standing postures and the three surfaces were compared using a paired t test or Wilcoxon signed-rank test.
p < .05 was considered statistically significant. All statistical analyses were performed using SPSS (version 22.0; IBM Corp., Armonk, NY, USA).
Results
Comparison of muscle activation of TA and GM between two standing postures and three standing surfaces
Muscle activation of TA and GM in six postural control tasks.

Data are presented as Mean ± SD. *p < .05, **p < .01, ***p < .001.
MVIC: maximum voluntary isometric contraction; TA: tibialis anterior; GM: gastrocnemius medial; BLS: Both-leg standing; SLS: Single-leg standing; FG: Flat Ground; SM: Soft Mat; BB: BOSU Ball.
Comparison of ankle muscle co-contraction between two standing postures and three standing surfaces
Co-contraction index of ankle flexor extensor muscle in six postural control tasks.
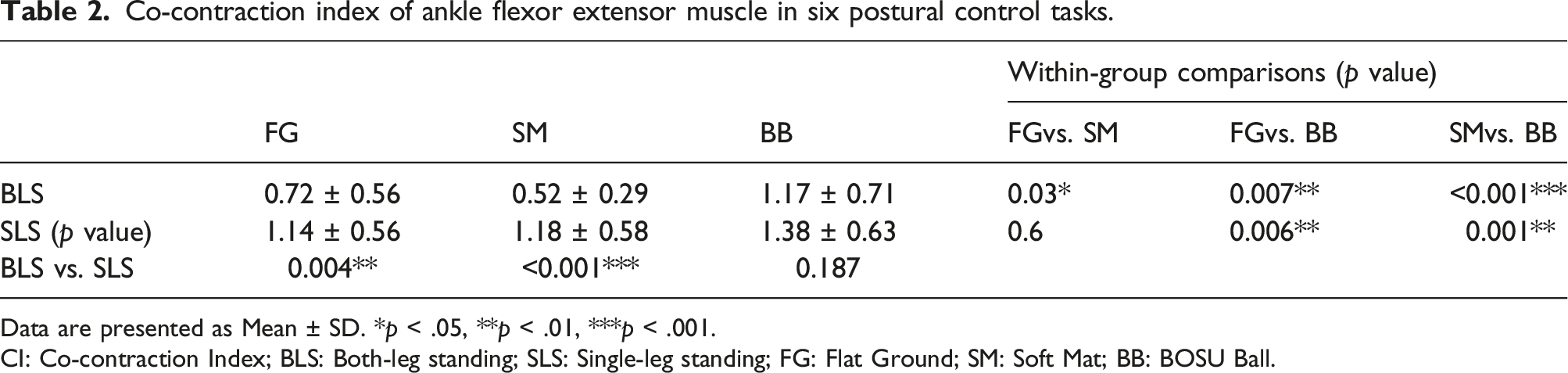
Data are presented as Mean ± SD. *p < .05, **p < .01, ***p < .001.
CI: Co-contraction Index; BLS: Both-leg standing; SLS: Single-leg standing; FG: Flat Ground; SM: Soft Mat; BB: BOSU Ball.

Comparisons of the co-contraction index of the ankle flexor extensor muscle during two standing postures on three surfaces. Comparisons of co-contraction index during (a) Both-leg standing, and (b) Single-leg standing on flat ground, soft mat, and BOSU ball surfaces. The Y-axis represents co-contraction index, and the X-axis represents three different surfaces.
Discussion
Interpretation
Musculoskeletal disorders, such as degenerative lumbar spondylolisthesis 21 and knee osteoarthritis, 22 remain a major burden worldwide. Some academics focus on the basic research of the pathogenesis of musculoskeletal diseases, 23 while others explore the optimization of surgical approaches. 24 Patients diagnosed with musculoskeletal disorders often present with diminished functionality in their lower limb musculature. 25 In the context of rehabilitation research, we investigated the effect of different levels of standing stability on the activation of the TA and GM muscles. We found that compared with BLS, there was a significant increase in the activation of the TA and GM muscles during SLS across all surfaces. The most significant increase is observed on the BB surface.
Comparison with previous studies revealed consistent findings regarding the influence of standing surface instability on ankle muscle activation.18,26,27 However, Hirono’s findings add nuance to this understanding, indicating that not all increases in instability result in further muscle activation; training methods and posture also play a role. 15 Wahl believed that not all increases in instability lead to further increases in muscle activation. 28 Based on these findings, our study meticulously observed the effects of the standing posture on muscle activation on various unstable surfaces, with results emphasizing the intricate response of muscle activation to different degrees of standing stability.
The central nervous system (CNS) may simplify motor production by activating muscles through a coordinated pattern known as co-contraction rather than by activating each muscle individually. 29 Munoz-Martel found that, during SLS, the co-contraction module predominantly involved the ankle muscles, which is consistent with our findings. 17
Further investigation revealed that under mild perturbations (both legs standing on the FG and SM), the activation level of the GM muscle was higher than that of the TA muscle. This may be attributed to specific morphological and anatomical characteristics, namely short muscle bellies, long tendons, and large insertion angles, which result in higher sensitivity of the GM muscle to mild perturbations than the TA muscle, leading to earlier activation. 30 As the perturbations intensified, the contribution of the TA increased continuously. There are two possible reasons for this. First, to achieve greater mechanical stability during standing, participants adopt larger ankle dorsiflexion to lower the center of mass, activating the TA muscle in this posture . 31 Second, apart from co-contraction with muscles such as the GM, the TA also has an inversion function. This may lead to the formation of new synergistic contraction patterns with muscles, such as the peroneus longus, contributing to stability.
In summary, the current study found that standing on surfaces with different stability levels could induce an increase in ankle muscle activation and co-contraction. We suggest that mild standing instability conditions tend to induce significant activation of the GM, which can produce more ankle plantar flexion force to maintain postural stability, whereas severe standing instability and postural stability are often maintained by altering muscle activation strategies.
Limitations
This study had several limitations. This study on healthy individuals can provide a reference for subsequent patient studies. This study mainly focused on the characteristics of the surface electromyography data and did not include a joint mechanical analysis. This study observed immediate changes and short-term effects of ankle muscle activity under different standing stability levels; however, no observations were made after several weeks of training. Further well-designed clinical trials are needed to establish better scientific evidence for the effects of different standing stability levels on ankle muscle activity combined with kinetic and kinematic parameters.
Conclusion
Our results showed that ankle plantar flexion and dorsiflexion muscle activation can be induced by increasing standing instability levels, such as SLS and unstable surfaces; SLS, in particular, is a simple way to create more influence on ankle muscle activation than unstable surfaces. Ankle CI during BLS tends to decrease under mild standing instability conditions and significantly increases under severe standing instability conditions. Variations in CI indicate that individuals can adopt different muscle control strategies based on their standing stability levels. The findings of this study provide insights into the application of balance recovery training and an understanding of the mechanism of ankle strategy during rehabilitation.
Supplemental Material
Supplemental Material - Surface electromyography analysis of ankle flexor and extensor activity under different standing stability levels
Supplemental Material for Surface electromyography analysis of ankle flexor and extensor activity under different standing stability levels by Fangchao Wu, Jiahe Zhang, Kezhen Yang, Yangzhen Li, Zetong Li, Tian Li and Jianhua Li in Journal of Orthopaedic Surgery
Footnotes
Acknowledgments
The authors thank all subjects who participated in this study and the physiotherapists who assisted the implement of protocol.
Author contributions
F.W. and Y.L. collected and analyzed the data and wrote the manuscript. K.Y., J.Z., and J.L. cleaned the data set and edited the manuscript. F.W., T.L., and J.L. reviewed and edited it. F.W. and Z.L. designed the research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zhejiang Medical Science and Technology Project (2022KY180).
Ethical statement
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the study was carried out without any commercial or financial relationships that may be served as a potential conflict of interest.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
