Abstract
Purpose
To understand the systemic effect of major spine surgery, we investigated stress, anabolic and catabolic hormonal levels and their association with interleukin 6 (IL-6) in patients undergoing elective lumbar spine fusion surgery.
Methods
Blood samples were collected preoperatively, and at 1, 3, 42, 90 days postoperatively (POD) from 49 patients who underwent elective lumbar spine fusion surgery.
Results
Serum concentration of cortisol was below the preoperative value at POD 1 but did not differ from the baseline values thereafter. Adrenocorticotropic hormone (ACTH) decreased at PODs 1 and 3. Testosterone decreased at PODs 1 and 3 in men, and at POD 3 in women. Sex hormone-binding globulin decreased at PODs 1 and 3 in both genders. No changes were observed in free testosterone or growth hormone concentrations. Insulin-like growth factor 1 increased significantly above the preoperative level at PODs 42 and 90 in women, and at POD 42 in men. IL-6 was significantly elevated at PODs 1 and 3. Increases in IL-6 from the preoperative level to POD 1 correlated significantly with decreases of cortisol at POD 1 but not with ACTH.
Conclusions
There were only short-term stress hormonal changes after elective lumbar spine fusion surgery. Cortisol changes after elective lumbar spine surgery are transient and might be partly cytokine induced and non-ACTH driven since there was association between cortisol and IL-6 but not with ACTH and IL-6.
Introduction
The stress hormone cortisol is an essential component of the reaction to stress induced by surgical trauma. High cortisol levels during acute stress contribute to the supply of extra energy to vital organs by acutely shifting carbohydrate, fat, and protein metabolism and by delaying anabolism. Moreover, cortisol affects the hemodynamic system by initiating intravascular fluid retention and enhancing inotropic and vasopressor responses to catecholamines and angiotensin II. In addition, the anti-inflammatory effects of cortisol prevent over-activation of the inflammatory cascade. During acute stress, plasma cortisol concentrations are substantially elevated, a phenomenon that has traditionally been explained by the elevation of cortisol production in the adrenal cortex driven by adrenocorticotropic hormone (ACTH) secreted from the pituitary gland. However, evidence has accumulated showing that ACTH is elevated only transiently after severe acute stress (trauma, sepsis or critical illness) whereas cortisol concentrations remain high.1,2
Low plasma ACTH in the presence of high plasma cortisol concentrations has been interpreted as non-ACTH-driven cortisol production, a process in which cytokines could play a role. 3 Alternatively, this phenomenon could be explained by reduced cortisol breakdown suppressing the production of ACTH via feedback inhibition. A study by Boonen et al. showed that cortisol production during a critical illness was only slightly higher than in healthy subjects. 2 Cortisol breakdown was substantially reduced, probably explaining the increased levels of plasma cortisol despite no increase in its production. Both ACTH and cortisol have returned to their normal levels as early as 4–6 days after surgery in patients who have undergone hepatectomy. 4 To our knowledge, no previous studies have investigated cortisol, ACTH and cytokine responses after elective lumbar spine surgery.
The main insulin counter-regulatory hormones are growth hormone, glucagon, adrenalin, noradrenalin and cortisol. Stress-induced endocrine responses ensure the adequate availability of glucose by activating gluconeogenesis, decreasing the insulin sensitivity of muscle cells and increasing the insulin sensitivity of organs predominantly relying on glucose, such as the brain and blood cells. 5
Insulin-like growth factor 1 (IGF-1) is an endocrine mediator of growth hormone (GH)-induced metabolic and anabolic actions, and it has paracrine and autocrine functions. Circulating IGF-1 level is mainly controlled by pituitary GH secretion. Age, gender, smoking status and dietary intake have an effect on IGF-1 levels by affecting GH secretion and GH sensitivity. Cytokines and genetic factors may also influence circulating levels of IGF-1. In general, acute critical illness is characterized by an increase in circulating GH levels and a decrease in IGF-1 levels, indicating relative GH resistance. At least partially, this GH resistance is thought to be responsible for the negative nitrogen balance observed in a critical illness. 6 The knowledge of GH and IGF-1 levels after elective lumbar spine surgery is sparse.
Lumbar spine fusion is performed by using an open midline approach in which the paraspinal muscles are detached from the spinous processes and retracted. Thereafter, the vertebrae are fixed together with transpedicular screws and rods and, if needed, with posterior lumbar interbody fusion is added. The authors have previously reported high interleukin 6 (IL-6) and CRP-response associated to spinal fusion surgery indicating the significant tissue trauma. 7 Perioperative tissue trauma undoubtedly causes an acute stress reaction, which might activate hormonal secretion. However, the mechanisms underlying postoperative hormonal changes, and precisely when the changes occur, remain unknown. Furthermore, it is not known whether elective lumbar spine surgery with stable hemodynamics causes similar hormonal responses than critical illness. Changes in serum hormonal levels caused by elective major lumbar spine surgery can impact on patient recovery and could explain the differences observed between individuals in their rates of recovery and complications. Moreover, it is possible that an association exists between cytokine reaction and hormonal levels induced by surgical trauma. The rise in cytokine levels in response to surgical trauma 7 could also impact hormonal levels after surgery.8–10
This study investigated changes in serum cortisol, ACTH, serum testosterone, sex hormone-binding globulin (SHBG), GH and IGF-1 concentrations during the postoperative trauma and tissue repair process induced by instrumented lumbar spine fusion. A further aim was to assess the associations between interleukin (IL-6) cytokine and hormonal levels.
Materials and methods
Ethical considerations and patient selection
The ethics committee of Tampere University Hospital approved the protocol. All methods were performed in accordance with the relevant guidelines and regulations including the Declaration of Helsinki. Patients aged at least 18 years who provided a signed informed consent and for whom successful blood samples had been taken at the different follow-up time points were included. Patients diagnosed with inflammatory diseases or who had severe cardiovascular, psychiatric, social or general health disorders were excluded from the study. Patients who have hyper- or hypocortisolism, hypo- or hypergonadism, GH deficiency or pituitary gigantism would be excluded according to medical history.
Patients
Altogether 52 consecutive patients with isthmic or degenerative spondylolisthesis or spinal stenosis who underwent an elective instrumented lumbar spine fusion in Tampere University Hospital were recruited for the study. The main indication for surgery was lower limb radicular pain. All operations were performed under general anesthesia in the operating theater. Glucocorticoid or immunomodulatory medication was not administered during the follow-up.
Questionnaires
Participants were asked to complete patient-reported outcome instruments and questionnaires on sociodemographic variables, duration of preoperative symptoms, and back and radicular pain at the time of admission. Disability was captured by the Oswestry disability index (ODI).11,12
Blood samples
Patient blood samples were obtained pre- and postoperatively between April 2010 and November 2011 in Tampere University Hospital, Finland. All samples were collected in the morning before breakfast (before 7.30 a.m.) on the day of surgery, at 1 and 3 days postoperatively in the hospital ward, and at 6 weeks and 3 months postoperatively in a clinical follow-up at the hospital outpatient clinic. Blood was drawn from antebrachial venae into serum vacuum tubes. Centrifuged serum samples were stored at −80C° until assayed with an Immulite1000 analysator. Cortisol, ACTH, IGF-1, GH, testosterone, and SHBG levels were analyzed at each needed time point. Interleukin 6 (IL-6) levels were also analyzed at each time point. Free testosterone was calculated with Anderson’s equation: serum free testosterone (pmol/l) = serum testosterone (nmol/l) × (2.28−1.38 × log (SHBG (nmol/l)/10)) × 10.
Statistics
Data are presented as means with standard deviations (SD), medians with interquartile ranges (IQR), 95% confidence intervals (95% CI) or as counts with percentages. Repeated measures for continuous outcomes (hormones) were analyzed using a generalized estimating equations (GEE) model with an unstructured correlation structure. The Friedman Test or Wilcoxon signed-rank test with post hoc Bonferroni correction was used for analyzing significant differences in hormone levels between each time point or group. Statistical significance was calculated using a permutation test or Fisher’s exact test, or Sidak-adjusted probabilities for baseline differences and IL-6 levels, respectively. Spearman’s correlation was used to obtain information on the associations of hormonal level with age, gender and time from surgery. Correlations between hormonal levels and cytokine IL-6 were also calculated. Analyses were performed using STATA 14.1 or SPSS 24.0.
Results
Clinical and sosiodemographic details, and baseline values for men and women.

ACTH: adrenocorticotropic hormone; IGF-1, BMI: body mass index; GH: growth hormone; insulin-like growth factor 1; L: litre; ml: millilitre; n: number of; nmol: nanomole; ng: nanogram; SD: standard deviation; SHBG: sex hormone-binding globulin; ODI: Oswestry disability index; VAS: visual analogue scale.
p-values calculated using permutation test or fisher exact.
Hormone levels
Serum cortisol concentration decreased only at POD 1 (p < 0.025) and returned to the preoperative level at POD 3 (Figure 1) for both genders combined. However, a significant decrease in ACTH was noted on PODs 1 (p < 0.001) and 3 (p < 0.001) but not on PODs 42 and 90. Changes in serum cortisol and adrenocorticotropic hormone (ACTH) concentrations at postoperative time points in the total group of men and women. The preoperative values have been set at zero (dashed line). Whiskers show 95% CIs. (*p < 0.05, **p < 0.001, ***p < 0.0001).
In men, serum testosterone showed a decrease on PODs 1 and POD 3 but returned to normal by POD 42 (p < 0.001). In women, testosterone significantly decreased on POD 3, but no change compared to baseline level was found in any of the other time points. SHGB concentration decreased in women at POD 1 and 3 (p < 0.001), and in men at POD 3 (p = 0.050). SHBG rose to its baseline level by POD 42 after surgery. However, free testosterone calculated by the Anderson’s equation did not differ from the baseline values in both genders (Figure 2). Changes in serum testosterone and sex hormone-binding globulin (SHBG) concentrations at postoperative time points in men and women. The preoperative values have been set at zero (dashed line). In addition, absolute values of calculated free testosterone (S-Testo-VI) are given at all five time points. Whiskers show 95% CIs. (*p < 0.05, **p < 0.001, ***p < 0.0001).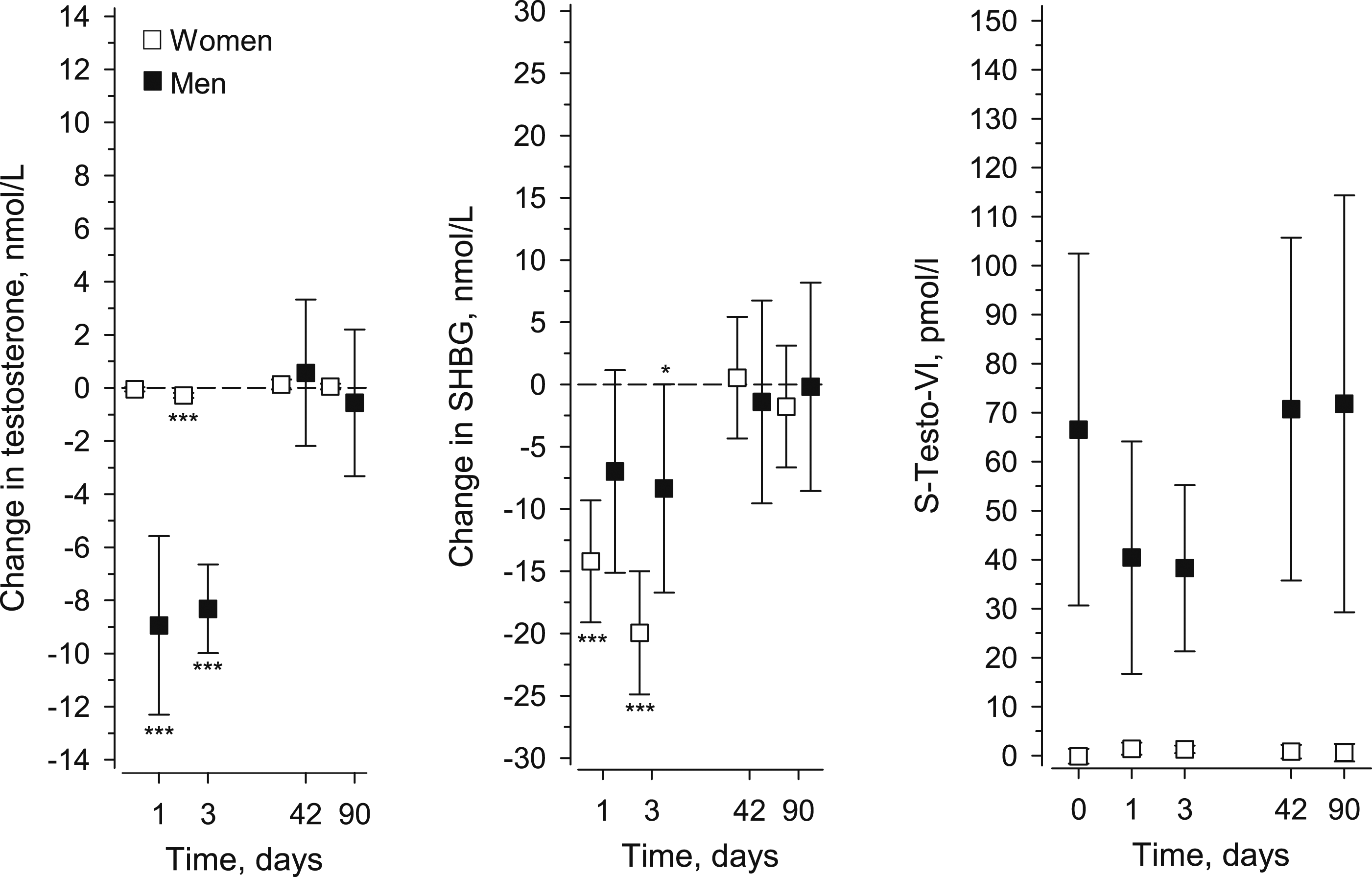
No significant changes were observed in men or women in serum GH concentrations (Figure 3). IGF-1 increased significantly above the preoperative level at PODs 42 (p = 0.043) and 90 (p = 0.008) in women and at POD 42 (p = 0.016) in men after surgery. Changes in serum growth hormone and insulin-like growth factor 1 (IGF-1) concentrations in men and women. The preoperative values have been set at zero (dashed line). Whiskers show 95% CIs. (*p < 0.05, **p < 0.001, ***p < 0.0001).
Correlation between IL-6 and hormones
IL-6 was significantly elevated on PODs 1 and 3 (p <0.001) (Figure 4) and showed a significant correlation with cortisol (p < 0.001) levels on POD 1 (Table 2). No other statistically significant associations were found between IL-6 and hormonal changes on PODs 1 and 3. Change in IL-6 concentrations for postoperative time points in the total group of men and women. The preoperative has been set at zero (dashed line). Whiskers show 95% CIs. (*p < 0.05, **p < 0.001, ***p < 0.0001). Correlation with IL-6 and hormone levels on POD 1 and 3. Note. Statistical significance calculated using Sidak-adjusted probabilities. ACTH: adrenocorticotropic hormone; GH: growth hormone; IGF-1: insulin-like growth factor 1; SHBG: sex hormone-binding globulin. *p < 0.05, **p < 0.01, ***p < 0.001.

Discussion
The present study studied short-term changes in serum stress hormone concentrations after instrumented lumbar spine fusion. Only small and transient cortisol responses returning to baseline within 72 h after lumbar surgery was seen in the current study. Interestingly, cortisol levels were below baseline in POD 1 and returned to normal thereafter. The use of stress doses of hydrocortisone for several days after surgery is a common perioperative practice for patients on glucocorticoid therapy, based upon early case reports of intraoperative adrenal crisis after abrupt withdrawal of glucocorticoids. Understanding the expected magnitude and duration of cortisol production after elective lumbal spine surgery is relevant, especially for patients with primary and secondary adrenal insufficiency and patients receiving chronic treatment with exogenous steroids. In these patients, surgical stress might require an increase of the glucocorticoid replacement dose to avoid a life-threatening adrenal crisis but the magnitude and duration of the stress reaction depends on the surgery. Our study suggests that supplemental exogenous stress glucocorticoids may not be needed after the third postoperative day in the patients undergone elective spinal surgery supporting the previous data. 13 Our study suggests that supplemental exogenous stress glucocorticoids may not be needed to meet the demands of perioperative stress supporting the previous data. 13
Prete et al. found in their meta-analysis that increased cortisol level continues to increase up to the POD 7 after surgery in association with age, sex and invasiveness of the surgical or anaesthetic technique. 13 Our results suggest that postoperatively ACTH is not the only factor affecting cortisol levels since ACTH levels were decreased on POD 1 and POD 3 while cortisol levels normalized. This might be explained by the cytokine-driven cortisol production 3 or reduced cortisol breakdown after surgery suppressing the production of ACTH via feedback inhibition. 2 These results broaden the current knowledge from critically ill patients to patients undergone elective lumbar spine surgery. To the best of the authors’ knowledge, post-surgical cortisol and ACTH levels have not previously been monitored after elective lumbar spine fusion surgery and for as long a period as in the present study.
GH and IGF-I have complex anabolic effects and are important regulators of muscle remodeling. 14 Cortisol and anti-diuretic hormone levels have been shown to increase during lumbar spine surgery and remain elevated at one-hour post-surgery. 15 In the present study GH concentration did not change postoperatively while IGF-1 was above baseline at 1.5–3 months after surgery. The increase in IGF-1 at 6 weeks and at 3 months could indicate a late anabolic effect after surgical trauma and healing process after bone grafting. Further studies would be needed to unravel the effect of IGF-1 on anabolic metabolism. IGF-1 levels are associated with age and gender, although to a lesser degree after age 40. In the present study, no gender differences were observed in the baseline levels of IGF-1. The authors used IGF-1, as all the patients were at an age where the IGF-1 reference values do not change drastically by gender.
The results of the present study show that lumbar spine surgery does not have an impact on free testosterone levels in men. Testosterone and SHBG levels should not be measured immediately after an operation, as the surgery can distort the levels of testosterone and SHBG. Hohman et al. investigated the effects of Nandrolone decanoate on recovery and muscle strength after total knee arthroplasty. 16 Their randomized controlled study found that the use of anabolic steroids led to faster recovery and better muscle strength compared to controls. 16 However, our results do not support the use of testosterone to improve recovery from elective lumbar spine surgery.
Interleukin-6 (IL-6) is a proinflammatory cytokine that is activated by acute surgical trauma. IL-6 could be associated with acute stress hormones.8–10 To the best of the authors’ knowledge, no previous studies have assessed the relationship between acute phase hormones and IL-6 levels. Jameson et al. stated that endogenous corticosteroids modulate the IL-6. 17 The results of the present study also indicate that there might be a postoperative association between IL-6 and cortisol. The results suggest that perioperative cortisol changes might be partly cytokine induced and non-ACTH driven since there was correlation between cortisol and IL-6 but not with ACTH and IL-6. There was no statistically significant correlation between ACTH and IL6. However, there was statistically significant but weak positive correlation between change in cortisol and IL-6 level from preoperative situation to POD one but not from preoperative situation to POD 3 (table 2). The positive correlation between change in IL6 and cortisol levels were probably explained by the subpopulation of patients with increasing cortisol levels and increasing IL-6 levels. Unfortunately, we did not measure cortisol, ACTH and IL6-levels during the day of operation. No association with the other hormones studied, such as GH and testosterone, was found. Further studies with larger cohorts are needed to corroborate this finding.
The prospective setting of this study enabled hormone measurements at distinct time points. The blood samples were obtained in real clinical settings both for ethical reasons and to ensure the minimum inconvenience to patients. The length of follow-up and intervals between measurements can be considered adequate and compare favorably with those reported in previous published studies.9,10,17 The study contained more women than men simply because the prospective unselective sample contained more women. A weakness of the study was the lack of cortisol, ACTH and IL6-levels during and immediately after the lumbar surgery since with those values we could have seen if there is hypercortisolism and disparity of cortisol and ACTH levels already during the surgery. Furthermore, the study does not provide evidence to identify the benefit of using testosterone after elective surgery. Future studies should use samples containing equal numbers of men and women and also stratified by age. As the number of spine surgery is rapidly increasing, 18 it is very important to investigate the effects of elective lumbar spine fusion surgery on hormonal environment and healing process.
Conclusion
The current study showed short-term changes in ACTH, cortisol, GH and free testosterone concentrations after elective lumbar spine fusion surgery. Surgical stress after instrumented lumbar spine fusion induces tissue trauma that elevates IL-6. However, cortisol levels and ACTH levels decreased instead of increased in POD 1 and cortisol levels returned to normal already on POD 3. The late elevation of IGF-1 seems to be associated with late tissue repair process. Further studies could investigate the associations of cytokines and hormones during the operation and whether inter-individual differences exist in hormonal responses to surgical stress and whether surgical stress affects surgical outcomes or complications.
Footnotes
Acknowledgments
The authors would like to thank Hannu Kautiainen for statistical consultation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by the Competitive State Financing of the Expert Responsibility Area of Tampere University Hospital.
