Abstract
Background
Dexamethasone is a corticosteroid with powerful anti-inflammatory effects. This study aimed to explore whether combining intravenous and topical dexamethasone could improve postoperative pain, swelling, and function recovery after total knee arthroplasty (TKA).
Methods
In this prospective, double-blind, randomized controlled study, 90 patients undergoing primary unilateral TKA were randomized into a dexamethasone group, which received dexamethasone (10 mg) by periarticular infiltration during surgery, as well as intravenous dexamethasone (10 mg) before tourniquet release and at 12 h postoperatively; or a control group, which received equal volumes of isotonic saline instead of dexamethasone. The primary outcome was postoperative pain, as assessed on the visual analogue scale (VAS). Secondary outcomes were postoperative consumption of morphine hydrochloride for rescue analgesia, postoperative swelling ratio of the thigh, knee, and tibia; functional recovery in terms of total range of motion (ROM) of knee and daily ambulation distance; postoperative inflammation biomarkers levels of C-reactive protein and interleukin-6; and postoperative complications.
Results
Resting VAS scores at postoperative 6, 12, and 24 h, and VAS scores during motion at postoperative 2, 6, 12, and 24 h were significantly lower in the dexamethasone group. The dexamethasone group also showed significantly less morphine consumption during the first 24 h after surgery and cumulatively during hospitalization, milder limb swelling at 24 and 48 h postoperatively, greater flexion and total ROM on postoperative day 1, and longer ambulation distance on postoperative days 1 and 2, and lower levels of inflammatory biomarkers on postoperative days 1 and 2. The dexamethasone group had significantly lower incidence of postoperative nausea and vomiting.
Conclusion
Compared with placebo, the combination of intravenous and topical dexamethasone can reduce pain, swelling, and inflammation after TKA, it also can improve functional recovery and reduce the incidence of postoperative nausea and vomiting.
Introduction
Total knee arthroplasty (TKA) is one of the most effective surgical interventions for end-stage degenerative knee disease. 1 However, more than 60% of patients undergoing TKA suffer moderate to severe pain after surgery.2,3 Inadequate pain management after surgery often prevents patients from enhanced recovery and increases the incidence of complications.4,5 In addition, postoperative lower limb swelling following TKA are one of the other important causes of dissatisfaction, and contribute to pain, inflammation, reduced range of motion, functional limitation and negative perception of enhanced recovery.6–10 It is reported that the incidence of significant joint swelling after TKA is close to 20%. 8
Enhanced recovery after surgery (ERAS) has been introduced to arthroplasty in recent years and aims to reduce patients’ stress response and postoperative complications, alleviate patients’ pain, and shorten the length of hospital stays in TKA.11,12 Dexamethasone, a corticosteroid with powerful anti-inflammatory and antiemetic effects, has been frequently used in enhanced recovery protocol of TKA.13–15 A recent Clinical Practice Guidelines of the American Association of Hip and Knee Surgeons reported that perioperative intravenous dexamethasone could reduce postoperative pain, opioid consumption, and nausea/vomiting after primary TKA. 16 Topical dexamethasone is less commonly used than intravenous administration. Recent studies have reported that topical dexamethasone can also improve early pain relief and reduce lower limb swelling after TKA.17–19 Up to now, the optimal administration protocol of dexamethasone remains unclear and no studies have explored the efficacy of combining intravenous and topical dexamethasone against postoperative pain, swelling and function recovery after TKA. We assumed that combining intravenous and topical dexamethasone could also reduce postoperative pain and swelling, and enhance recovery after TKA. This prospective, double-blind, randomized controlled trial was conducted to verify this hypothesis.
Materials and methods
This study was approved by the Clinical Trials and Biomedical Ethics Committee of our institution, and written informed consent was obtained from all subjects participating in the trial.
Patient recruitment and randomization
This study recruited osteoarthritis patients who underwent primary unilateral TKA at our institution between February 2021 and April 2022 and who had an American Society of Anesthesiologists (ASA) functional status of I–III. We excluded patients who were diagnosed with rheumatoid, traumatic, or septic arthritis; or who had a knee flexion deformity of ≥30°, a varus-valgus deformity of ≥30°, or known allergies to the drugs used in the present study. We also excluded patients with diabetes mellitus, with a history of open knee surgery, knee infection, analgesic drug dependence or neuromuscular disorder.
All patients were randomly classified into two groups by Investigator one using a computer-generated list of random numbers. Patients in the dexamethasone group received dexamethasone (10 mg) by periarticular infiltration during surgery, as well as intravenous dexamethasone (10 mg) before tourniquet release and at 12 h postoperatively. Patients in the control group received equal volumes of isotonic saline instead of dexamethasone at the same timepoints.
Prior to surgery, Investigator one ensured that the same anesthesiologist prepared all intraoperative experimental drugs in the central pharmacy (see “Standardized surgical procedure”). The surgeon was blinded to the treatment group. Investigator one prepared the postoperative experimental drugs in the pharmacy and injected them in the ward. Investigator 2, who assessed outcomes, was blinded to the treatment group, as was Investigator 3, who performed statistical analysis.
Preoperative management
The base-line patient characteristics were recorded at admission. On the day before surgery, celecoxib (200 mg) was administered twice as a preemptive analgesic.
Standardized surgical procedure
All surgical procedures in this study were performed by the same surgeon at our institution. After general anesthesia, surgery was performed by making a midline skin incision with a medial parapatellar approach. During surgery, cemented prostheses (DePuy Synthes, New Brunswick, NJ, USA) were used. The surgeon performed periarticular infiltration analgesia using 0.2% ropivacaine, 2.0 μg/mL epinephrine, and 0.1 mg/mL dexamethasone for the dexamethasone group, or 0.2% ropivacaine and 2.0 μg/mL epinephrine for the control group. Prior to prosthesis implantation, 20 mL of the analgesic cocktail was injected into the posterior aspect of the capsule, and another 20 mL of the cocktail was used as infiltration analgesic in the medial and lateral collateral ligaments. After prosthesis implantation, quadricep and retinacular tissues were infiltrated with 20 mL of the cocktail, while fat and subcutaneous tissues were infiltrated with another 40 mL of the cocktail. Drainage tubes were not used before the wound was sutured. From skin incision to wound closure, a pneumatic tourniquet was administered at a pressure of 240 mmHg.
Postoperative management
After awakening from general anesthesia, patients were sent to the bed ward, and an ice compress was applied around the incision. Celecoxib (200 mg) and sustained-release oxycodone hydrochloride tablets (10 mg) were administered twice daily to control postoperative pain. If the patient was unable to tolerate the pain, define as whenever the pain score at rest was ≥4 or during motion was ≥6, 10 mg of morphine hydrochloride was injected subcutaneously as rescue analgesia.
Outcomes and follow-up
The primary outcome was postoperative pain at rest and during motion, when patients were asked to flex their knee as much as possible. Patients reported the level of pain along a visual analogue scale (VAS) from 0 to 100 mm, 20 where 0 mm indicated no pain and 100 mm extreme pain. Pain at rest and during motion was measured at 2, 6, 12, 24 and 48 h after surgery.
Secondary outcomes included postoperative consumption of morphine hydrochloride for rescue analgesia, postoperative limb swelling, functional recovery, levels of inflammatory biomarkers, and complications.
Postoperative consumption of morphine hydrochloride for rescue analgesia during the first 24 h after surgery and cumulatively during hospitalization were compared.
Postoperative limb swelling was measured at 12, 24 and 48 h after surgery at three positions: thigh, specifically the 10 cm proximal to the upper pole of the patella; knee, specifically the upper pole of the patella; and tibia, specifically the part where the lower leg showed the largest circumference. The ratios of postoperative circumferences to the preoperative circumference were calculated.21,22
Functional recovery was measured in terms of total range of motion (ROM) of knee and daily ambulation distance. After surgery, the flexion and extension of knee was measured using a protractor, three times per day at 6 h apart, and the best value was used as the day's value. Total ROM was defined as the sum of flexion and extension. Postoperative ambulation started on the morning of postoperative day 1. During hospitalization, after breakfast, patients were requested to walk as far as possible in a single attempt with a walking aid, and the distance was recorded as the daily ambulation distance.
Fasting blood samples were taken on the morning of postoperative days 1 and 2. Samples were assayed for two inflammatory indicators, C-reactive protein (CRP) and interleukin-6 (IL-6).
Complications were recorded, and these included postoperative nausea, vomiting, wound ooze, delayed wound healing (defined as mild wound dehiscence or inflammation, hematoma, or effusion around the wound), postoperative infection, postoperative chronic pain (defined as a VAS score ≥40 mm during daily activities at 3 months after surgery), and venous thrombotic events.
Data were collected on each patient during hospitalization and at 3 months after surgery on an outpatient basis. At follow-up, VAS scores, limb swelling ratio, functional recovery and postoperative complications were assessed.
Statistical analysis
Sample size was based on power analysis of a pilot study involving 50 patients not enrolled in the main study. In order to achieve a difference of 10 mm in inter-group VAS scores during motion at 24 h postoperatively, we calculated that each group would require at least 37 individuals in order to detect a statistically significant difference between the two groups at a two-sided alpha level of 0.05 and power of 90%. To adjust for dropouts, we aimed to enroll 45 patients in each group.
Statistical analysis was performed using SPSS 26.0. The normality of data was assessed using histograms and quantile-quantile plots. Continuous data were presented as mean and standard deviation. Categorical data were presented as numbers or percentages. Inter-group differences in normally distributed data were assessed for significance using Student’s t test; differences in skewed and ordinal data, using the Mann-Whitney U test; and differences in categorical data, using Pearson's chi-squared test or Fisher's exact probabilities test. Differences were considered significant if p < .05.
Results
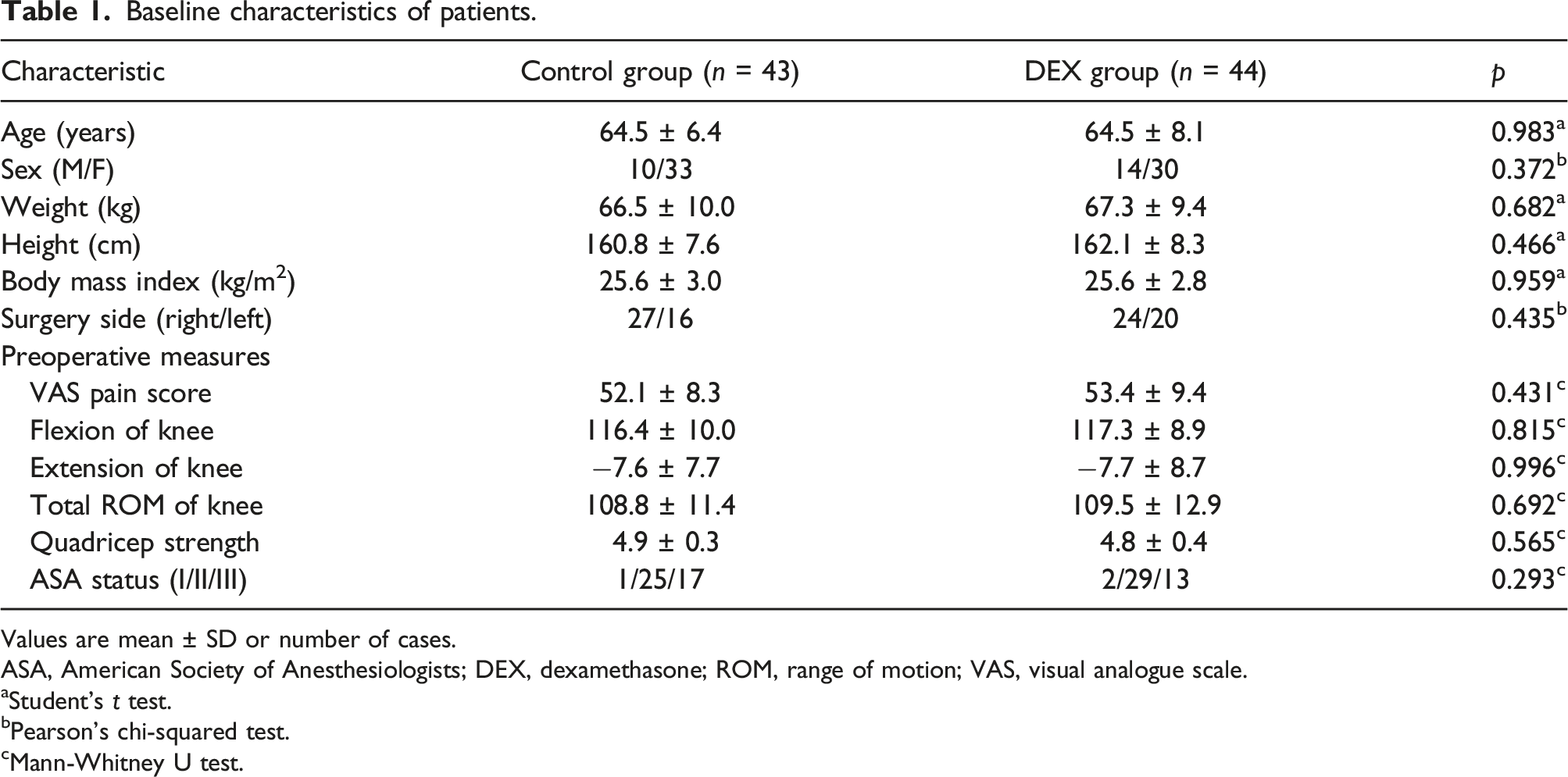
A total of 161 patients were assessed for eligibility, of whom 34 did not meet the eligibility criteria and another 37 were unwilling to give consent. The remaining 90 were randomized into two groups. During postoperative outcome assessments, two patients in the control group and one in the dexamethasone group refused to participate in the outpatient follow-up at 3 months after surgery, so they were excluded from the analysis. In the end, we analyzed 43 patients in the control group and 44 in the dexamethasone group (Figure 1). The two groups showed no significant differences in characteristics before surgery (Table 1). Flow diagram of patient selection and analysis. DEX, dexamethasone. Baseline characteristics of patients. Values are mean ± SD or number of cases. ASA, American Society of Anesthesiologists; DEX, dexamethasone; ROM, range of motion; VAS, visual analogue scale. aStudent’s t test. bPearson’s chi-squared test. cMann-Whitney U test.
Primary outcome: VAS score
Resting VAS scores were significantly lower in the dexamethasone group than the control group at 6, 12 and 24 h after surgery, while VAS scores during motion were significantly lower at 2 h, 6 h, 12 h and 24 h after surgery (Figures 2 and 3). Mean postoperative visual analogue scale (VAS) scores for pain at rest. Error bars show standard deviation. *p < .05 versus the control group (Mann-Whitney U test). DEX, dexamethasone. Mean postoperative visual analogue scale (VAS) scores for pain during motion. Error bars showed standard deviation. *p < .05 versus the control group (Mann-Whitney U test). DEX, dexamethasone.

Secondary outcomes
Postoperative morphine consumption
Postoperative morphine consumption.

Values are mean ± SD.
DEX, dexamethasone.
aMann-Whitney U test.
bPearson’s chi-squared test.
Postoperative limb swelling ratios
Limb swelling ratios after total knee arthroplasty.
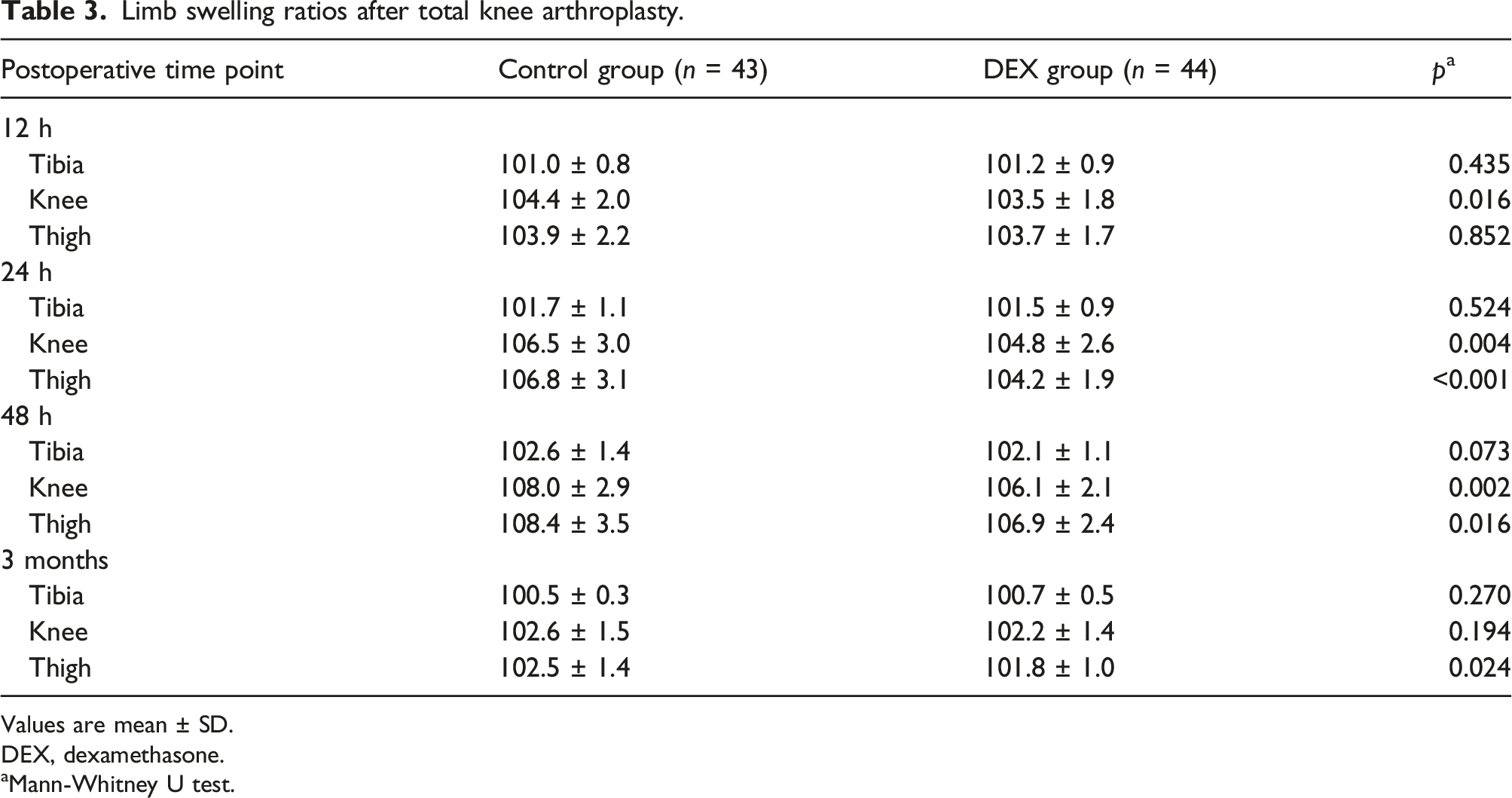
Values are mean ± SD.
DEX, dexamethasone.
aMann-Whitney U test.
Functional recovery
Functional recovery after total knee arthroplasty.

Values are mean ± SD.
DEX, dexamethasone; ROM, range of motion.
aMann-Whitney U test.
Inflammatory indicators
Inflammatory indicators before and after total knee arthroplasty.

Values are mean ± SD.
DEX, dexamethasone.
aMann-Whitney U test.
Complications
Complications after total knee arthroplasty.

Values are number of cases (percentage).
DEX, dexamethasone.
aPearson’s chi-squared test.
bFisher’s exact probabilities test.
Discussion
Dexamethasone, as a classic part of enhance recovery protocol of TKA, its optimal administration methods remain unclear. 16 We explored the efficacy of combining intravenous and topical dexamethasone against postoperative pain, swelling, and function recovery after TKA. The most important finding of the present study was that combining intravenous and topical dexamethasone can reduce pain, swelling, and inflammation after TKA, it also can improve functional recovery and reduce the incidence of postoperative nausea and vomiting, compared with placebo.
Improving postoperative analgesia and knee function after TKA are the major goals for recovery.12,23,24 The dexamethasone group showed significantly lower VAS scores than the control group on postoperative day 1, and the absolute reduction in VAS scores during motion at postoperative 12 h and 24 h exceeded the reported minimal clinically important difference. 25 A recent systematic review included 570 randomized clinical trials on pain management after total hip and knee arthroplasty reported that the median minimal clinically important differences for pain scores were absolute 15 mm at rest (IQR: 10–20) and 18 mm during motion (IQR: 10–20) on a 0–100 mm VAS. 25 With the pain alleviated, the demand for rescue analgesia was significantly reduced in the dexamethasone group. In addition, patients in the dexamethasone group had better function recovery including flexion and total ROM of knee, and ambulation distance. The absolute change in total ROM on postoperative day 1 also exceeded the reported minimal clinically important difference. 26 A previous study has reported that an increase of 10° for total ROM signifies a clinically important improvement. 26 We were unable to compare other outcomes to any thresholds of clinical significance because we failed to find such reports in the literature.
We also found that dexamethasone could reduce lower limb swelling and inhibit inflammation. In our study, levels of two well-established indicators of systemic inflammation, CRP and IL-6,18,27 were significantly lower in the dexamethasone group on postoperative days 1 and 2. Indeed, dexamethasone is known to inhibit phospholipase A2, which reduces production of pro-inflammatory derivatives of arachidonic acid. 28 The efficacy of dexamethasone on reducing pain and lower limb swelling may be related to its powerful ability of anti-inflammation. Previous studies have reported that intravenous dexamethasone can reduce postoperative nausea and vomiting.16,29,30 As expected, our results suggested that combining intravenous and topical dexamethasone could also reduce the incidence of postoperative nausea and vomiting.
Previous three randomized controlled trials compared multiple doses of intravenous dexamethasone to a single dose of dexamethasone in TKA.13,31,32 All three studies reported decreased pain and opioid consumption in the early postoperative period compared to a single dose.13,31,32 Therefore, we chose to administer multiple doses of dexamethasone intraoperatively and 12 h after surgery. However, there is significant heterogeneity in the dose of dexamethasone administered in the current literature. For example, in previous studies, the dose of intravenous dexamethasone administered perioperatively ranged from 4 mg to 20 mg, 16 which may have very different efficacies and the risk of complications. In the present study, we chose a median 10mg as a single dose. Further studies are needed to determine the optimal dose of dexamethasone to optimize its clinical effects while minimizing risks associated with its use. Up to now, there is no gold standard for the perioperative use of dexamethasone in patients undergoing TKA. In addition to the dose, the number of doses, timing, and duration of dexamethasone treatment still need to be further explored.
The contraindications and risks associated with dexamethasone use in TKA remain unknown. In the present study, we excluded patients with diabetes mellitus for concern of affecting their blood glucose levels. However, no studies to date have directly studied any potential implication of administering dexamethasone to TKA patients with diabetes mellitus. Future studies are warranted to investigate if it is safe to use dexamethasone in patients with diabetes mellitus. These patients will require longer follow-up than the perioperative period to see if dexamethasone administered to patients with diabetes mellitus may further increase their already elevated risk for periarticular infection. 33 The use of dexamethasone did not increase the incidence of complications in our study. However, it is unclear if dexamethasone used in TKA are associated with an increased risk of other medical complications that have not been previously reported in the literature, such as avascular necrosis or adrenal-related complications. Future studies are required to investigate whether dexamethasone increase the risks of these medical complications.
As a strong anti-inflammatory drug, the role of dexamethasone has been widely explored in patients undergoing TKA. A lot of studies have reported that perioperative intravenous dexamethasone can reduce postoperative pain, opioid consumption, and nausea/vomiting.13,14,30–32,34,35 A recent study compared intravenous and topical dexamethasone for TKA. This study found that topical administration of dexamethasone provided better clinical outcomes on postoperative pain management and knee swelling early after TKA, while intravenous dexamethasone was more effective in decreasing blood inflammatory biomarkers and preventing postoperative nausea. 17 So far, no studies have explored the efficacy of combining intravenous and topical dexamethasone for TKA. Our study is the first study to combine intravenous and topical dexamethasone in patients undergoing TKA. However, we used placebo as control and we did not compare combining intravenous and topical dexamethasone with intravenous dexamethasone and topical dexamethasone directly. This is the major limitation of this study. Further studies are needed to compare these different routes of dexamethasone directly.
Our study presents several limitations. First, as mentioned above, we compared combining intravenous and topical dexamethasone with placebo, without direct comparisons with intravenous and topical dexamethasone. Further studies are needed to compare combining intravenous and topical dexamethasone with intravenous dexamethasone and topical dexamethasone directly. Second, the sample was small and may not have been sufficiently powered for secondary outcomes. Third, patients were discharged approximately 48 h after surgery, reflecting routine practice at our medical center, and we did not collect data between discharge and follow-up at 3 months. Fourth, this study included only patients with osteoarthritis, so the results may not be generalizable to patients with other primary diseases requiring TKA.
Conclusions
Compared with placebo, the combination of intravenous and topical dexamethasone can reduce pain, swelling, and inflammation after TKA, as well as improve functional recovery and reduce the incidence of postoperative nausea and vomiting in the early period after surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Clinical Trials and Biomedical Ethics Committee of Dafang People’s Hospital and written informed consent was obtained from all subjects participating in the trial. The clinical trial registration number was ChiCTR200004106 (Date of registration: December 17, 2020).
