Abstract
Background
Glucocorticoids have been widely used in perioperative period for postoperative pain relief after total knee arthroplasty (TKA). However, the optimal administration protocols of glucocorticoids remain controversial. This study aims to compare the efficacy of glucocorticoids between intravenous and periarticular injection on clinical outcomes.
Methods
A total of 114 patients were randomly assigned to intravenous (IV) group (n = 57) and periarticular injection (PI) group (n = 57). The IV group received 10 mg dexamethasone intravenously and the PI group received periarticular injection of 10 mg dexamethasone during the procedure. The clinical outcomes were assessed using visual analogue scale (VAS), knee society score (KSS), range of motion (ROM), knee swelling, inflammation markers and complications after TKA.
Results
The VAS score during walking at 2nd day postoperatively was lower in the PI group compared with the IV group (2.08 ± 1.45 vs 2.73 ± 1.69, p = .039), and there was no significant difference at the other time points of VAS score in two groups. The inflammation markers, knee swelling, knee ROM and KSS score were not statistically different. Vomiting and other complications occurrence were not significantly different between the two groups.
Conclusions
Intraoperative periarticular injection of glucocorticoids has similar analgesic effect compared to intravenous in the postoperative period following TKA and may be even more effective on the second postoperative day. In addition, periarticular injection of glucocorticoids does not impose an excess risk or complication on patients.
Introduction
Total knee arthroplasty (TKA) has proven to be a reliable and effective method for advanced stages of knee osteoarthritis. However, the moderate to severe postoperative pain associated with TKA is one of the most commonly therapeutic problems, with an incidence rate of 30-60%.1–3 Despite recent developments in pain control, postoperative pain remains a significant clinical challenge after TKA. Inadequate management of postoperative pain can lead to delayed recovery, prolonged hospital stays, and decreased patient satisfaction.4,5
Glucocorticoids have been widely used in perioperative period for postoperative pain relief and the prevention of postoperative nausea and vomiting (PONV) due to their powerful anti-inflammatory and antiemetic effects.6–16 Currently, the administration protocols of glucocorticoids include intravenous administration and periarticular injection. Previous studies have suggested that intraoperative intravenous dexamethasone have the advantages of relieving postoperative pain, reducing the consumption of opiates and decreasing the level of early inflammation markers.7,14,17–19 An alternative administration is periarticular injection of glucocorticoids which has also been reported in recent years due to the efficacy in reducing postoperative pain.6,8,11,15 However, the ideal administration protocols of glucocorticoids remain controversial.
At present, there are very few randomized controlled trials of high-level evidence comparing these two administration protocols directly. Therefore, we wondered whether the intraoperative intravenous and periarticular injection of glucocorticoids have different clinical outcomes, including postoperative pain management, prevention of PONV, inflammation, knee swelling, knee function recovery, consumption of opiates and the incidence of complications following primary TKA in this prospective, randomized and controlled study.
Materials and methods
Study design
The study was a prospective, randomized and controlled trial comparing the efficacy of dexamethasone between intravenous and periarticular injection on clinical outcomes following primary TKA. This study was approved by the Human Ethics Committee of *** and was registered with the Chinese Clinical Trial Registry (registration number ChiCTR2000033490). Written informed consent was provided by all patients.
Participants
A total of 163 patients scheduled to undergo unilateral primary TKA were recruited between June 2020 and June 2021 in the Center of Orthopedics Surgery. We excluded patients: (1) < 18 years old; (2) allergy to dexamethasone; (3) daily use of glucocorticoid or strong opioid drugs; (4) with history of serious heart disease (5) with renal insufficiency, active peptic ulcer or rheumatoid arthritis; (6) glycosylated hemoglobin >8; (7) with neurological or psychiatric disorders that may affect pain or cognitive impairment. Ultimately, 114 patients were randomly assigned to two groups (Figure 1). Study flow chart.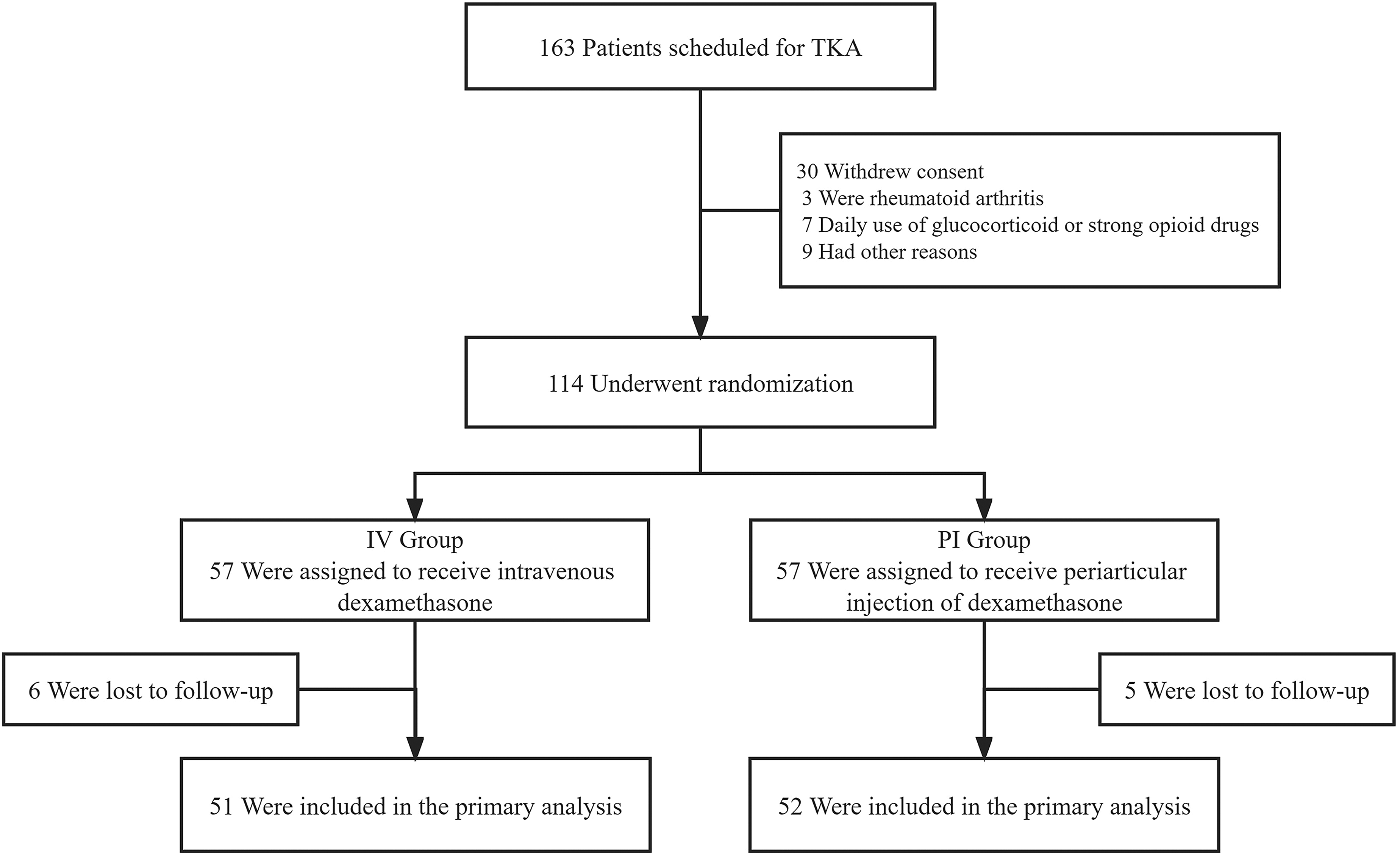
Randomization and blinding
A computer-generated randomization list for concealed allocation with a 1:1 ratio was prepared by an independent statistician. Eligible patients were assigned a sequential study number and randomized into two groups. A random allocation was concealed in consecutively numbered, opaque and sealed envelopes by a staff member not involved in the trial. The envelopes were opened on the day of the surgery to determine the treatment grouping and the trial drug was prepared by a nurse not involved in the trial. The patients, surgeons and data collectors were all blinded to patient group assignment.
Procedure
Interventions
Following randomization, the participants are divided into intravenous (IV) group or periarticular injection (PI) group. In the IV group, the cocktail including ropivacaine and flurbiprofen was injected at multiple points around the knee before prosthesis insertion, including synovium, joint capsule, posterior capsule periosteum and fascia, and 10 mg dexamethasone was injected intravenously with 5% glucose and sodium chloride as the solvent. The PI group received periarticular injection of cocktail including 10 mg dexamethasone, ropivacaine and flurbiprofen around the knee before prosthesis fixation at multiple points, and 10 mL of 5% glucose and sodium chloride was injected intravenously.
Preoperative and postoperative medications
All patients followed the same perioperative regimen. Preoperative administration of celecoxib 200 mg per day was given to all patients undergoing knee replacement. On the operative day and the first day after surgery, 40 mg of parecoxib was injected intravenously twice a day. On the third day after the surgery, the patient took celecoxib 200 mg twice a day. If the pain could not be relieved, the tramadol dosage was used and recorded according to the need of the patient. If the nausea and vomiting symptoms could not be relieved, ondansetron was injected intravenously according to the needs of the patients. The dosage of ondansetron was recorded, and the total dosage should not exceed 16 mg.
Surgery and rehabilitation
All patients were anesthetized by adductor nerve block combined with general anesthesia. All TKAs were performed by one of three senior surgeons using a standard medial parapatellar approach with the posterior stable prosthesis (RP, Depuy, Johnson & Johnson, USA). Tourniquets are used only from the fixation of the prosthesis to the closure of the wound. Drainage tube was placed 24 h after operation to reduce postoperative hematoma. All patients in this study were managed by standard clinical pathway. The standard rehabilitation plan is as follows: postoperative day (POD) 1-4: quadriceps femoris setting training, walking with a walker, active range of motion training; POD 5-16: the patient is discharged home or sent to rehabilitation center.
Study assessments
The pain was evaluated at 6h, 24h, 2nd, 3rd and 4th day after operation, and the rest time was at least 30min before the test. Records were made at each time point in the following order: rest (supine) and walking. Using the 100 mm visual analogue scale (VAS; 0, no pain, 100, the most severe pain imaginable) to assess knee pain when rest and walking (5 m) assisted by a walker. The levels of interleukin-6 (IL-6), C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) in blood were measured before operation, POD 1 and POD 3 to reflect the inflammatory reaction. The circumference of 1 cm near the upper edge of patella was measured before operation and POD 3. The range of knee motion (ROM) and knee society score (KSS) were measured by angle measuring instrument before operation, POD 1 and POD 3. The number of patients with nausea and vomiting within 48 h after surgery was recorded. In addition, the cumulative consumption of ondansetron was recorded. The daily cumulative amounts and the total consumption of narcotic drugs were converted into milligram equivalent oxycodone for statistical analysis.
The pain score, functional scale, range of knee motion, wound and complications were evaluated in the outpatient department 2 weeks and 3 months after operation. The definition of wound complications and any wound that is still draining during follow-up or any surgical site that needs to be returned for flushing and debridement. Other complications included deep vein thrombosis, pulmonary embolism, periprosthetic infection, and imaging evidence of avascular necrosis, which we recorded at each follow-up.
Statistical analysis
According to a systematic review study by, 20 a sample size of 51 patients per group would be required to detect a clinically relevant differences in pain scores with a mean difference of 1.3 points and a standard deviation of 2.0 points at an α level of 0.025 and a β value of 0.10. Considering the 10% attrition due to cancellation of surgery or expected withdrawal, we selected 114 sample sizes (57 in each group).
All statistical analyses were performed using Statistical Package for Social Sciences version 23.0 (SPSS, Chicago, IL, USA). Quantitative variables were depicted as mean and standard deviation while categorical data were described as number with percentage. The continuous data of demographic characteristics and clinical outcomes were analyzed using independent-samples ttest or Mann-Whitney U test according to the normality of data. And the categorical data were analyzed using chi-square or Fisher exact test. The level of statistical significance was set at p value less than 0.05.
Results
Baseline characteristics
Characteristics of the patients at baseline.

Abbreviations: Body mass index (BMI); Intravenous (IV); Periarticular injection (PI).
Visual analog score
Preoperative and postoperative pain evaluation.
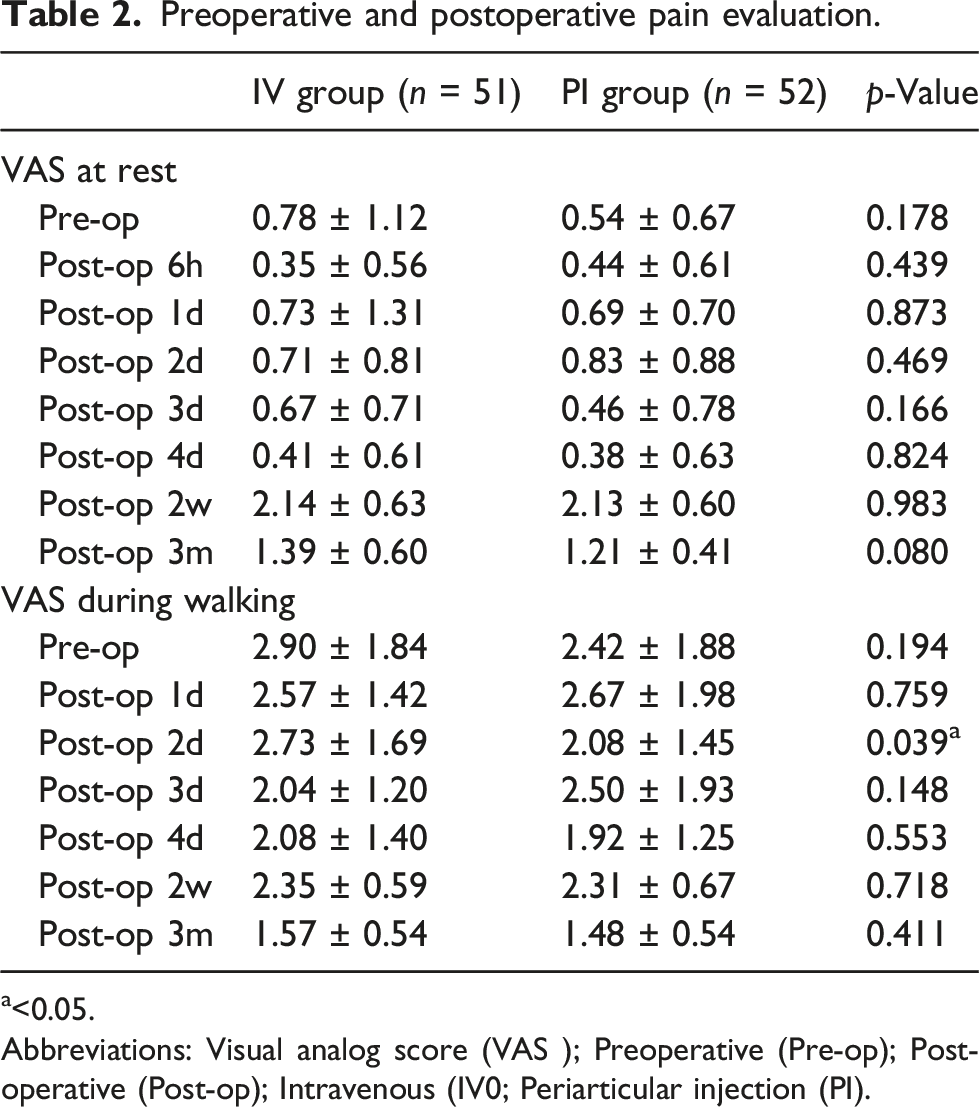
a<0.05.
Abbreviations: Visual analog score (VAS ); Preoperative (Pre-op); Post-operative (Post-op); Intravenous (IV0; Periarticular injection (PI).

Visual analog scale (VAS) pain score at rest (a) and during walking (b) between the intravenous (IV) group and periarticular injection (PI) group during the perioperative period. * indicates a significant difference between the two groups (p < .05).
Knee function and inflammatory indicators
Preoperative and postoperative knee function.

Abbreviations: Range of motion (ROM ); Knee society score (KSS ); Preoperative (Pre-op); Post-operative (Post-op); Intravenous (IV ); Periarticular injection (PI).
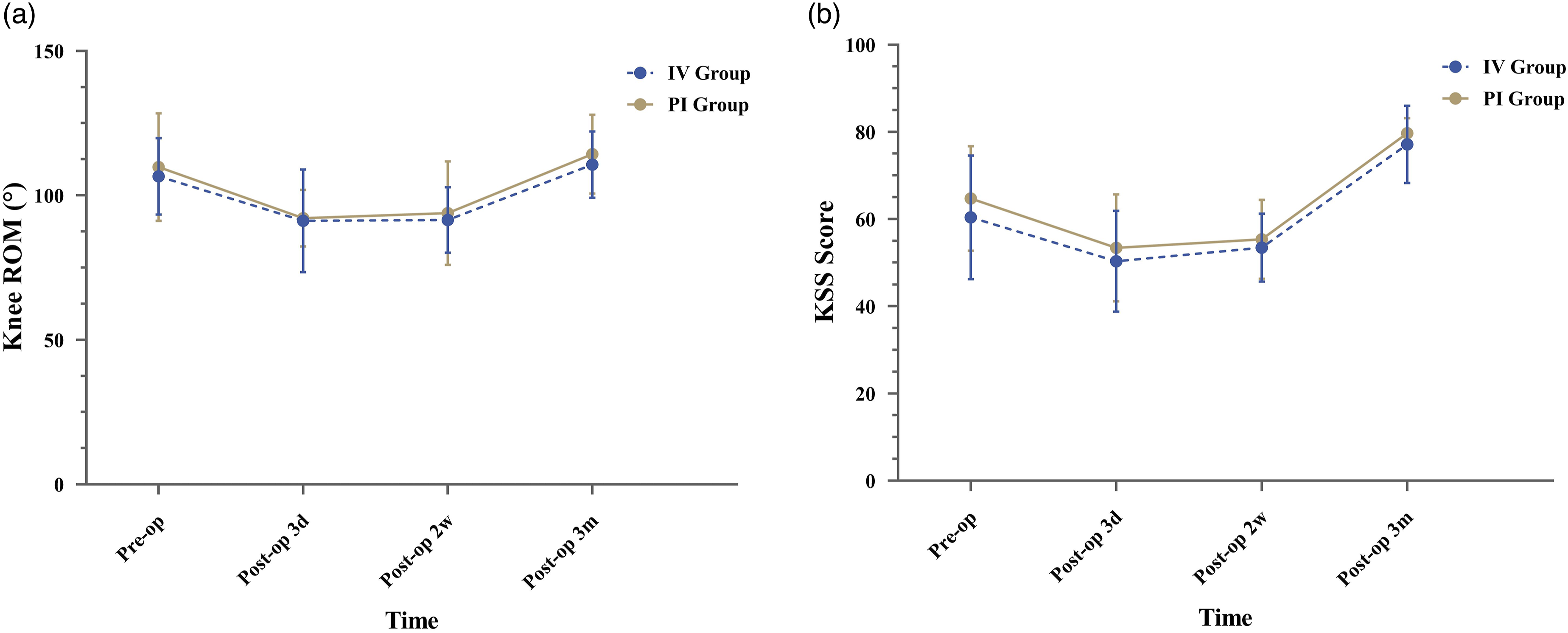
Range of motion (ROM) of knee (a) and knee society score (KSS) (b) between the intravenous (IV) group and periarticular injection (PI) group during the perioperative period.
Preoperative and postoperative inflammatory indicators.

Abbreviations: C-reactive protein (CRP ); interleukin-6 (IL-6); Erythrocyte sedimentation rate (ESR ); Preoperative (Pre-op); Post-operative (Post-op); Intravenous (IV); periarticular injection (PI).

The levels of interleukin-6 (IL-6) (a); C-reactive protein (CRP) (b) and erythrocyte sedimentation rate (ESR) (c) between the intravenous (IV) group and periarticular injection (PI) group during the perioperative period.
Postoperative complications
Postoperative complications.

Abbreviations: not available (N/A), intravenous (IV ); Periarticular injection (PI).
Discussion
This study was conducted to reveal whether the intraoperative intravenous and periarticular injection of glucocorticoids have different clinical outcomes following primary TKA and which administration route may be more recommended. In the present study, we found that the VAS score during walking at 2nd day postoperatively was lower in the PI group compared with the IV group (p < .05), and the other time points of VAS score in two groups were no significant difference. Additionally, there was no significant difference in knee swelling, knee ROM, KSS score, inflammation markers and complications occurrence between the two groups after TKA.
Intravenous and periarticular injection of glucocorticoids have been proven to be effective way to relieve pain after artificial joint replacement, but the best way to use glucocorticoids to relieve pain in TKA has not been determined. According to our outcomes, intraoperative periarticular injection of dexamethasone resulted in more significant pain relief in the early postoperative period after TKA. 21 argued that the pain scores at rest and during walking were significantly lower in patients who received periarticular injection of glucocorticoids than those who received intravenous administration of glucocorticoids in the early post-TKA period, which is consistent with our speculation. However, in the Hatayama’s study, the intravenous administration group received 10 mg dexamethasone but the periarticular injection group received a 40 mg triamcinolone acetonide, which might have contributed to the difference in results. 21
IL-6 is widely associated with inflammation, which has a vital role in the process of neutrophils and macrophage activation during surgery and trauma, and IL-6 can induce the production of CRP in hepatocytes.22,23 Previous studies revealed that the serum inflammatory cytokines including interleukins (ILs), C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) might be related to postoperative pain24–28. 28 suggested that serum inflammatory cytokines are positively correlated with acute postoperative pain following primary TKA. In the present study, there were no significant differences in the mean IL-6, CRP and ESR at 1st days and 3rd days postoperatively between the two groups, showing the similar anti-inflammatory effect of dexamethasone in IV group and PI group in the early postoperative period. 21 suggested that the mechanism of pain relief may be a direct inhibitory effect of glucocorticoids on signal transduction in pain C-fibers rather than the reduction in inflammation. 29 showed that IL-6 and CRP in the early postoperative period after TKA were significantly higher in topical dexamethasone group, but the postoperative pain scores were significantly lower in topical group comparing with intravenous group, which was not consistent with our results.
In addition, the inflammatory responses after surgical injury contribute to the knee swelling and impaired function of knee.30,31 Anti-inflammatory treatment can reduce knee swelling effectively, especially at the early postoperative stage. 31 reported that preoperative intravenous methylprednisolone was able to effectively reduce knee swelling within 24 h after TKA. However, 32 showed that a reduction in knee swelling after TKA was reported in 65.4% of patients who received periarticular injection of glucocorticoids. In this study, we found the similar outcomes between the two groups on reducing the knee swelling, the ROM of knee and the KSS score evaluated at 3rd days postoperatively. These results illustrate that the intravenous and periarticular injection of dexamethasone play the same role in improving knee swelling and knee function recovery after TKA.
Although glucocorticoids have obvious benefits in the perioperative period of artificial joint replacement, its application in the perioperative period of total joint replacement has not become a routine due to the concern of related side effects. However, these side effects are only related to the long-term use of glucocorticoids. Numerous studies have revealed that there was no significant increase of adverse reactions for patients who used glucocorticoids in perioperative period.33–35 The complications including PONV, delayed wound healing, deep vein thrombosis, pulmonary embolism, periprosthetic infection, avascular necrosis and the consumption of ondansetron and opioids were similar between IV group and PI group. The incidence and severity of side effects are related to the dosage, time, dosage form and injection site. Periarticular injection can reduce the total dosage of glucocorticoids and reduce systemic adverse reactions, such as transient facial flushing, excitement, sleep disorders, elevated blood pressure or blood sugar. 36
This study had several limitations. First, patients in this study were from a single center with a short period. Second, no placebo group was set in the study, and blank control was lacking. Third, all patients underwent adductor block, and the pain scale may be masked in the early postoperative period. However, we had to implement adductor nerve block to minimize pain and nausea after TKA due to ethical considerations.
Conclusions
In conclusion, this study supports that intraoperative periarticular injection of glucocorticoids has similar analgesic effect compared to intravenous in the postoperative period following TKA and and may be even more effective on the second postoperative day. In addition, periarticular injection of glucocorticoids does not impose an excess risk or complication on patients.
Footnotes
Acknowledgements
We thank our study participants and their family.
Author’s note
The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Major Science and Technology Project of Guangdong Province, grant number 2015B020225007; Natural Science Foundation of Guangdong Province, grant number 2018A0303130206 and 2021A1515011008; Guangdong Medical Science and Technology Research Fund, grant number A2019150; Scientific Research Project of Guangdong Traditional Chinese Medicine Bureau, grant number 20221008 and Starting fund of National Natural Science Foundation of Guangdong Provincial People’s Hospital, grant number 8207050645 and 8217051081.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the human ethics committee of Guangdong Provincial People’s Hospital (reference number No. GDREC2018522H (R1)).
Informed Consent
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets used and/or analyzed during the current study can be available from the corresponding author upon reasonable request.
