Abstract
Background
The optimal dosing of aspirin (ASA) monotherapy for prophylaxis after total joint arthroplasty is debatable. The objective of this study was to compare two ASA regimens with regards to symptomatic deep venous thrombosis (DVT), pulmonary embolism (PE), bleeding, and infection 90 days after primary total hip arthroplasty (THA) and total knee arthroplasty (TKA).
Methods
We retrospectively identified 625 primary THA and TKA surgeries in 483 patients who received ASA for 4 weeks post-op. 301 patients received 325 mg once daily (QD) and 324 patients received 81 mg twice daily (BID). Patients were excluded if they were minors, had a prior venous thromboembolism (VTE), had ASA allergy, or received other VTE prophylaxis drugs.
Results
There was a significant difference in rate of bleeding and suture reactions between the two groups. Bleeding was 7.6% for 325 mg QD and 2.5% for 81 mg BID (p = .0029 Χ2, p = .004 on multivariate logistic regression analysis). Suture reactions were 3.3% for 325 mg QD and 1.2% for 81 mg BID (p = .010 Χ2, p = .027 on multivariate logistic regression analysis). Rates of VTE, symptomatic DVT, and PE were not significantly different. The incidence of VTE was 2.7% for 325 mg QD and 1.5% for 81 mg BID (p = .4056). Symptomatic DVT rates were 1.6% for 325 mg QD and 0.9% for 81 mg BID (p = .4139). Deep infection was 1.0% for 325 mg QD and 0.31% for 81 mg BID (p = .3564).
Conclusion
Low-dose ASA in patients with limited comorbidities undergoing primary THA and TKA is associated with significant lower rates of bleeding and suture reactions than high dose ASA. Low-dose ASA was not inferior to higher dose ASA for the prevention of VTE, wound complications, and infection 90 days postoperatively.
Keywords
Introduction
It is well known that total hip arthroplasty (THA) and total knee arthroplasty (TKA) are safe and successful surgeries which improve patients’ quality of life. As projections for the number of primary THA and TKA cases completed per year continue to increase through 2050 – 330% and 380%, respectively – the importance of optimizing these procedures and limiting complications is paramount. 1
Although it is an infrequent complication, symptomatic venous thromboembolism (VTE) has historically been one of the most common causes for hospital readmission following THA and TKA, with rates ranging from 40%–60% in cases without prophylaxis.2–7 With prophylaxis, rates of symptomatic VTE during the 90 days following THA and TKA are reported to be up to 15% for deep vein thrombosis (DVT) and 3% for pulmonary embolism (PE).8,9
As a result of the costly and potentially fatal consequences of VTE, the American Association of Hip and Knee Surgeons (AAHKS) and the American Academy of Orthopedic Surgeons (AAOS), in conjunction with the American College of Chest Physicians (ACCP), have all recognized both mechanical and chemical prophylaxis as valid methods for VTE prophylaxis following total joint arthroplasty.3,10–12 While there is consensus amongst the three societies that prophylaxis is standard of care, the recommended pharmacologic agents and treatment modalities for minimizing VTE differ greatly.
The selection of the appropriate prophylactic agent necessitates a balance between efficacy and safety. There are five major classes of chemical anticoagulation: Vitamin K antagonists (VKA), heparins, direct thrombin inhibitors, factor Xa inhibitors, and aspirin (ASA). Compared to other anticoagulants, ASA remains an appealing choice for arthroplasty surgeons. 13 Its main advantages include low cost, ease of administration, no need for routine blood monitoring, decreased risk for postoperative wound drainage/complications, hematoma, and periprosthetic joint infection (PJI), and even a decrease in mortality.8,14–17 The proven efficacy and safety of ASA in reducing the incidence of VTE following THA and TKA has been consistently demonstrated across a large body of research which spans over four decades.6,10–12 The comparable effectiveness of ASA to other medications such as warfarin and direct oral anticoagulants is a major factor in its success as the most widely used chemoprophylactic agent in the United States for arthroplasty patients.18–22 In a recent poll of approximately 600 AAHKS members, ASA was the most popular agent used for VTE prophylaxis (86% of respondents). 23
Beginning in 2012, the AAOS and the ACCP jointly recommended the use of ASA monotherapy for the prophylaxis of VTE after primary THA and TKA – 325 mg BID for 6 weeks after surgery.3,11,24 It should be noted that these recommendations were based on low level evidence due to the lack of comparative studies at that time.17,25,26 Moreover, current recommendations from both AAOS and ACCP have not yet clarified the optimal prophylactic ASA dose following primary THA and TKA in preventing symptomatic VTE.
In the current surgical literature, the most commonly used ASA doses are 81 mg BID (low-dose) and 325 mg BID (high-dose).27–29 However, recent data suggest that lower doses of ASA may be have similar levels of efficacy, while lowering concerns for safety.30,31 In addition, lower doses of ASA may be more effective than higher doses in lowering the risk of hemorrhagic stroke and death following carotid endarterectomy, decreasing gastrointestinal (GI) adverse effects, and interfering less with natural antithrombotic mechanisms at the endothelial level.32–35
The prophylactic ASA dose for post-operative primary THA and TKA patients remains up for debate. The main objective of this retrospective study is to determine the efficacy of a low dose ASA protocol consisting of 81 mg BID for VTE prophylaxis following primary THA and TKA when compared to a higher dose protocol of 325 mg QD. The secondary objective was to evaluate the safety between the two dosing regimens by comparing the adverse events of bleeding (GI and hematoma formation), wound complications, deep infection (namely acute PJI), and return to the operating room (OR) within 90 days following initial THA or TKA. We hypothesized that low dose ASA would result in fewer adverse events relating to bleeding and wound complications compared to high dose ASA, without a significant difference in VTE rates.
Methods
Following approval from the Institutional Review Board (2021-00,441), a retrospective cohort study was conducted at a tertiary, suburban, academic institution identifying consecutive patients who underwent primary THA and TKA by a single fellowship-trained surgeon over a two-year period between 21 October 2019 and 21 October 2021. All de-identified data were collected using Microsoft Excel software by three of the authors (AD, SB, and SB). As this study was a retrospective review, patients were not randomized into experimental groups. Informed consent was not obtained for this study, as it was deemed exempt by our Institutional Review Board.
Patient population
Patients were identified using the following Current Procedural Terminology (CPT) codes: 27,130 for primary total hip arthroplasty and 27,447 for primary total knee arthroplasty. Our cohort consists of 625 primary surgeries (323 THA and 302 TKA) among 483 consecutive patients during the above time frame. Inclusion criteria included use of ASA doses of 325 mg QD or 81 mg BID for postoperative chemical VTE prophylaxis for 4 weeks. Patients were excluded if they were less than 18 years old, had a prior history of VTE event, ASA allergy, were receiving another agent for VTE chemoprophylaxis, or receiving any other anti-thrombotic medication during the immediate four-week postoperative period (including anti-platelets). Patients with coagulopathy but no history of VTE were not excluded unless they were already receiving an agent for chemoprophylaxis. Patients with significant cardiac history (prior MI, arrhythmia or valvular disease) were excluded. All patients undergoing bilateral procedures were treated with alternative anticoagulation – warfarin, apixaban, or rivaroxaban – and were therefore excluded from this study (Figure 1). Patient inclusion/exclusion criteria.
The variables collected by manual chart review for this study included: ASA dosage (either 325 mg daily or 81 mg BID), baseline patient demographics; including age, gender, body mass index (BMI), American Society of Anesthesiologists (ASA) score, Charlson comorbidity index (CCI), method of anesthesia (general or spinal), smoking status, and COVID-19 status.
Venous thromboembolism prophylaxis protocol
All patients were placed on a four-week regimen of VTE chemoprophylaxis following surgery. Before October 2020, patients with standard risk for VTE at our institution received 325 mg of ASA QD starting the evening following surgery. Starting in October 2020, the dosage and timing of ASA was changed to 81 mg BID and continued for the same four-week duration.
Mechanical prophylaxis by intermittent pneumatic compression stockings was used in all cases and applied at the end of surgery and continued throughout the entire hospitalization period. All patients began BID inpatient physical therapy within 24 hours of surgery and continued this regimen until their discharge from the hospital.
Outcomes assessed
Symptomatic VTEs occurring within 90 days of TKA or THA were identified from patient medical records, which included a thorough review of institutional administrative and radiological databases, as well as a review of clinical records of patient-provider communications and encounters. DVT events were diagnosed by ultrasound and PE events were diagnosed by a computerized tomography scan. The timing of VTE was determined based on either the date of diagnostic imaging or on patient-provider phone calls confirming the diagnosis. During the COVID pandemic, patients had the ability to communicate through telehealth visits or directly come to the office. All VTE events were diagnosed after the patient had been discharged home and within the first 6 weeks of follow-up.
Major bleeding events were defined by the 2010 criteria for major bleeding in surgical patients as defined by the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. 36 For our study group, major bleeding events were identified from review of patient medical records.
We additionally identified 90-days complications such as GI bleeding, wound complications (dehiscence, delayed healing, drainage), and acute PJI via query of the electronic medical record using International Classification of Disease, 9th and 10th Revision (ICD-9 and ICD-10) diagnosis and procedure codes. The ICD-9 and ICD-10 codes for bleeding complications were 578 and K92.2 for GI hemorrhage, and 531.90 and K25.9 for gastric ulcers. The ICD-9 and ICD-10 codes for acute PJI were 996.6 and T84.5, respectively.
Statistical analysis
Descriptive data are represented as mean ± standard deviation for continuous data or counts (%) for categorical data. An initial analysis revealed all data sets to be normally distributed. Subsequently, unpaired t-tests were used to test for significant differences between continuous variables and Χ2 tests for categorical variables. All descriptive statistical analyses were performed using Prism v9 (Graphpad Inc.). p-value less than 0.05 were considered to be statistically significant.
A series of multivariate logistic regression models were used to examine the effect of ASA dose (81 mg BID vs 325 mg QD) on several postsurgical outcomes (i.e., separate models predicting: bleeding, PE, DVT, suture reactions, infections, and return to OR), adjusting for common risk factors for postoperative complications: age, BMI, type of surgery (THA vs TKA), Charlson Comorbidity Index score, type of anesthesia (general vs spinal), and smoking status.
A power analyses was conducted with G*Power, Version 3.1.9.4 (Faul et al., 2007). We assumed small effect size (d = 0.20) and we set our alpha and beta values at α = 0.05, β = 0.80. This power analysis indicated a sample of N = 113 would be appropriate for our proposed logistic regression models, thus we expect to have sufficient power for our proposed analyses with the current sample of N = 625.
Results
Patient demographics and characteristics between different aspirin protocols.

BMI, body mass index; ASA, American Society of Anesthesiologists; CCI, Charlson Comorbidity Index; THA, total hip arthroplasty; TKA, total knee arthroplasty.
Aspirin 325 mg QD Regimen
There was a total of 301 patients in this cohort, of which 51% (154) underwent THA. The mean age at index surgery for those receiving ASA 325 mg QD was 66.4 ± 9.2 years (42 – 89), 61% (184) were female, and the mean body mass index (BMI) was 30.9 ± 5.8 (20–53.7). Seventy-one percent of patients (212) in this cohort denied ever smoking tobacco or nicotine products. Thirteen patients (4.3%) in this cohort underwent general anesthesia during their respective arthroplasty procedure. Of these thirteen patients, six had contraindications to spinal per the anesthesia team and seven patients were converted to general anesthesia after unsuccessful spinal attempts. The mean American Society of Anesthesiologists and Charlson Comorbidity Index scores for this cohort were 2.6 ± 0.5 and 2.9 ± 1.6, respectively.
Aspirin 81 mg BID regimen
There was a total of 324 patients in this cohort, of which 52% (169) underwent THA. The mean age at index surgery for those receiving ASA 81 mg BID was 65.2 ± 9.8 years (23 – 91), 56% (181) were female, and the mean BMI was 30.7 ± 5.7 (18.7 – 48.5). Seventy-one percent of patients (230) in this cohort denied ever smoking tobacco or nicotine products. Thirty-one patients (9.6%) in this cohort underwent general anesthesia during their respective arthroplasty procedure. Of these 31 patients, 24 had contraindications to spinal per the anesthesia team and seven patients were converted to general anesthesia after unsuccessful spinal attempts. The mean American Society of Anesthesiologists and Charlson Comorbidity Index scores for this cohort were 2.6 ± 0.5 and 2.9 ± 1.4, respectively.
Venous thromboembolism
Ninety-day complication rates stratified based on aspirin prophylaxis dose.
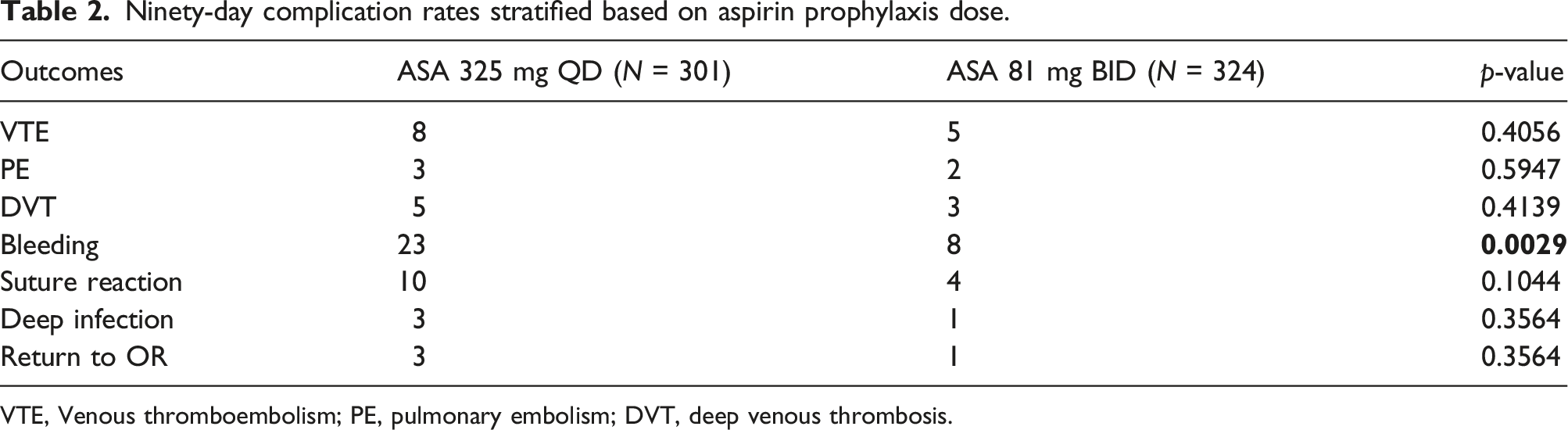
VTE, Venous thromboembolism; PE, pulmonary embolism; DVT, deep venous thrombosis.
Major bleeding events
The total incidence of documented major bleeding events at the incision site during this study period between both ASA protocols was 5.0% (31/625 patients). There was a statistically significant difference in the incidence of major bleeding events between groups: 7.6% in the 325 mg QD group and 2.5% in the 81 mg BID group (p = .0029). These events were all reported as postoperative hematomas that were managed with observation. None of the patients who developed major bleeding events within our study cohorts were taken back to the OR. Additionally, two separate patients within the ASA 325 mg QD group developed upper GI (UGI) bleeds within 2 weeks postoperatively.
Surgical site complications/infection
The overall incidence of infection and subsequent return to OR was 0.64% (4/623 patients). There was no statistically significant difference in the incidence of deep infection and return to OR between groups: 1.0% in the 325 mg QD group and 0.31% in the 81 mg BID group (p = .3564). Three of the four patients underwent two-stage exchanges while the last patient was successfully treated with a single-stage exchange.
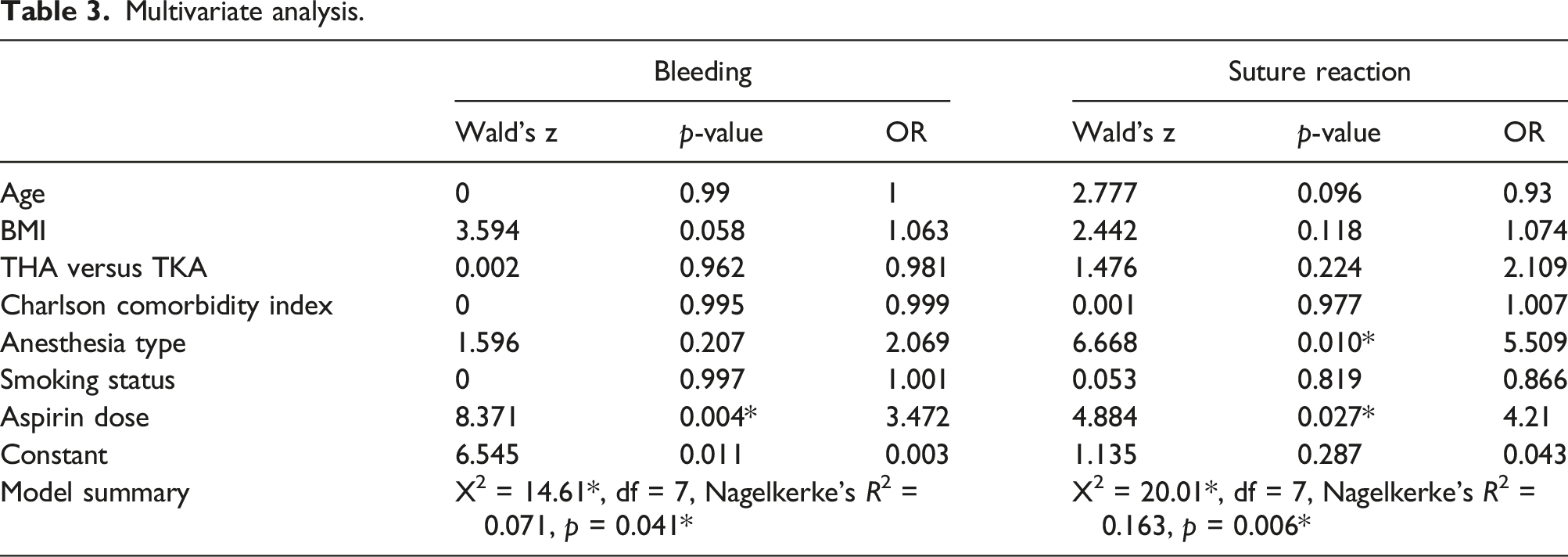
Multivariate logistic regression analysis
Multivariate analysis.
Discussion
This study sought to identify any differences in symptomatic VTE, bleeding, and wound complication rates/infection within 90 days following primary THA and TKA between patients taking ASA 325 mg QD versus ASA 81 mg BID. Our results indicate a significant decrease in bleeding related complications and suture reactions in the low dose ASA group compared to the high dose ASA group, with no concomitant difference in deep infection rates. We found no significant differences in rates of VTE, symptomatic DVT, or PE between the low dose ASA and high dose ASA groups.
In total joint arthroplasty, the administration of postoperative ASA has been demonstrated to be effective and safer than other anticoagulation regimens in the prevention of VTE after THA and TKA.4,20,28,37 The risk of VTE has dropped dramatically in over the last two decades (0.6 – 1.1%) in conjunction with the improvement of peri-operative protocols.38–43 To date, few studies have evaluated the efficacy and safety of different ASA regimens in the prevention of VTE following THA and TKA, making it difficult to determine differences in the efficacies of chemoprophylactic modalities and further confounding attempts to optimize postoperative treatments and recovery.27,30
The change in postoperative protocol from a higher ASA dose to a lower dose at our institution was implemented based on the lack of compelling evidence regarding the traditionally recommended higher dosages of ASA.11,17 Our institution believed that this change was clinically appropriate as it was based on a growing number of studies concluding that the lower ASA dose was a safe and effective alternative for primary THA and TKA.17,35,44,45.
The present study’s findings demonstrate the efficacy of low dose ASA for VTE chemoprophylaxis when utilized in patients without prior history of VTE events or ASA allergy, and without concurrent additional VTE prophylactic agent or anti-thrombotic use. The rate of VTE between the two doses was not significantly different and remained at < 2.7% within the high dose cohort and <1.5% within the low dose cohort (p = .41). Our study also demonstrated the potential harm of high dose ASA, which had greater than three times the rate of major bleeding events (p = .004, OR 3.47). While there was no difference in the rate of deep infection between different dosage groups (p = .35), there was a more than four-fold increased risk of suture reactions in the high dose ASA group (p = .027, OR = 4.2). Overall, our findings are consistent with the available literature on the efficacy of low dose ASA in the prevention of VTE events after THA and TKA, and offer additional insight into potential downsides of high-dose protocols.46,47
We found a significant increase in the rate of utilization of general over spinal anesthesia in the low dose ASA group. It is unclear why this trend was demonstrated, as our institution favors neuraxial anesthesia when not contraindicated. One possible explanation is that the high dose ASA group had more chronologic overlap with the first wave of the COVID-19 pandemic. There may have been a more aggressive effort to limit aerosolizing procedures like intubation during this time which resulted in a disproportionate number of spinal anesthetic cases compared to the later low dose ASA period. Unfortunately, our hospital’s anesthesia documentation does not specify the rationale behind their chosen modality to either confirm or to refute this speculation.
This study is not without some limitations. First, the retrospective design of the study may have resulted in inaccuracy in data collection. Although all efforts were made to utilize available databases and clinical records, to ensure that ninety-day follow-up was complete for all patients, our reported VTE incidence suggests a good capture rate. Additionally, our data may only be generalizable within patient populations with similar exclusion criteria. Another limitation to this study is that we only looked at symptomatic VTEs. Therefore, future prospective studies may be warranted to see the effects of these doses on both symptomatic and asymptomatic VTEs.
Ultimately, VTE prophylaxis should be individualized for each patient by assessing their respective risk factors for developing a symptomatic VTE event against the potential impact of excessive anticoagulation on bleeding. Our findings suggest that low dose aspirin appears to be an effective alternative to high dose regimens, with statistically similar VTE rates, but with a lower risk of major bleeding events within 90 days postoperatively. We advocate that our data serve as a springboard for further studies into other ASA chemoprophylaxis regimens in order to find the most optimal balance between VTE protection and postoperative bleeding sequelae.
Conclusion
The results of this study reaffirm the efficacy of low dose ASA as VTE prophylaxis in patients with limited comorbidities undergoing primary TKA and THA and reveal some risks of high dose ASA including increased rates of major bleeding events and suture reactions. Low dose ASA was also non-inferior to high-dose ASA for prevention of wound complications, and infection for 90 days postoperatively. Further studies are warranted to assess other potential low dose ASA regimens which can be utilized in postoperative joint arthroplasty patients to further minimize risks of bleeding and suture reactions while also adequately preventing VTE events.
Footnotes
Acknowledgements
The authors would like to acknowledge Adrian Torres for his help with data collection in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
