Abstract
Background
Our previous study reported that when a respective 20 mL of ropivacaine was used for adductor canal block (ACB) combined with infiltration between the popliteal artery and capsule of the posterior knee (IPACK) block for patients undergoing total knee arthroplasty (TKA), the minimum concentration of 0.275% could provide successful block in almost all patients. Based on the results, the primary objective of this study was to investigate the minimum effective volume (MEV90, defined as the volume which can provide successful block in 90% of patients) of ACB + IPACK block.
Methods
This double-blind, randomized dose-finding trial was based on a biased coin up-and-down sequential design, where the volume of ropivacaine administered to a given patient depended on the previous patient’s response. The first patient received 15 mL of 0.275% ropivacaine for ACB and again for IPACK. If the block failed, the next subject received a 1 mL higher volume for ACB and IPACK, respectively; otherwise, the next subject received either a 1 mL lower volume (probability of 0.11) or the same volume (probability of 0.89). The primary outcome was whether the block was successful. Block success was defined as the patient did not suffer significant pain and did not receive rescue analgesia within 6 h after surgery. Then the MEV90 was estimated by isotonic regression.
Results
Based on analysis of 53 patients, the MEV90 was 17.99 mL (95% CI 17.47–18.61 mL), MEV95 was 18.48 mL (95% CI 17.45–18.98 mL) and MEV99 was 18.90 mL (95% CI 17.38–19.07 mL). Patients whose blocks succeeded had significantly lower NRS pain scores, lower morphine consumption, and shorter hospitalization.
Conclusion
0.275% ropivacaine in 17.99 mL respectively can provide successful ACB + IPACK block in 90% of patients undergoing TKA. The minimum effective volume (MEV90) of ACB + IPACK block was 17.99 mL.
Introduction
Total knee arthroplasty (TKA) is one of the most common surgical procedures in the United States. 1 However, more than 60% of patients suffer moderate to severe pain after TKA.2,3 Inadequate pain management can delay postoperative recovery and increase risk of immobility-related complications such as venous thromboembolism (VTE).4,5 The advent of multimodal pain protocols has significantly decreased postoperative pain and length of hospital stay associated with TKA.6,7
Peripheral nerve block is one of the key techniques of multimodal pain protocols after TKA.8,9 At present, extensive studies have explored the analgesic efficacy of combining adductor canal block (ACB) and infiltration between the popliteal artery and capsule of the posterior knee (IPACK) block after TKA. 10 Some researchers have recommended this combination to block only sensory nerves as an important method for postoperative analgesia in enhanced recovery TKA protocols.1,11-13
Both ropivacaine and bupivacaine are long-acting amide local anesthetics commonly used for peripheral nerve block. 14 However, ropivacaine produces less suppression of motor fibers and has a significantly higher threshold for cardiovascular and central nervous system toxicity than bupivacaine.15,16 Therefore, we chose ropivacaine as the research object of this study.
The optimal dose of ropivacaine for ACB + IPACK block is unclear. The dose is determined by both concentration and volume. Our previous study reported that when a respective 20 mL of ropivacaine was used for ACB + IPACK block for patients undergoing TKA, the minimum concentration of 0.275% could provide successful block in almost all patients. 14 Our previous study explored the minimum effective concentration. 14 However, the knowledge gap was that the minimum effective volume (MEV90, defined as the volume which can provide successful block in 90% of patients)
Of ropivacaine used for ACB + IPACK block was still unclear. Therefore, the minimum effective dose was also unclear. Previous studies have reported volumes from 15-30 mL.11,17-21 The risk of local anesthetic systemic toxicity should always be contemplated very carefully with all regional anesthesia techniques. 22 A dose-finding study is required for the optimization of the ACB + IPACK block, thereby ensuring administration of the appropriate dose of local anesthetic together with preservation of maximum clinical efficacy.
Based on our previous results, the primary objective of this study was to investigate the minimum effective volume (MEV90, defined as the volume which can provide successful block in 90% of patients) of ropivacaine for ACB + IPACK block in patients undergoing TKA. The secondary objectives were (1) to investigate MEV95 and MEV99 at the same time; (2) to compare postoperative numerical rating scale (NRS) pain scores, morphine consumption, and time to hospital discharge between patients whose blocks succeeded with those which failed; and (3) to record any adverse events that occurred during surgery or during hospitalization.
For our preview plan, we planned to conduct a double-blind, randomized dose-finding trial to accomplish our objectives.`
Materials and Methods
This study was designed as a double-blind, randomized dose-finding trial. This study was approved by the Clinical Trials and Biomedical Ethics Committee of our institution and written informed consent was obtained from all subjects participating in the trial.
Patient recruitment
Patients eligible for the trial complied with all of the following requirements to be included: 1. Diagnosed with osteoarthritis; 2. American Society of Anesthesiologists (ASA) functional status of I–III; 3. With normal quadriceps strength; 4. Scheduled for primary unilateral TKA under general anesthesia; 5. 4 ≤ NRS pain score in daily life ≤6; 6. A knee flexion deformity of ≤15°; 7. A varus-valgus deformity of ≤15°.
Patients eligible to one or several of the criteria listed below were not enrolled in the trial: 1. Known allergies to the drugs being used in this study; 2. With a history of open surgery of knee; 3. With a history of knee infection; 4. Drug addiction; 5. With recognized neuromuscular disorders; 6. Unable to communicate verbally.
Trail design
This study was designed as a double-blind, randomized dose-finding trial to estimate MEV90 for ultrasound-guided, single-injection ACB + IPACK block in patients undergoing TKA. This type of trial does not require a control group. The drug volume was assigned using a biased coin up-and-down sequential design, where the volume of ropivacaine administered to a given patient depended on the previous patient’s response. The first patient to be recruited received 15 mL 0.275% ropivacaine for ACB and 15 mL 0.275% ropivacaine for IPACK. If the block was successful in the first patient, the next patient was randomized to receive either the same ropivacaine dose (at a probability of 0.89) or 1 mL lower ropivacaine volume for ACB and IPACK, respectively (at a probability of 0.11). If, however, the block failed in the first patient, the next patient received 1 mL higher ropivacaine volume for ACB and IPACK, respectively. The maximum volume was set at 30 mL. If the block failed in a patient who received this maximum volume, the next patient did not receive a higher volume.
The sequential allocation of biased coin up-and-down sequential design was carried out using a computer-generated list of random numbers prepared by a statistician with Microsoft Excel (Redmond, WA, USA). Investigator one accessed the list and prepared the corresponding syringes of ropivacaine in the central pharmacy, adding epinephrine (2.0 μg/mL) to all ropivacaine formulations. Then Investigator one carried the syringes to the operating room. In this way, the patient, anesthesiologist, surgeon, and postoperative caregiver were blinded to volume allocation.
Analgesic procedures
One anesthesiologist performed all ACB and IPACK procedures in the operating room for all patients 30 min before general anesthesia. After subcutaneous infiltration with 1 mL of 2% lidocaine, ACB and IPACK block were performed with the patient in the supine position. For ACB (Figure 1), a high-frequency linear-array ultrasonic transducer (Anesus ME7, Mindray, Shenzhen, China) was used to scan the middle of the thigh, halfway between the inguinal crease and patella, in order to identify the adductor canal, superficial femoral artery, sartorius, adductor longus, and adductor magnus. The site of injection was chosen as the anterolateral hyperechoic structure of the artery (saphenous nerve and nerve to vastus medialis). Once the site was located, a 21-gauge, 100-mm needle (Pajunk, Geisingen, Germany) was introduced in-plane in a lateral to medial direction. Correct needle placement was confirmed using 3 mL of isotonic saline, then the ropivacaine was injected. Ultrasound-guided adductor canal block. AL, adductor longus; FA, femoral artery; SM, sartorius muscle; SN, saphenous nerve; VM, vastus medialis; line, needle insertion point.
IPACK was performed using the same ultrasonic transducer mentioned above (Figure 2). The anesthesiologist identified the popliteal artery at the popliteal crease and moved cephalad just beyond the femoral condyles, to where the condyles merged with the shaft of the femur. The tibial and peroneal nerves superficial to the popliteal artery were located, and when the space between the femur and popliteal artery was located, the needle was inserted in-plane in a medial to lateral direction. The tip was positioned at the middle of the femur, near the lateral border near the periosteum. Then 5–10 mL of ropivacaine was injected to ensure adequate spread to the lateral end of the femur. As the needle was withdrawn, the rest of the ropivacaine was injected along the femur, such that ropivacaine infiltrated incrementally into the area between the artery and femur, ending at the medial end of the femur. Ultrasound-guided infiltration between the popliteal artery and capsule of the posterior knee block. FB, femoral bone; PA, popliteal artery; SN, saphenous nerve; line, needle insertion point.
Surgery and concomitant medication
On the day before surgery, celecoxib (200 mg) was administered twice as a preemptive analgesic. Patients were instructed to fast for 8 hours before surgery and to drink 100 mL of a clear, pure carbohydrate liquid 2 hours before surgery. All surgeries in this study were performed by the same surgeon under general anesthesia. General anesthesia was performed as follows: after pure oxygen inhalation, the following anesthetics were administered intravenously: Midazolam, 2 mg/kg; Propofol, 2 mg/kg; Sufentanil, 0.3 μg/kg; and Cis-atracurium, 0.2 mg/kg. Patients were then intubated and given an inhaled anesthetic (Sevoflurane, 1-1.5 MAC). Surgery was performed by making a midline skin incision with a medial parapatellar approach. During the surgery, cemented prostheses (DePuy Synthes, New Brunswick, USA) were used, but not pneumatic tourniquets. Drainage tubes were not used before the wound was sutured. Flurbiprofen (50 mg) was administered 20 min before the end of the surgery to prevent postoperative pain, along with Tropisetron (5 mg) to prevent postoperative nausea and vomiting.
After regaining consciousness, patients were sent to the post-anesthesia care unit, where Investigator 2 (postoperative caregiver) administered an opioid add-on to the multimodal pain treatment whenever the pain score at rest exceeded 3, in accordance with routine procedures at our hospital. The patients whose pain score exceeded three simultaneously received intravenous Sufentanil (5 μg).
After awakening from anesthesia, patients were sent to the bed ward, and an ice compress was applied around the incision. Patients did not receive any oral analgesics
Within 6 h after surgery. After postoperative 6 h, oral celecoxib (200 mg) and pregabalin (150 mg) were administered twice daily to control postoperative pain. If the patient was unable to tolerate the pain, a further 5 mg of morphine hydrochloride as rescue analgesia was injected subcutaneously.
Outcome assessment
The primary outcome was whether the block was successful. Block success was defined as the patient did not suffer significant pain and did not receive rescue analgesia within 6 h after surgery. The assessments were performed by Investigator 2 (postoperative caregiver), who was blinded to volume allocation. Pain at rest was assessed in the post-anesthesia care unit, at 2 h, 4 h, and 6 h after surgery. If the pain score at rest exceeded three in a NRS from 0 to 10 23 at any time point, the patient was recorded as experiencing block failure. Pain during motion (flex the knee as much as possible) was assessed at 2 h, 4 h, and 6 h after surgery. If the pain score during motion exceeded five at any time point, the patient was recorded as experiencing block failure. In addition, if the patient received rescue analgesia (received Sufentanil in the post-anesthesia care unit or morphine hydrochloride in the bed ward) within 6 h after surgery, the patient was recorded as experiencing block failure.
Secondary outcomes included NRS pain scores at rest and during motion at postoperative 24 and 48 h, postoperative morphine consumption, and time to hospital discharge. The discharge criteria of patients included: adequate pain control on oral pain medication, independent transfer, ambulation alone for at least 200 feet, and the ability to climb stairs. These outcomes were compared between patients whose blocks succeeded with those which failed. In addition, any adverse events that occurred during surgery or postoperative recovery in hospital were recorded. Secondary outcomes were assessed by Investigator 2 (postoperative caregiver) who was blinded to volume allocation.
Bias control
In this study, the ACB and IPACK procedures were performed by a specific senior anesthesiologist, the surgery by a specific senior surgeon, and the outcome assessment by a specific investigator to reduce potential bias. In addition, we recruited a homogenous group of patients to make the results more consistent and stable as to whether the nerve block was successful or not. For example, we recruited a group of patients with similar preoperative pain and only mild knee deformity.
Statistical analysis
Based on previous studies,24,25 we estimated that we would need to observe at least 45 successful ACB + IPACK blocks in order to estimate MEV90. Thus, we decided to recruit patients until we had achieved this.
Data were analyzed statistically using R statistical software (R Foundation for Statistical Computing, Vienna, Austria). MEV90 was calculated using isotonic regression, and the 95% confidence interval (CI) was derived by bootstrapping.26,27 Similar procedures were used to estimate the minimum effective volumes to produce a successful block in 95% or 99% of patients (MEV95, MEV99). We used the dose estimator μ3, defined as the interpolated dose whose probability of effect was estimated to be 0.9.
Secondary outcomes were analyzed statistically using SPSS 26.0 (IBM, Chicago, IL, USA). The normality of data was assessed using histograms and quantile-quantile plots. Continuous data were presented as means and standard deviations. Inter-group differences in normally distributed data were assessed for significance using Student’s t test; differences in skewed and ordinal data, using the Mann–Whitney U test. Differences were considered significant if p < 0.05.
Results
Baseline characteristics of patients
Patient characteristics.
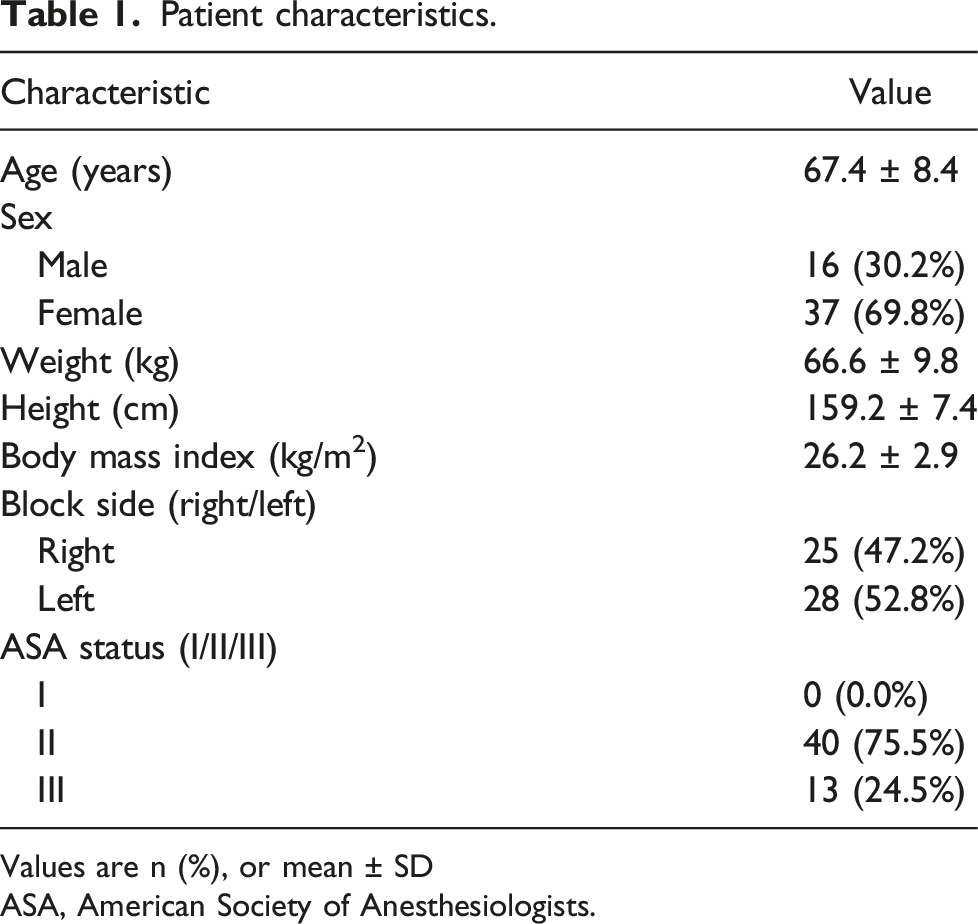
Values are n (%), or mean ± SD
ASA, American Society of Anesthesiologists.

Graph of successful ( ) and failed ( ) blocks with different ropivacaine volumes. The horizontal line is the calculated minimum effective volume of ropivacaine providing successful ACB + IPACK block in 90% of patients (MEV90); error bars represent 95% confidence interval.
Results regarding primary objective
Based on analysis of the 53 patients, the MEV90 was 17.99 mL (95% CI 17.47–18.61 mL).
Results regarding secondary objectives
Observed response rates.

Values are n or percentage.
Secondary outcomes.

Values are mean ± SD
Mann–Whitney U test.
NRS: numerical rating scale.
During the nerve block, no events of vascular puncture were observed; after nerve block, none of the patients suffered nausea, vomiting or dyspnea before general anesthesia. After surgery, none of the patients suffered diminished quadricep strength, residual paresthesia, or residual neural deficits. None of the patients showed signs of local anesthetic intoxication during the trial.
Discussion
The analgesic efficacy of ACB + IPACK block remains controversial. Some studies indicate that it can significantly improve analgesic and functional outcomes following TKA,11,12,19,28 while other studies have reported that it is not superior to ACB alone.21,29 This discrepancy may reflect the use of different doses of local anesthetic, highlighting the need for a dose-finding study. Before this study, we conducted a study to estimate the minimum effective concentration for ropivacaine in ACB + IPACK block. We found that single injection of 0.275% ropivacaine in 20 mL respectively (containing 2.0 μg/mL of epinephrine) can provide successful ACB + IPACK block in almost all patients undergoing TKA. Therefore, we chose 0.275% ropivacaine in this study. Based on this result, we further calculated a MEV90 of 17.99 mL (95% CI 17.47–18.61 mL).
The volumes of ropivacaine used for ACB + IPACK block ranged from 15 mL to 30 mL in previous studies,11,17-21 so we chose the lowest 15 mL as the initial volume of this trial. In clinical practice, sympathetic nervous system modulators such as epinephrine is often added to local anesthetic to potentiate the intensity and duration of local anesthetic. 30 Thus, epinephrine was added to ropivacaine in this study. At present, there is no definitive clinical evidence to support that the addition of local infiltration analgesia to ACB + IPACK block can improve analgesic outcomes. 10 To avoid interference between the efficacy of ACB + IPACK block and local infiltration analgesia, thus local infiltration analgesia was not performed during surgery.
Several methodological approaches can be used to investigate the efficacy of a local anesthetic agent. The Dixon and Massy up-and-down design is the classic strategy to estimate the median effective dose (ED50). 31 However, the ED50 offers little clinical relevance. The biased coin up-and-down sequential design can determine higher quantiles (ED90 and ED95) directly, which provide useful clinical knowledge. In the biased coin up-and-down sequential design, volume assignment is carried out in a sequential and interactive method, which avoids that patients are randomized to doses with toxicity risk and seeks to volume treatments around the suitable doses.24,32 This is the advantage of using biased coin up-and-down sequential design to determine the MEV90 for ACB + IPACK block.
The doses of ropivacaine used for ACB + IPACK block.

ACB: adductor canal block.
IPACK: infiltration between the popliteal artery and capsule of the posterior knee.
To our knowledge, we provide the first estimate of minimum effective dose for ropivacaine in ACB + IPACK block. In future clinical practice and studies, researchers can refer to our results. We expect this finding to provide valuable information and supply a contribution to the evidence concerning ACB + IPACK block. In a broader context, we hope to promote an opioid-sparing multimodal pain management for TKA.
In our study, after completing the nerve block, none of the patients suffered nausea, vomiting or dyspnea before general anesthesia. During the process of this trial, no patients suffered diminished quadricep strength, residual paresthesia, residual neural deficits, or local anesthetic intoxication. It indicated that the dose of ropivacaine used in this study was safe.
Our results should be interpreted with caution in light of several limitations. First, the success rate can vary depending on the investigators’ definition of block success, caution should be exercised when comparing different studies. Second, the multimodal pain regimen used in this study may be different from that in other medical centers or other regions. For example, local infiltration analgesia was not used in this study. Different multimodal pain regimens may affect the results. Future work should address these limitations, as well as verify and extend our findings for a range of multimodal pain regimens.
Conclusions
This biased coin up-and-down sequential design-based trial provides evidence that ultrasound-guided single injection of 0.275% ropivacaine in 17.99 mL respectively (containing 2.0 μg/mL of epinephrine) can provide successful ACB + IPACK block in 90% of patients undergoing TKA. Our findings may be useful for standardizing future clinical practice and research toward optimizing pain management after TKA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 1.3.5 Project of Sichuan University West China Hospital. Grant ID: ZYJC18040.
Ethical approval
This study was approved by the Clinical Trials and Biomedical Ethics Committee of Sichuan University West China Hospital, and written informed consents were obtained from all participants. The clinical trial registration number was ChiCTR2100052731 (Date of registration: 4 November 2021). This trial was prospectively registered.
Clinical trial registration number
ChiCTR2100052731 (Date of registration: 4 November 2021). This trial was prospectively registered.
