Abstract
Purpose
Nontuberculous mycobacteria periprosthetic joint infection (NTMPJI) is a rare complication of hip or knee joint arthroplasty. The experience for outcomes of NTMPJI treatment is still limited. The objective of this study was to investigate the outcome of hip or knee nontuberculous mycobacteria periprosthetic joint infection following treatment with two-stage exchange arthroplasty.
Material and Methods
From 1995 to 2020, 12 patients with NTMPJI were treated with two-stage exchange arthroplasty at our institution. We collected and analyzed variables including demographic data, comorbidity, microbiological data, treatment outcome and antibiotic formula in bone cement.
Results
Mycobacterium abcessus (n = 6) and Mycobacterium chelonae (n = 2) constitute the majority of the cases. Five patients had early-onset PJIs and the other seven patients were late onset. The success rate of two-stage exchange arthroplasty was 66.7% (8 of 12). Three patients experienced infection relapse, and one patient had soft tissue compromise complication. Post-operative antibiotic therapy may not improve the success rate (4 of 6 cases, 66.7%). Based on in vitro study, the most commonly used effective antibiotic in bone cement spacer for nontuberculous mycobacteria was amikacin.
Conclusions
nontuberculous mycobacteria is a rare cause of PJIs and should be suspected especially in relatively immunocompromised patients. Resection arthroplasty with staged reimplantation is the preferred approach. Prolonged post-operative antibiotic therapy before reimplantation may not improve the success rate. Delayed revision surgery may not be needed and can be performed once C-reactive protein level is normal after a drug holiday.
Introduction
Prosthetic joint replacement for improving the life quality of individuals with degenerative joints has been increasingly practiced. Arthroplasty mostly achieves successful outcomes. However, periprosthetic joint infections (PJIs) still constitute a highly challenging complication. Infections may complicate 1%–5% of prosthetic joints, 1 and according to a previous study conducted in our hospital, the most common agents are Staphylococcus aureus and coagulase-negative staphylococci. 2 Infections related to less commonly identified organisms such as fungal and mycobacterial species may also occur. A study reported the clinical outcome of the treatment of tuberculosis (TB) PJIs, and the success rate was still inadequate. 3 Compared with Mycobacterium tuberculosis, nontuberculous mycobacteria (NTM) have lower virulence and exist in environments such as water or soil. The identification of NTMPJI is often complicated by a lack of clinical suspicion, delayed treatment and there are only a few case reports of treatment of NTMPJI. In addition, some of the NTM can be classified as rapidly growing mycobacteria (RGM), which require <7 days to grow on culture. This characteristic is more like bacterial growth which may be quite different from mycobacteria infection. RGM usually cause skin and soft tissue infections, especially in immunocompromised individuals which are difficult to be cured.4,5 Thus, we sought to investigate the treatment outcome of PJI caused by NTM.
Patients and methods
We conducted a retrospective case series study which includes patients who received two-stage exchange arthroplasty caused by NTM at the Department of Orthopedic Surgery in our hospital between January 1995 and October 2020. Institutional review board approval was obtained from our hospital’s Medical Foundation Institutional Review Board (IRB no.201601034B0 and no.201403539B0D001). And for this type of retrospective study, informed consent was waived by our hospital’s Institutional Review Board that approved the study. 15 patients were identified, and three patients were excluded due to pyogenic arthritis. Finally, 12 patients presenting with hips or knees periprosthetic joint infection were treated with two-stage exchange arthroplasty and enrolled in the outcome analysis.
All patients received resection arthroplasty with antibiotic-loaded spacers by different orthopedic surgeons. This procedure was standardized that included radical debridement, removal of the prosthesis, and temporary implantation with antibiotic-loaded bone cement spacer (Figure 1. Mobile hip antibiotic spacer: A 53-year-old male patient has left hip nontuberculous mycobacteria periprosthetic joint infection. Two stage revision arthroplasty was performed with a temporary antibiotic loaded hip spacer) (Figure 2. Mobile knee antibiotic spacer: A 62-year-old female has left knee nontuberculous mycobacteria periprosthetic joint infection and septic loosening of prosthesis. Two stage revision arthroplasty was performed with a temporary antibiotic loaded knee spacer), and administration of short-term systemic antimicrobial agents postoperatively during admission period .
6
The antibiotic formula or whether to prescribe oral antibiotics were decided according to the clinician’s discretion. A 53 years old male patient has left hip nontuberculous mycobacteria periprosthetic joint infection. Two stage revision arthroplasty was performed with a temporary antibiotic loaded hip spacer. A 62 years old female has left knee nontuberculous mycobacteria periprosthetic joint infection and septic loosening of prosthesis. Two stage revision arthroplasty was performed with a temporary antibiotic loaded knee spacer.

We routinely arranged 18 sets of culture including culture for aerobic bacteria, anaerobic bacteria, fungus and mycobacterium species. We aspirated synovial fluid and took synovium and granulation tissue for deep tissue culture. And the diagnosis of PJIs caused by NTM was confirmed by positive culture results for the organisms collected from periprosthetic tissue specimens or joint aspirates, using Löwenstein-Jensen medium with up to 3 months of incubation. Reimplantation of the prosthesis was performed after successful treatment. Treatment success was defined as well functioning joint and normal C-reactive protein level (CRP) without using anti-mycobacterium drugs, and there was no relapse of NTM infection after reimplantation for at least 1 year of follow-up. Treatment failure was defined as any of the following: 1 infection relapsed with original microorganism, the infection status remained positive, or permanent excision arthroplasty due to uncontrolled infection; 2 unable to have reimplantation because of soft tissue compromise.
We reviewed all the medical records of the patients enrolled in this study. Demographic characteristics and data regarding duration of symptoms, surgical intervention, organism identification, initial C-reactive protein (CRP) level, comorbidity, antibiotic formula in bone cement, and treatment outcome were collected.
Results
Demographic data and clinical outcomes of patients with periprosthetic joint infections due to nontuberculous mycobacteria.
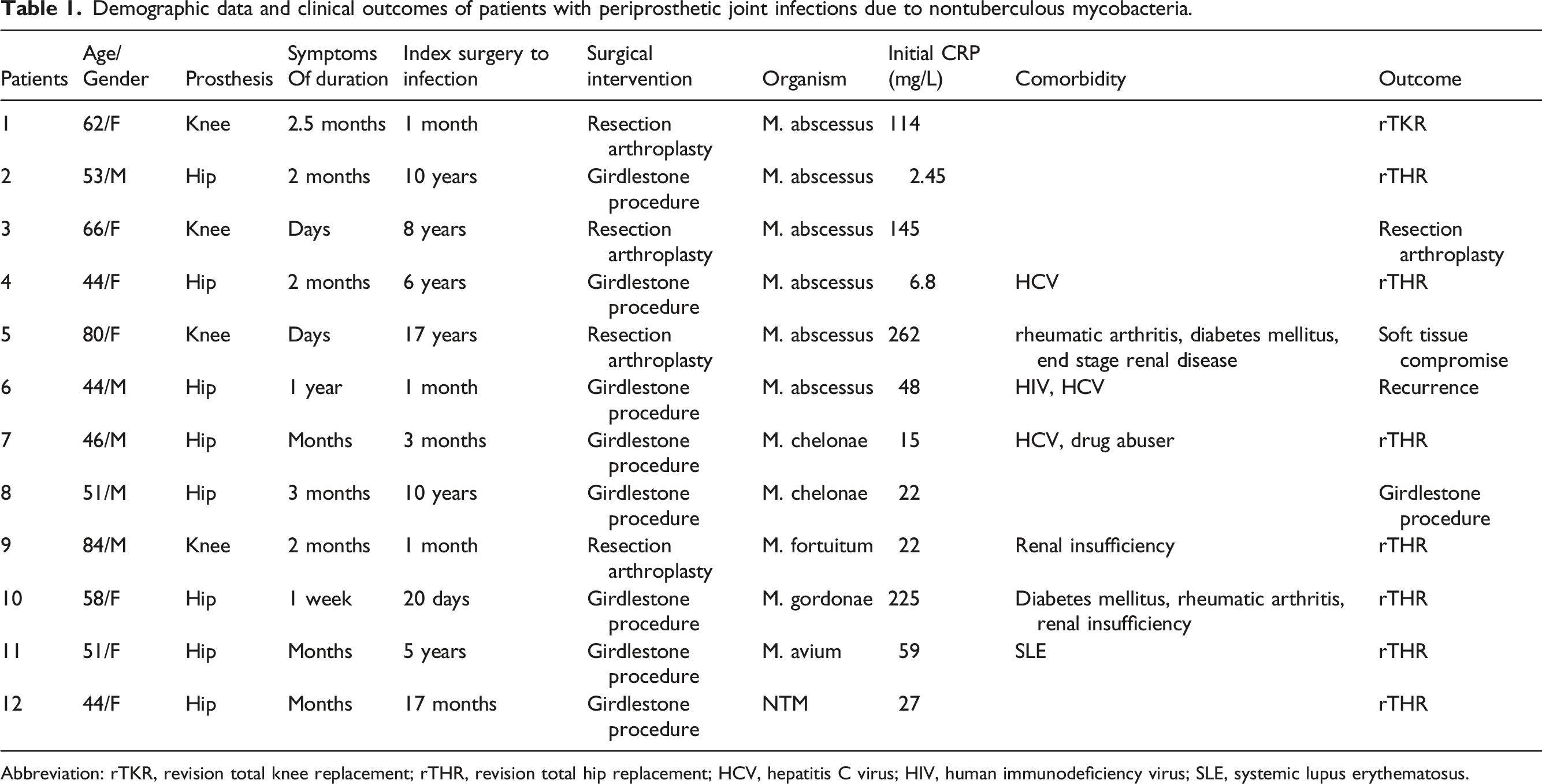
Abbreviation: rTKR, revision total knee replacement; rTHR, revision total hip replacement; HCV, hepatitis C virus; HIV, human immunodeficiency virus; SLE, systemic lupus erythematosus.
Selected antimicrobial regimens for nontuberculous mycobacteria.

After surgical intervention, five patients received repeat resection arthroplasty because their PJIs were not controlled or relapsed. Reimplantation was performed in eight patients, with the reimplantation time ranging from 1.5 months to 4.5 years after prosthesis explantation. The four patients who did not undergo reimplantation were infected with Mycobacterium abscessus or Mycobacterium chelonae. In addition, of the four patients, one never received revision arthroplasty because of skin necrosis and another did not receive revision arthroplasty because of infection induced by multiple PJI pathogens.
Discussion
Despite the high surgical volume of arthroplasty, NTM are still a rare cause of PJIs, although the pathogens are widely distributed in the environment, such as in water or soil. Moreover, most of the papers describing NTM are case reports. This paper reports the largest series to date, to our knowledge, of NTM as a cause of PJIs. Although the guideline for treating PJI is already well established, the rarity of the NTM as a PJI pathogen still made us facing difficulty to treat patients. This include lacking the experience of knowing clinical presentation, antibiotic usage in spacers, duration of treatment needed and timing for revision. We report our clinical experience with the treatment modality and surgical outcomes for PJIs caused by NTM. Atypical mycobacteria are usually classified using the Runyon criteria on the basis of the growth rate and pigmentation of colonies grown in vitro: Group I includes slow-growing photochromogens such as Mycobacterium kansasii and Mycobacterium marinum. Group II includes slow-growing scotochromogens such as M. gordonae and Mycobacterium szulgai. Group III includes nonchromogens such as M. avium intracellulare and Mycobacterium ulcerans. Group IV includes RGM such as Mycobacterium fortuitum, M. chelonae, and M. abscessus, which have the ability to produce growth on agar within 1 week. 7
M. fortuitum, M. chelonae, and M. abscessus constitute the majority of RGM that cause human diseases; they are major causes of cutaneous, pulmonary, and nosocomial infections.8,9 Our findings are consistent with those in the literature. Six cases were caused by M. abscessus, two by M. chelonae, one by M. fortuitum, two by a slow-growing group (M. gordonae and M. avium), and one by a pathogen not being classified into a subgroup (Table 1). Our patients were relatively young; only four patients were aged >60 years. The majority of cases involved avascular necrosis or hip osteoarthritis secondary to dysplasia. However, our patients were still relatively immunocompromised. Half of our patients may have had underlying comorbidities such as hepatitis C virus infection, human immunodeficiency virus infection, renal insufficiency, rheumatoid arthritis, systemic lupus erythematosus, or intravenous drug abuse. Our findings are quite different from those of a previous study that revealed that a majority of the cases involved immunocompetent individuals. 8 Although the route of developing NTMPJIs cannot be clearly identified, we think that immune compromised may be a major concern of causing these infections. We believe that young patients with a relatively immunocompromised state should still be aware of infections induced by NTM.
Similar to most cases of PJIs caused by bacteria, the CRP level in our study was often elevated. However, in two of our cases, CRP levels were within the normal range, signifying that infections induced by NTM may be insidious. According to our literature review, early onset of infections induced by NTM is attributed to intraoperative contamination from tap water used during implantation, and late onset of PJIs is attributable to direct inoculation of NTM.8,10,11 In our study, five patients had early-onset PJIs (within 3 months), which we considered to be attributable to intraoperative contamination during implantation. The other seven patients developed PJIs a couple of years after their index surgery, but the route for NTM seeding was still unclear. The most likely reason for this is microtrauma from the skin, according to our literature review.5,12
The current approach to treating PJIs due to NTM involves resection arthroplasty in combination with effective antimycobacterial therapy.10,13–15 Patients with a retained prosthesis may experience relapse because of architecturally complex mature biofilm formed by mycobacteria.8,16–18 In our institute, all patients underwent two stage resection arthroplasty with antibiotic spacers, but only six patients received postoperative antibiotics for NTM. However, of the 12 patients, 8 were determined to be free of relapse and have received revision surgery. The reason we chose two stage exchange arthroplasty rather than one stage is that one stage exchange arthroplasty need effective post-operative antibiotic treatment for at least 6 weeks.19,20 The mean culture period for confirming NTM infection in our study is about 2 weeks. So the effective antibiotic usage for one stage exchange arthroplasty may be delayed. In addition, the indications for one stage exchange arthroplasty include absence of immunocompromised and significant soft tissue or bony compromise and concurrent acute sepsis. Multidrug resistant or atypical organism may need two stage exchange arthroplasty. 21 In our series, most of the patients are relatively immunocompromised, and NTMs are also atypical pathogen, thus two stage exchange arthroplasty is better than one stage for this type of infection. We speculate that the overall success rate (66.7%), which is comparable to the TB infection rate in our previous study, 3 may be related to the effectiveness of local antibiotics released from the spacers. Use of antibiotic-loaded spacers is necessary to prevent joint contracture.14,15 In addition, reoperation for spacer replacement may often be required because diagnosing infections induced by NTM in the first surgery is usually difficult. Thus, we routinely sent mycobacterial smears and cultures for all PJI cases to improve the diagnosis rate. As revealed by our previous study, 22 antibiotic-loaded spacers have a persistent release effect that leads to no difference in the overall success rate of postoperative short-term or long-term antibiotic therapy. Although post-operative oral antibiotics may not improve the overall success rate (66.7%, 4 of 6), we noticed that success rate can be ameliorated in RGM group (66.7% vs 33.3%). Because the growth characteristics of RGM are similar to those of bacteria, the use of locally released antibiotics may still be an effective treatment strategy. Iinfections induced by RGM are often resistant to several groups of antimycobacterials. According to results of in vitro antimycobacterial activity testing, amikacin, cefoxitin, and clarithromycin are effective antibiotics and are thus commonly used in spacers or post-operatively in our institute.23,24 Thus, the regimen of antibiotic-loaded spacers used in our study mainly included vancomycin and ceftazidime, with amikacin or tigecycline being used added to cover most of the possible pathogens.
Staged reimplantation constituted our treatment goal for PJIs. In our study, no evidence of relapse was obtained in eight patients who underwent reimplantation. The other four patients never received revision surgery. The timing for reimplantation was based on normal CRP levels after a drug holiday or resolution of clinical symptoms. Thus, in our study, reimplantation was performed from 1.5 months to 4.5 years after prosthesis explantation according to the clinical findings. According to our literature review, prolonged antibiotic treatment for complete remission of infections induced by NTM is necessary before reimplantation, but the optimal duration is still not well defined.8,12,13,24,25 The review also indicated that a longer delay in reimplantation (>6 months) may also result in a more favorable outcome. However, in our experience, reimplantation within 6 months could also be successful once the infection has resolved, as indicated by normal CRP and negative culture results. This study also included two immunocompromised patients; in these patients, the pathogens belonged to slow-growing species. A previous case report showed that the treatment outcome of a PJI caused by M. avium intracellulare is variable and similar to that of infections caused by NTM species, which need a prolonged course of antimycobacterial treatment. 26 Nevertheless, we successfully treated the included patients by using our protocol, which did not involve prolonged administration of antibiotics before reimplantation; this may be due to the effectiveness of the local antibiotics in the spacers.
This study has several limitations. First, this was a retrospective review performed over an extended period of time due to the rarity of NTMPJI which resulted in small sample size and made it impossible to perform a randomized trial. Second, the variability factors due to different operating surgeons that include type of antibiotic used in cement, duration of oral antibiotics, and interval between resection arthroplasty and reimplantation. Finally, some of the patients were lost to follow-up in our hospital that data might have been lost. However, we believe our findings still provide valuable outcome data concerning the use of two-stage exchange arthroplasty for treating NTMPJI despite these limitations.
In conclusion, NTM are a rare cause of PJIs but should be considered in the list of possible pathogens, especially in relatively immunocompromised patients or those with uncontrolled infections despite receipt of broad-spectrum antibiotics. We suggest that explantation of an infected prosthesis and temporary articulation with an antibiotic-loaded spacer that contains effective antimycobacterials is the key to treating NTMPJIs. In addition, post-operative antibiotic may improve the treatment outcome for RGM group. Although the timing of reimplantation is still not well defined, we believe that revision surgery can be performed once the CRP level is normal and no clinical sign of infection is present after a drug holiday. And with accurate diagnosis and proper surgical treatment, NTMPJI could be cured.
Footnotes
Author’s contributions
All authors had access to the data and participated in preparation of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Chang Gung Memorial Hospital Grant (CORPG3G0541 and CORPG5G0021) for supporting this research.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Consent to participate
For this type of retrospective study, formal consent is not required.
Consent for publication
Chih-Hsiang Chang hereby declares that I participated in the study. I have read the final version and give my consent for the article to be published.
Availability of data and material
All data generated during this study are included in this article.
