Abstract
Introduction
The COVID-19 pandemic has captured the attention of healthcare systems worldwide, and testing remains a mainstay of preoperative testing before all surgical procedures, including shoulder arthroplasty. Other viral illnesses, specifically influenza, while still having severe complications, are either overshadowed by testing for COVID-19 or not tested at all.
Methods
A multi-institutional cohort study using the TriNetX network examined patients undergoing total or hemi shoulder arthroplasty from January 2020 to March 2024. Matched cohorts were created: one with influenza and one with COVID-19 infection, 14 days before surgery.
Results
A total of 3233 and 18,209 patients were included in the influenza and COVID-19 cohorts, respectively. The matched cohorts both consisted of 3232 patients. Patients in the influenza cohort had a significantly higher risk of sepsis (95% CI [1.264, 2.528],
Discussion
Patients with recent influenza infections prior to shoulder arthroplasty have a greater risk of medical complications, with no difference in implant-related complications compared to COVID-19, emphasizing the need for perioperative patient counseling and surgical planning.
Keywords
Introduction
COVID-19, from 2019 to the present times, has profoundly affected healthcare around the world, including patients undergoing elective and non-elective orthopedic surgery. The specific effects of the pandemic on elective procedures, namely shoulder arthroplasty (SA), have yet to be rigorously investigated. Some studies have shown changes in the prevalence of elective SA,1–3 while others have identified an increase in outpatient SA procedures. 4 Johnson et al. 5 reported no difference in clinical outcomes in pre-COVID patients versus post-COVID patients.
Preliminary research in this field has demonstrated that patients diagnosed with COVID-19 2 weeks before total joint arthroplasty were at increased risk of pneumonia, acute myocardial infarction, sepsis within 90 days, and 30-day mortality. 6 Stevens et al. 7 found similar outcomes in range of motion and strength between the COVID-19 and control groups undergoing reverse total SA (rTSA). Another study comparing a 2019 cohort to a 2020 cohort showed that postoperative follow-up and physical therapy for SA were delayed in the 2020 group; however, this delay did not result in differences in complication or readmission rates compared to those of the 2019 cohort. 8
Amidst this collection of nuanced information, it is crucial to understand how COVID-19 specifically impacts SA surgeries. Chokshi and Somerson 9 have outlined how COVID-19 increases the postoperative risk of complications in SA. However, the current literature does not compare COVID-19 with other similar, common viruses to evaluate relative impact. Moreover, COVID-19 has become the dominant viral infection screened in preoperative workups, overshadowing other viral infections like influenza, which have also been recognized to increase the risk of complications across all surgeries. 10
Preoperative screening for COVID-19 became standard practice in 2020,11,12 and by 2021, new studies updated screening strategies to be more selective. 13 A thorough review of the current literature suggests that similar screening measures are not routinely implemented across healthcare systems for influenza. Despite influenza's well-recognized impact on postoperative outcomes, it has not been consistently incorporated into preoperative viral screening protocols, highlighting a notable gap in current surgical practice.14,15 The present study directly compared postoperative outcomes in patients with COVID-19 and influenza infection undergoing total SA (TSA) and rTSA. Utilizing a multi-network database, we aimed to assess postoperative outcomes in these two groups.
Methods
Study design and data source
This retrospective cohort study utilized TriNetX, a comprehensive database containing de-identified electronic medical records from 89 healthcare organizations within the research network. The database was queried to identify patients who underwent SA from 1 January 2020 to 27 March 2024, assessing 90-day postoperative medical outcomes and two-year implant complications. Due to the use of de-identified patient records, this study was exempt from Institutional Review Board approval.
Cohort selection
Patients who underwent SA were divided into two cohorts: those infected with influenza within 14 days prior to SA and those infected with COVID-19 within 14 days prior to SA, based on prior studies demonstrating the significance of this 14-day interval (Leeds). 16 Propensity score matching was conducted using logistic regression on the TriNetX platform, utilizing nearest-neighbor techniques with a tolerance level of 0.01 to balance demographics and comorbidities, including diabetes, tobacco use, obesity, osteoporosis, alcohol-related disorders, chronic obstructive pulmonary disease, chronic kidney disease, liver diseases, and heart failure. These factors were selected based on prior studies demonstrating that they are established risk factors influencing orthopedic surgical outcomes (Dlott et al., 17 Gustafsson et al., 18 Kamps et al., 19 and Mihalko et al. 20 ).17–20 Other variables, such as vaccination status and symptom duration, were considered but could not be matched for within the TriNetX system.
Study outcomes
The primary outcomes of this study are categorized into major medical complications within 90 days post-SA and implant-related complications within 2 years post-SA. Within the first 90 days, we assessed sepsis, infection, wound disruption, pulmonary embolism (PE), deep vein thrombosis (DVT), myocardial infarction (MI), stroke, readmission, anemia, pneumonia, renal failure, transfusion needs, and opioid analgesic usage. For the two-year follow-up, the outcomes included periprosthetic fracture, prosthetic joint infection (PJI), prosthetic dislocation, mechanical loosening, revision SA , and injury of the brachial plexus. These outcomes were analyzed using chi-squared exact tests for categorical variables and univariate regressions within the matched cohorts.
Statistical analysis
Relative risk ratios (RRs) and 95% confidence intervals for all outcomes were calculated using the advanced analytics tools provided by TriNetX, which supports robust data analysis on large-scale, de-identified datasets. Significance was assessed with a
Compliance and ethical considerations
This study utilizes de-identified data, compliant with HIPAA and other relevant regulations, ensuring adherence to stringent data privacy regulations. Given the de-identified nature of the data, the study received an exemption from full IRB review.
Results
Demographic and clinical characteristics
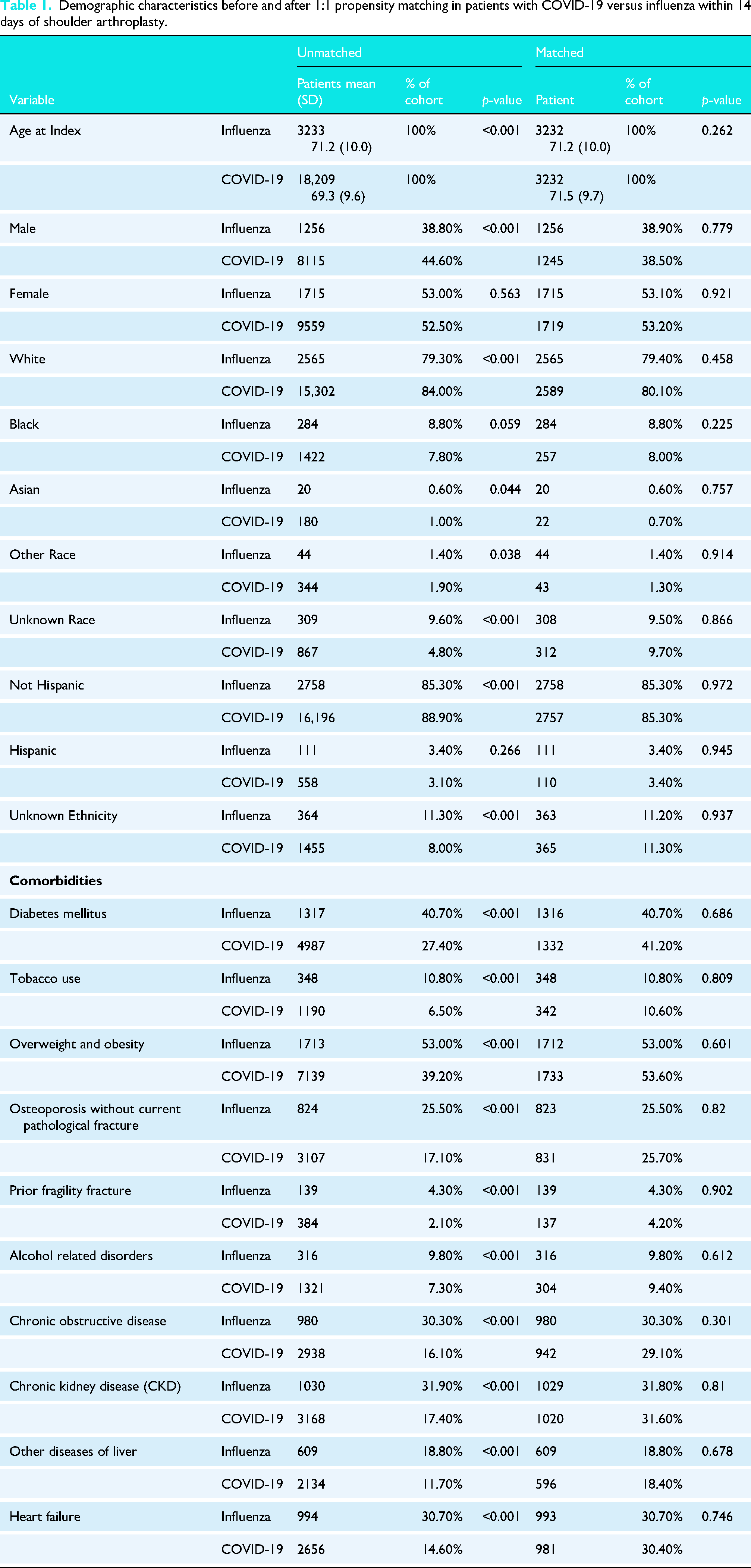
The study identified 21,442 patients who underwent SA within 14 days of either a COVID-19 or influenza infection, consisting of 3233 influenza patients and 18,209 COVID-19 patients. After 1:1 propensity score matching, 3232 pairs were included. The matched cohorts had a mean follow-up of 886 days (influenza) and 869 days (COVID-19) with standard deviations of 644 and 655 days, respectively. Before matching, the COVID-19 cohort was younger, predominantly White, non-Hispanic, and had fewer comorbidities. Post-matching, these differences were no longer significant. Gender distribution was nearly identical (38.80% males in the influenza group vs. 38.50% in the COVID-19 group), as were rates of diabetes mellitus (40.70% in both cohorts) and chronic kidney disease (31.80% vs. 31.60%), along with racial and ethnic distributions. Detailed cohort characteristics before and after matching are in Table 1.
Demographic characteristics before and after 1:1 propensity matching in patients with COVID-19 versus influenza within 14 days of shoulder arthroplasty.
Medical complications
Significant differences were observed in the rates of various 90-day medical complications post-SA between the influenza and COVID-19 cohorts. Sepsis occurred in 3.3% of influenza patients compared to 1.9% in COVID-19 patients (
Medical complications within 90 days of surgery in patients diagnosed with influenza or COVID-19.
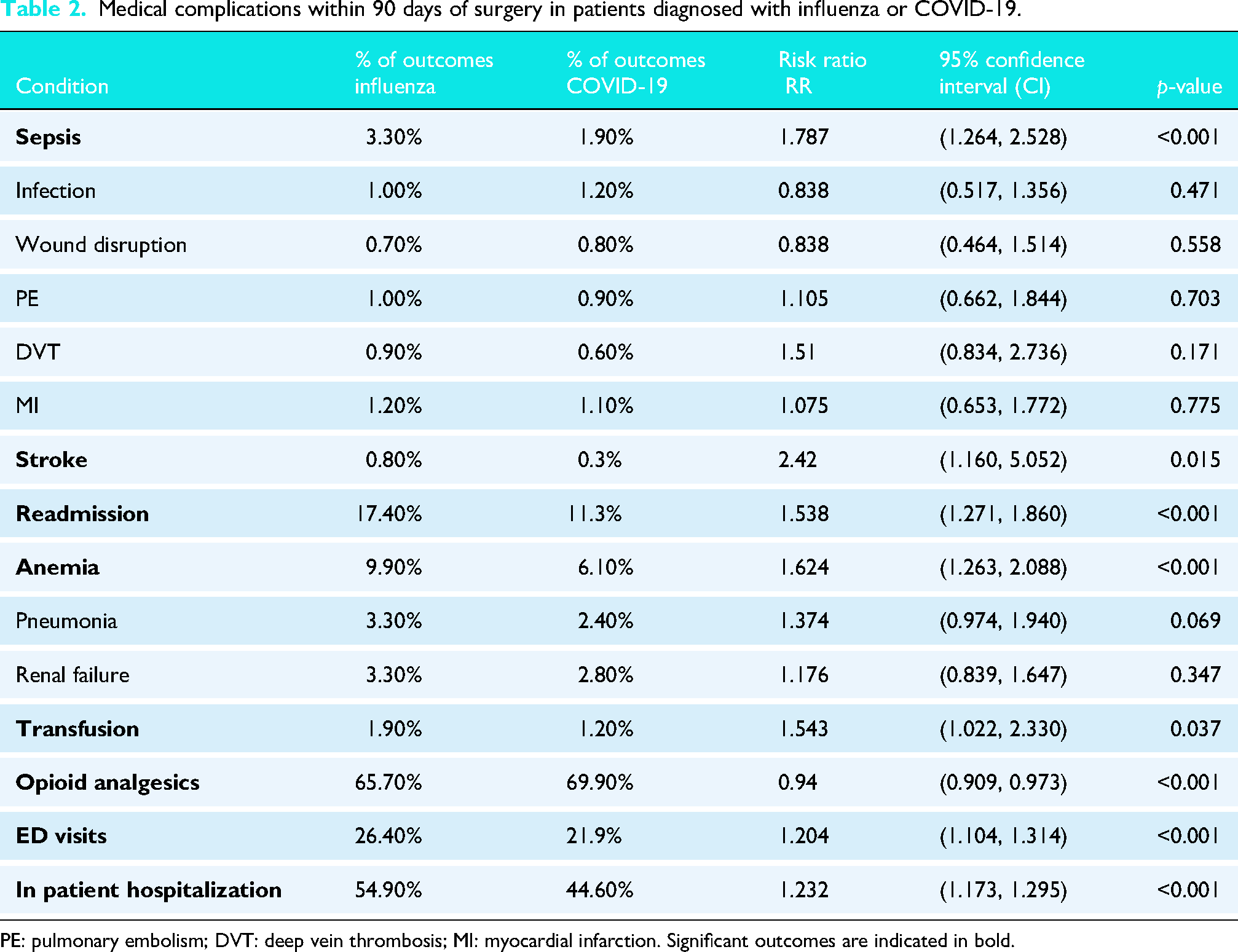
PE: pulmonary embolism; DVT: deep vein thrombosis; MI: myocardial infarction. Significant outcomes are indicated in bold.
Mechanical complications
Over the 2-year follow-up, the analysis of mechanical complications revealed no significant differences between the influenza and COVID-19 patients. Rates of periprosthetic fracture (0.80% vs. 1.20%,
Mechanical complications within 2 years of surgery in patients diagnosed with influenza or COVID-19.
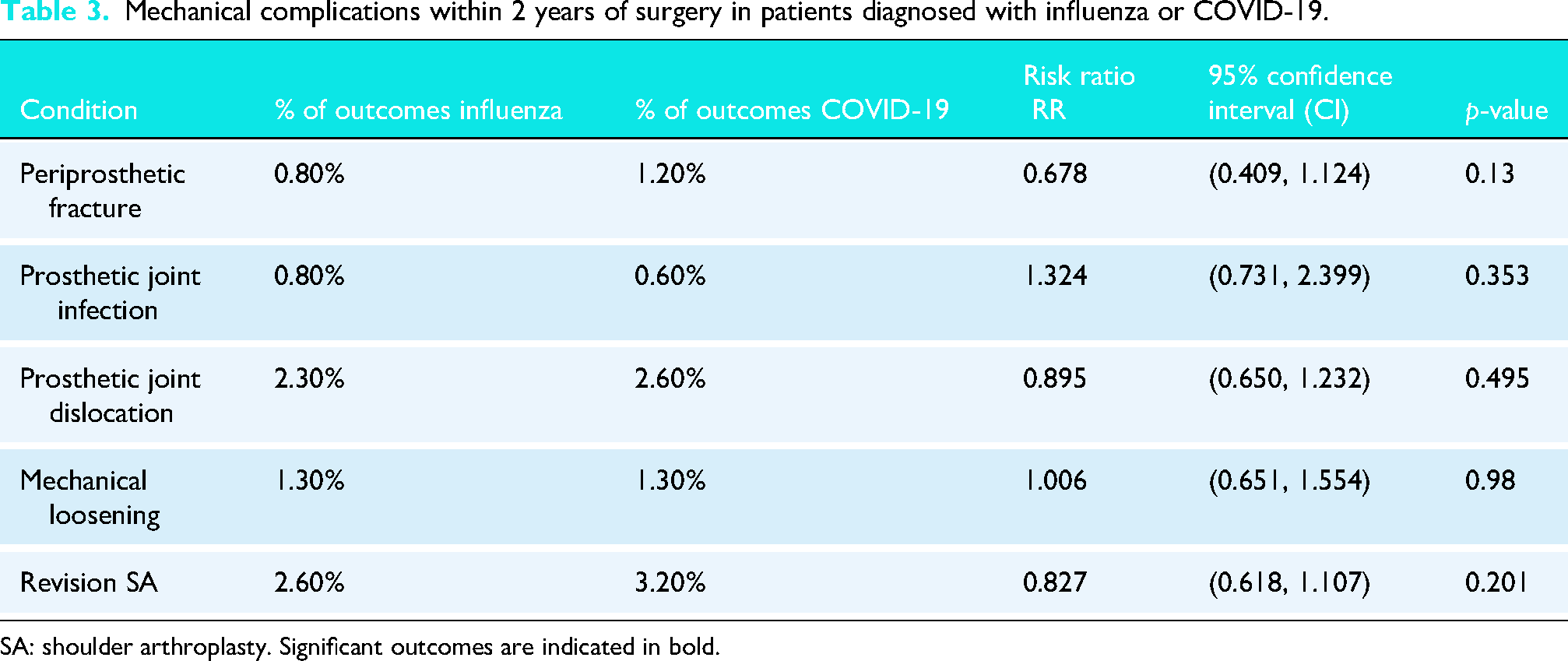
SA: shoulder arthroplasty. Significant outcomes are indicated in bold.
Discussion
This study compared outcomes of COVID-19 and influenza patients to identify differences in medical and mechanical complications. Our findings reveal that SA patients with recent influenza diagnoses had significantly higher risks of sepsis, stroke, readmission, anemia, transfusion, ED visits, and inpatient hospitalization within 90 days of SA compared to those with recent COVID-19 diagnoses. Conversely, patients with COVID-19 were at greater risk for opioid analgesic use during the same period.
Much of the literature in this area has focused on the outcomes of patients during the pandemic or those with COVID-19. No studies have specifically compared outcomes of SA in COVID-19 and influenza patients. To our knowledge, there is only one study assessing the rate of postoperative complications following surgery in patients diagnosed with influenza preoperatively. Lam et al. 10 characterized the postoperative consequences of influenza, finding that patients with a history of influenza 1–7 days before surgery had a higher risk of pneumonia, septicemia, acute renal failure, and urinary tract infection compared to uninfected patients. Additionally, just two studies examine the relationship between preoperative COVID-19 diagnoses and postoperative complications in arthroplasty surgeries. Pincavitch et al. 6 reported increased risk in knee and hip arthroplasty patients who had a recent (within 60 days) COVID-19 diagnosis. Similarly, Chokshi and Somerson 9 reported higher odds of sepsis, ED visits, hospital admissions, periprosthetic fracture, superficial wound infection, DVT, PE, and revision surgery within 60 days of TSA. While there are studies investigating either influenza or COVID-19, ours is the first to directly compare outcomes between the two using a 14-day preoperative infection interval. Our findings indicate that having a COVID-19 diagnosis prior to SA is not associated with worse outcomes than influenza, suggesting clinicians should factor in these differential risks when developing and implementing patient care plans.
While our study does not determine the optimal timing for surgery, it highlights the need for further research into whether delaying surgery for patients with elevated preoperative risk could improve outcomes. Future studies could investigate whether extending the preoperative period affects postoperative complications and patient outcomes. In a comprehensive international study conducted in October 2020, researchers found a significant increase in postoperative mortality among patients who had been diagnosed with COVID-19 within 7 weeks prior to their surgery. 21 However, this increased risk was not observed in patients who had recovered for 7 weeks or more, except in those who continued to exhibit symptoms beyond this period. 21 Conversely, Lam et al. 10 identified an association between influenza within 14 days preoperatively and an increased risk of postoperative complications, especially when the infection occurred within 7 days prior to major surgeries—defined as procedures requiring general, epidural, or spinal anesthesia and hospitalization lasting more than 1 day. The American Society of Anesthesiologists also recommends considering the severity of the initial infection, potential risk of ongoing symptoms, comorbidities, frailty status, and surgery complexity when deciding the optimal waiting period for surgery. 22 Generally, waiting at least 7–14 days after an influenza infection and 7 weeks after a COVID-19 infection is supported by literature.10,21 However, no studies have examined the effects of COVID-19 vaccines or new variants, nor are there studies specifically investigating the impact of influenza on SA surgeries. Thus, no definitive optimization strategy for SA has been suggested by research at this time. More investigation is needed to better characterize risk profiles for these infectious diseases in patients undergoing SA, especially in the context of short-term and long-term complications.
This study is not without limitations. Our analysis benefits from a large patient cohort, enabling the ability to control patients medically. Data from multiple institutions bolsters generalizability. Yet, limitations include difficulty distinguishing SA types, lack of granularity in data hindering factors like bone loss quantification, and inability to track care transitions outside the TriNetX network and patient-reported outcomes. Billing practices introduce potential errors. Missing data on surgery indication, implant type, cement use, and medication adherence are also acknowledged. It is important to recognize that this study did not account for differences in viral variants, temporal seasons, vaccination status, treatment approaches, illness severity, or changes over time during the 2020–2024 study period, all of which may influence outcomes. Additionally, the COVID-19 cohort was much larger, reflecting real-world testing practices in which COVID-19 screening was performed more routinely, including in asymptomatic patients. In contrast, individuals who tested positive for influenza were typically symptomatic, which could influence clinical presentations and outcomes. Furthermore, because multiple postoperative outcomes were evaluated simultaneously, the potential for a Type I error is increased.
The results of our study suggest a significantly higher risk of postoperative complications such as sepsis, stroke, readmission, anemia, transfusion, ED visits, and inpatient hospitalization during the SA perioperative period for influenza patients. These results highlight the importance of contextualizing preoperative risk for SA candidates with recent influenza infection, while recognizing that further research is needed before specific clinical recommendations can be made.
Level of Evidence
Level III; Retrospective Case-Control Comparison using Large Database; Prognosis Study.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
