Abstract
Background and objectives
Chondrogenesis of bone marrow mesenchymal stem cells (BMSCs) exerts great function during the pathogenesis of osteoarthritis (OA). Studies have reported the association of plexin B1 (PLXNB1) with OA pathogenesis. In this study, the upstream mechanism and function of PLXNB1 in this disease were explored.
Methods
Flow cytometry was applied to test BMSC characterization. Chondrogenic differentiation of BMSCs was evaluated by Alcian blue staining. The expression of PLXNB1, miR-362-5p, miR-501-5p, miR-1827, miR-500-5p was measured using RT-qPCR analysis. The protein levels of PLXNB1, Aggrecan, and Silent information regulator factor 2-related enzyme 1 (SIRT1) were determined by western blotting. Binding relationship between miR-362-5p and PLXNB1 was confirmed using bioinformatics analysis and luciferase reporter assay. The in vivo model of OA was established in Sprague-Dawley rats which received medial meniscus instability surgery. For histopathological examination, cartilage tissues in the knee joint of rats were stained with hematoxylin and eosin. Micro-CT analysis was employed to observe the changes of morphometric indices including average trabecular separation, average trabecular thickness, and bone volume fraction.
Results
BMSCs were identified to possess the characteristics of mesenchymal stem cells. PLXNB1 was observed to be highly expressed during chondrogenic differentiation of BMSCs and PLXNB1 overexpression promoted BMSC chondrogenic differentiation. Mechanically, PLXNB1 was targeted by miR-362-5p. In rescue assays, miR-362-5p reversed the effects of PLXNB1 on chondrogenic differentiation of BMSCs. In the in vivo experiments, upregulated PLXNB1 expression alleviated joint injury of OA rats. Additionally, overexpressed miR-362-5p and downregulated PLXNB1 expression levels were detected in OA rats.
Conclusion
MiR-362-5p promotes OA progression by suppressing PLXNB1.
Introduction
As a chronic and degenerative joint disorder, osteoarthritis (OA) affects the health and life quality of middle-aged and elderly people worldwide. 1 Clinically, OA is manifested as joint dysfunction, stiffness, and pain. Osteophyte formation, synovial inflammation, subchondral bone remodeling, and progressive cartilage degradation, are the primary pathophysiological characteristics of OA.2,3 At present, common methods for OA treatment include surgical arthroplasty and administration of hyaluronic acid, cyclooxygenase inhibitors, and non-steroidal anti-inflammatory drugs. Nevertheless, these approaches can partially relieve pain and may provoke severe complications. 4 Hence, exploration of effective and safe therapeutic measures to prevent OA is of great importance.
Among different types of stem cells, adult mesenchymal stem cells (MSCs) seem to be appropriate candidates for cell therapy based approaches due to easy access, high proliferation rate, immunomodulatory properties, angiogenic activities, differentiation capacity, anti-oxidative effects, and anti-hypertrophic scar properties. 5 BMSCs also have the potential to regenerate chondrocytes and form cartilaginous tissues, and they are widely utilized for bone remodeling and cartilage repair in clinical applications. 6 In recent years, BMSC transplantation has become a therapeutical strategy for OA treatment. 7
Plexin B1 (PLXNB1), is a cell surface receptor widely expressed in neural, hematopoietic, and endothelial cells. Plexin B1 belongs to a large family of plexin proteins which bind to ligands called semaphorins. 8 PLXNB1 is expressed in bone marrow stromal/osteoblast precursor cells. 9 Notably, a previous report mentioned that PLXNB1 was under-expressed in osteoarthritic bone. 10 Additionally, Hopwood et al. 10 reported that PLXNB1 was downregulated in the trabecular bone of OA patients. However, its specific role in OA remains unknown.
MicroRNAs (miRNAs) are a group of single-stranded noncoding RNAs with 18–24 nucleotides in length. Through binding to the 3′ untranslated region (3′ UTR) of target mRNAs, miRNAs are able to control their expression and limit their protein-coding abilities. 11 An increasing magnitude of studies have revealed that the physiological and developmental processes of OA can be modulated by miRNAs. 12 Especially, to date, multiple miRNAs have been validated to regulate chondrogenesis, thereby participating in cartilage-associated disorders, including OA. For example, miR-130b interacting with SOX9 inhibits BMSC chondrogenesis. 13 MiR-132-3p enhances MSC differentiation into chondrocytes by regulating ADAMTS-5. 14 MiR-410 targets Wnt3a to boost BMSC chondrogenesis. 15 Nevertheless, the upstream miRNAs that regulate PLXNB1 to mediate cartilage differentiation of BMSCs in OA have not been investigated.
In this report, the functional role of PLXNB1 in BMSC chondrogenic differentiation and cartilage repair in OA rats was explored. Additionally, the related mechanism of PLXNB1 in OA was also investigated. This study may deepen our understanding of OA etiology and provide a potential therapeutic method for OA treatment.
Material and methods
Isolation and identification of rat BMSCs
All experiments were approved by the Animal Experimentation Ethics Committee of Wuhan Hospital of Traditional Chinese Medicine. As previously described, 16 2-week-old Sprague-Dawley (SD) rats (Vital River Co. Ltd, Beijing, China) were chosen for the isolation of BMSCs. Briefly, after the rats were sacrificed, tibia bones and femur were collected. Subsequently, Dulbecco’s modified Eagle medium (DMEM; Gibco, NY, USA) was used to flush out the bone marrow. After the bone marrow was subjected to centrifugation, the supernatant was discarded and cells were resuspended in DMEM containing 10% fetal bovine serum (FBS; Gibco). Then, the cells were treated with 6 mL of Percoll separation solution (1.073 g/cm3) and were centrifugated at 800× g for 20 min. The cells in the middle yellow-brown ring cloud layer were collected and washed with phosphate-buffered saline (PBS) twice. A low-sugar DMEM that contained 0.1% FBS was used to resuspend and culture cell pellets in a saturated humidity incubator with 5% CO2 at 37°C. After 5 days, the medium was changed. Thereafter, the medium was changed every 3 days. The adherent cells were sub-cultured according to a 1:2 ratio when cell confluence reached 80%–90%. BMSCs at the third generation with good growth conditions were made into single-cell suspension. For the characterization of BMSCs, flow cytometry analysis was employed to identify the cell surface markers (CD34, CD45, CD44, and CD29). Cells were washed in PBS and incubated with primary antibody against human CD34 (ab81289, abcam), CD45 (ab25386, Biolegend), CD44 (ab25340, Biolegend) and CD29 (ab27947, Biolegend) for at 4°C for 30 min, followed by incubation with FITC I-labeled goat anti-mouse secondary antibody for half an hour. At last, the expression of f these surface antigens on BMSCs was analyzed by Coulter EPICS-XL flow cytometry (CA). Windows MDI 2.8 software (Scripps Institute, CA) was utilized to create corresponding histograms.
Chondrogenic induction of BMSCs
According to the previous literature, 17 BMSCs at the third passage were cultured in chondrogenic induction media (50 mg/mL Vitamin C, 1 mg/mL BSA, 40 mg/mL proline, high glucose DMEM, 100 mg/mL sodium pyruvate, and 10% FBS), followed by addition with 100 nM dexamethasone in the absence or presence of TGF-β1 (10 ng/mL).
Alcian blue staining
Alcian blue staining was conducted at 0, 7, and 14 days after chondrogenic induction. In brief, BMSCs were fixed with 4% paraformaldehyde and then stained with 0.1% Alcian blue 8GX (MICXY REAGENT, Chengdu, China) for 40 min. The absorbance at 590 nm was measured. A flatbed scanner (Cannon, USA) was employed to detect signals and Image-Pro Plus software (Media Cybernetics Inc., Bethesda, MD) was used to quantify the staining intensity. 18
Cell transfection
MiR-362-5p mimic was used to overexpress miR-362-5p with NC mimic as negative control. Full-length PLXNB1 was inserted into the pcDNA3.1 vector to elevate PLXNB1 with pcDNA3.1 as negative control. All plasmids used in this study were purchased from Genechem (Shanghai, China). When the cell confluence reached 80%, a total of 50 nM pcDNA3.1/PLXNB1, 50 nM miR-362-5p mimic or 50 nM of their corresponding negative controls were transfected into BMSCs using Lipofectamine 2000 (Invitrogen, CA, USA) at 37°C for 48 h based on supplier’s guidance. The transfection efficiency was examined by RT-qPCR after 48 h. All plasmids were synthesized by GenePharma (Shanghai, China).
Establishment of OA animal model
Vital River Co. Ltd (Beijing, China) provided 80 seven-week-old SD rats (male, 210–250 g). Rats were caged in a clean and open facility with controlled temperature (23 ± 3°C and 45%–65% humidity). After acclimation under a 12/12 h light/dark cycle with free access to water and food ad libitum for 1 week, a rat model with OA was established as per the previously delineated method. 19 The Institutional Animal Care and Use Committee of Wuhan Hospital of Traditional Chinese Medicine approved all the experimental protocols and the experimental procedures were performed according to the recommendations and guidelines of the National Institutes of Health. All rats were randomly divided into four groups (n = 20/group): Sham + AAV-NC, Sham + AAV-PLXNB1, OA + AAV-NC and OA + AAV-PLXNB1. After anesthetization with isoflurane, OA in rats were induced by performing medial meniscus instability surgery on the right knee. The sham-operated rats underwent similar surgical procedure without removing the ligament or meniscus. To overexpress PLXNB1 in cartilage tissues of rats, the adeno-associated virus (AAV) serotype 2 containing PLXNB1 vector or empty vector were constructed and packaged by Genechem Company (Shanghai, China). After surgery, rats in the Sham + AAV-PLXNB1 and OA + AAV-PLXNB1 received AAV-PLXNB1 (1 × 109 PFUs in a total volume of 5 μL) via intra-articular injection. Rats in the Sham + AAV-NC and OA + AAV-NC groups received equivalent volume of AAV-NC in the same way. After 8 weeks, all rats were euthanized. Then, the knee joints of rats was collected.
Histology
Right knee joints of rats were exposed to 4% paraformaldehyde for fixation for 24 h, followed by treatment with CalciClear slow solution [10% (w/v) EDTA, pH 7.4] for decalcification for 6 weeks. Subsequently, the tissue samples were paraffin-embedded, sectioned, dewaxed in xylene, rehydrated in gradient ethanol, and stained with hematoxylin and eosin (H&E) according to standard procedures. Modified Mankin’s score was calculated based on previous scoring criterion. 20
Micro-CT evaluations
Micro-CT analysis was performed to observe changes in subchondral bone structure. Briefly, Micro-CT (Bruker microCT N.V., Kontich, Belgium) was utilized to capture the images of knee joints. Meanwhile, microtomographic data from 3-D morphometry was used to determine quantitative morphometric indices including average trabecular separation [Tb. Sp, mm], average trabecular thickness [Tb. Th, mm], and bone volume fraction [BV/TV, %].
RT-qPCR
Primer sequences for RT-qPCR.

miR-326-3p: PMID: 33515352.
miR-330-5p: PMID: 34712659.
miR-362-5p: PMID: 24899157.
miR-150-5p: PMID: 32453709.
Western blotting
RIPA lysis buffer (PH0316, PHYGENE, Fuzhou, China) was utilized to isolate proteins from BMSCs and cartilage tissues. The BCA protein quantitative kit (Yanjing Biotechnology Co., Ltd, Shanghai, China) was employed for the quantification of total proteins. Denaturation was achieved by boiling the protein samples for 5 min following concentration adjustment. The supernatant liquid was added with equal volume of 2 × SDS-PAGE loading buffer. Subsequent to be mixed well and heated at 95°C, the protein was electrically transferred onto PVDF membranes. The membranes were blocked with TBST solution supplemented with 3% BSA for 1 h and then incubated with the diluted primary antibodies at 4°C with overnight. The membranes were then incubated with the diluted secondary antibody (ab102248, 1/1000) after being rinsed with TBST five times. Abcam (Cambridge, MA, USA) provided all the antibodies used in our study. Protein bands were visualized with an ECL detection kit (Bio-Rad, Hercules, CA, USA) and quantified with Image Lab software (Bio-Rad). The primary antibodies used are as follows: PLXNB1 (1:1000, ab39717, Abcam, USA), SIRT1 (1:1000, ab189494, Abcam), Aggrecan (1 μg/mL, ab3778, Abcam), β-actin (1 μg/mL, ab8226, Abcam). β-actin served as loading control.
Bioinformatic analysis
Potential miRNAs (miR-362-5p, miR-330-5p, miR-362-5p, miR-452-3p, miR-500-5p, miR-150-5p) of PLXNB1 were identified by miRDB (https://www.mirdb.org/) (screening condition: target score >80). The binding sites between miR-362-5p and PLXNB1 3′UTR were predicted by Targetscan (https://www.targetscan.org/).
Luciferase reporter assay
The wild type (Wt) or mutant type (Mut) sequence of PLXNB1 3′ UTR containing the putative binding site of miR-362-5p was subcloned into the pmirGLO vector (E1330, Promega, USA) to generate PLXNB1 3′UTR-Wt/Mut reporters. Following the manufacturer’s instructions, cell transfection was conducted using Lipofectamine 2000 (Invitrogen). MiR-362-5p mimic or NC mimic was co-transfected with pmirGLO-PLXNB1 3′UTR-Wt or pmirGLO-PLXNB1 3′UTR-Mut into BMSCs. After 48 h of transfection, the luciferase activity (Firefly/Renilla) of each group was assessed using Luciferase Reporter Assay System (Promega). 22
Statistical analysis
SPSS 18.0 software (SPSS Inc., USA) was used for data analysis. Experimental data obtained from at least triplicate trails are shown as the mean ± standard deviation (SD). Differences between groups were tested using independent sample t test and multigroup comparisons were achieved using one-way analysis of variance, followed by Tukey’s post hoc test. p value less than 0.05 was set as the threshold for statistical significance.
Results
Identification of BMSCs and high PLXNB1 expression during BMSC chondrogenesis
BMSCs were obtained from bone marrow that was isolated from rat tibia and femur through gradient centrifugation. To identify the immunophenotypic characteristics of isolated BMSCs, molecular markers (CD44, CD29, CD45 and CD34) on BMSC surfaces were analyzed by flow cytometry. According to Figure 1(a), BMSCs were positive for the mesenchymal stem cell markers CD44 and CD29 and negative for the hematopoietic cell markers CD45 and CD34. Moreover, BMSCs displayed a spindle-like shape under a light microscope (Figure 1(b)). To induce chondrogenic differentiation of BMSCs, BMSCs were cultured in chondrogenic induction media. At 0, 7, and 14 days after chondrogenic induction, Alcian blue staining was conducted to test BMSC chondrogenesis at 620 nm. As Figure 1(c), Alcian blue staining intensity in BMSCs was enhanced with the induction time increasing, furthering confirming the chondrogenic potential of BMSCs. Western blotting and RT-qPCR results manifested that the protein and mRNA levels of PLXNB1 in BMSCs were time-dependently increased after chondrogenic induction (Figure 1(d)). Identification of BMSCs and high PLXNB1 expression during BMSC chondrogenesis. (a) Flow cytometry analysis for detecting the surface markers on BMSCs. (b) Morphology of BMSCs under a light microscope. (c) After 0, 7 or 14 days of chondrogenic induction, alcian blue staining was conducted for detecting the chondrogenic potential of BMSCs. (d) Western blotting for determining aggrecan and SIRT1 protein levels in BMSCs after 0, 7 or 14 days of chondrogenic induction. *p < 0.05, **p < 0.01, ***p < 0.001.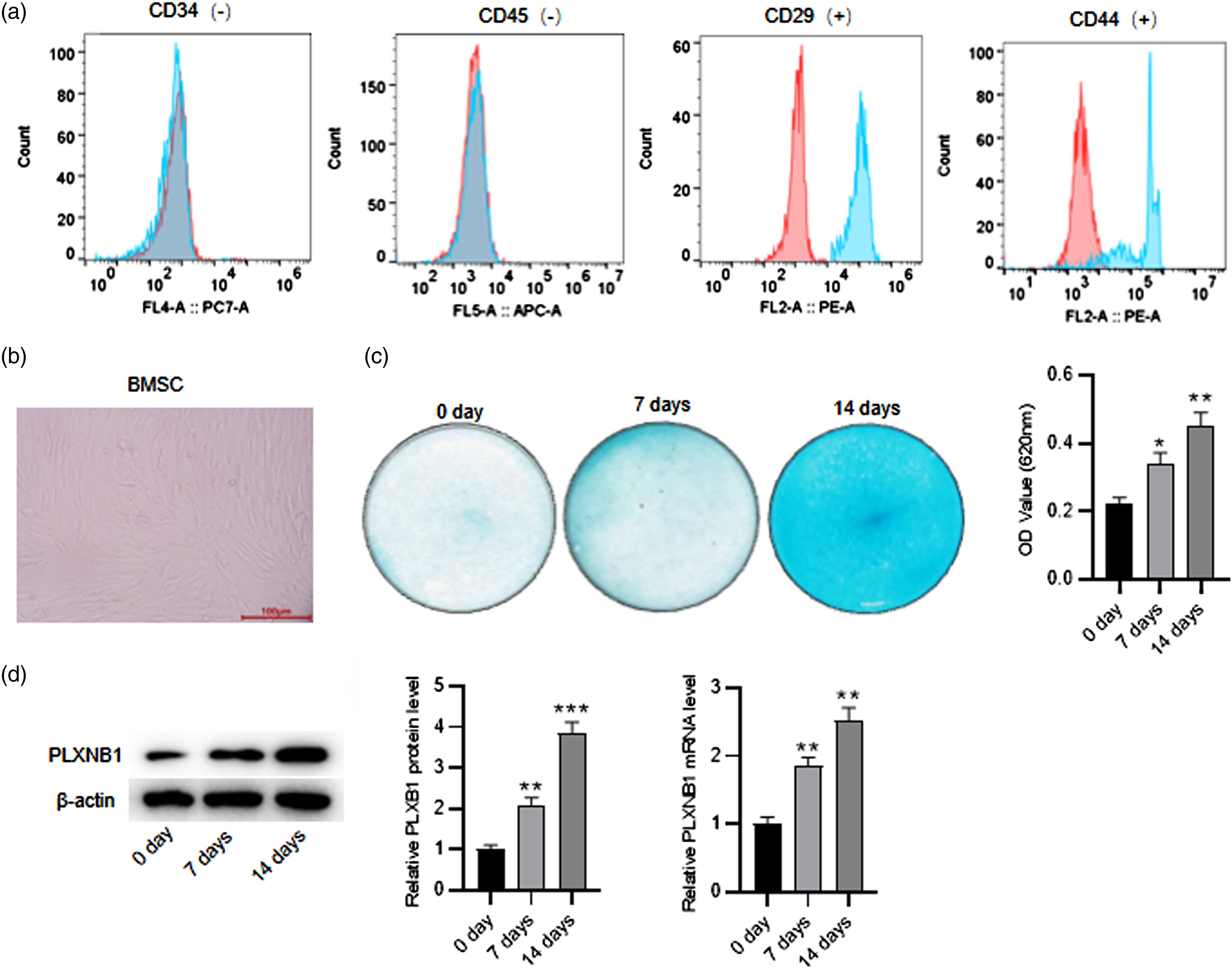
PLXNB1 overexpression enhances cartilage differentiation of BMSCs
To figure out the biological role of PLXNB1 in modulating BMSC chondrogenic differentiation during OA development, we effectively upregulated PLXNB1 at the mRNA and protein levels by transfection of pcDNA3.1/PLXNB1 into BMSCs after 14 days of chondrogenic induction (Figure 2(a) and (b)). Alcian blue staining showed that the staining intensity in induced BMSCs was enhanced by overexpressing PLXNB1 (Figure 2(c)). Accumulating evidence demonstrates that SIRT1 promotes cartilage formation.
17
As Figure 2(d) depicted, the protein levels of SIRT1 and Aggrecan (a cartilage marker) in induced BMSCs were elevated by PLXNB1 upregulation. PLXNB1 overexpression enhances cartilage differentiation of BMSCs. (a and b) Western blotting and RT-qPCR for evaluating the overexpression efficiency of pcDNA3.1/PLXNB1 in BMSCs. (c) Alcian blue staining was conducted for detecting the chondrogenic potential of induced BMSCs after the indicated transfections. (d) Western blotting for determining aggrecan and SIRT1 protein levels in induced BMSCs after the indicated transfections. *p < 0.05, **p < 0.01, ***p < 0.001.
MiR-362-5p targets PLXNB1
To figure out the underlying mechanism of PLXNB1 in regulating IL-1β-induced chondrocyte dysfunction, the upstream miRNAs of PLXNB1 were investigated. Potential miRNAs (miR-362-5p, miR-330-5p, miR-362-5p, miR-452-3p, miR-500-5p, miR-150-5p) of PLXNB1 were identified at the miRDB website. Among these candidates, only miR-362-5p expression was time-dependently reduced in BMSCs after 0, 7, or 14 days of chondrogenic induction (Figure 3(a)). As revealed by RT-qPCR, transfection of miR-362-5p into BMSCs resulted in an elevation of miR-362-5p expression (Figure 3(b)). Moreover, overexpression of miR-362-5p induced decreased PLXNB1 mRNA and protein levels in BMSCs (Figure 3(b) and (c)), indicating that miR-362-5p negatively regulated PLXNB1. Subsequently, the binding site between miR-362-5p and PLXNB1 3′UTR was predicted by Targetscan (Figure 3(d)). Luciferase reporter assay was employed for the validation of their binding. Upregulation of miR-362-5p in BMSCs markedly reduced the luciferase activity of vectors containing wild type PLXNB1 but exerted no significant influence on that of vectors containing mutant type PLXNB1 (Figure 3(e)). MiR-362-5p targets PLXNB1. (a) RT-qPCR for measuring miR-362-5p, miR-501-5p, miR-1827, and miR-500-5p levels in BMSCs after 0, 7, or 14 days of chondrogenic induction. (b and c) Western blotting and RT-qPCR for evaluating the overexpression efficiency of miR-362-5p and the impact of miR-362-5p on PLXNB1 expression in BMSCs. (d) The binding site between miR-362-5p and PLXNB1 3′UTR predicted by targetscan. (e) Luciferase reporter assay for confirming the interaction between miR-362-5p and PLXNB1 3′UTR. *p < 0.05, **p < 0.01.
PLXNB1 elevation counteracts the suppressive impact of miR-362-5p overexpression on BMSC chondrogenesis
Rescue assays were conducted to disclose whether the miR-362-5p/PLXNB1 axis mediates BMSC chondrogenesis. Alcian blue staining revealed that miR-362-5p enhancement reduced staining intensity in BMSCs after 14 days of chondrogenic induction, which was neutralized by upregulating PLXNB1 (Figure 4(a)), suggesting that PLXNB1 attenuated the repressive impact of miR-362-5p on chondrogenic potential of BMSCs. Western blotting manifested that PLXNB1 upregulation mitigated the repressive influence of miR-362-5p elevation on the protein levels of Aggrecan and SIRT1 in induced BMSCs (Figure 4(b)). PLXNB1 elevation counteracts the suppressive impact of miR-362-5p overexpression on BMSC chondrogenesis. (a) After 14 days of chondrogenic induction, alcian blue staining was conducted for detecting the chondrogenic potential of BMSC transfected with miR-NC, miR-362-5p, or miR-362-5p+PLXNB1. (b) Western blotting for determining aggrecan and SIRT1 protein levels in induced BMSCs after the indicated transfections. *p < 0.05, **p < 0.01, ***p < 0.001.
Overexpression of PLXNB1 alleviates cartilage tissue injury and improves subchondral bone structure of OA rats
In this section, we further probed into the effect of PLXNB1 overexpression in OA in vivo. As shown by H&E staining, morphologically normal cartilage tissues of knee joints were observed in sham-operated rats (Figure 5(a)). In comparison to sham-operated rats, OA rats displayed significant histopathological changes in cartilage tissues, including aggregately distributed chondrocytes, visible cartilage fissures, and rough surface of cartilage tissues. In contrast, these histologic alterations in OA were mitigated by injection with AAV-PLXNB1 (Figure 5(a)). As Figure 5(b) illustrated, OA rats had a significantly higher Modified Mankin’s score than sham-operated rats, whereas the OA + AAV-PLXNB1 group presented a remarkably lower injury score than the OA + AAV-NC group (Figure 5(b)). Moreover, reduction in Aggrecan and SIRT1 protein levels was detected in OA rats compared to sham-operated rats (Figure 5(c)). On the contrary, OA-induced decrease in Aggrecan and SIRT1 protein levels was markedly partially offset by injection of AAV-PLXNB1 vectors (Figure 5(c)). Micro-CT analysis was also used to observe changes in subchondral bone structure. As Figure 5(d) and (f) delineated, morphometric indices including BV/TV, Tb.Th, and Tb.Sp were evidently changed in OA rats in comparison with sham-operated rats, while this change was partially offset by AAV-PLXNB1 injection. Overexpression of PLXNB1 alleviates cartilage tissue injury and improves subchondral bone structure of OA rats. (a) H&E staining for observing histopathological characteristics of rat cartilage tissues of indicated groups (scale bar = 100 μm). (b) Modified Mankin’s score of indicated groups. (c) Western blotting for determining aggrecan and SIRT1 protein levels in OA rats and sham-operated rats. (d–f) Micro-CT analysis for detecting BV/TV, Tb. Th, and Tb. Sp of indicated groups. ***p < 0.001 versus the Sham + AAV-NC group;
###
p < 0.001 vs. the OA + AAV-NC group.
The expression of miR-362-5p and PLXNB1 in cartilage tissues of OA rats
As Figure 6(a) and (b) depicted, compared to sham-operated rats, OA rats exhibited high miR-362-5p expression and low PLXNB1 expression in their cartilage tissues. Injection of AAV-PLXNB1 had no impact on miR-362-5p expression and increased PLXNB1 expression in cartilage tissues of OA rats in comparison to those of sham-operated rats (Figure 6(a) and (b)). In addition, a negative correlation between miR-362-5p and PLXNB1 expression was observed in cartilage tissues of OA rats, as presented by Pearson correlation analysis (Figure 6(c)). The expression of miR-362-5p and PLXNB1 in cartilage tissues of OA rats. (a and b) RT-qPCR for evaluating the levels of miR-362-5p and PLXNB1 in rat cartilage tissues of indicated groups. (c) Pearson correlation analysis for analyzing correlation between miR-362-5p expression and PLXNB1 expression in cartilage tissues of OA rats (n = 20). **p < 0.01, ***p < 0.001.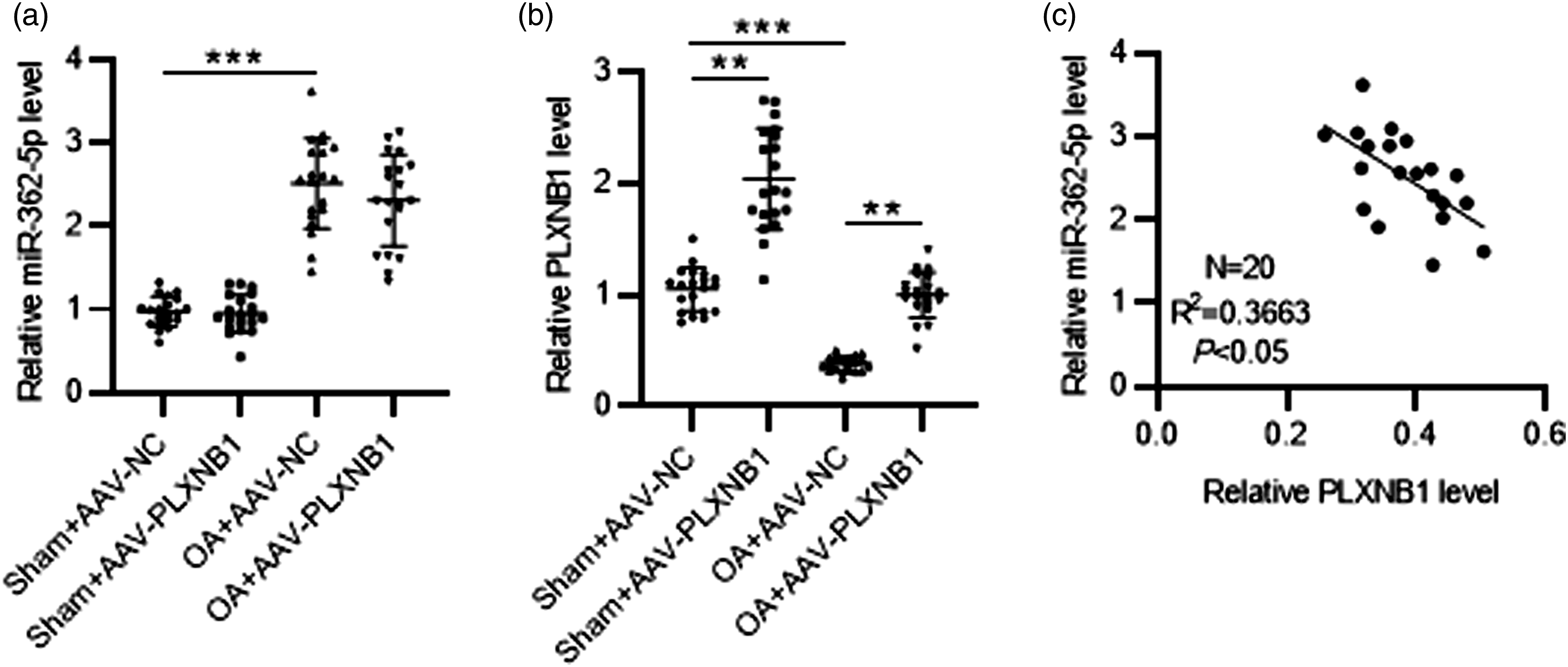
Discussion
Osteoarthritis is an inflammatory joint disorder with a high prevalence, pathologically characterized with articular cartilage destruction. 23 Moreover, chondrocyte differentiation and apoptosis and extracellular matrix degradation are the key contributors to articular cartilage degradation. Nonetheless, the present therapeutic agents and surgical approaches cannot eradicate OA. 24 Thus, it is urgent to find novel and effective therapeutic means for OA treatment. Major efforts, such as using BMSCs, have been made in hopes of achieving cartilage repair or OA treatment, which shed light on solving the medical dilemma through regenerative strategies.25,26 BMSCs show pluripotency in differentiating into many cell types. OA therapy with intra-articular transplanted BMSCs has shown potential in facilitating cartilage regeneration with chondrogenic differentiation capabilities, anti-inflammatory effects, and immunomodulatory properties.27–29 Therefore, the change and its molecular mechanism of BMSCs differentiation into chondrocytes, and chondrocyte metabolic alterations are important research targets for OA. The current report for the first time revealed that PLXNB1 targeted by miR-362-5p mitigated cartilage injury of OA rats and promoted cartilage differentiation of BMSCs.
In the initial step, we identified the immunophenotypic characteristics of BMSCs by flow cytometry analysis. As expected, BMSCs were negative for the hematopoietic cell markers CD45 and CD34 and positive for mesenchymal stem cell markers CD44 and CD29, suggesting that the isolated BMSCs possessed the characteristics of MSCs. Subsequently, BMSCs were cultured in chondrogenesis induction medium and Alcian blue staining validated the chondrogenic potential of BMSCs. SIRT1 is a nicotinamide adenine dinucleotide-dependent enzyme silent information regulator 2 type 1 deacetylase that has chondroprotective effects.30,31 It is an epigenetic regulator that plays a role in cartilage protection, and the mechanisms involved mainly include regulation of extracellular matrix expression, regulation of bone homeostasis, and anti-catabolic, anti-inflammatory, and anti-apoptotic processes, among others.30–32 Loss of SIRT1 function can induce apoptosis and aggravate osteoarthritis progression. 33 It also can elevate cartilage-specific genes including Aggrecan and COII2a to facilitate cartilage formation. 34 In the current investigation, we observed that the protein levels of SIRT1 and Aggrecan were upregulated in BMSCs with the induction time increasing, further verifying the chondrogenic capability of BMSCs. All these data illustrated that BMSCs in our study successfully differentiated into chondrocytes. An in vivo rat model of OA was also constructed by utilizing medial meniscus instability surgery. Compared to sham-operated rats, OA rats exhibited obvious cartilage tissue injury and pathological structural changes of subchondral bone including narrowing of joint space and calcification. Furthermore, alterations in morphometric indices including BV/TV, Tb. Th, and Tb. Sp and decrease in SIRT1 and Aggrecan protein levels were observed in cartilage tissues of OA rats. These results demonstrated the successful establishment of a rat model of OA.
PLXNB1 is a member of plexins which bind to ligands called semaphorins. 35 Depletion of PLXNB1 represses lipopolysaccharide-triggered chondrocyte injury, thereby attenuating OA pathogenesis. 36 A previous report also showed low PLXNB1 expression in the trabecular bones of OA patients. 10 Consistently, our study also indicated that PLXNB1 was under-expressed in cartilage tissues of OA rats. Functionally, PLXNB1 overexpression alleviated cartilage tissue injury and improved subchondral bone structure of OA rats. Upregulated PLXNB1 enhanced cartilage differentiation by increasing SIRT1 and Aggrecan protein levels in cartilage tissues. BV/TV, Tb. Th, and Tb. Sp were improved in OA rats after AAV-PLXNB1 injection. In the in vitro experiments, PLXNB1 was overexpressed in BMSCs after 7 or 14 days of chondrogenic induction, implying the potential role of PLXNB1 in regulating BMSC chondrogenesis during OA pathogenesis. Moreover, PLXNB1 elevation upregulated SIRT1 and Aggrecan protein levels and enhanced chondrogenic potential of induced BMSCs, suggesting that PLXNB1 overexpression boosted BMSC chondrogenesis. Thus, this report demonstrated that PLXNB1 could alleviate OA pathogenesis in vivo and in vitro.
Cumulative evidence has disclosed that aberrantly expressed miRNAs are closely related to the initiation or development of OA.37–39 Mechanistically, although miRNAs lack protein-coding capacity, they can modulate the expression of target mRNAs by binding to the 3′UTR of mRNAs. 40 Interestingly, the miRNA-mRNA regulatory mechanism is found in regulating chondrogenic differentiation in OA. For example, miR-30a potentiates MSC chondrogenesis by suppressing Delta-like four level. 41 MiR-132-3p boosts rat MSC chondrogenesis by binding to ADAMTS-5. 42 MiR-134 represses BMSC chondrogenic differentiation via targeting SMAD6. 43 Here, miR-362-5p were identified to target PLXNB1 and negatively regulated PLXNB1 expression in BMSCs. Studies show that miR-362-5p is associated with the pathology of gestational diabetes mellitus 44 and Marfan’s syndrome. 45 However, no evidence previously elucidated the role of miR-362-5p in OA. In our study, miR-362-5p was downregulated in BMSCs after chondrogenic induction. Moreover, OA rats also exhibited low expression of miR-362-5p and a negative expression correlation between miR-362-5p and PLXNB1 in their cartilage tissues. In vitro rescue assays, the suppressive impact of miR-362-5p overexpression on BMSC chondrogenesis was counteracted by PLXNB1 elevation. Therefore, miR-362-5p might contribute to OA by inhibiting cartilage differentiation.
However, it has some limitations. First, we only focused on a single upstream miRNA of PLXNB1 to elucidate the mechanism through which PLXNB1 modulates chondrogenic differentiation. A target gene can be targeted by numerous miRNAs and modulate multiple signaling pathways. Therefore, more molecules and signaling pathways should be investigated for elucidating the functions and mechanisms of PLXNB1. Additionally, although the in vivo model we used correlates with the pathological phenotype of OA, these experiments cannot fully replicate the combination of factors that contribute to the development of OA. Accordingly, more suitable animal OA models are required to improve the accuracy of our findings.
To conclude, this study demonstrated that PLXNB1 targeted by miR-362-5p promotes BMSC chondrogenesis and ameliorates OA pathogenesis. Our report highlights the potential application of the PLXNB1 in the targeted treatment of cartilage-related disorders. Therefore, this study may contribute to the development of new approaches that allow manipulation of differentiation outcome of BMSCs mediated by PLXNB1 for engineering of neocartilage.
Supplemental Material
Supplemental Material - MiR-362-5p inhibits cartilage repair in osteoarthritis via targeting plexin B1
Supplemental Material for MiR-362-5p inhibits cartilage repair in osteoarthritis via targeting plexin B1 by Qian Li, Ping Xu, Chi Zhang and Yang Gao in Journal of Orthopaedic Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Key Projects of Wuhan Municipal Health Commission (Approval number: WZ17A10), Wuhan Municipal Health Commission Project (Approval number: WZ19Z01), Wuhan Municipal Health Commission Project (Approval number: WZ21C23).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
