Abstract
Antidepressant use is common in women. However, characteristics associated with duration of use remain unclear. We conducted a descriptive study using data from the 1973-1978 and 1946-1951 birth cohorts of the Australian Longitudinal Study on Women’s Health linked to antidepressant dispensing records from the Pharmaceutical Benefits Scheme, July 2012 to December 2019 to examine use patterns and their associated characteristics. Early discontinuation and prolonged antidepressant use were common, a significant deviation from current guideline recommendations. There were 5553 episodes of antidepressant use in 4416 women, with a mean estimated duration of first-time use of 664 days. One in four women had only one prescription filled, while 32% continued treatment beyond two years, with 14% continuing use at five years. Age and previous antidepressant use impacted duration of use, with a shorter average duration of use and a higher proportion of discontinuation for each subsequent episode after the first prescription.
Keywords
What We Already Know
Antidepressants are among the most prescribed medications in Australia. Females are more likely to be using antidepressants than males.
There is a global trend toward prolonged duration of antidepressant use.
What This Article Adds
More than half of women remain on antidepressants beyond guideline recommendations (one year of use), with 14% continuing use at five years based on findings from the Australian Longitudinal Study on Women’s Health.
One in four women discontinued treatment after one prescription.
Age and previous use of antidepressants impact duration of use, with a shorter average duration of use and a higher proportion of discontinuation for each subsequent episode after the first prescription.
Introduction
Antidepressant use is common in Australia, especially among women. In 2020-2021, antidepressant expenditure on the Pharmaceutical Benefits Scheme (PBS) and Repatriation PBS (RPBS) was $212 million, 1 with escitalopram and sertraline the sixth and eighth most prescribed medication. 2 Internationally, Australia is one of the highest consumers of antidepressants among the Organisation for Economic Co-operation and Development countries. 3
Antidepressants are commonly prescribed for affective disorders, but efficacy may be limited in patients with mild or moderate depressive symptomatology. 4 There is also ongoing debate about whether antidepressants are superior to placebo for treating more severe depression. 5 Evidence suggests treatment for depression over a decade is not linked to any superior clinical outcomes, and may be linked with poor outcomes, including multiple relapses. 6 A study by Bockting and colleagues found that continuous antidepressant use over two years was linked to higher rates of symptom recurrence (60%-63%) compared with patients who discontinued antidepressant treatment after achieving remission. 7
The Therapeutic Guidelines (Australia) and the National Institute for Health and Care Excellence (UK) currently recommend a six- to 12-month treatment duration for patients with a single episode of uncomplicated depression, with regular review to determine the ongoing indication of antidepressant.8,9 However, antidepressant use frequently extends beyond the recommended timeframe, with average duration of use among Australian concession card holders approximately four years. 10 Unnecessary long-term antidepressant use raises concerns about potentially avoidable medication misadventure from side effects, drug interactions, and emerging evidence of all-cause and cardiovascular disease mortality.11,12 One in three long-term antidepressant users in Australian general practice are in remission with no clear reason for continued use, indicating an opportunity for treatment re-evaluation and discontinuation. 13
Given Australian women are twice as likely as Australian men to be taking antidepressants, 14 we used linked PBS data from the Australian Longitudinal Study on Women’s Health (ALSWH) to examine variation in the duration of new antidepressant prescribing by episode of use between July 2012 and December 2019. Before July 2012, dispensing was only recorded in the PBS item reports if the medication cost more than the general co-payment threshold. So, focusing our analysis on PBS antidepressant prescribing after July 2012 means ours is the first study to include both concession and nonconcession card holders. We also explored whether sociodemographic characteristics and common health comorbidities affected patterns of antidepressant use.
Methods
The ALSWH, an ongoing study of women’s health and well-being, commenced in 1996 with three cohorts (born 1973-1978; 1946-1951; 1921-1926) and a fourth cohort recruited in 2012-2013 (born 1989-1995), totaling approximately 56 000 women. 15 Women recruited in 1996 were randomly selected from the Australian Medicare database at the time; these women have been routinely surveyed every three years since recruitment. The original cohorts were oversampled from rural and remote areas to improve representation. 15 The youngest cohort (women born 1989-1995) has been surveyed annually between 2013 and 2017, with a sixth survey in 2019. The ALSWH has been determined to be a reasonable representation of Australian women through comparison with census data. 16 Survey data from ALSWH are linked to various administrative health services–related databases, including the PBS, an Australian government program subsidizing medications for Australian citizens and permanent residents. Additional information regarding ALSWH is available on the study website (http://www.alswh.org.au). 15
Study Population
Of the four ALSWH cohorts, women from the 1973-1978 and the 1946-1951 cohorts were included in this study as both demonstrated a high proportion of antidepressant dispensing and/or higher mental health service use. The 1921-1926 cohort was not included due to reported low mental health care use 17 and low proportion of antidepressant dispensing, with only 14.7% of participants having one dispensing in 2013 and 6.8% in 2019. The 1989-1995 cohort was also not included as these women were of reproductive age, 17 to 30 years between 2012 and 2019, with a significant proportion likely to have experienced pregnancy during the study period. As depression and anxiety affect 10 to 15 of every 100 pregnant women and symptoms generally improve for a significant proportion postpartum (up to a year after delivery), this would introduce sampling bias.18,19 The PBS data were linked to participant survey data, excluding women who did not consent to administrative data linkage. In this study, we investigated PBS antidepressant dispensing after July 2012, so both concession and nonconcession card holders were included. Concession card holders are entitled to PBS medication subsidies, whereas nonconcession card holders’ medication is not subsidized unless the cost exceeds a PBS co-payment threshold. Before July 2012, the PBS database only included dispensing of subsidized medications. We analyzed the PBS-linked data across 7.5 years from July 2012 to December 2019. We captured only new antidepressant users by excluding any users with antidepressant dispensing records during a three-month washout period (July-September 2012). Data from women born between 1946 and 1951 were used to compare a three- and a six-month washout period using Kaplan-Meier curves. As the pattern of use between the two washout periods was similar, it was determined that a three-month washout period would be sufficient to identify and exclude prevalent antidepressant users at the start of the medication observation period.
Antidepressant Use
We used the Anatomical Therapeutic Chemical code N06A, 20 to identify antidepressant dispensing, excluding lithium and amitriptyline, as both are mainly indicated for conditions other than depression. All other antidepressants were included in the analysis. We extracted each episode of each patient’s antidepressant use from the first to the last antidepressant dispensing and assumed that patients continued their medication for 28 days after the dispensing. In classifying a new episode of antidepressant use, we relied on the last dispensed date as recorded in the PBS data, without adding the additional 28 days from the previous date of dispensing. This approach means that even a gap of five months between dispensing a 28-day supply of medication would still be counted as a single episode. Switching was considered when defining an antidepressant use episode. Our objective was to evaluate each use episode, recognizing that many patients may trial different antidepressants during their treatment period. Thus, even if different antidepressants were dispensed within a six-month period, we still classified them as part of the same episode of use.
When a patient had two supplies of the same antidepressant of the same strength, it was assumed that the medication would be taken consecutively, and the estimated duration of use was adjusted based on the total number of supplies dispensed. For example, if a patient had three dispensings of escitalopram 20 mg on the same day, and it was their last dispensing, we assumed treatment continued for 28 × 3 = 84 days beyond the day of dispensing. However, when the supplies involved different strengths or different antidepressants, it was assumed that the medications were used concomitantly. This was relevant where patients were prescribed higher strengths (e.g., venlafaxine 225 mg daily) or combined antidepressant therapies, such as an SSRI alongside mirtazapine. Finally, when a script was received prior to the previous supply being fully used, we still counted the last dispensing date as the relevant date. There are various reasons patients might seek an early supply, such as misplacing medication or a dosage change. In these cases, it was reasonable to disregard scripts received before the prior supply was exhausted and to focus solely on the last dispensing date.
Covariates
We explored the effect of patient demographic characteristics on antidepressant treatment duration, including education (university degree versus no university degree), country of birth (Australian born versus born overseas), ability to manage on available income (easy or not too bad versus difficult or impossible), and place of residence (metropolitan versus rural/remote) based on the Modified Monash Model (MMM). 21 We also explored the association between duration of antidepressant use and common co-morbidities, specifically one or more of cardiovascular disease, diabetes, or cancer.
Statistical Analysis
We reviewed antidepressant dispensing records and recorded the start and end dates of each dispensing, accounting for any antidepressant switching. We calculated the mean estimated duration of treatment in days. Up to three episodes of antidepressant use per participant were recorded during the follow-up period. Differences of duration of use using the Kaplan-Meier estimator were compared across each of the covariates. Participants were censored if they were still using antidepressants by the end of the follow-up period, or they had died (while still on antidepressants). Otherwise, they were defined as discontinued. Four models were analyzed with the Cox regression analysis: an unadjusted model, a model adjusted for participants’ cohort, a model adjusted for co-morbidities, participants’ cohort and area of residence and the last model adjusted for co-morbidities, participants’ cohort, area of residence and all remaining covariates (mentioned above). The covariate “co-morbidities” was included due to the close relationship between other chronic health and mental health conditions. For survey variables, we used data available from the survey closest to 2019—this included survey 9 (2019) for women born between 1946 and 1951 and survey 8 (2018) for women born between 1973 and 1978. 15 Kaplan-Meier analysis was performed using only data from the first episode of use, including for all covariates, except when the episode of use was the independent variable. Log-rank tests were performed to analyze the differences observed in the Kaplan-Meier analysis. A P-value of .05 was used as the cutoff for the significance level. All statistical analyses were performed using Stata version 17.0.
Ethical Approval
The ALSWH has ongoing ethical approval from both the University of Queensland and the University of Newcastle’s Human Research Ethics Committees (EC00144 [the University of Newcastle HREC] / EC00456/7 [Ratified by the University of Queensland HREC]). The PBS data linkage was approved by the Australian Institute of Health and Welfare. This specific project was approved by the ALSWH Data Access Committee in 2019, with an extension granted in 2022.
Results
We included 4416 eligible participants (2643 from women born 1973-1978 and 1773 from women born 1946-1951), with 5553 episodes of antidepressant use (3535 from women born 1973-1978 and 2018 from women born 1946-1951). A participant eligibility flowchart is available in supplemental material. There were 1942 episodes of antidepressant use censored at the end of follow-up, that is, still on the treatment.
Collectively, the average estimated duration for the first episode of antidepressant use was 665 days (1.83 years) or 886 days (2.43 years) after excluding those who only had a single dispensing. Women born between 1973 and 1978 tended to have a shorter average duration of use (624 days) than women born between 1946 and 1951 (726 days), approximately 3.5 months longer for the older women. The estimated average duration of use was shorter for second and third episodes of use. Half of the women remained on an antidepressant for longer than one year, one in three (32%) continued for two years, one in four (24%) for three years, one in five (19%) for four years, and one in seven (14%) for at least five years. Twenty-nine percent of women born between 1946 and 1951 had a single antidepressant prescription dispensed compared with 24% of the women born between 1973 and 1978 (25% overall). Forty-four percent of women born between 1973 and 1978 remained on treatment after one year compared with 47% of women born between 1946 and 1951 (Table 1).
Episodes and Duration of Antidepressant Use.

Figure 1a shows the estimated duration of antidepressant use by episode of use using the Kaplan-Meier graph. The proportion of patients filling only one prescription was higher with each subsequent episode of antidepressant use (log-rank test, P < .001). Figure 1b shows the estimated duration of antidepressant use by cohort (1b), and each covariate using Kaplan-Meier graphs (1c-g).
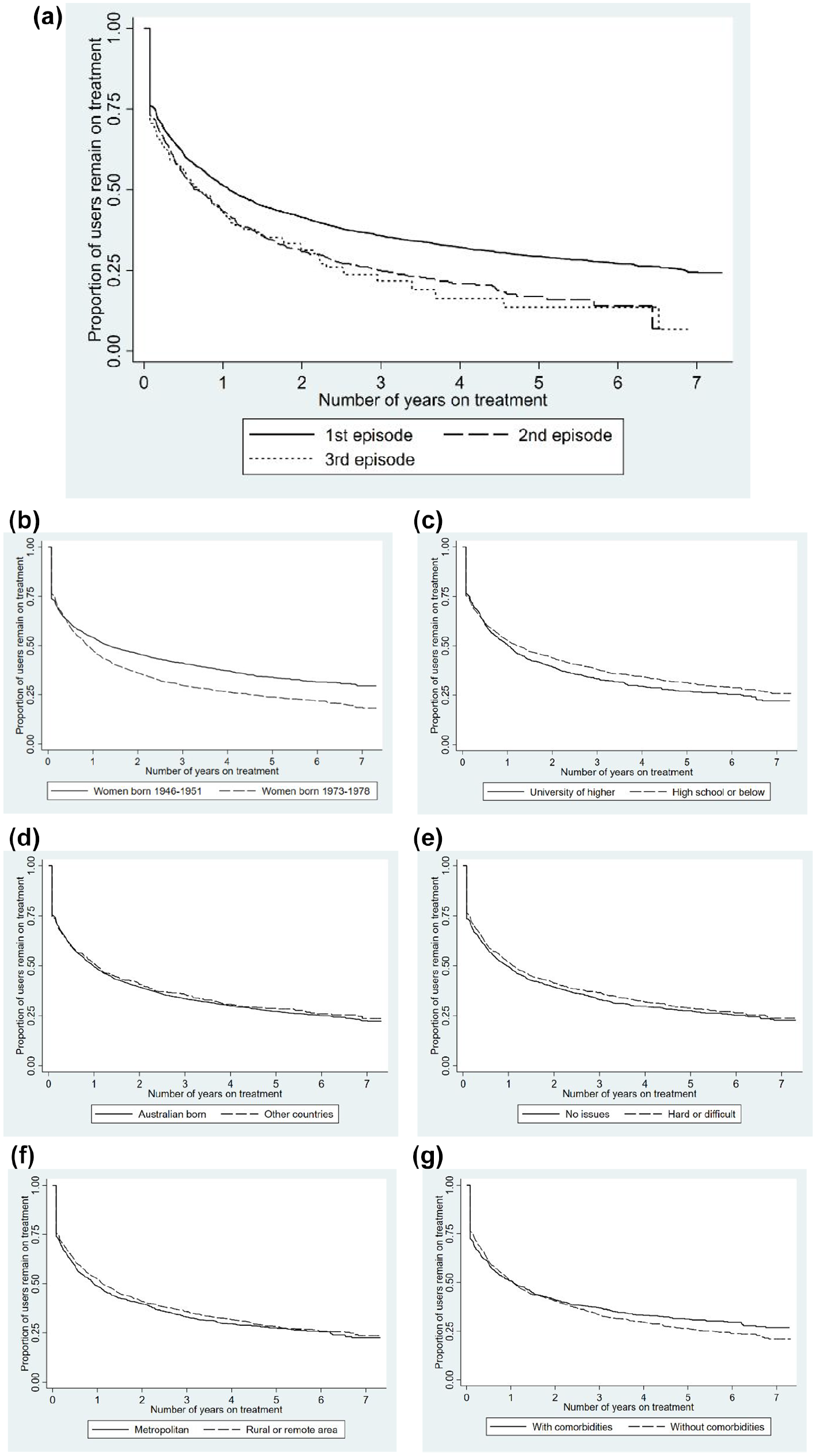
(a) Episode of antidepressant use (P < .001). (b) Patient cohort (age)a (P < .001). (c) Educationa (P = .06). (d) Country of birtha (P = .59). (e) Ability to manage on available incomea (P = .23). (f) Area of residencea (P = .22). (g) Physical health co-morbiditiesa,b (P = .28).
Cox regression analyses, with the univariate model and three adjusted models, are described in Table 2. The univariate model showed a significant association between duration of use, cohort, and episode of use. Adjusted model 1 showed women born 1973-1978 were associated with a hazard ratio (HR) for discontinuing antidepressants of 1.21 (95% confidence interval [CI] = 1.13-1.29, P < .01) compared with women born 1946-1951 (reference group). However, this was not supported when other factors were considered (models 2 and 3). Adjusted model 1 showed the HR associated with a second episode was 1.24 (95% CI = 1.13-1.36, P < .01) for discontinuing antidepressants and 1.25 (95% CI = 1.05-1.47, P < .01) in women with a third episode of antidepressant use. This remained significant after adjusting for the area of residence in the adjusted model 2. However, this association only remained significant in the adjusted model 3 for patients with a second episode of use (HR = 1.21, 95% CI = 1.04-1.40, P = .02) but not with a third episode of use (HR = 1.34, 95% CI = 0.98-1.81, P = .06). No other covariates were statistically significant in the adjusted models.
Cox Regression Analysis for the Effect of Cohort, Episode of Care and Patient Characteristics on the Probability of Discontinuing Antidepressant Treatment.

Abbreviations: HR, hazard ratio—a HR greater than 1 suggests a longer duration of antidepressant use compared with the reference group.
Area of residence was adjusted due to the ALSWH study design as rural residents were sampled at twice the rate of metropolitan residents.
Discussion
Our longitudinal study linking PBS antidepressant dispensing records with ALSWH data indicates that many Australian women are taking antidepressants beyond the guideline-recommended duration of six to 12 months.8,9 Conversely, one quarter of women filled only one prescription. This suggests both overuse and underuse of antidepressants.
Half of new users remained on their antidepressant for more than one year, one in three continued treatment beyond two years, and one in seven beyond five years. The mean duration of antidepressant use from our study (2.2 years, excluding participants with a single dispensing) was shorter than findings in another Australian study by Kjosavik and colleagues (4.1 years). 10 However, their mean treatment duration differed by age-band: two years in patients below 24 years, and up to four years thereafter. Study differences could be due to their use of a 10% PBS sample of both genders, a longer baseline but shorter (four-year) analysis period, and the inclusion of only concession card holders. While there may be a clinical need for prolonged antidepressant use in patients with complicated mental health conditions, widespread long-term use is concerning. There are adverse health outcomes (such as early mortality) associated with prolonged antidepressant use. 12 Therefore, regular reviews are warranted to assess the need for continuation, with antidepressants deprescribed if they are no longer indicated and cessation is appropriate. In addition, early discontinuation of antidepressants by 25% of women represents a missed opportunity for quality use of medicines. This could be due to poor adherence, poor drug tolerability, inappropriate initiating dose, or patients simply feeling better. 22 It suggests clinical support and close monitoring of patients’ experience is warranted at treatment initiation.
Women from the 1946-1951 cohort had a longer treatment duration than those in the 1973-1978 cohort. This is consistent with Burton and colleagues’ study that used a large, representative data set of new antidepressant users in the United Kingdom, where older participants (>65 years) were less likely than younger participants (36-65 years & <35 years) to discontinue antidepressants over a 12-month study period. 23 Antidepressant use is common in older people, and infrequent deprescribing of antidepressants may be one of the contributors to clinically inappropriate long-term antidepressant use. 24
For each subsequent episode of use, the duration of use was reduced and a higher proportion of discontinuation after the first prescription. While this differs from the findings of Burton et al, 23 our results align with studies reporting a reduced duration of persistence in patients recommencing psychotropic medications, including antidepressants.25,26 As reports of antidepressant persistence have largely focused on the first episode of use, with limited investigation of medication use patterns in restarters, further investigation is warranted to determine the potential predictors of successive treatment interruptions. Nonetheless, this represents opportunities for health professionals to target this population to discuss the role of lifestyle and access to nonpharmacological strategies while addressing patient concerns that may act as a barrier to adherence and persistence.
In the present study, one in four women had only one prescription filled. Early discontinuation may represent a lack of therapeutic benefit and/or unpleasant side-effects.23,25,27 Yet, this medication use behavior is frequently observed in practice and remains unaddressed. Poor communication between health professionals and patients could lead to patients having unresolved concerns and hamper their acceptance of treatment, resulting in early discontinuation. 28 Given that patients commonly have questions regarding their antidepressants due to information gaps and safety concerns, 29 health professionals, especially those in primary health settings, are in a position to provide clear guidance and adopt a shared decision-making model at antidepressant commencement. By proactively addressing any information gaps and concerns, early discontinuation may be avoided in those who may benefit from antidepressant treatment. Future research should focus on overcoming the barriers for early antidepressant discontinuation.
Despite previous literature suggesting that sociodemographic variables can affect antidepressant utilization, 14 the sociodemographic variables examined in our study did not show any association with duration of antidepressant use, except for age. This is consistent with findings by Burton et al 23 who determined that greater socioeconomic deprivation had little influence on continuation of newly initiated antidepressant treatment (up to 12 months of observation). This suggests that a patient’s sociodemographic background may have a more pronounced impact on the decision to commence antidepressants and less effect on duration of use.
In our PBS data set spanning 7.5 years, physical health co-morbidities, specifically cardiovascular disease, diabetes and/or cancer, have no evidence to support an association with duration of antidepressant use. While this is also consistent with findings reported by Burton and colleagues, it was unexpected. 23 Mental health often has a bi-directional relationship with chronic physical health illnesses, for example, patients with multimorbidity are twice as likely to have depression compared with those without multimorbidity. 30 We chose this “co-morbidity” grouping to compare our findings with that of Burton’s large primary care UK database cohort 12-month study of all patients with a newly initiated course of eligible antidepressant treatment. 23 It remains unclear whether this lack of association in both studies was due to these co-morbidities being grouped together, potentially masking the effect of each co-morbidity. Further analysis with co-morbidities (as separate variates) or functional health scores, such as the SF-36 scale, may clarify this correlation.
Limitations
The epidemiological nature of our data meant that we could not identify whether prolonged antidepressant use was clinically justified. Our estimates of duration of use are likely underestimates, women could continue their antidepressant beyond the seven-year study period. Likewise, PBS data are available for concession card holders only prior to 2012, so ultra-long-term antidepressant use, such as treatment for more than a decade or even longer, could not be investigated. Moreover, as we had a wash-out period to only include new antidepressant users, those who had been on treatment for considerably longer were not captured in this study. As we were unable to collect data before July 2012, this may not be true first-time users. However, the longitudinal nature of our data and its strong resemblance to international studies citing long-term antidepressant use23 -25 suggest the robustness and validity of our findings. As this study was descriptive, further analyses could consider examination with competing risk analyses to account for those who died. The noncontinuous age range of the two cohorts investigated in this study raises the possibility that there may be differences in duration of antidepressant use in women in other age groups. Last, we utilized dispensing records for this study, which may not represent actual use of antidepressants.
Conclusion
Some Australian women remain on antidepressants well beyond guideline recommendations, while others prematurely discontinue treatment. This mismatch between guideline recommendations and actual duration of use is of public health concern, due to the increased health care burden of poor antidepressant treatment outcomes, risk of medication harm, and the associated financial impact. In our results, age and previous episodes of antidepressant use were found to be associated with average duration of use, whereas level of education, physical health co-morbidities, country of birth, place of residence, and ability to manage on available income were not. Duration of use was shorter for each subsequent episode of use. Further research is needed to determine not only the health impacts of long-term antidepressant use but also safe and effective tapering strategies to support clinicians and their patients to cease antidepressants. Consideration should also be given to developing patient-centered antidepressant treatment goals and management plans to ensure patients have clear expectations of their treatment initiation and duration.
Supplemental Material
sj-docx-1-aph-10.1177_10105395251321316 – Supplemental material for Patterns and Duration of Antidepressant Use in Australian Women: Findings From the Australian Longitudinal Study on Women’s Health
Supplemental material, sj-docx-1-aph-10.1177_10105395251321316 for Patterns and Duration of Antidepressant Use in Australian Women: Findings From the Australian Longitudinal Study on Women’s Health by Edgar Poon, Maria Donald, Mieke van Driel, David Pache, Samantha Hollingworth, Xenia Dolja-Gore and Treasure McGuire in Asia Pacific Journal of Public Health
Footnotes
Acknowledgements
The research on which this paper is based was conducted as part of the Australian Longitudinal Study on Women’s Health by the University of Queensland and the University of Newcastle. The authors are grateful to the Australian Government Department of Health for funding and to the women who provided the survey data. They acknowledge the Australian Government Department of Health for providing PBS data, and Aged Care data, and the Australian Institute of Health and Welfare (AIHW) as the integrating authority. They would also like to acknowledge Ms Alison Griffin from the QIMR Berghofer Medical Research Institute for her support in graphical output.
Author Contributions
E.P., M.D., T.M., D.P., S.H., and M.v.D. contributed to the study design. E.P. conducted the data collection. E.P., M.D., T.M., D.P., S.H., M.v.D., and X.L.G. discussed the results. X.L.G. and E.P. performed the statistical analysis. E.D. created the first draft of the manuscript. All authors reviewed and approved the final manuscript.
Data Availability
The data that support the findings are available from the Australian Longitudinal Study of Women’s Health and Pharmaceutical Benefits Scheme. Restrictions apply to the availability of these data, which were used under license and ethics approval for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The Australian Longitudinal Study of Women’s Health (ALSWH) has ongoing human research ethics approval from both The University of Queensland and The University of Newcastle’s Human Research Ethics Committees (EC00144 [The University of Newcastle HREC] / EC00456/7 [Ratified by The University of Queensland HREC]). PBS data linkage was approved by the Australian Institute of Health and Welfare. This specific project was approved by the ALSWH Data Access Committee in 2019, with an extension granted in 2022.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
