Abstract
Salivary gland carcinomas are a group of heterogeneous tumors of different histological subtypes, presenting relatively low incidence but the entire variable of types. Although novel treatment options for salivary gland carcinomas patients’ outcomes have improved, the treatment of this type of cancer is still not standardized. In addition, a significant number of patients, with a lack of optimal treatment strategies, have reduced survival. In the last two decades, a plethora of evidence pointed to the importance of autophagy, an essential catabolic process of cytoplasmatic component digestion, in cancer. In vitro and in vivo studies highlight the importance of autophagy in salivary gland carcinomas development as a tumor suppressor or promoter mechanism. Despite the potential of autophagy in salivary gland carcinomas development, no therapies are currently available that specifically focus on autophagy modulation in salivary gland carcinomas. In this review, we summarize current knowledge and clinical trials in regard to the interplay between autophagy and the development of salivary gland carcinomas. Autophagy manipulation may be a putative therapeutic strategy for salivary gland carcinomas patients.
Introduction
A salivary gland cancer (SGC) is a rare malignancy at the head and neck region with diverse in pathology and origin. 1 The number of carcinoma types has increased from 5 in the 1972 World Health Organization (WHO) classification edition to 22 in the 2017 one, and, at the same period, the number of benign tumors has increased from 4 to 11.2,3 SGCs are divided into those arising from the major and those arising from the minor salivary glands. The most common site of SGCs arising from the major salivary glands is parotid glands followed by sublingual and submandibular ones. Less than 23% of all salivary gland tumors arise from minor salivary glands.4–6 The majority of SGCs arising from the minor SGCs are located in the oral cavity with a hard palate to be the most frequent subside.4,7–9 In addition, the vast majority of major salivary gland tumors (75% and 85% are in the parotid gland, and 70%–80% of these are benign) are benign; more than 80% of tumors arising from the minor salivary glands are malignant.10,11 Based on the Epidemiology, End Results (SEER) program and Surveillance of the National Institutes of Health, SGCs incidence in the United States during the last 30 years trended upward from 1.1 to 1.3 cases per 100,000 individuals, with a male-to-female ratio of ∼1.6:1. The incidence rate accelerated after age 50 years to more than 7 per 100,000 aged 70 years and older.12,13
The most common SGC type is mucoepidermoid carcinoma (MEC) accounting for approximately 35% of all malignancies of the major and minor salivary glands in general.
2
It is characterized by components of mucin-producing, intermediate-type, and squamoid cells with cystic and solid growth patterns.
14
Other SGC types are adenoid cystic carcinoma (AdCC), polymorphous adenocarcinoma (PAC), and salivary duct carcinoma (SDC), and acinic cell carcinoma (AciCC).
14
AdCC (∼10% of all salivary malignancies) is a tumor from the parotid, submandibular, and mostly minor salivary glands.
15
Mutations in different genes such as
Types, origin, genes alterations, and relative percentage of salivary gland malignancies.

SGC: salivary gland cancer: MEC: mucoepidermoid carcinoma; AdCC: adenoid cystic carcinoma; PAC: polymorphous adenocarcinoma; SDC: salivary duct carcinoma; AciCC: acinic cell carcinoma.
The autophagosome formation in macroautophagy mechanism
Autophagy is a well-characterized catabolic pathway for various cellular functions. 23 It is a lysosome-dependent process which associates with cell homeostasis through degradation and adaptive responses to a plethora of stressful conditions. 24 Three forms of autophagy are commonly described: macroautophagy, microautophagy, and chaperone-mediated autophagy. 25 The key step of macroautophagy is the formation of double-membrane structures called autophagosomes which are fused with lysosomes to create the autophagolysosome. Autophagolysosome is a fundamental structure responsible for digesting proteins and dysfunctional cellular organelles. 25 It is known that the role of autophagy is extended from cellular homeostasis to tumor development.23,24,26 In the last decade, several studies have highlighted the controversial role of autophagy in tumorigenesis. Tumor cells have been found to require high basal levels of autophagy for critical cellular functions like proliferation. 27 It appears that the basic levels of autophagy are associated with tumor types, stage of malignancy, growth factors, oncogenes, and tumor suppressors. 25 In already established tumors, the role of autophagy is to keep the energy and metabolic balance within the hypoxic tumor regions. 28 Furthermore, the increasing levels of autophagy in the tumor have also been associated with immunosurveillance and immunosuppression of tumor microenvironment. 29
Various morphological changes characterize the macroautophagy process. In the first step of macroautophagy, which is called initiation or nucleation, the phagophore, a double-membrane structure is formed through the activation of the class III PI3K–Beclin-1 complex. 25 The elongation is the next step in the macroautophagy process. This step is characterized by the arising of phagophore from different double-membrane organelles such as endoplasmic reticulum (ER), Golgi, and mitochondria. The phagophore starts to enclose the cytosolic cargos, leading to the formation of the autophagosome. The formation of phagophore is highlighted by different autophagy-related genes (ATGs), p62/SQSTM1 (an adaptor protein responsible for the docking of specific cargoes) and the lipid-modification of protein-light chain 3 (LC3), LC3-I to LC3-II. The maturation step and the following fuse step include the autophagosome formation, which eventually fuses with lysosomes to form autolysosomes. Finally, during the degradation step, lysosomal/vacuolar hydrolases digest autolysosomal products and release them into the cytosol. 30 The distinct steps of macroautophagy are presented in Figure 1.

Autophagic modulators as therapeutic options in salivary gland cancer. A plethora of different interventions are available to modulate autophagy at different steps of autophagy process. Five distinct steps have already been characterized: nucleation, elongation, fusion, or degradation step. The majority of autophagy modulators target the first step (initiation) and/or the fusion between lysosome and autophagosome. Several clinical trials already use agents that modulate (inhibit or activate) autophagy. For additional details in clinical trials, please refer to Table 2.
ATGs have a key role in the regulation of autophagy. Thirty-six different ATGs have already been identified. 31 Autophagosome is formed through the interaction of unc-51-like kinase (ULK) complex (ATG13, ATG101, ULK1/2, and family-interacting protein) and class III PI3K. 32 The initiation of autophagy is regulated by vacuolar protein sorting–associated protein 34 (Vps34) (a class III PI3K) that is bound to Beclin-1, and other ATG proteins. 33 It is well known that mammalian target of rapamycin complex (mTOR) inhibits the formation of ULK complex and consequently the autophagy initiation.34,35
LC3 is a crucial component of the second ubiquitin-like pathway. The microtubule-associated protein 1-light chain 3 (LC3-I) conjugates to the lipid phosphatidylethanolamine (PE) by ATG7 and ATG3 to form the membrane-bound LC3-II. 36 The membrane-bound form of LC3, LC3-II, is recruited to both sides of the autophagosomal membrane. During maturation, lysosomal-associated membrane protein 2 and Ras-related protein Rab-7a promote autophagosome fusion with endocytic and lysosomal compartments to form an autolysosome. 37 LC3-II on the cytoplasmic domain of the autolysosome can be dilapidated by ATG4 and recycled, whereas proteins located on the internal surface of the autophagosome and autophagic cargo are processed for degradation by lysosomal enzymes.38,39 Despite the extensive research regarding the controversial role of autophagy in various types of cancer, less knowledge is available on the impact of autophagy in SGCs.
Current therapy for SGCs
During the last years, different therapeutic approaches included radiotherapy (RT), surgery and chemotherapy were used for SGCs depended on the clinical patient status. 40 For patients with high-grade disease and/or locally advanced SGC of major glands, adjuvant RT proved more beneficial compared to those who undergo surgery alone. 41 As it was mentioned above, the main role of autophagy is to maintain cellular homeostasis and energy balance of the cells and may be activated due to stressful conditions as a response to RT and chemotherapy.
RT, as any other treatment against cancer, could cause adverse side effects such as damage and dysfunction of normal salivary gland tissue. Several autophagy modulators, such as the derivative of rapamycin and temsirolimus (CCI-779), 42 were examined in order to ameliorate the side effects caused by RT and improve the physiological function of salivary glands.
Chemotherapy
Systemic therapy has been proven beneficial for recurrent and/or metastatic SGCs (which is characterized by rather frequent local recurrence and distant metastasis) while surgery or RT to treat localized disease. The rarity of the disease is the most important barrier to design clinical trials in order to investigate the efficacy of systemic therapy. Chemotherapy remains the most important approach for systemic therapy of symptomatic locally recurrent and/or metastatic disease.43,44 The first-line therapy for patients with advanced disease is cisplatin and 5-fluorouracil (FU) or capecitabine (CAP) (cisplatin, doxorubicin, and cyclophosphamide) with the most effective combinatorial scheme still remains 5-FU, doxorubicin, and cisplatin.45–48
Autophagy and RT
As it was mentioned previously, autophagy acts as a key survival mechanism during several stressful conditions. It is well known that salivary glands are exposure to non-diseased salivary tissues to therapeutic radiation during the treatment of head and neck cancer (HNC). Radiation increases the acute and chronic loss of normal salivary function and consequently leads to a severe diminishment in the quality of life for these patients.49–51 Several studies using knockdown models of autophagy in different cell types have identified that loss of normal autophagic function, consequently leads to increased DNA damage, dysregulation of the cell cycle, and finally cell death, following treatment with chemotherapeutic agents and radiation. 52
Target therapy
Due to the poor results with chemotherapy, novel therapeutic approaches are required for SGCs. Epidermal growth factor receptor family (EGFR and HER2), KIT, and androgen receptors (ARs) are the most promising target for individualized therapies in SGCs and their expression is linked with the histogenetic origin of the tumor cells. Different agents such as cetuximab, imatinib, trastuzumab, and gefitinib have already been used in order to explore new treatment for SGCs, but on account of the rare incidence of this type of cancer, the number of cases available on targeted therapy for analysis is relatively small. 43
Consequently, therapeutic options for SGCs are limited and personalized treatment is virtually non-existent. In the last decade, it has been identified that the catabolic process of autophagy has a key role in the onset and progression of different types of cancers. 53 The role of autophagy in tumorigenesis is controversial, although autophagy promotes tumor progression in later stages; it acts also as a tumor-suppressive mechanism in early stages of tumorigenesis.54,55 Therefore, it is crucial to determine the exact role of autophagy at different stages of SGC development for future autophagy-based therapeutic approaches.
New approaches in SGC therapy
Because there is a lack of effective standard treatments and due to the irreversible side effect of RT and surgical disfigurement for patients with SGC, the antitumor activity of immunotherapy is a notable finding and justifies further research of immune checkpoint inhibitors in patients with SGC.
Sridharan et al. 56 identified that no patients with adenoid cystic carcinoma (ACC) having a significant expression of PD-L1, but PD-L2 is expressed in over 60% of the primary SGCs and 73% of the metastatic disease. In addition, it has also reported that chemoradiation increases the number of CD8+ effector T cells (which express TIM-3, LAG-3, or PD-1) in the peripheral blood. Pembrolizumab (which targets the programmed cell death protein 1 or PD-1 receptor of lymphocytes) was tested for SGCs in KEYNOTE-028 study, but it showed no responses in ACC subtype. 57 The safety and the antitumor activity of pembrolizumab in SGC patients with high expression of PD-L1 were tested in the phase Ib KEYNOTE-028. In this study, pembrolizumab appears to have modest activity with an overall response rate (ORR) of 12%, a median progression-free survival (PFS) of 4 months, and a median overall survival (OS) of 13 months. 57 Moreover, an ongoing phase II clinical trial is comparing pembrolizumab with or without radiation for recurrent or metastatic ACC (NCT03087019).
More recently, several other pathways have been provided on the molecular characterization of each subtype of SGCs. Vascular endothelial growth factor receptor (VEGFR) and fibroblast growth factor receptor-1 (FGFR1) appear to be overexpressed in ACC and may be a potential therapeutic approach. Several agents that target these pathways—such as sunitinib, sorafenib, nintedanib, axitinib, regorafenib, and/or pazopanib—have already been studied in ACC and appeared to show some clinical benefits, with ORR in the range of 6%–10%, and prolonged stable disease (SD) (>6 months) in up to 85% of unselected patients. 58 More recently, the new generation multi-kinase inhibitor lenvatinib that is active against VEGFR2, FGFR1-3, KIT, RET, and platelet-derived growth factor receptor (PDGFR) α and β demonstrated an ORR of 12%–16% in two phase II studies, with 25%–27% of patients with ACC achieving at least a 20% reduction in size of targeted tumor lesions.59,60 In addition, tumors with overexpression of C-KIT and HER2 with several inhibitors including imatinib, trastuzumab, dasatinib, and lapatinib failed to show activity in ACC, likely due to the lack of an underlying gene amplification and/or driver mutation. 58
The role of autophagy in normal salivary gland function
Flodby et al.
61
used
The
Ligation of the major excretory duct of the submandibular gland (SMG) prevents the outflow of saliva, and extended obstruction leads to acinar cell and glandular atrophy. In SG, both mTOR pathways and autophagy are activated and following sustained duct ligation, and thus, both mechanisms of action are associated in SG atrophy and survival, albeit possibly targeting a different population of cells.
67
Moreover, daily treatment with rapamycin, a well-known mTOR inhibitor and consequently autophagy activator, delayed ligation-induced SG atrophy in mice models.
68
Ligation-induced acinar cell apoptosis was delayed in autophagy-impaired acinar cells in SMGs. Furthermore, in vitro analyses highlighted that treatment with autophagy inhibitors such as bafilomycin A1 and/or chloroquine or knockout of the
The role of autophagy in SGC development
The dysregulation of autophagy has been associated with a variety of human diseases, including vascular, pulmonary, intestinal, infectious, metabolic, and neurodegenerative diseases, as well as in aging and a variety of cancer types.25,70,71 Autophagy has a significant but controversial role in tumor development. 72 At the early stage, tumorigenesis has an initial protective role against cell differentiation, apoptosis, and proliferation of cancer cells. In already established tumors and in later stages, it demonstrates such role in aid of tumor cells. 73
The impact of autophagy in SGC tumorigenesis
Several studies in the field of SGC support the hypothesis that autophagy acts as a protective mechanism under different conditions. It is well known that several solid tumors are characterized by increasing levels of hypoxia. 74 CoCl2, a hypoxia inducer molecule, activates autophagy in in vitro study with ACC-M cell line through HIF1α/BNIP3 signaling pathway, and it may be a potential target for the future treatment of AdCC. Inhibition of autophagy arrested tumor invasion induced by hypoxia in this cell line. 75 In a recent study, autophagy has a cytoprotective role in AdCC under treatment with obatoclax (also known as GX15-070, an inhibitor of the Bcl-2 family of proteins). Obatoclax increased the expression autophagy marker of Beclin-1 and transiently increased ATG5 expression at the early stage of the disease. Furthermore, inhibition of autophagy triggered apoptotic cell death in ACC-M cells. 76 It is well known that cisplatin (DDP)-based adjuvant chemotherapy is widely used in the treatment of AdCC.77,78 Jiang et al. 79 and Ma et al. 80 supported that cisplatin inhibited mTOR cascade and it activated autophagy as a cytoprotective mechanism of cancer cells against chemotherapy. The combination treatment for cisplatin (DDP)-based adjuvant chemotherapy (which induced autophagy) and 3-methyladenine (3-MA) as it was described in this study was assessed as a useful approach to anticancer treatment by both in vitro and in vivo experiments in salivary adenoid cystic carcinoma (SACC). 79 In addition, ACC-M cell line appears to have an increased sensitivity to cis-diamminedichloroplatinum (CDDP), suggesting that inhibition of CDDP-depended autophagy enhanced cytotoxicity in human ACC. 80 Thus, inhibition of cisplatin-based autophagy induction might be a promising chemotherapeutic strategy for adjuvant chemotherapy in AdCC. 80
A recent study supported that AdCC did not depend on neo-angiogenesis and was probably associated with autophagy. Immunohistochemistry for this type of SGC revealed a strong expression of autophagy marker Beclin-1 in tumor cells. 81 Both Beclin-1 and LC3B play an essential role in AdCC pathogenesis as well as in the development of chemotherapy resistance. 82 Furthermore, expression of the autophagy-related protein Beclin-1 proved a favorable independent prognostic factor for AdCC. Such results identify autophagy as a regulator of pathogenesis in the salivary gland AdCC. 83
A recent study highlighted the impact of autophagy in PAC tumor growth. Both autophagy markers Beclin-1 and LC3 were expressed in this rare type of tumor, suggesting that PAC autophagy is a key mechanism to support tumor growth. 84
The role of autophagy as a tumor suppressor mechanism in SGCs
In contrast with the general concept of autophagy as a resistance and cytoprotective mechanism in SGCs, a plethora of studies in SGCs models identified autophagy also as a pro-apoptotic mechanism. In a recent study, LC Jiang et al. 85 suggested the autophagy marker Beclin-1 as a promising prognostic factor for clinical outcomes in head and neck AdCC patients. In more detail, patients with higher expression levels of Beclin-1 appear to have better overall survival with better clinical characteristics. Moreover, in the same study, Beclin-1 expression showed a statistically significant correlation with histological growth pattern and grade of tumor. 85 Autophagy can be induced pharmacologically in salivary glands AdCC through the use of mTOR inhibitors such as temsirolimus as it was suggested in recent study. 86 The specific mTOR inhibitor temsirolimus triggered autophagy in AdCC xenograft mice models and led to suppression of tumor development in this kind of SGC model. The results of this study highlighted the suppressive mechanism of autophagy against AdCC development. 86
Histone deacetylases (HDACs) are essential enzymes that regulate epigenetic modification and catalyze the removal of acetyl from lysine residues in histones and non-histone proteins.87,88 It appears that inhibition of HDAC7 activates autophagy as it was identified through increasing levels of autophagic markers, the number of acidic vesicular organelles, LC3B II levels, and decrease in p62 levels. This led to growth inhibition through G2/M phase cell cycle arrest and trigger apoptosis through autophagy in MEC cells. 88 In addition, silence of the same gene reduced c-Myc expression and increased p27 expression in MEC cells. Therefore, HDAC7-depended autophagy might be a putative chemotherapeutic target against MEC. 89 In another study with salivary gland (A-253) cancer cells and mouse salivary gland cancer cells (SALTO), (+)-GOS (a naturally occurring polyphenolic) decreased cell proliferation through autophagy induction and apoptotic cell death. 90
Noxa (a BH3-only group of the Bcl-2 family) is characterized as a p53-inducible gene that is associated with DNA damage, autophagy, and apoptosis.91,92 L Liang et al. 93 proved that Noxa appeared to be downregulated in human AdCC tissues and it was negatively correlated with tumor histological grade, the risk for lung metastases, more aggressive phenotype and significantly poorer overall patients’ survival. Furthermore, ectopic Noxa expression in AdCC cell line triggered autophagy as it was identified through the accumulation of the LC3-II, Beclin-1, Atg5, and Atg12 and decreased cell proliferation. Collectively, these results identify Noxa as a suppression mechanism of AdCC progression through the induction of autophagy. 93
Vigorous research in autophagic cell death led to the development of several agents that trigger apoptotic cell death through autophagy induction in SGCs models. Among other drugs, isoliquiritigenin (ISL), a flavonoid derived from licorice, appeared to increase autophagy and apoptotic cell death through inhibition of mTOR in AdCC. 94 This may be representing an important mechanism in cancer chemotherapy by ISL. In addition, zoledronic acid (a third generation bisphosphonate) appeared to reduce clonogenic survival of SACC-83 cell line through upregulation of reactive oxygen species, autophagy, and apoptotic cell death. Thus, zoledronic acid should be a promising drug for the treatment of AdCC. 95 Moreover, survivin, an inhibitor of apoptosis protein (IAP) family is overexpressed in most cancers including head and neck squamous cell carcinoma (HNSCC) but not in normal tissues. 96 YM155, an inhibitor of survivin, suppressed tumorigenesis of AdCC through the induction of autophagy and apoptotic cell death. It appeared that YM155 triggered autophagy in an Erk- and mTOR-dependent manner. 97 Accordingly to the autophagy-depended SGC development, autophagy modulators may represent a novel approach to treat SGC patients alone or in combination with other chemotherapeutic approaches (radio- and/or chemotherapy).
Clinical trials
Many studies are already in the clinical phase using a plethora of molecules that modulate autophagy in different stages. Figure 1 shows different agents that modulate autophagy (inducers or inhibitors) in each step of the autophagy process.
It is well known that P3K/AKT/mTOR signaling pathway acts as an autophagy inhibitor. 98 The phase II clinical study uses the AKT inhibitor MK2206 in treating patients with progressive, recurrent, or metastatic AdCC (ClinicalTrials.gov no. NCT01604772). In early phase I trial studies, everolimus, an mTOR inhibitor, was used in order to restore salivary gland function in participants with locally advanced HNC after concurrent chemotherapy and radiation (NCT03578432).
Several studies have already highlighted the effect of anti-EGFR monoclonal antibodies (MoAbs) in autophagy initiation. It appeared that different anti-EGFR MoAbs, such as cetuximab and panitumumab, activated autophagy in several types of cancer. 99 Furthermore, many studies supported that acquired-resistance to anti-EGFR MoAbs biochemically converge into Ras/Raf/Mek/Erk and PI3K/AKT/mTOR pathways.100,101 In the light of this knowledge, the investigators of clinical trial NCT01637194 studied the side effects and the best dose of cetuximab when combined with everolimus in treating patients with metastatic or recurrent colon cancer or HNC. In another phase I/II clinical study, PI3K inhibitor BKM120 was combined with cetuximab in patients with recurrent or metastatic HNC. This study will identify the side effects and the best dose of PI3K inhibitor BKM120 when given together with anti-EGFR monoclonal antibody cetuximab (ClinicalTrials.gov no. NCT01816984). In addition, the phase II clinical trial studied cetuximab in order to define how well it worked when given together with sorafenib in patients with refractory, recurrent, and/or metastatic HNC. It appeared that sorafenib treatment induced autophagy, which might suppress tumor growth. 102
In the last few years, many immunomodulating molecules have already been developed and show significant efficacy. The Food and Drug Administration (FDA) has approved immune checkpoint inhibitors, such as ipilimumab (an anti-CTLA-4 MoAbs), nivolumab, and pembrolizumab (anti-PD-1 MoAbs) for several types of cancer, like melanoma, lung cancer, and renal cell carcinoma.103,104 Moreover, it suggested that checkpoint inhibitors were associated with autophagy modulation. 99 The goal of a phase II study is to evaluate the efficacy of nivolumab in combination with ipilimumab in patients with recurrent and/or metastatic (R/M) SGCs (ClinicalTrials.gov no. NCT03172624). Furthermore, two more clinical studies NCT03749460 and NCT03146650 try to investigate the combinatorial effect of nivolumab and ipilimumab in patients with SGCs.
However, paclitaxel, a well-known chemotherapeutic agent appears to block autophagy through inhibited phosphorylation of Vps34 at T159 amino-acid residue and blocking autophagosome–lysosome fusion. 29 In more details, the Vps34 complex, which consists of Vps34, Vps15, ATG6, and ATG14 in yeast and Vp34, Vps15, Beclin-1, the WD domain protein Ambra1, and the endophilin Bif-1 in mammals, is a key regulator for autophagy machinery.105–107 Many studies are already in clinical phase II using paclitaxel. In phase II clinical study (NCT01969578), the investigators combine different chemotherapeutic agents including paclitaxel (carboplatin + paclitaxel) in patients with SGC in order to evaluate the efficacy and safety of chemotherapy versus androgen deprivation therapy (ADT) in patients with recurrent and/or metastatic AR expressing SGCs. In another study (NCT00002632), paclitaxel displayed a moderate effect in SGCs of MEC and AdCC histology. The poor response rate in adenoid cystic carcinoma is consistent with prior reports in this chemoresistant histologic subtype 108 and the direct and/or indirect inhibition of autophagy by paclitaxel may have an essential role in the efficacy of this drug in SGCs. In addition, the phase II clinical trial (NCT01316757) tries to evaluate the beneficial effect of co-treatment of paclitaxel with several different agents, for patients with metastatic or recurrent HNSCC. A phase I trial (NCT01847326) studies the side effects and best dose of paclitaxel albumin-stabilized nanoparticle formulation when given together with carboplatin followed by chemoradiation in treating patients with recurrent HNC. Moreover, in a randomized phase III clinical trial to compare the effectiveness of methotrexate with that of paclitaxel in treating patients who have advanced HNC that cannot be treated with cisplatin.
Several autophagy modulators appear to show side effects that range from mild to serious reactions. The most well-known autophagy inducers, mTOR inhibitors or rapalogs, appear to be insensitive to the hypoxic region of tumors. Rapalog, a conventional chemotherapeutic drug, is more effective in patients with benign tumors with cytostatic rather than cytotoxic effect. However, therapy with rapalogs suffers from significant side effects due to non-tissue and cell specificity such as dermatological, respiratory, metabolic, hematological, and renal toxicities. 109 On the contrary, the use of hydroxychloroquine (HCQ) (a well-described autophagy inhibitor) at low doses and in short time periods shows rare incidents of side effects. However, at higher doses and longer time period, some serious side effects such as retinal toxicity have been observed. 110 In addition, patients with autoimmune diseases (i.e. arthritis or lupus) appear to tolerate HCQ treatment well with rare reports of systemic reactions. 111 Table 2 presents clinical trial with agents that modulate autophagy in SGCs.
Clinical studies with autophagy modulators for SGC patients.
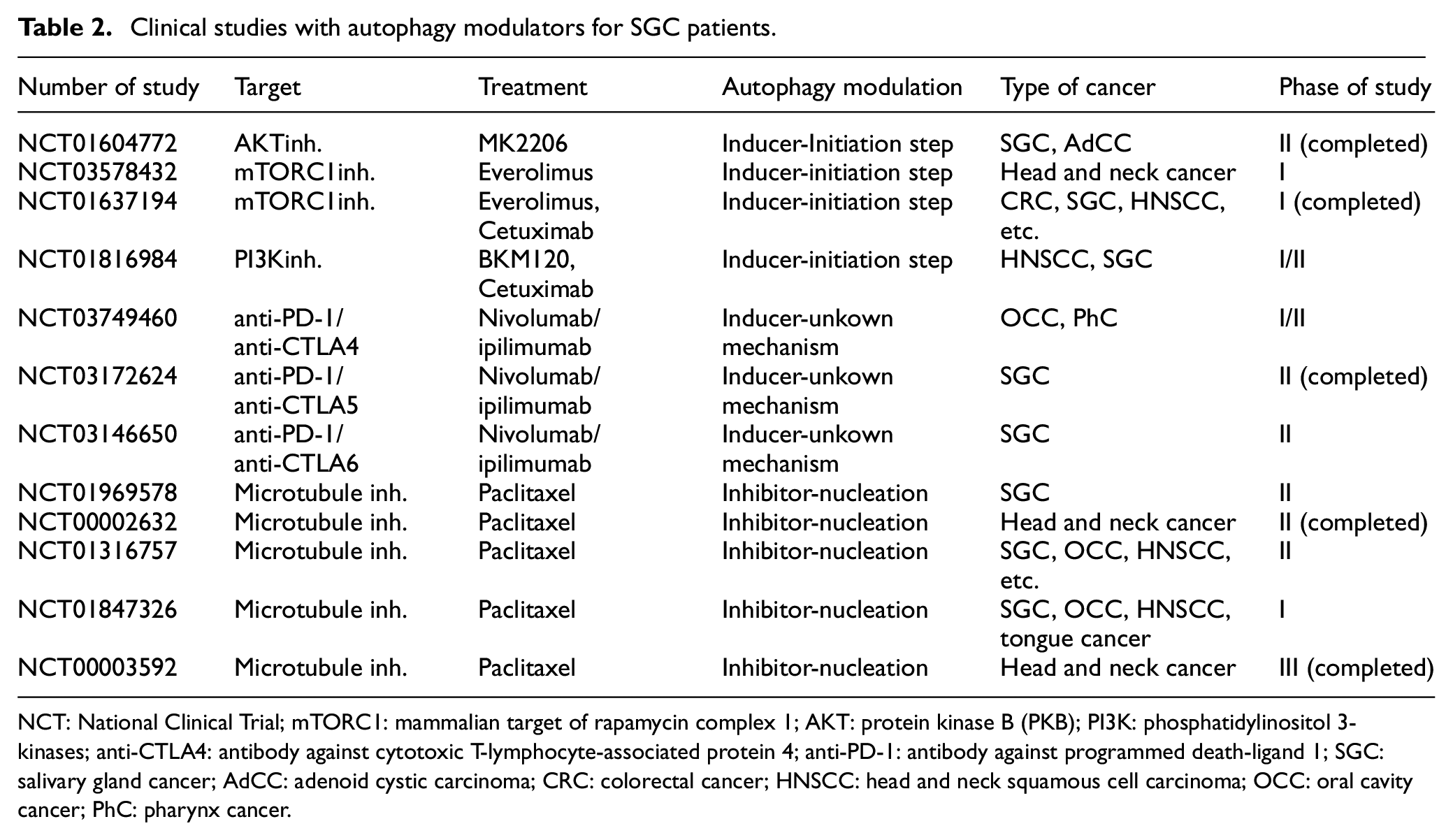
NCT: National Clinical Trial; mTORC1: mammalian target of rapamycin complex 1; AKT: protein kinase B (PKB); PI3K: phosphatidylinositol 3-kinases; anti-CTLA4: antibody against cytotoxic T-lymphocyte-associated protein 4; anti-PD-1: antibody against programmed death-ligand 1; SGC: salivary gland cancer; AdCC: adenoid cystic carcinoma; CRC: colorectal cancer; HNSCC: head and neck squamous cell carcinoma; OCC: oral cavity cancer; PhC: pharynx cancer.
Conclusion
It is well known that the main role of autophagy is strongly associated with the degradation of the dysfunctional cellular organelles and proteins. A plethora of studies in the field of autophagy and cancer highlights the controversial role of autophagy as either tumor suppressor or promoter mechanism in different types of cancer including SGC. Several in vitro and in vivo studies in SGCs have associated autophagy with the development and progression of this type of cancer. Autophagy markers such as Beclin-1 and LC3 appeared to associate with a different stage in SGCs. Beclin-1 expression showed a statistically significant correlation with histological grade. Furthermore, small agents have been identified to trigger apoptotic cell death through the induction of autophagy in SGCs cell models. In light of in vitro and in vivo results on SGCs, together with the available pharmaceutical tools, several clinical trials are now underway in this area and some of them have already given encouraging results for patients with SGCs. It will be interesting for future studies in SGCs to focus on the different circumstances that autophagy are triggered. Finally, it would also be interesting to combine small molecules that modulate autophagy with other therapeutic approaches, including chemotherapy, immunotherapy, and radiation, in order to determine the beneficial effect of this putative therapeutic approach for SGC patients.
Footnotes
Author contributions
E.K., P.S., M.T., M.V.K., and S.T. made substantial contributions in the conception, design, and interpretation of the data. S.T. and M.V.K. made substantial contributions in drafting the manuscript and revising it critically for valuable intellectual content. E.K., P.S., and M.T. made substantial contributions to acquisition, analysis, and interpretation of data. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work. The manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. The order of authors listed in the manuscript has been approved by all of us. All authors have no consideration of intellectual property associated with this work and there are no impediments to publication, including the timing of publication, with respect to intellectual property. All authors have followed the regulations of their institutions concerning intellectual property.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was co-funded by the European Union (ESF) and Greek National Funds through the Operational Program “Human Resources Development, Education and Lifelong Learning,” project title “Immunohistochemical Markers for the Detection of Angiogenesis and Biological Behavior of Salivary Gland Tumours” (project code: 14999).
