Abstract
The abundance and location of CD8+ tumor-infiltrating lymphocytes demonstrate important facets of the anticancer immune response. CD8-expressing lymphocytes have been used in immunotherapy for multiple cancers. This study aims to determine the association between the abundance and localization of CD8+ tumor-infiltrating lymphocytes and clinical outcomes of Wilms’ tumor. This retrospective study employed 42 pediatric patients diagnosed with Wilms’ tumor. CD8+ tumor-infiltrating lymphocyte counts were calculated based on the mean percentage of stroma occupied by CD8+ lymphocytes at the center and the invasive border of the tumor using immunohistochemistry. CD8+ tumor-infiltrating lymphocyte counts were significantly higher in the center and the invasive border of the early-stage tumor samples. CD8+ tumor-infiltrating lymphocytes in the invasive border and tumor center positively correlated with tumor invasion, regional lymph node invasion, histological type, metastasis, and stage of the tumor. A high CD8+ tumor-infiltrating lymphocyte scores at the invasive margin of the tumor correlated with low tumor recurrence. Low CD8+ tumor-infiltrating lymphocyte scores in the two tumor regions correlated with poor prognosis and shorter disease-free survival. Overall, these findings show that patients with high CD8+ tumor-infiltrating lymphocytes are associated with better clinical outcomes. Therefore, measuring the abundance of CD8+ tumor-infiltrating lymphocytes may be useful in predicting response to cancer immunotherapies.
Introduction
CD8+ lymphocytes express T-cell receptors to identify specific antigens. These antigens bind to class I major histocompatibility (MHC) molecules inside the cell before being transported to the cell surface where they are recognized by the T cell and destroyed. 1 The T-cell receptor binds to the class I MHC molecule via the CD8 glycoprotein, which binds to the constant portion of the class I MHC molecule. 2
CD8+ and CD4+ lymphocytes comprise the major components of the tumor immune microenvironment. These cells limit neoplastic cell growth and suppress tumor cell infiltration, primarily via the destruction of neoplastic cells 3 by the death receptor pathway, 4 degranulation pathway, 5 and secretion of tumor necrosis factor-α (TNF-α), interferon gamma (IFN-γ), and other cytokines.6,7 Numerous studies have highlighted the presence of CD8+ tumor-infiltrating lymphocytes (CD8+ TILs) in different parts of solid tumors, such as the tumor center, tumor stroma, or invasive border of the tumor.8–12
The recent decades have seen significant advances in the field of cancer immunotherapy as a promising model for the treatment of cancer. CD8+ TILs play a critical role in the clinical outcome of cancer patients.13–16 However, CD8+ lymphocytes are useful biomarkers of the efficacies of immunotherapies for several cancers. 17 An increase in the population of Wilms’ tumor-infiltrating CD8+ lymphocytes may correlate with beneficial clinical outcomes of immunotherapy. Thus, we try to determine the association between the abundance and localization of CD8+ TILs and clinical outcomes of Wilms’ tumor.
Materials and methods
Patient data
This retrospective study was conducted on 42 consecutive patients who underwent radical nephrectomy and complete surgical tumor staging for Wilms’ tumor at the department of surgery, Ali Asghar Pediatric Hospital, Iran University of Medical Sciences between March 2011 and June 2017. The study protocol was approved by the Ethics Committee of the Iran University of Medical Sciences (Issuance of ethics certificate with code IR.IUMS.REC 1394.8723215108 and complied with the guidelines embodied in the Helsinki Declaration). All the guardians or parents of the patients’ agreed to the experimental procedures and provided written informed consent. All data and personal information were anonymized. The inclusion criteria included the availability of follow-up of patients associated with optimal surgical specimens for pathological assessment and immunohistochemistry. Immunocompromised patients, patients with chronic diseases, patients subjected to neoadjuvant chemotherapy before surgery, and patients with incomplete data and unsuitable pathology paraffin blocks were excluded. All data were obtained from the pathology department of the hospital. Tumors were staged into five categories based on the TNM classification (pathological tumor, node, and metastasis) using the American Joint Committee on Cancer staging system (8th edition). Pathologic specimens from each patient were reviewed and confirmed independently by two pathologists. The final follow-up date was 31 June 2017, and all cases were examined. The minimum follow-up period for each patient was 26 months.
Immunohistochemistry method
Representative paraffin-embedded tissue blocks were punched (5 µm) onto glass slides to construct the tissue microarray (TMA). Immunohistochemistry for CD8+ lymphocytes was performed using a primary antibody against human CD8 (clone C8/144B, dilution 1:100) according to the manufacturer’s protocol (DakoCytomation, Glostrup, Denmark). Briefly, the tissue sections were deparaffinized, rehydrated using graded ethanol, incubated with 0.3% hydrogen peroxide for 35–40 min, and blocked using 10% bovine serum albumin–containing buffer. Slides were incubated with the primary antibody at 4°C overnight followed by incubation with the horseradish peroxidase–conjugated secondary antibody (Thermo Scientific, Waltham, MA, USA) at room temperature for 1 h. Positive staining was visualized using 3,3’-diaminobenzidine tetrahydrochloride (DAB) substrate (Gene Tech, Shanghai) and counterstained with hematoxylin. We ignored areas associated with immune cell infiltration in vessels, lymph nodes, and lymphatic, necrotic tissue, or necrosis-adjacent regions. Tonsil tissue was used as a positive control for CD8+ lymphocytes, whereas the negative control was prepared by replacing the primary antibody with a non-immune immunoglobulin of the same isotype. Positive and negative controls were included for each replicate. Two independent investigators examined the sections using a digital slide scanner microscope (Nikon DR-Si2). Immunohistochemical staining of CD8 was independently evaluated by two pathologists who were blinded to the pathological characteristics and patients’ outcomes. The interobserver agreement was 0.81 (κ statistic can take values from −1 to 1, and the range of 0.81–0.99 shows near-perfect agreement).
Immunohistochemistry evaluation (scoring system)
Immunostaining was used to measure the expression of CD8 in the cytoplasm and membrane of lymphocytes. We counted the CD8+ lymphocyte content using ten representative high-power microscopic fields (40× magnification) of the intratumor region and invasive border of the tumor. The average cell numbers for each area were scored as follows: 0 (average number 0), I (average numbers 1–19), II (average numbers 20–49), and III (average numbers >50). This formulated the CD8+ lymphocyte infiltration score (CD8+ TIL score) (Figure 1(a)–(d)).
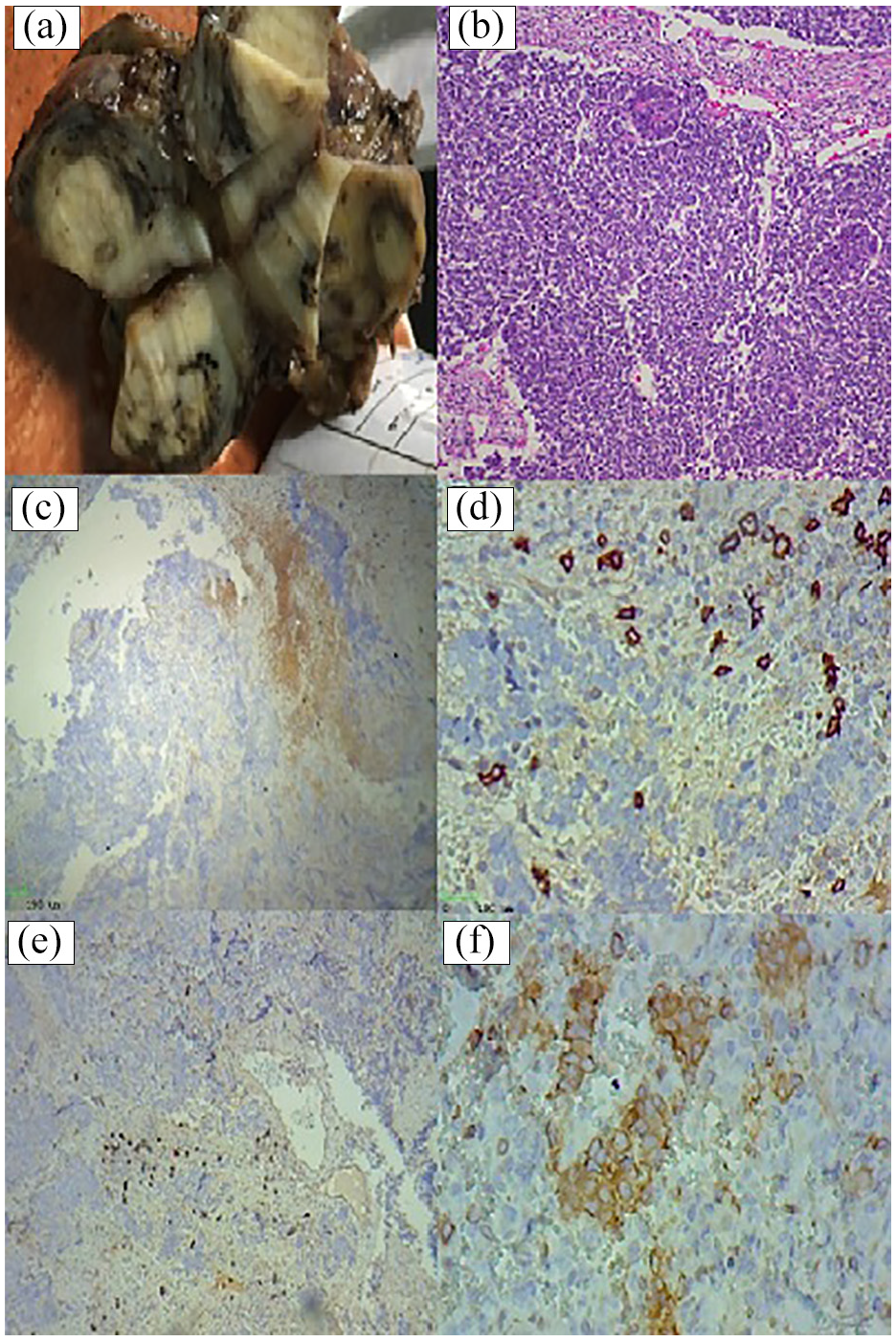
A 4-year-old boy with Wilms’ tumor. Macroscopic examination shows a well-demarcated creamy solid mass measuring 9 cm × 7.5 cm × 6 cm in upper lobe of right kidney (a). Microscopic examination represents an encapsulated mass with undifferentiated blastema on fibroblast-like stroma and epithelial elements (B) (hematoxylin and eosin (H&E) 100×) (b). Wilms’ tumor tissues showing a high CD8+ T-cell lymphocyte expression at invasive marginal border of tumor (c, d) (immunohistochemistry (IHC) original magnification 100×, 200×). Wilms’ tumor tissues showing intratumoral a high CD8+ lymphocyte expression (e, f) (IHC original magnification 100×, 200×).
Statistical analysis
The clinical data were analyzed using standard statistical tests. Pearson’s chi-square method or Fisher’s exact test was used to determining the correlation between CD8+ TIL score and clinicopathological factors. The CD8+ TIL scores were analyzed using the Spearman correlation test. Survival analysis and prognosis performed using the Kaplan–Meier curves were based on specific cut-offs to illustrate differences in disease-free survival (DFS). DFS was defined as the time between primary surgical treatment and date of relapse. DFS was monitored during the last follow-up for patients who survived without disease recurrence or on the day of non-cancer-associated death. Data analysis was performed using the SPSS version 22.0 (Chicago, IL, USA). p < 0.05 was considered as statistically significant.
Results
We retrospectively analyzed 42 patients with Wilms’ tumor for 72 months of follow-up data approximately (mean 59.4 ± 11.6). Twenty-seven boys and 15 girls were included in this study. The male to female ratio was 1.8. The mean age of male and female patients was 28 months (ranged from 1 to 56 months) and 31 months (ranged from 1 to 49 months), respectively. The clinicopathologic characteristics of the patients are summarized in Table 1.
Patients and clinicopathological characteristic of Wilms’ tumor.

Most of the patients (55%) represented biphasic (blastoma and epithelial) histologic subtype of the tumor with favorable histology (69%) in the early stage (36%). During follow-up of the patients, 30 patients (71.5%) were DFS, seven patients (16.5%) presented with recurrence, and five patients (12%) presented with metastasis who suffered from high-stage tumors (≥stage III). None of the patients died during the initial years of follow-up. The CD8+ TIL score at the invasive border of tumor and tumor center area was significantly lower in larger tumors (>4 cm; p < 0.04 and p < 0.05, respectively), ureter invasion (p < 0.01 and p < 0.02, respectively), capsular invasion (p < 0.03 and p < 0.03, respectively), renal vein invasion (p < 0.05 and p < 0.03, respectively), renal sinus invasion (p < 0.04 and p < 0.01, respectively), nodal invasion (p < 0.05 and p < 0.02, respectively), anaplastic tumor (p < 0.03 and p < 0.03, respectively), adjacent organ invasion (p < 0.02 and p < 0.05, respectively), and advanced stages (p < 0.05 and p < 0.02, respectively). The association between CD8 expression and clinicopathological characteristics of Wilms’ tumor in invasive border and tumor center locations is summarized in Table 2.
The association between CD8 expression and clinicopathological characteristic of Wilms’ tumor in invasive border and tumor center locations.

TIL: tumor-infiltrating lymphocyte.
Patient age, gender, and histology did not significantly affect the CD8+ TIL scores in the two tumor locations (p > 0.05). The CD8+ TIL score in the two tumor regions was significantly lower in patients with recurring tumors (p < 0.04 and p < 0.05, respectively), metastasis (p < 0.05 and p < 0.01, respectively), and shorter DFS (p < 0.03 and p < 0.05, respectively; Table 2). In contrast, the CD8+ TIL scores were higher in the two tumor locations during the early stage of the tumor without recurrence or metastasis. There was a positive correlation between the high CD8+ TIL score and the favorable prognosis in all the patients. Patients with high CD8+ TIL scores survived longer than patients with low CD8+ TIL scores (Figure 2(a)–(d)). The main characteristics of patients enrolled for survival analysis of Wilms’ tumor are summarized in Table 3.
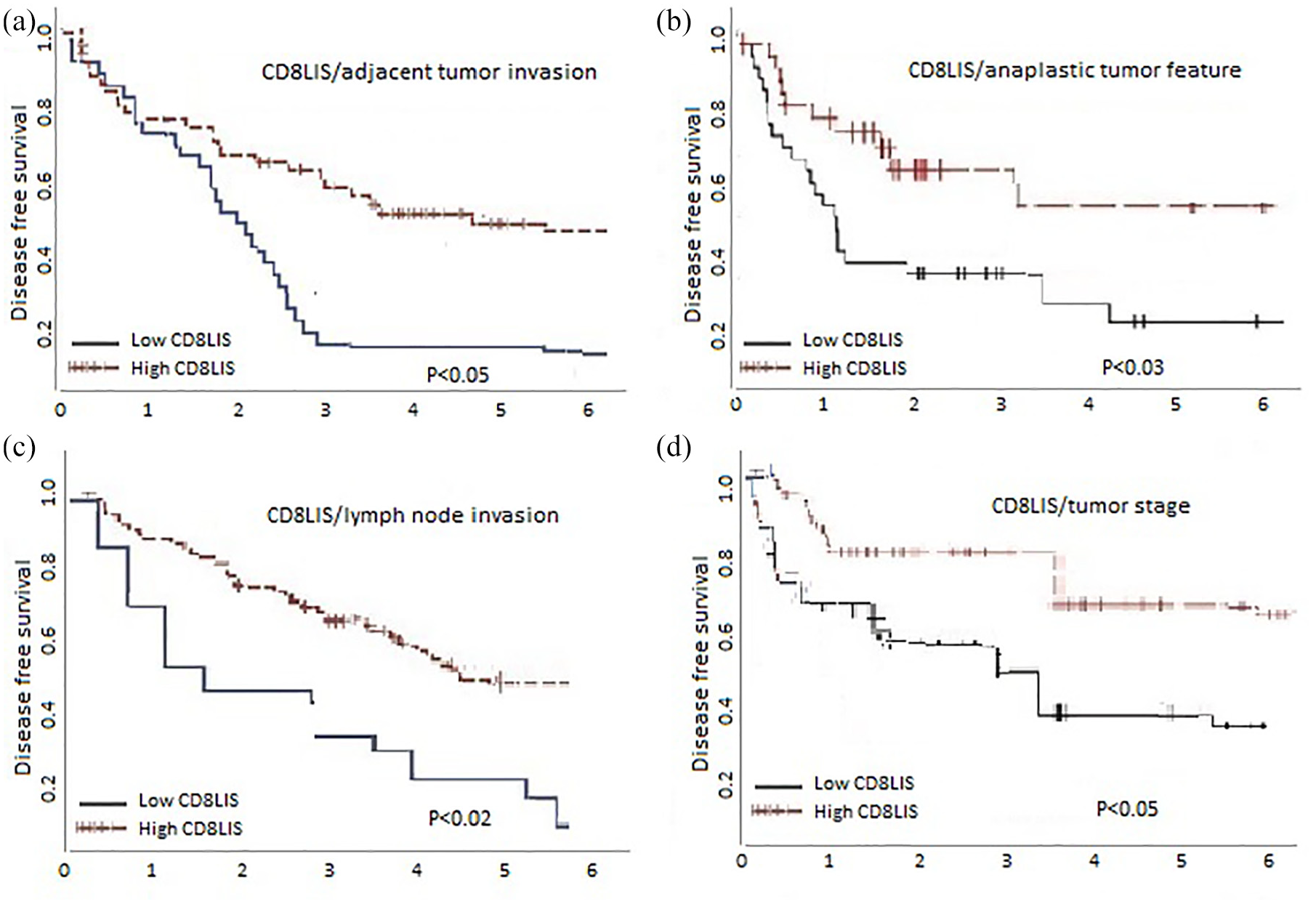
Disease-free survival of Wilms’ tumor based on association with high versus low CD8+ lymphocytes response with adjacent tumor invasion (a), anaplastic tumor feature (b), lymph node invasion (c), and tumor stage (d) of the patients (n = 42). The disease-free survival (DFS) of patients with higher CD8+ lymphocyte infiltration score (CD8+ TIL) does significantly differ from patients with low CD8+ TIL on patients with adjacent tumor invasion, anaplastic tumor feature, lymph node invasion, and advance tumor stage. Kaplan–Meier curves of the probability of DFS based on percentile cut-offs of CD8+ lymphocyte count per mm2. Values below the percentile cut-off are depicted by a black line and values above the percentile cut-off are depicted by a red line.
The main characteristics of patients enrolled for survival analysis of Wilms’ tumor.

SD: standard deviation.
Our findings indicate better clinical outcomes for Wilms’ tumor in patients with high CD8+ TIL scores in the center and the invasive border of the tumor. Thus, CD8+ TIL score may serve as a biomarker for developing novel immunotherapeutic approaches in the future.
Discussion
Wilms’ tumor is the most common malignant renal tumor in children. Patients with early-stage tumors of favorable histology exhibit longer overall survival. 18 There is limited knowledge of the correlation between TILs and the prognosis of patients with solid cancers. TILs were evaluated based on the recommendations of the International TILs Working Group in Solid Tumors. 19
A high abundance of TIL correlates with improved patient survival. 20 However, the subset of TILs that play a critical role in the progression and prognosis of solid cancer remains to be understood. 21 Majority of TILs in solid tumors comprise T lymphocytes, which include CD4 helper and CD8 cytotoxic lymphocytes. 22 Accordingly, during the adaptive immune reaction that suppresses tumor progression, CD8+ lymphocytes are integral in destroying neoplastic cells and trigger apoptosis. 23 Previous studies have shown that CD8+ lymphocytes have a protective prognostic effect in many cancers, such as colorectal, 24 lungs, 25 esophageal, 26 ovarian, 27 renal, 28 pancreatic, 29 liver, 30 breasts,31–33 and skin cancers. 34
In this study, we found that CD8+ TILs correlated with DFS. However, the effect of CD8+ TILs on the DFS of patients with Wilms’ tumor depended on their abundance and location. The univariate analysis reported that patients with increased abundance of CD8+ T cells showed longer survival, also confirmed using the Kaplan–Meier curves. However, these findings are in contrast with those of Catacchio et al., 35 who reported a strong infiltration of intratumoral CD8+ T cells correlated with poor survival in patients with breast cancer. This could be attributed to the invasiveness of breast cancer that is different from the invasiveness of Wilms’ tumor in children.
Chen et al. 17 reported that the degree of infiltration of CD8+ TILs correlates with the clinicopathological stage of lung sarcomatoid carcinoma. The mean overall survival of patients with a high abundance of CD8+ TILs is higher than that of patients with a low abundance of CD8+ TILs. These results suggest that a high density of CD8+ lymphocytes has antitumor benefits and plays an important role in improving patient survival. This is following the findings of this study and other reports, including Al-Shibli et al. 36 and Mohamed et al. 37 for breast cancer, Shibutani et al. 38 for colorectal cancer, and Brambilla et al. 39 for non-small cell lung cancer.
The past few decades have seen advances in the research on Wilms’ tumor along with progress in the effective treatment of this cancer. However, not all children are cured and further research is warranted on enhancing the rate of therapeutic efficacy. Holl et al. 40 analyzed the immune response of five patients with Wilms’ tumor before undergoing chemotherapy: activated CD8 and CD4 T cells infiltrated the tumor. This indicates that patients with Wilms’ tumor are susceptible to immunotherapy like specific adult malignancies. Activated CD8+ TILs play a critical role in antitumor immunity. Cancer immunotherapy has significantly improved the survival of adults with malignancies. 41 A few studies have investigated the immune microenvironment of Wilms’ tumor in an attempt to address the critical role of immunotherapy. This study aimed at understanding the importance of TILs in predicting the clinical outcome of patients with Wilms’ tumor. However, future studies should employ a larger patient cohort and a longer follow-up period to confirm the role of immunotherapy in these patients.
In conclusion, the results of this study indicate that CD8+ TILs have clinical significance in antitumor activity against human Wilms’ tumor. The presence of CD8+ TILs within the center or invasive margin of the tumor correlated with favorable clinical outcomes. The prognostic value of CD8+ TILs in Wilms’ tumors might be useful to devise personalized immunotherapy in the future.
Footnotes
Acknowledgements
The authors thank all the patients for their participation in this study.
Author contributions
K.M. and M.R. contributed to conception and design. S.M. helped in the collection of tissue samples from patients who went to surgery and the acquisition of data. M.R. involved in analyses, interpretation of data, and statistical analyses. N.M. helped in the drafting of the manuscript. M.R. was involved in study supervision. All authors involved in the revision of the manuscript and have approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This retrospective study was confirmed in the Ethics Committee of Ali Asghar Children Hospital.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written consent was taken from the parents and the patients’ guardians at the time of admission as a conventional approach before study inclusion.
Availability of data and materials
The data generated or analyzed during this study included in this published article.
