Abstract
Introduction:
Metastasis-associated in colon cancer 1 (MACC1), one of the prognostic markers for colonic and other tumours was noted to be overexpressed in retinoblastoma (Rb) Y79 cancer stem cells. This prompted us to evaluate its expression in primary Rb tumour and serum samples with clinicopathologic correlation. The interacting partner, c-MET was also evaluated in primary tumour tissues to explore the activation of MACC1 signaling.
Methodology:
This study was done following institutional review board approval from participating institutes. Semiquantitative gene expression for
Results:
High expression of
Conclusion:
Overexpression of MACC1 and c-MET in retinoblastoma tissues, specifically those with risk factors for metastasis, suggests its role in proliferation and possibly in invasion. However, the current data do not support it to be a clinical prognostic marker in retinoblastoma tumours. The inverse serum expression is an intriguing finding, which warrants further studies especially in retinoblastoma.
Introduction
Retinoblastoma (Rb) is the most common childhood intraocular malignancy initiated by a mutation within the retinoblastoma (
Metastasis-associated in colon cancer 1 (MACC1) was first identified in colon cancer using differential display real-time polymerase chain reaction (RT-PCR) by Stein and colleagues.11,12 It has been further studied in several cancers such as breast, ovarian and gastric cancer as an important prognostic indicator for systemic metastasis and metastasis-free survival.13–19 MACC1 has been reported to promote tumour cell invasion and metastasis via the hepatocyte growth factor (HGF)/c-MET pathway.11,20 In colon cancer, Stein and co-workers reported the overexpression of MACC1 in cytoplasm and that it translocated into the nucleus following HGF treatment acting as a transcriptional regulator for receptor tyrosine kinase, MET. It was therefore implied that MACC1-facilitated activation of the HGF/c-MET pathway led to increased invasive and metastatic ability of colon cancer cells.11,21,22
A comprehensive review has also shown that MACC1 is observed in other human normal tissues such as intestine, stomach, pituitary gland, kidney and trachea showing highest expression, followed by pancreas, mammary gland, bone marrow (BM), ovary, lung, heart, liver and B-lymphoblasts.
21
Dunlevy and co-workers observed that MACC1, earlier named as 7a5 protein, was expressed in ARPE-19 and Y79 cells, neural retina and retinal pigment epithelium tissue extracts.
23
Similarly, we observed the overexpression of
Methods
Sample collection
This study was carried out with approval from the institutional review board (IRB) (LEC 01-18-005) of LV Prasad Eye Institute, Asian Institute of Gastroenterology and University of Hyderabad (UH/IEC/2018/2). Informed consent was obtained prior to the collection of samples. Fresh Rb tumour tissue samples (
Semiquantitative polymerase chain reaction
Total RNA was isolated from fresh Rb tissues (
Immunohistochemistry
Immunolocalization of MACC1 was done in Rb tissues (n = 22) and BM aspirates (n = 3), and c-MET was carried out in Rb tissues (n = 17). Briefly, the enucleated Rb eye was fixed with 10% fresh formalin, processed and embedded in paraffin. The BM aspirates from cases with proven Rb metastases were collected in EDTA blood collection tubes, gently mixed and were transferred into a petri dish. They were further fixed by adding 10% fresh formalin, processed and embedded in paraffin. Thin 3 µm sections were taken on silane-coated glass slides and used for immunostaining with the primary antibodies – MACC1 (Abcam, MA, USA) and c-MET (Santa Cruz Biotechnology, Inc., CA) as per previously published protocol. 25 The sections were visualized under a light microscope, and the staining was assessed blindly by two experienced ocular pathologists (G.K.V. and D.K.M.).
Semiquantitative immunoreactivity scoring (IRS) system was used to score the intensity of immunohistochemistry (IHC) staining as per previously published established protocol. 26 Briefly, all the immunostained sections were graded as follows: grade 4 for positively stained cells >80%; grade 3 for positively stained cells between 51% and 80%; grade 2 for positively stained cells between 10% and 50%; grade 1 for positively stained cells <10%; and grade 0 for no positively stained cells. Furthermore, the intensity of the staining was assessed visually under a light microscope by two experienced ocular pathologists blinded to the clinical data and were scored as 0 (no colour reaction); 1 (mild reaction); 2 (moderate reaction); and 3 (intense reaction). A composite score ranging between 0 and 12 was achieved by multiplying the grade and the intensity. An IRS score between 0 and 1 was considered negative; 2 and 3 was considered mildly positive; 4–8 was considered moderately positive; and 9–12 was considered strongly positive.
MACC1 serum analysis by enzyme-linked immunosorbent assay
MACC1 protein expression in serum samples of Rb patients with intraocular tumour (
Statistical analysis
All statistical analyses were done using the GraphPad Prism software (La Jolla, CA, USA), and the results were presented as mean values ± standard deviation. Multiple group comparisons were assessed by the one-way analysis of variance with Tukey’s multiple comparison tests.
Results
Clinical details
The clinical details of the Rb cases are provided in the supplementary Table 1. Mean age of patients (
Gene expression analysis

Evidence of overexpression of
Correlation of clinicopathological parameters with MACC1 expression (tissue and serum) in retinoblastoma.
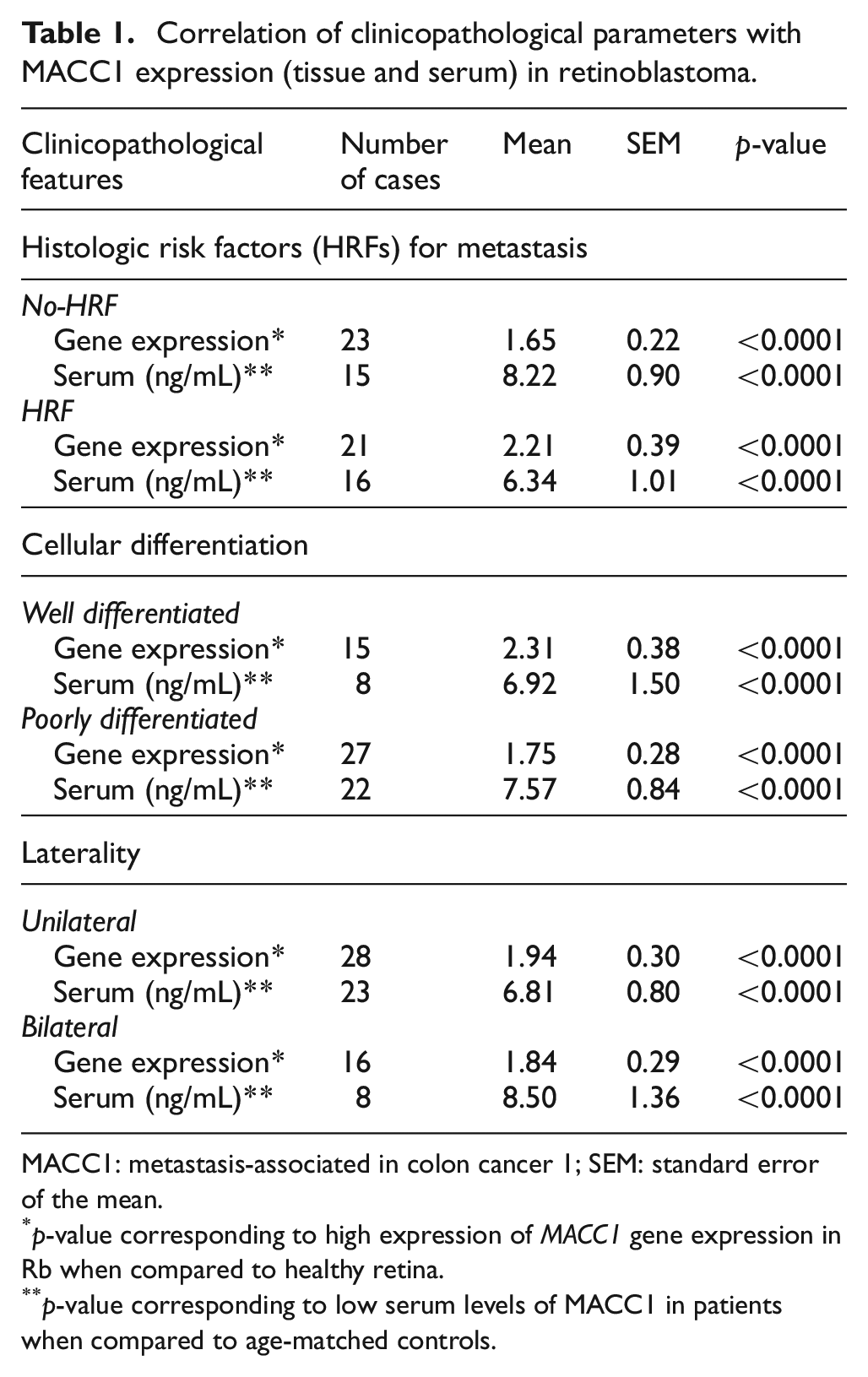
MACC1: metastasis-associated in colon cancer 1; SEM: standard error of the mean.
Immunolocalization of MACC1 in Rb tissues
Immunolocalization showed MACC1 expression in the cytoplasm and with many cells showing nuclear membrane and nuclear positivity in HRF cases (Figure 2(a)); however, in No-HRF cases, the expression was predominantly cytoplasmic (Figure 2(b)). The expression varied from moderate to high in both HRF and No-HRF cases. Mean IRS scoring revealed that the MACC1 expression was significantly higher in cases with HRF than compared to No-HRF cases (
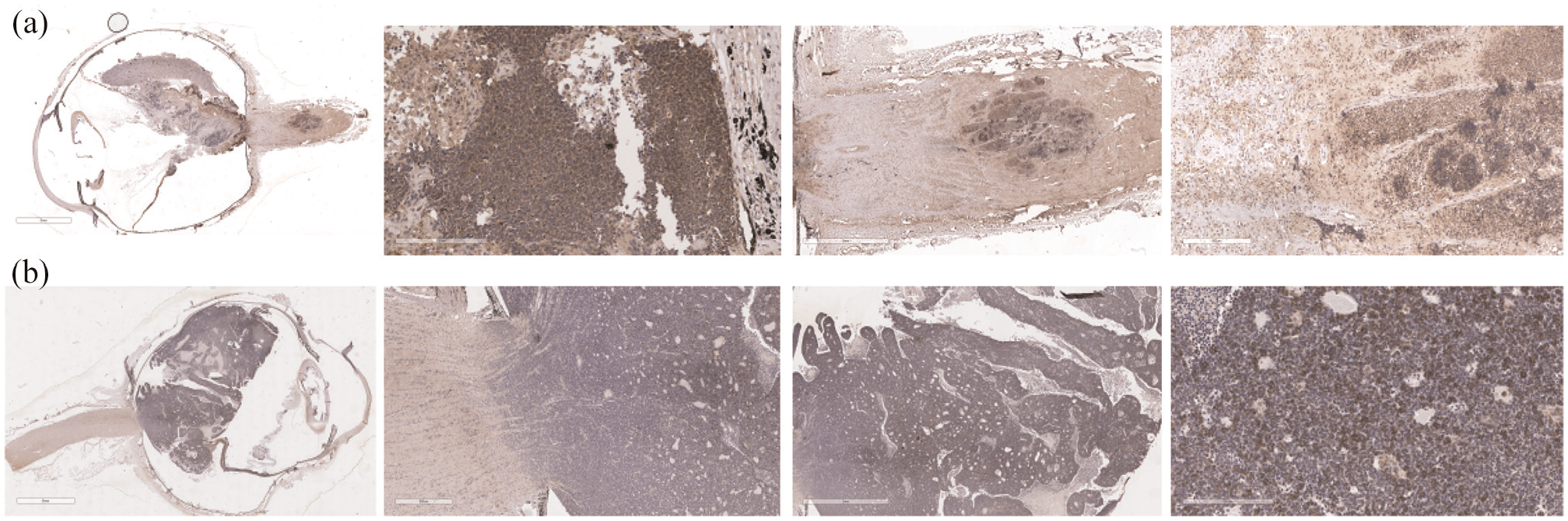
Immunolocalization of MACC1 in Rb tissues. Rb tumour cells show intense immunoreactivity to MACC1, with (a) HRF cases showing nuclear, membrane and cytoplasmic expression and (b) No-HRF cases showing predominantly cytoplasmic expression.

IRS score of MACC1 tissue immunolocalization.

Immunolocalization of MACC1 in Rb bone marrow positive cases: Cell block of the bone marrow aspirate with tumour cells (*) stained with haematoxylin and eosin under (a) low and (b) high magnifications. MACC1 positive tumour cells (*) within bone marrow under (c) low and (d) high magnifications.
MACC1 expression was noted as a nuclear staining in the healthy inner retinal layers, nuclei of limbal epithelial cells and cytoplasmic expression was observed within the healthy corneal epithelium, optic nerve, retinal pigment epithelium and choroid (Figure 5(a)–(e)).

Immunolocalization of MACC1 in healthy ocular tissues: MACC1 expression was observed in healthy (a) corneal epithelium, (b) limbus, (c) neural retina, (d) retinal pigment epithelium (RPE) and choroid and (e) optic nerve (10×).
Immunolocalization of c-MET in Rb tissues
c-MET was highly expressed in all cases, with variable staining patterns, localized to cytoplasm, nuclear membrane and nucleus (Figure 6(a) and (b)). Staining pattern was predominantly nuclear in HRF cases whereas it was localized to cytoplasm in No-HRF cases. Mean IRS scoring revealed that the c-MET expression was significantly higher in cases with HRF than compared to No-HRF cases (

Immunolocalization of c-MET in Rb tissues and other ocular tissues: (a) Rb cells expressed c-MET with the HRF cases showing nuclear, membrane and cytoplasmic expressions (10×) and (b) No-HRF cases showed less expression of c-MET with predominant cytoplasmic expression (10×, 20×). c-MET was also shown to be expressed in healthy (c) corneal epithelium, (d) limbus, (e) neural retina and (f) optic nerve (4×, 10×).

Semiquantitative IRS score of c-MET tissue immunolocalization.
Serum levels of MACC1 in retinoblastoma
Serum analysis of MACC1 protein level in Rb patients when compared to healthy age-matched control samples revealed that there is a significantly lower expression in Rb cases (7.25 ± 0.70 ng/mL) (

MACC1 serum analysis in Rb: (a) HRF cases (
Discussion
Retinoblastoma, one of the most common malignant ocular tumours, is known for its aggressive nature and rapid intra and extraocular spread.1,4,27 While the clinical and histologic prognostic factors are reasonably well identified, further insights into understanding the mechanisms of Rb tumour progression and metastasis would help pave the way in designing therapies. This study focused on exploring one such potential biomarker for metastasis first identified in colonic tumour, MACC1, which was also highly expressed in primary Rb tumours. This study reports the novel finding of MACC1 overexpression in Rb tumours, more so in cases with HRFs for metastasis than those without HRF. Immunolocalization of MACC1 and its downstream regulator c-MET revealed both cytoplasmic as well as nuclear staining; however, nuclear staining correlated with high risk factor for metastasis, hinting at its possible role in proliferation and invasion in Rb tumours.
MACC1 was first identified by Stein and co-workers in colon cancer who observed that MACC1 mRNA levels could be an independent prognostic indicator for the formation of metastasis and tumour relapse.
19
While all the cases showed high expression of MACC1, it was significantly higher in HRF than No-HRF cases suggesting that it may have a role in invasion and proliferation thus reiterating the need for a longer follow-up. In a 10-year follow-up study in colorectal cancer, primary tumours that showed higher expression of MACC1 eventually developed distant metastases than those tumours with low expression.
29
A concordant result was also observed in hepatocellular carcinoma (HCC), where MACC1 expression was high in vascular invasive HCC.
30
Another observation which is not clearly understood (may or may not affect prognosis) is that
We also checked for other key pathways deregulated with
An interesting observation of this study was the high expression of MACC1 in normal ocular cells such as the neural retina, retinal pigment epithelium, limbal and corneal epithelium. Dunlevy and Koppelman 23 also showed similar expression of MACC1 protein in the extracts from these normal ocular tissues. A comprehensive analysis of MACC1 expression in a large array of normal tissues has shown a range of its expression with the highest in the intestine, trachea, pituitary gland, stomach and kidney. 21 Moderate to low expression was observed in the pancreas, BM, ovary, lung, liver, heart and mammary tissues. This evidence strongly suggests that MACC1 has a normal role in the growth and proliferation of cells and in neoplastic tissue, the overexpression of this protein leads to abnormal proliferation and promotes metastasis. Although the scope of this study did not include other childhood tumours, it is possible that being a childhood tumour, there could be an inherent overexpression of this marker in the ocular tissues studied.
One of the intriguing findings in this study is that the serum expression of MACC1 was observed to be significantly lower in primary Rb patients when compared to the healthy controls. Serum MACC1 expression in three cases with clinically proven extraocular metastasis also showed significantly lower expression when compared to primary Rb and control samples. This is in contrast to the studies in colonic tumours which highlight the reliability of using MACC1 as a serum marker for predicting metastasis. Interestingly, Tan and co-workers showed that in benign breast cancer patients, the serum MACC1 levels were similar to the healthy controls and lower than that of cases with metastasis suggesting that pathway may be activated after the metastatic cascade is triggered. 36 Wang et al. 37 showed that the plasma mRNA levels of MACC1 in patients with benign non-small cell lung cancer (NSCLC) were similar to that of healthy controls whereas the cases with proven metastasis, the levels were significantly higher. These studies and the results from our study partially hint at the temporal expression of MACC1 during the course of tumorigenesis and metastasis. Interestingly, our analysis revealed that in age-matched controls (healthy paediatric serum samples), MACC1 levels were significantly higher when compared to the normal adult serum samples. We believe that this rise in MACC1 serum concentration could be associated with other normal functions of this protein such as cell growth, stem cell maintenance and development.19,21
The overexpression of MACC1 in primary Rb tumours, especially in cases with HRF and poorly differentiated tissues, hints at the activation of MACC1/c-MET pathway for promoting metastasis within the tissue. To the best of our knowledge, this is the first report that identifies a differential regulation of MACC1 pathway in Rb tumour tissues and serum during tumorigenesis and metastasis. We believe that analysing the serum MACC1 levels in a larger cohort would add more value to the findings in the future and define the use of MACC1 as a prognostic marker in Rb.
In conclusion, this study reports the novel finding of constitutive overexpression of MACC1 and c-MET in Rb primary tissues suggesting its role in proliferation and possibly in invasion. While the tissue overexpression implies it to be a promising marker for predicting metastasis, the serum levels in this study do not support it to be a prognostic clinical marker. Further studies, specifically with regard to the downstream molecules in MACC1/c-MET pathway, may throw light on its association with proliferation and metastasis.
Supplemental Material
sj-xlsx-1-tub-10.1177_1010428320975973 – Supplemental material for Overexpression of metastasis-associated in colon cancer 1 in retinoblastoma
Supplemental material, sj-xlsx-1-tub-10.1177_1010428320975973 for Overexpression of metastasis-associated in colon cancer 1 in retinoblastoma by Rohini M Nair, Varsha Prabhu, Radhika Manukonda, Dilip K Mishra, Swathi Kaliki and Geeta K Vemuganti in Tumor Biology
Footnotes
Acknowledgements
The authors thank Asian Institute of Gastroenterology (AIG) for their collaboration and for providing technical support (control samples). The authors also thank Sreedhar Boyenpally and Chenchu Naidu G (LVPEI) for providing the technical support in the collection, processing and generation of retinoblastoma histological sections.
Author contributions
The study was conceptualized by G.K.V., and the experiments were designed by R.M.N., V.P. and G.K.V. The experiments were conducted by R.M.N., V.P. and R.M. Validation of experiments and data interpretation were done by R.M.N., V.P., R.M. and G.K.V. Clinical and histological data analyses were done by R.M.N., V.P., R.M., D.K.M., S.K., and G.K.V. All the authors contributed to the discussion of the results and approval of the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by academic research grants from the DST-PURSE (University of Hyderabad), UPE-II (University of Hyderabad), UGC-UKIERI (Thematic Partnership 2017–2020) and Hyderabad Eye Research Foundation (HERF). R.M.N. was supported by the UGC, Government of India (Senior Research Fellowship), and V.P. and R.M. are supported by the UGC-UKIERI grant. The funding body had no role in the design of the study, collection, analysis, and interpretation of data and in writing the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
