Abstract
Although it could be speculated that almost everything has been said concerning the use of statins in cancer therapy, statins as anticancer drugs have both committed supporters and opponents, for whom the dispute about the legitimacy of statin use in cancer treatment seems never to be clearly resolved; every year more than 300 reports which deepen the knowledge about statins and their influence on cancer cells are published. In this mini-review, we focus on the latest (since 2015) outcomes of cohort studies and meta-analyses indicating statin effectiveness in cancer treatment. We discuss attempts to improve the bioavailability of statins using nanocarriers and review the effectiveness of statins in combined therapies. We also summarise the latest results regarding the development of mechanisms of resistance to statins by cancer cells and, on the other hand, give a few examples where statins could potentially be used to overcome resistance to commonly used chemotherapeutics. Finally, special attention is paid to new reports on the effect of statins on epithelial–mesenchymal transition.
Introduction
Statins are a group of drugs commonly used in the treatment of cardiovascular diseases. These drugs are competitive inhibitors of 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR), one of the first enzymes that take part in cholesterol de novo synthesis, and are able to inhibit both cholesterol and, in further steps, isoprenoid synthesis.
Since the early 1990s, it has been known that statins also have anticancer properties, but to what extent statin use is beneficial in cancer treatment remains a subject of debate. The effect of statins on cancer cells in vitro is well known and has been reviewed, among others, by us. 1 The main problem regarding statin use in therapies is low bioavailability and toxicity at high doses, which are required for effective cancer treatment. Despite that, every year more than 300 reports are published that lead to a better understanding of the molecular mechanisms underlying statin actions and to give an unequivocal answer regarding the legitimacy of statins use in cancer therapy.
In this review, we have attempted to gather the latest reports (since 2015), in five sections, which reflect the ongoing trends and questions in the anticancer activity of statins.
Statins in meta-analyses and cohort studies
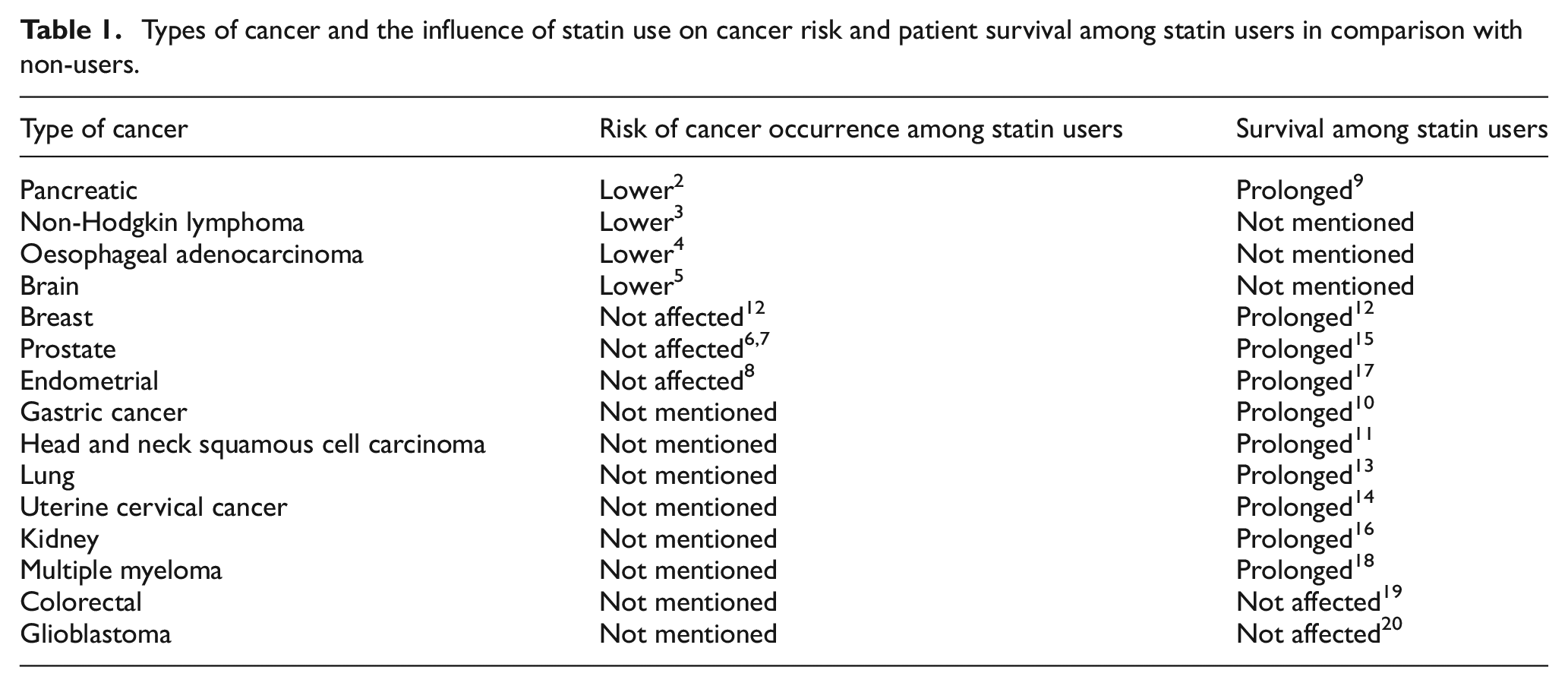
The vast majority of currently published studies on the application of statins in cancer therapies concern retrospective cohort studies or meta-analyses of data obtained for specific types of cancers in observational studies. In general, treatment with statins that preceded cancer diagnosis lowers the risk of cancer development and prolongs the overall survival in comparison with statin non-users or patients who started taking statins after a cancer diagnosis. For instance, studies have shown that the use of statins before a diagnosis is associated with a lower risk of pancreatic cancer, 2 non-Hodgkin lymphoma, 3 oesophageal adenocarcinoma 4 and brain cancer 5 but not breast cancer, 12 prostate cancer6,7 or endometrial cancer. 8 Also, regular statin use before diagnosis modestly prolonged the survival of patients in comparison with non-users or non-regular users in pancreatic cancer, 9 gastric cancer, 10 head and neck squamous cell carcinoma, 11 breast cancers, 12 lung cancer, 13 uterine cervical cancer, 14 prostate cancer, 15 kidney cancer, 16 endometrial cancer 17 and multiple myeloma, 18 but not in colorectal cancer 19 or glioblastoma 20 (see Table 1).
Types of cancer and the influence of statin use on cancer risk and patient survival among statin users in comparison with non-users.
More detailed studies drew attention to some other important points. For lung 21 and breast 22 cancer it was shown that lipophilic statins are associated with decreased cancer-specific and all-cause mortality, whereas hydrophilic statins have very weak protective effects. In the same study, it was noted that patients with more than 4 years of follow-up do not show a significant correlation between statin use and cancer-specific mortality or all-cause mortality, whereas groups with less than 4 years of follow-up still showed the protective effect of statins. 22
Other studies showed that specific histologic types of cancer may demonstrate differential susceptibility to statin treatment. This effect was observed for ovarian cancer 23 and lung cancer: adenocarcinoma and squamous-cell carcinoma patients had decreased mortality risk in comparison with small-cell lung carcinoma patients. The effect was also dependent on the dose of statin used. 24
Overall, it is worth noting that despite the fact that retrospective studies show protective effects of statins, prospective clinical studies have mostly not been successful. It may be due to the fact, that prospective statin studies omit some or all of the factors identified in preclinical studies that are likely to be necessary for statins to be efficacious. Moreover, it does not seem appropriate to assume that statins should be used in cancer treatment in the same way as they are used for the treatment of cardiovascular diseases. 25
New carriers of statins
The main problem faced by possible future statin therapies is the low oral bioavailability of the lipophilic drugs, which does not exceed 24%. Because statins have a high affinity to albumins, their bioavailability is usually lower than 10%, 26 and it is not possible to use statins in high doses due to their overall toxicity. To overcome this obstacle, many attempts have been made to design proper drug carriers that enable the delivery of statins in high doses and are characterised by low self-toxicity.
Many different materials, methods of drug loading and many different types of nanocarrier delivery systems have been tested in order to establish the most efficient method of statin delivery in a high dose that would effectively inhibit cancer progression and at the same time would not cause harmful side effects.
For instance, simvastatin was successfully encapsulated in calcium alginate nanocapsules formed on the interface of polysorbate 20 non-ionic surfactant solution, giving high drug loading efficiency.
27
There are also a few studies showing that free statins are less effective in vitro against cancer cells than their encapsulated forms. For example, simvastatin encapsulated in nanostructured lipid carriers that contain PEG/glycerides was effective against MCF7 cells.
28
Rosuvastatin and atorvastatin encapsulated in biodegradable polymeric micelles made of triblock copolymer PCL-PEG-PCL were cytotoxic for MCF7 cells.
29
Atorvastatin incorporated in nanostructured lipid carriers had lower IC50 values than free atorvastatin in these cells.
30
Alpha-lipoic acid nanoparticles loaded with simvastatin induced a cytotoxic effect in MCF7 cells.
31
Nanostructured lipid carriers made of alpha-lipoic acid, ellagic acid and fluvastatin were effective in prostate cancer treatment in vitro.
32
Polymeric nanocapsules made of poly-lactide-co-glycolide (PLGA) polymer loaded with rosuvastatin induced apoptosis in HepG2 cells.
33
Also, simvastatin conjugated to PLGA to form nanoparticles further modified by the incorporation of superparamagnetic iron oxide nanoparticles (SPIONS) giving a hybrid nanocarrier system, was effective against prostate cancer cells.
34
A very promising attempt was made by Wu et al.
35
who developed a nanocarrier system of a cholic acid core and star-shaped polymer consisting of poly(
Liposomes, another form of statin carriers, have been extensively studied during the past few years. It was shown in in vivo experiments that simvastatin delivered in a form of long-circulating liposomes may reduce murine colon carcinoma by both an inhibitory effect on tumour growth and suppressive actions on tumour angiogenesis and inflammation. 38 Other studies have shown that liposomes with simvastatin targeted selectively to cancer cells via monoclonal antibodies conjugated to liposome surfaces are highly effective in vitro in treatment of breast cancers overexpressing HER2 39 and EGFR, 40 although in vivo experiments showed a moderate effect. 40 This result could be due to the dose of simvastatin being too low, or it may suggest that statins should be used in combined therapies rather than as a single anticancer agent. In fact, it was shown that liposomes co-loaded with simvastatin and doxorubicin targeted towards prostate cancer cells overexpressing HER2 may lead to 80% inhibition of tumour growth in an in vivo model. It was also shown that simvastatin and doxorubicin may exert a synergistic, antiangiogenic effect. 41
Combined therapies
Difficulties in statin administration and debates on the application of statins as single agents in different types of cancer therapies contributed to the development of combined therapies.
In our previous review, 1 we listed a few promising in vitro studies of the concomitant use of statins with drugs that are routinely used in cancer therapy. The results of the latest research led us to extend that list (see Table 2).
Promising new anticancer combined therapies that involve statins.

DMXAA: 5,6-dimethylxanthenone-4-acetic acid; VEGF: vascular endothelial growth factor; EGFR: epithermal growth factor receptor; TKIs: tyrosine kinase inhibitors.
For instance, in vitro studies demonstrated a synergistic effect of pentoxifylline, a drug used to treat muscle pain in peripheral artery disease, and simvastatin in breast cancer cells of triple-negative phenotype. 42 Both in vitro and in vivo results confirmed that the combination of atorvastatin and aspirin exerts a stronger inhibitory effect on growth and stimulates apoptosis in prostate cancer cells than either drug alone. 43 Another in vitro study showed that simvastatin, together with the anti-angiogenic agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA), is able to suppress the aggressive phenotype of melanoma cells co-cultured with tumour-associated macrophages under hypoxia-mimicking conditions. 44 Furthermore, in vivo studies on rats after resection of glioblastoma showed 40% tumour volume reduction after oral administration of lovastatin, atorvastatin and pioglitazone, an anti-diabetic drug, 45 although, in the cited study, no survival improvement could be demonstrated. Another common drug used for the treatment of type II diabetes, metformin, when combined with atorvastatin, showed stronger effects on inhibition of cell growth and migration, together with apoptosis induction in vitro and inhibition of tumour growth in vivo. 51 There was also a study showing that simvastatin in conjunction with vemurafenib, a BRAF (B-Raf proto-oncogene) inhibitor used in melanoma treatment, and selumetinib, a BRAF and MEK inhibitor used in lung and colorectal cancer treatment, demonstrates additional growth suppression via inhibition of isoprenoid synthesis. 46
There is also some evidence from retrospective or case-control studies that statins in some types of cancer may act synergistically with anticancer drugs widely used in clinical practice. For instance, patients with metastatic renal cell carcinoma who were statin users demonstrated improved overall survival compared to non-users, but only if they received therapy targeting vascular endothelial growth factor or mammalian target of rapamycin and not when they were treated with interferon-α (IFN-α). 47 Furthermore, it was shown that statins might potentially enhance the effects of tyrosine kinase inhibitors (EGFR-TKIs) and chemotherapy in patients with non-small-cell lung cancer as it correlated with reduced risk of death and longer median progression-free and overall survival. 48 In patients with unresectable pancreatic cancer treated with erlotinib and gemcitabine, additional statin use was associated with stabilisation of the long-term tumour growth. 49 Statins were also shown to enhance the efficacy of venetoclax, an inhibitor of BCL-2 used in the treatment of primary leukaemia and lymphoma cases. 50 The authors of the study pointed to the fact that also in retrospective analyses of three clinical studies of chronic lymphocytic leukaemia, background statin use was associated with an enhanced response to venetoclax. Furthermore, statins in combination with the above-mentioned metformin were associated with reduced all-cause and prostate cancer mortality in a population-based retrospective cohort study. 52 Finally, quite surprisingly it was found that statins may reduce the risk of cardiotoxicity induced by anthracyclines and trastuzumab used in the treatment of HER2-positive breast cancer. 53
However, in patients with advanced cancer and a survival prognosis lower than 2 years, the addition of statins to standard anticancer therapy does not appear to improve overall or progression-free survival. 54 Also, a phase II study showed no increase in cetuximab efficacy in colorectal cancer patients with KRAS mutation when simvastatin was additionally prescribed. 55
Statins and mechanisms of resistance
Extensive studies on the effect of statins on cancer cells have led to additional observations. It was noted that some histological types of cancer are more sensitive to statin treatment than others. This observation led to a search for possible mechanisms of resistance that cancers develop towards statins or other cholesterol-lowering drugs. One such mechanism was observed for prostate cancer. 56 It was shown that in mice with induced prostate tumour, after simvastatin or ezetimibe treatment alone, no changes in tumour growth were noted despite promising in vitro results. The combination of those two drugs even accelerated tumour growth. For ezetimibe, a lowered serum cholesterol level was observed concomitantly with a higher level of tumour cholesterol. Also, induction of low-density lipoprotein (LDL) receptor messenger RNA (mRNA) was observed in tumour cells for ezetimibe alone or in combination with simvastatin. This result may suggest that induction of the LDL receptor may be one of the possible mechanisms of resistance that prostate tumours use to counteract the therapeutic effects of lowering serum cholesterol.
Another mechanism was observed for breast cancer. 57 In the case of breast cancers unresponsive to statin therapy, atorvastatin treatment led to enhanced accumulation of lipids in cytoplasmic lipid droplets (LDs). Dysregulation of lipid uptake and efflux as specific mechanisms associated with differences in LD accumulation was excluded, but upregulation of genes involved in unsaturated fatty acid metabolism, stearoyl-CoA desaturase, cholesterol biosynthesis, and HMGCR was associated with atorvastatin insensitivity. Upregulation of genes involved in cholesterol biosynthesis and HMGCR as a mechanism of cells’ resistance was confirmed and extended also by another study. It was shown 58 that the downregulation of HMGCR expression by siRNA in epithelial and mixed epithelial-mesenchymal cells improved cells’ sensitivity to atorvastatin’s growth inhibitory effect. That effect is mainly associated with blockade of isoprenoid synthesis.
On the other hand, it was also observed that the introduction of statins to ongoing cancer therapy makes it possible to abolish cell resistance to commonly used chemotherapeutics. Increased expression of proteins associated with de novo cholesterol biosynthesis was demonstrated in mammospheres enriched with cancer stem cells, isolated from xenograft tumours developed from estrogen receptor-negative breast cancer patients. 59 Cancer stem cells exhibit resistance to conventional therapy, and their presence correlates with shorter relapse-free survival. However, inhibition of the cholesterol biosynthesis pathway reduced mammosphere formation and pointed to statins as possible therapeutic agents in breast cancer, in combination with chemotherapy.
Apart from that, statins were also reported to be agents that may resensitise HER2+-resistant breast cancer cells to the therapy by inhibition of the mevalonate pathway.
60
It was shown that in HER2+ breast cancer cells resistant to lapatinib alone or in combination with trastuzumab, the activity of the mevalonate pathway is increased. The use of statins or
Effect of statins on epithelial–mesenchymal transition of cells
One of the mechanisms leading to metastases and further development of cancer disease is epithelial–mesenchymal transition (EMT). As a result of this process, epithelial cells lose their polarity and ability to retain cellular adhesion, gain migratory properties and become multipotent mesenchymal stem cells, or cancer stem-like cells. Mesenchymal cells can differentiate into a variety of cell types, giving rise to cancer recurrences.
It was shown that statins may inhibit EMT in different types of cancer. Studies on mechanisms involved in this process have been conducted on, among others, prostate cancer, 61 oesophageal cancer, 62 ovarian cancer 63 and breast cancers, 64 all pointing to statins as agents effectively inhibiting EMT. The inhibitory effect of statins on cancer stem-like cells is largely due to the fact that those cells are highly dependent on the mevalonate pathway. The mevalonate pathway not only affects EMT but also influences remodelling of the cytoskeleton and cell motility, changes cell polarity and modulates mesenchymal–epithelial transition (MET) (reviewed by Likus 65 ). Treatment with statins probably may affect all above-mentioned processes in cancer stem-like cells. Moreover, statins may sensitise treated stem cells to chemotherapeutics such as doxorubicin 64 and, because of that, they seem to be good candidates for adjuvant therapy protecting against metastases. This hypothesis is in agreement with observations from epidemiological studies showing that statins, for example, in breast cancer, reduce cancer-specific mortality rather than the incidence of primary cancer. 12 Moreover, it was shown in vitro that atorvastatin may affect the proliferation of mesenchymal, but not epithelial, breast cancer cells at the metastatic site. In the same study, in an ex vivo three-dimensional (3D) model, atorvastatin was shown to block stimulated emergence of dormant breast cancer cells. In vivo, in two independent models of breast cancer metastasis to liver and to lung, atorvastatin reduced proliferation of the metastatic, but not the primary, tumour cells. 66 Interestingly, in a large panel of cancer cell lines of multiple cancer types, it was shown that enrichment of EMT features in cells is associated with increased sensitivity to statins, which is independent from prenylation of RAS family proteins. 67
Conclusion
The presented cohort studies and meta-analyses show that prolonged statin use may be associated with decreased cancer incidence and/or decreased cancer-specific mortality, depending on tumour type. However, on the basis of results from epidemiological studies, we may only draw general conclusions. Difficulties in statin administration at high doses, which are still unsolved, are the reason why it cannot be stated unequivocally whether statin application as single agents in cancer treatment is justified.
Growing evidence shows that statins exert greater effects when they are administered as nanocarriers, especially targeted towards defined cellular targets. Nevertheless, more advanced research should be performed to obtain a suitable carrier and to determine the dose of statins delivered via a nanocarrier that is effective in inhibition of tumour growth.
Statins appeared to be very promising in combined therapies: they may act synergistically with some types of widely used chemotherapeutics. It should be very carefully investigated which types of therapeutics would be the most suitable for such treatment. Statins were shown to reduce the resistance of cancer cells to other anticancer drugs; however, cells may develop several mechanisms making them statin-resistant. It is very likely that the use of statins as adjuvant drugs in the treatment of metastatic cancers would be particularly beneficial.
In conclusion, it seems that the use of statins in cancer therapy should be considered in the context of combined therapies, especially in terms of their use in the treatment of metastatic cancers, and not as individual therapeutic agents.
Footnotes
Author contributions
L.M. collected and analysed data and wrote the original draft. A.C. and A.F.S. reviewed and edited the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Science Centre, Poland Project No. 016/21/B/NZ7/01070, and The Polpharma Scientific Foundation, Warsaw, Poland (project no 4/XVI/2017).
