Abstract
A key tool for monitoring breast cancer patients under neoadjuvant treatment is the identification of reliable predictive markers. Ki67 has been identified as a prognostic and predictive marker in ER-positive breast cancer. Ninety ER-positive, HER2 negative locally advanced breast cancer patients received letrozole (2.5 mg daily) and cyclophosphamide (50 mg daily) with/without Sorafenib (400 mg/bid daily) for 6 months before undergoing surgery. Ki67 expression and tumor size measured with caliber were determined at baseline, after 30 days of treatment and at the end of treatment. Patients were assigned to a clinical response category according to Response Evaluation Criteria in Solid Tumors, both at 30 days and before surgery and further classified as high-responder and low-responder according to the median variation of Ki67 values between biopsy and 30 days and between biopsy and surgery time. The predictive role of Ki67 and its changes with regard to clinical response and survival was analyzed. No differences in terms of survival outcomes emerged between the arms of treatment, while we observed a higher percentage of women with progression or stable disease in arm with the combination containing Sorafenib (20.5% vs 7.1%, p = 0.06). Clinical complete responders experienced a greater overall variation in Ki67 when compared with partial responders and patients with progressive/stable disease (66.7% vs 30.7%, p = 0.009). High responders showed a better outcome than low responders in terms of both disease-free survival (p = 0.009) and overall survival (p = 0.002). ΔKi67 score evaluated between basal and residual tumor at definitive surgery showed to be highly predictive of clinical complete response, and a potential parameter to be used for predicting disease-free survival and overall survival in luminal breast cancer treated with neoadjuvant endocrine-based therapy.
Introduction
Neoadjuvant systemic therapy (NST) has become a mainstream approach in breast cancer treatment. Aside from helping the achievement of disease local control with breast conserving surgery, NST allows prompt evaluation of tumor response and guides therapy adjustments accordingly. Furthermore, NST allows to test new therapeutic compounds and to monitor the impact of treatment on the biological, molecular, and pathological characteristics of the tumor, thus providing invaluable information on the mechanisms of action of anticancer drugs.1–4 Monitoring the treatment response allows to assess if the cytotoxic treatment is effective in increasing the disease-free survival (DFS) and the overall survival (OS) to provide information on the mechanisms of action of the anticancer drugs and to identify intermediate endpoints of treatment response. 5
Pathological complete response (pCR), Ki67 tumor expression value, and the changes induced by treatment as well as SUV variation on PET-TC have been identified as potential surrogate endpoints of treatment efficacy, that is, observational variables that can replace the true outcome of interest in clinical studies and routine. 5 Moreover, recent studies have indicated that Ki67 and pCR in NST are independent predictors of DFS and OS.6–8
NST in breast cancer was originally limited to locally advanced inoperable disease but has been extended first to operable disease and later to earlier-stage tumors.9,10 Therapeutic strategy strongly depends on molecular classification; ER status is the most successful predictive biomarker for endocrine therapy. A number of clinical trials in the recent years evaluated the efficacy of aromatase inhibitors (AIs) over tamoxifen. In the neoadjuvant setting, AIs have shown to be more effective than tamoxifen, with a response rate between 40% and 60%.11–13 Among the AIs, letrozole (LET) showed an overall response rate of about 80%; 14 additionally, 12 months of LET based-therapy resulted to be more effective in overall response and complete response (CR) than 4 or 8 months’ therapy. 15
In an integrated approach setting, hormonal therapy may cause a reduction in cell proliferation and this may be counter-productive to chemotherapy, which has an effect on high proliferating cells. This “issue” can be bypassed by administration of chemotherapy in a metronomic regimen (low-dose metronomic chemotherapy (LDM)). 16 The combination of LET and metronomic cyclophosphamide (CYC) has already been reported by Bottini et al. 14 in a Phase II study safely conducted on elderly breast cancer patients. In view of the action of LDM on the endothelial vasculature, Bazzola et al. 17 hypothesized a synergism with other anti-angiogenic drugs and designed a Phase II study to address this question, comparing the combination of LET and CYC with LET-CYC plus Sorafenib, a serin-threonine kinase, RAF-1 inhibitor with anti-angiogenic activity, in breast cancer patients. The triplet combination was well tolerated and effective in reducing tumor size, Ki67 and VEGF-A. 17
As these promising results warranted further studies, we conducted a study evaluating the combination of LET, metronomic administration of CYC, and Sorafenib with a focus on clinical response, on the tumor proliferation, and how they may affect the DFS and OS.
Materials and methods
Study design and patients selection
Ninety postmenopausal women (mean age 66.6 ± 8.6) with ER positive, HER2 negative breast cancer were included in this prospective, open-label, single-center, randomized Phase III study. Eligible patients had T2-4, N0-N2, M0 breast cancer, uni-dimensionally measurable by objective examination according to RECIST criteria (Response Evaluation Criteria in Solid Tumors), and performance status 0–2 according to ECOG (Eastern Cooperative Oncology Group). Women were randomly assigned 1:1 to receive LET 2.5 mg daily and metronomic oral Cyc 50 mg daily with (Arm B; n = 45) or without (Arm A, n = 45) Sorafenib 400 mg/bid daily for 6 months before undergoing surgery.
Written consent was provided by each participant. The study was approved by the Val Padana Ethics Committee (Eudract No. 2007-006208-39). The study was prematurely closed due to an unexpected high number of progressions in Arm B (with sorafinib); from the ethical point of view it was decided to interrupt the recruitment.
Proliferation index
Proliferation index was tested with the KI67 expression; Ki67 was evaluated by immunohistochemistry at three different time points. Tissue was obtained from patients from an incisional biopsy performed at presentation, from tru-cut biopsy performed after 30 days of treatment and at definitive surgery. Immunohistochemistry was performed on paraffin-embedded tumor samples; Ki67 staining was performed using standard protocols as described in a previous article. 18 Briefly, an antigen retrieval step was performed by heating a tissue section in a citrate buffer. The primary antibody applied was mouse monoclonal Mib-1 (Dako, Glostrup, Denmark), dilution 1:30, 1 h incubation at RT; biotinylated horse anti-mouse IgG and avidin–biotin–peroxidase complex were applied as a staining method (Vectastatin ABCkit; Vector Laboratories, Inc., Burlingame, CA). A solution containing hydrogen peroxide (0.06% v/v) and diamino-benzidine4 HCL (DAB; 0.05 v/v) was used as chromogen.
Percentage variation of proliferation index has been calculated as follows: ΔKi67 (%) short variation (baseline—30 days) = (Ki67 baseline − Ki67 30 days/Ki67 baseline) × 100; ΔKi67 (%) intermediate variation (30 days—surgery) = (Ki67 30 days − Ki67 surgery/Ki67 30 giorni) × 100; ΔKi67 (%) long variation (baseline-surgery) = (Ki67 baseline − Ki67 surgery/Ki67 baseline) × 100.
Response assessment
Primary tumor size was measured with a caliber by a clinician at three time points: enrollment, after 30 days, and at the end of treatment (before surgery). Early clinical response (eCR)—between baseline and 30 days—and presurgical clinical response—between baseline and surgery—was assessed according to RECIST criteria (Version 1.1). 19 Lesions were scored as follows: CR (disappearance of all target lesions), partial response (PR, decrease of ≥30% in the sum of the longest diameter of target lesions), stable disease (SD, does not meet the criteria for CR, PR or progression disease (PD)), and PD (an increase in tumor size of ≥20% in the sum of the longest diameter of target lesions). Progression-free survival (PFS) and OS were, respectively, defined as the time from the date of surgery to the date of appearance of metastasis or death and as the time from the date of surgery to the date of death by any cause. 20
Statistical analysis
Characteristics of the study population are described using means ± standard deviation or median and range (minimum–maximum values) for continuous variables, depending on the distribution’s shape. Data were tested for normal distribution using the Shapiro–Wilk test. Categorical variables were summarized with absolute frequencies and percentages; cross-tabulations were generated to compare frequency distributions and chi-square or Fisher’s Exact test, when appropriate, were used to assess possible associations.
Analyses were performed to test for differences among Ki67 median values at three different time points (baseline, 60 days, and at surgery) using Friedman’s test for paired data and the post hoc analysis performed by the Wilcoxon test applying the Bonferroni adjustment for multiple comparisons. Differences among percentage variations of Ki67 at three different time points (short, intermediate, and long) were evaluated by Kruskall–Wallis test for independent variables and post hoc analysis with the Mann–Whitney test applying the Bonferroni adjustment for multiple comparisons. The association between clinical response (total, partial, non-responding) with respect to continuous variables (Ki67, Ki67 variation) was assessed by Kruskall–Wallis test. DFS and OS were estimated using the Kaplan–Meyer method and differences between the curves were tested for significance by the Log-rank test. All statistical analyses were performed using the R (the R Foundation for Statistical Computing; Version 3.0.3, library “survival”). A p value less than 0.05 was considered statistically significant.
Results
From 2009 to 2013, 90 women were enrolled onto the trial; 45 were randomly assigned to receive only LET-CYC (Arm A) and 45 were assigned to receive LET-CYC plus Sorafenib (Arm B). The trial was interrupted due to the occurrence of posttreatment progression but all patients completed the planned 6 months of therapy. Patients’ characteristics enrolled into the trial are detailed in Table 1.
Patients’characteristics.
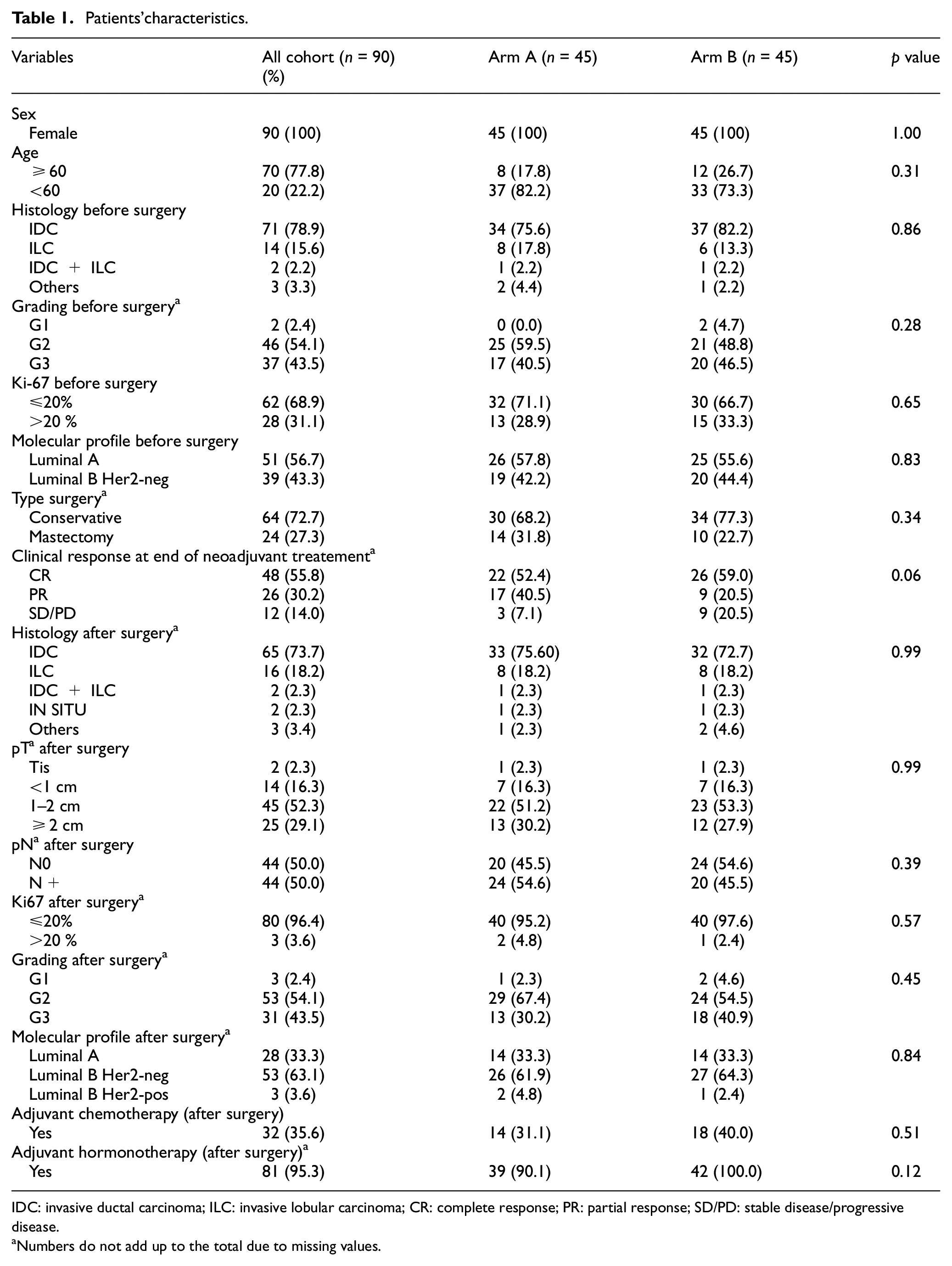
IDC: invasive ductal carcinoma; ILC: invasive lobular carcinoma; CR: complete response; PR: partial response; SD/PD: stable disease/progressive disease.
Numbers do not add up to the total due to missing values.
Treatment response
Data on early clinical response (after 30 days of therapy) were available for 77 of the 90 patients: only one patient registered a CR (1.3%), 22 patients had PR (28.6%), and 54 had either clinical SD or clinical progression (70.1%). The arm of treatment did not significantly influence early clinical response (Pr = 0.71, Fisher’s Exact test).
At the end of treatment assessment, clinical response data was available for 86 patients, 4 patients were missing either basal or posttreatment assessment. None of the 86 patients showed a complete pathological response; however, 55.8% had a complete clinical response (n = 48), 30.2% had partial clinical response (n = 26) and 14.0% had SD or clinical progression (n = 12, Table 1). A greater number of patients in Arm B experienced disease stability or progression (p = 0.06, chi-squared test, Table 1). Even if Sorafenib-treated women had a median age greater than the control arm (69.2 vs 63.8, p = 0.003), age was not associated with early or late clinical response (p = 0.40, one way ANOVA, p = 0.15, Student’s t-test, respectively). Tumor classification at diagnosis (Luminal A or Luminal B type) was not associated with presurgical clinical response (p = 0.94).
Early and presurgical clinical response
Treatment response changes significantly between early (30 days) and presurgical evaluations (p < 0.001, Stuart Maxwell test for paired data). Among the 75 patients for whom data are available, 25 women (33.3%) who had eSD/ePD and 15 with ePR (20%) registered a CR at the end of treatment. In all, 17 patients (22.7%) had an improved response (from eSD/PD to PR), 17 patients (22.7%) maintained clinical response between 30 days and end of treatment evaluation, whereas only one woman (Arm B, 1.3%) worsened her response, going from early PR to stable/progressive disease.
Treatment response rate at 30 days and end of treatment was statistically correlated with the treatment arm (p < 0.001, Stuart Maxwell test for paired data). Clinical response classification between 30 days and end of treatment was not changed for 16.7% of patients in Arm A and 28.2% of patients in Arm B. A greater percentage of patients in Arm A experienced an improvement of clinical response (83.3% vs 69.2%) even if this difference did not reach statistical significance (p = 0.27, Fisher’s Exact test).
Change in clinical response during and after treatment was evaluated in relation to the Ki67 variation. Our population was divided into three groups: no change between early and presurgical response, change to CR, and change to PR; the patient who progressed was excluded from the statistical evaluation. A greater variation of Ki67 between basal and 30 days was observed in the patients who achieved a CR from PR/PD-SD at 30 days (p = 0.625); however, the comparison between the three groups is not statistically significant (p = 0.11, Kruskall–Wallis test). The comparison between the two groups with an experience of tumor change (to CR or to PR) resulted in a difference at the limit of statistical significance (p = 0.05, Mann–Whitney test).
Survival analysis
Survival analysis was performed on 79 women (11 excluded: 4 due to lack of information on the clinical response and 7 due to lack of information on the follow-up). Median follow-up was 55.6 months and eight deaths by any cause occurred.
There were not any significant differences between arms of treatment in terms of DFS (p = 0.84) and of OS (p = 0.74).
Clinical response and survival
There are no significant differences in terms of DFS and OS with regard to early clinical response but survival analysis according to presurgical clinical response was performed and revealed significant differences between groups (p = 0.015 log rank test, Figure 1). Survival at 60 months (5 years) was significantly greater in women with clinical CR than in partial responders and patients with stable or PD: 98% vs 66% of women with PR and 65% of women with stable or progressive disease. Five deaths occurred during the evaluation period and 12 women had progressive disease, 3 of which died, for a total number of events of 17.

Overall survival (OS) and disease-free survival (DFS) according to clinical response (RECIST criteria).
DFS was also evaluated in relation to clinical response: 21 events occurred, 19 patients progressed, and 2 died without prior recurrence. Although no significant differences were registered between clinical response groups (p = 0.10), patients with SD or PD experienced recurrence early compared with patients with CR (Figure 1). Indeed DFS at the 2-year time point was 95% in the CR group and 60% in the SD/PD group.
Proliferation index: correlation with clinical response and survival
Proliferation index values decrease significantly in both arms of treatment (p < 0.001) but no statistically significant differences were observed between the two arms (p = 0.39, linear mixed effects model for repeated measurements (Figure 2)). Overall, the Ki67 values comparison before and after therapy shows that the number of patients with high proliferative index (values of Ki67 > 20%) decreases significantly (28 pre vs 3 post), while the number of patients with Ki67 ≤ 20% increases from 61 to 80 (p < 0.001 chi-square test). Ki67 expression was available at baseline for 90 patients, at 30 days’ time point for 49 patients and at surgery for 83 patients. Ki67 values decreased significantly (p < 0.001 Friedman’s test) between baseline (median value 16.5 (2–70)) and 30 days (median value 6 (0.9–50)) and between baseline and surgery (median value 5 (0–30)) (Figure 3). Reduction between 30 days and surgery was not statistically significant (p = 0.3; Wilcoxon test adjusted for multiple comparisons).
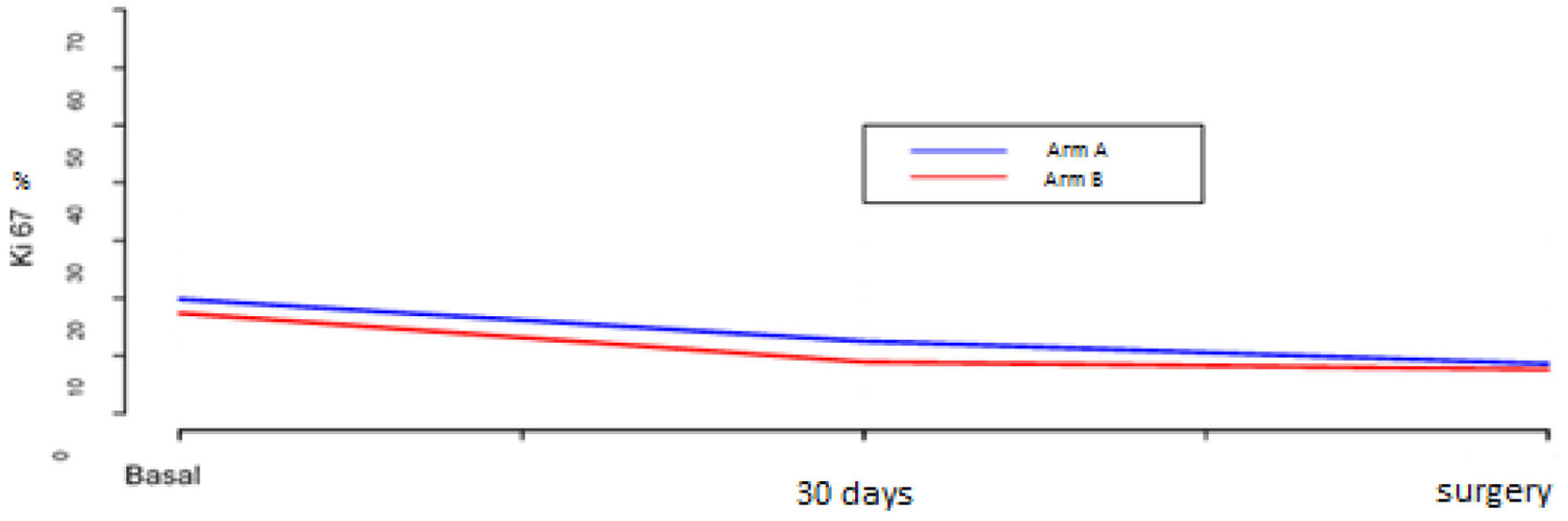
Plot of the Ki67 mean values at different times according to arm of treatment. Arm A received Letrozole + Cyclophosphamide, Arm B receive Letrozole, cyclophosphamide and Sorafenib.

Ki67 values at different time points.
Ki67 values and percentage variations were evaluated in relation to early and presurgical CR. Early response was not associated with proliferation index variation, irrespective of the time of evaluation (baseline, 30 days, and surgery). Conversely, Ki67 values at surgery are significantly lower in clinical responders (CR) in comparison to SD/PD patients (p = 0.008). Therefore, percentage variation of Ki67 between baseline and surgery was evaluated in relation to late clinical response. Patients who had a complete clinical response registered a considerably greater Ki67 variation compared with patients who had SD or PD (66.7% vs 28.0%, p = 0.003, Mann–Whitney test) but no differences were noted between PR and SD/PD (p = 0.09) nor between CR versus PR (p = 0.11).
To evaluate the impact of Ki67 changes on survival, we chose the median percentage of variation of Ki67 between time points as a cutoff to define high versus low responders. For the early variations we chose ΔKi67 50%, for presurgical variations we chose ΔKi67 60%. At first, early variations were explored and the population was divided into early low responders ΔKi67 < cutoff value, n = 24) and early high responders (ΔKi67 ≥ cutoff value, n = 25). The results showed no statistical difference in terms of DFS and OS (p = 0.76, log-rank test both; data not shown). Thereafter, we focused on presurgical clinical response and ΔKi67 (%) long variation (baseline-surgery). In all, 36 patients were classified as low responders (ΔKi67 < cutoff value) and 44 as high responders (ΔKi67 ≥ cut-off value). At 5 years, DFS was significantly longer in the group with the highest variation of Ki67: 92% (95% CI: 77%–97%) in the high responders group versus 60% (95% CI: 41%–75%, p = 0.002 Log Rank test, Figure 4) in the low responders. Similarly in terms of OS, the high response group had a better prognosis in comparison to the low response group (p = 0.009 Log-Rank test, Figure 5), and OS at 5 year was 92% (95% CI: 77%–97%) in the high response group and 60% (95% CI: 41%–75%) in the low response group.

Disease free survival (DFS) according to ΔKi67 between baseline and surgery. Low response (ΔKi67 < 60%) N = 36, high response (ΔKi67 ≥ 60%) N = 44.

Overall survival (OS) according to ΔKi67 between baseline and surgery. Low response (ΔKi67 < 60%) N = 36, high response (ΔKi67 ≥ 60%) N = 44.
Discussion
Monitoring treatment response has become a key factor in managing cancer patients and this is even truer in the neoadjuvant setting. 2 While endocrine therapy represents the most tailored NST for women with ER-positive breast cancer, previous studies have suggested that response rate and survival could be improved in postmenopausal, luminal, breast cancer patients with the concomitant administration of metronomic chemotherapy (CYC).14,16,17 In our study, we explored the efficacy of the LET–CYC combination with or without Sorafenib, a serine-threonine kinase inhibitor that has shown anti-angiogenic activity due to the interaction with VEGFR-2 and PDGFR-β. None of the patients enrolled in the study achieved a pCR. Usual characteristics associated with increased pCR rate are age <40 years, high expression of Ki67, ER-negative, triple-negative subtype, HER2 positive disease, ductal histology, and high nuclear grade tumors; 21 these features are not well represented in our study population. This fact could perhaps explain the low pCR rate. Pathologic CR is not often achievable with neoadjuvant systemic therapy (NST) especially in luminal breast cancer, but it is reported that even a reduction in tumor size affects the clinical response. In our trial, more than half of the enrolled patients had a complete clinical response (55.8%). The experimental combination of Sorafenib plus LET-CYC did not demonstrate superiority in comparison to LET-CYC alone; however, a higher number of non-responders (SD or PD) was identified in the Sorafenib-treated group at the end of treatment, leading to the premature closing of enrollment.
Early assessment of clinical response (eCR, after 30 days of therapy) showed no sufficient correlation with survival endpoints (DFS and OS, data not shown), nor with proliferation index and its percentage variation. Nevertheless, eCR was a useful intermediate tool to determine disease status and treatment efficacy. Statistically, significant differences were noted between the arms of treatment (p < 0.001, Stuart Maxwell Test for paired data). Overall, 53.3% of the evaluable patients with ePR, eSD, or ePD achieved a complete clinical response after treatment, 22.7% improved their response between early and presurgical assessment, 22.7% maintained it, and only one patient (Arm B, 1.3%) worsened the response. The improvement of clinical response was more evident in Arm A compared to Arm B (p = 0.27, Fisher’s Exact Test).
The right Ki67 cutoff is currently still being debated, with values ranging from 12% to 25%. 22 The accepted threshold value according to the latest indications from the St Gallen expert panel is 20%: 6 beyond this value the tumor is considered proliferative and consequently more aggressive. Proliferation index was assessed at three time points, allowing for monitoring throughout the whole treatment period.
Neoadjuvant treatment significantly lowered the quote of proliferating cells affecting Ki67 measurements as only three patients had Ki67 greater than 20% at the end of treatment. In more detail, we found the most significant variation of Ki67 between the first 30 days of treatment and till the end of treatment (p < 0.001 Friedman’s test), whereas there was no significant differences between measurements at 30 days and presurgery (p = 0.3 Wilcoxon test adjusted for multiple comparisons). The establishment of a Ki67 decrease trend in the first treatment period preludes to a clinical improvement over the entire period (change from ePR/ePD/eSD to CR, p = 0.625). Therefore, it seems that a decrease with greater slope in the first 30 days may represent a valid predictive indicator of treatment response, even in the presence of a non-complete eCR. Patients with luminal breast cancer with a relatively low- to mid-risk disease, as in our study, benefit from this early response prediction, as it increases the chances of a conservative surgery.
Percentage decrease of proliferation index is strongly associated with clinical response: the greater the Ki67 variation the greater the probability of clinical CR after treatment. In fact, patients who performed worse in terms of clinical response (PD/SD patients) recorded the lower percentage variation of Ki67. These results suggest that strict monitoring of the proliferation index could help the clinician with firsthand information on therapy efficacy. 23 Furthermore, achieving a clinical CR also fosters longer OS, as demonstrated by the survival rate: after 5 years 98% of women with CR are still alive. Even if these data are not statistically significant, patients with CR tend to relapse or progress later than SD/PD patients: DFS at 5 years is 95% for CR versus 60% in SD/PD. Given the correlations between proliferation index, clinical response to treatment, and survival, in our study we calculated the ΔKi67 between baseline and end of treatment and used the median value to discriminate between high- and low-responder patients. High responders recorded a significantly longer DFS (p = 0.009 Log-Rank test, Figure 3), meaning that a decrease of Ki67 between baseline and end of treatment greater than 60% lengthens the recurrence time. ΔKi67 showed consistency not only as a predictive factor but also as a prognostic marker for HR-positive patients, as confirmed in the literature. 24 High Ki67 value on residual tumor after treatment, rather than at baseline, has a negative prognostic value, as patients record a higher distant metastasis recurrence rate and poorer DFS and OS. 25 In our study, the decrease of the proliferation index is linked to a better prognosis (p = 0.002 Long-rank test, Figure 2), as previously reported by Von Minckwitz et al., 26 with a 5-year survival rate, 32% greater in high responders than in low responders. Proliferation index on residual tumor holds inarguably a prognostic importance, but our results suggest that greater attention should be given to the percentage of reduction of Ki67, rather than focusing merely on a fixed value. In the era of personalized medicine, this would certainly be a more comprehensive and patient-oriented approach.
Therefore, even in the presence of a pathological residue, a reduction in the proliferative index indicates a less aggressive tumor, a more stable response over time with a longer survival.
Conclusion
Neoadjuvant hormone-based treatment has shown clinical and biological activity with a 55.8% complete clinical response overall in our study. Clinical CR correlates with a lower risk of disease progression and a greater OS, and 5-year survival was improved in complete responders. Cellular proliferation measured by Ki67 levels can be safely used as an predictive clinical marker. Moreover, ΔKi67 between baseline and surgery time demonstrated both a predictive and prognostic value as it allows to discriminate between responders and non-responders and correlates with a better outcome in terms of both DFS and OS.
Footnotes
Authors’ contributions
AI drafted and revised the manuscript and monitored data; CP drafted and SPC and MB revised the manuscript; DG conceptualized, supervised, and validated the research; FG performed all statistic analyses; MC provided consultation on procedures; CS, MM, GA, SA, MS, AC, CA, and AB actively enrolled the patients, provided informed consent and collected data; and OB, VC, and MD helped in collecting data missing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Written consent was provided by each participant. The study was approved by the Val Padana Ethics Committee (Eudract Number 2007-006208-39).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
