Abstract
Feline invasive mammary carcinomas are characterized by their high clinical aggressiveness, rare expression of hormone receptors, and pathological resemblance to human breast cancer, especially triple-negative breast cancer (negative to estrogen receptor, progesterone receptor, and epidermal growth factor receptor type 2). Recent gene expression studies of triple-negative breast cancers have highlighted their heterogeneity and the importance of immune responses in their biology and prognostic assessment. Indeed, regulatory T cells may play a crucial role in producing an immune-suppressed microenvironment, notably in triple-negative breast cancers. Feline invasive mammary carcinomas arise spontaneously in immune-competent animals, in which we hypothesized that the immune tumor microenvironment also plays a role. The aims of this study were to determine the quantity and prognostic value of forkhead box protein P3-positive peritumoral and intratumoral regulatory T cells in feline invasive mammary carcinomas, and to identify an immune-suppressed subgroup of triple-negative basal-like feline invasive mammary carcinomas. One hundred and eighty female cats with feline invasive mammary carcinomas, treated by surgery only, with 2-year follow-up post-mastectomy, were included in this study. Forkhead box protein P3, estrogen receptor, progesterone receptor, Ki-67, epidermal growth factor receptor type 2, and cytokeratin 14 expression were assessed by automated immunohistochemistry. Peritumoral regulatory T cells were over 300 times more abundant than intratumoral regulatory T cells in feline invasive mammary carcinomas. Peritumoral and intratumoral regulatory T cells were associated with shorter disease-free interval and overall survival in both triple-negative (ER–, PR–, HER2–, N = 123 out of 180) and luminal (ER+ and/or PR+, N = 57) feline invasive mammary carcinomas. In feline triple-negative basal-like (CK14+) mammary carcinomas, a regulatory T-cell–enriched subgroup was associated with significantly poorer disease-free interval, overall survival, and cancer-specific survival than regulatory T-cell-poor triple-negative basal-like feline invasive mammary carcinomas. High regulatory T-cell numbers had strong and negative prognostic value in feline invasive mammary carcinomas, especially in the triple-negative basal-like subgroup, which might contain a “basal-like immune-suppressed” subtype, as described in triple-negative breast cancer. Cats with feline invasive mammary carcinomas may thus be interesting spontaneous animal models to investigate new strategies of cancer immunotherapy in an immune-suppressed tumor microenvironment.
Keywords
Introduction
Mammary carcinomas that spontaneously arise in female cats are characterized by their high frequency,1,2 clinical aggressiveness,3–8 and poor therapeutic responses. 9 Feline mammary carcinomas (FMCs) are considered to be mostly triple-negative, that is, negative to estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor type 2 (HER2)7,10–16 and basal-like (positive to basal cytokeratins). 17
Cats with FMCs represent one of the rare immunocompetent animal models for human breast cancer. This is fundamental, as the immune tumor microenvironment is crucial in subtyping triple-negative breast cancer and might also serve as a therapeutic target. Triple-negative basal-like breast cancers have been subdivided into basal-like immune-suppressed (BLIS) and basal-like immune-activated (BLIA) carcinomas by gene expression analysis, 18 or equivalent subtypes, such as the C2 (basal-like enriched, immune-suppressed) and C3 (basal-like enriched, adaptive immune response) clusters according to Jézéquel et al.,19,20 or the immunomodulatory subtype according to the study by Lehmann et al.21,22
Among immune cells of the tumor microenvironment, regulatory T cells (Tregs) play a critical role in immune tolerance and deficiency of anti-tumor immunity,23–25 and Tregs identified by FoxP3 immunohistochemistry on paraffin-embedded archival samples have been associated with poor prognosis in breast cancer patients.26–30 The transcription factor FoxP3 (forkhead box protein P3), critical for the development and function of Tregs, is considered to be the most specific marker of CD4+ CD25+ Tregs by immunohistochemistry, in humans31–33 and in cats.34,35
The objectives of our study were (1) to quantify intratumoral and peritumoral FoxP3+ Tregs in FMCs, (2) to assess their relationships with other clinical and pathological features, (3) to investigate their prognostic significance in FMCs, and (4) to better characterize triple-negative basal-like FMCs, that is, to identify a BLIS phenotype.
Materials and methods
Animals and inclusion criteria
This retrospective study included 180 cats with invasive mammary carcinomas, which have been previously described. 36 Female cats were eligible for inclusion if they had an invasive mammary carcinoma, if they were treated solely by surgery, and if follow-up was available for at least 2 years post-mastectomy.
Owners’ written consent was obtained prior to inclusion. Approval was obtained from the local animal welfare and ethics committee “Comité d’Ethique en Recherche clinique et épidémiologique Vétérinaire d’Oniris” (CERVO), of the Nantes Atlantic College of Veterinary Medicine, Food Science and Engineering (Oniris, France).
Recorded epidemiologic and clinical data included age at diagnosis, breed, medical and reproductive history, the location of the mammary carcinoma, and clinical stage according to the modified World Health Organization staging system,37,38 as previously described. 36 Distant metastasis assessment, performed by veterinary practitioners using medical imaging (thoracic radiography and/or abdominal ultrasound), was recorded as M0 (absence of distant metastasis at diagnosis), M1 (presence of distant metastases), or MX (absence of information because owners declined medical imaging for financial reasons). Follow-up methodology is described in Supplementary Methods.
Histopathology
After 1–4 days of formalin fixation, samples were embedded in paraffin blocks and were cut into 3-µm-thick sections for hematoxylin–eosin–saffron (HES) staining. In the case of multifocal (within a given mammary gland) or multicentric (within different mammary glands) invasive mammary carcinomas, the largest one was selected for analysis. Recorded histological data were the histological types, lymphovascular invasion (LVI), histological grade according to the Elston and Ellis grading system for human breast cancer, 39 which has been validated in FMCs, 40 presence/absence of central necrosis or squamous differentiation, and tumor-associated lymphohistiocytic inflammation, as previously described. 36
The pathologic tumor (pT) size was measured on histological sections as the greatest tumor diameter, in millimeters. This was achieved on a single slide for tumors of <25 mm (inner dimension of a histological cassette), or on two adjacent slides for tumors of 25–50 mm in diameter. Tumors greater than 50 mm in diameter could not be precisely measured.
The pathologic nodal (pN) stage was first assessed on HES-stained slides and designated pN+ for the presence of metastatic cells within the draining lymph node, whatever their numbers. Thus, a pN+ nodal stage could correspond to isolated tumor cells (<0.2 mm in diameter), micro-metastases (0.2–2.0 mm in diameter), or macrometastases (>2.0 mm in diameter). When the lymph node was free of metastatic cells on HES-stained slides (N = 24 cases), pancytokeratin immunohistochemistry was performed (4 out of 24 cases were then reclassified as pN+). Absence of nodal metastatic cells was referred to as pN0. The nodal stage was pNX when the lymph node had not been sampled for histopathology.
Immunohistochemistry
Automated immunohistochemistry (Benchmark XT, Ventana Medical Systems, Roche Diagnostics) was used as previously described36,40 and further detailed in Supplementary Methods, to detect p63 (myoepithelial marker that differentiates invasive from in situ breast carcinomas),41,42 cytokeratins (for isolated tumor cell detection in draining lymph nodes), LMO2 (lymphatic endothelial cell marker in cats, used to confirm LVI in uncertain cases), ER-alpha, PR, HER2, the proliferation marker Ki-67, cytokeratin 14 (CK14), and FoxP3.
The thresholds for positivity were 10% for ER and PR as previously reported for canine,43–45 feline, 36 and human 46 mammary carcinomas, 20% for Ki-67, 47 and 15% for CK14. HER2 was scored according to recommendations for breast cancer. 48
The 180 invasive FMCs were classified as luminal (ER+ and/or PR+, any HER2 score) or triple-negative (ER–, PR–, HER2 score 0 to 2+),10,17,49 and triple-negative basal-like carcinomas were defined as ER–, PR–, HER2–, and CK14+.
Nuclear FoxP3 expression in lymphocytes located within mammary carcinomas allowed identifying intratumoral Tregs, which were counted in 10 representative high-power fields (HPFs; at 400× magnification), separately as Tregs in direct contact with tumor cells (ITcontact Tregs, surrounded by carcinoma cells, Figure 1(a)) and Tregs of the intratumoral stroma (ITstroma Tregs, surrounded by collagen, cancer-associated fibroblasts, neocapillaries, and other inflammatory cells, Figure 1(a)). Peritumoral Tregs located at the invasive edge of FMCs (Figure 1(b)) were counted in one representative HPF. Nuclear FoxP3 expression in neoplastic cells was only observed in two cases, concerned a very low proportion of cancer cells, and was not further investigated.

FoxP3 expression in tumor-infiltrating lymphocytes of feline invasive mammary carcinomas: (a) Intratumoral FoxP3+ regulatory T cells were counted separately in direct contact with tumor cells (within circle, ITcontact Tregs) and in the intratumoral stroma (within square, ITstroma Tregs). (b) Peritumoral FoxP3+ regulatory T cells. The dotted line represents the borders of the mammary carcinoma. Peroxidase-DAB revelation system. Original magnification 400×. Bar = 20 µm.
Statistical analyses
Statistical analyses were conducted using the MedCalc® statistical software (Ostend, Belgium). Chi-squared tests (for discontinuous variables) and one-way analyses of variance (for continuous variables) were used to compare the clinicopathologic characteristics of Treg-enriched and Treg-poor FMCs. The Kaplan–Meier method and log-rank tests were used in univariate survival analyses, and Cox proportional hazard models for multivariate survival analyses. The results are reported using the hazard ratio (HR), its 95% confidence interval (95% CI), and the p-value of each covariate. The prognostic cutoffs for Treg numbers were determined by receiver operating characteristic curve analysis calculated for 2-year cancer-specific survival. A p-value of <0.05 was considered significant.
Results
Patient characteristics
The cohort comprised 180 previously described 36 female cats with invasive mammary carcinoma. Briefly, the mean age at diagnosis was 11.1 ± 2.7 years, most of the patients were European (short- or long-haired) domestic cats (154 out of 180, 85%), and most were intact females (N = 112, 62%). Twenty-six FMCs (14%) were multiple. The pT size was ≥20 mm in 95 cases (53%). Fifty-six percent of the patients (101 out of 180) had a positive pN stage (pN+), and 8 out of 180 (4%) had distant metastases (M1) at diagnosis.
Only 49 out of 180 FMCs (27%) were ER positive, 13 out of 180 (7%) were PR positive, and none of the FMCs overexpressed HER2. Fifty-seven FMCs (32%) were luminal, of which 8 were luminal-A (Ki-67 index of <20%), and 49 were luminal-B (Ki-67 ≥ 20%), and 123 out of 180 FMCs (68%) were triple-negative, of which 93 (76%) were triple-negative basal-like (CK14+) FMCs.
Numbers of FoxP3+ Tregs
The mean ITcontact Treg numbers within FMCs were 6 ± 11/mm2 (median: 1.3; range: 0–100). Most of the carcinomas (148 out of 180, 82%) contained at least 1 ITcontact Treg. At a threshold of ≥2 ITcontact Tregs/mm2, 81 FMCs (45%) were considered rich in ITcontact Tregs, including 31 out of 57 luminal FMCs (54%), and 50 out of 123 triple-negative FMCs (41%).
The mean ITstroma Treg numbers within FMCs were 7 ± 9/mm2 (median: 3.6; range: 0–68). Most of the carcinomas (165 out of 180, 92%) were infiltrated with ITstroma Tregs. At a threshold of ≥6 ITstroma Tregs/mm2, 64 FMCs (36%) were considered rich in ITstroma Tregs, including 26 out of 57 luminal FMCs (47%) and 38 out of 123 triple-negative FMCs (31%).
The mean peritumoral Treg numbers around FMCs were 1064 ± 800/mm2 (median: 926; range: 0–4307). Most of the carcinomas (175 out of 180, 97.2%) contained peritumoral Tregs. At a threshold of ≥575/mm2, 126 FMCs (70%) were considered rich in peritumoral Tregs, including 43 out of 57 luminal FMCs (75%), and 83 out of 123 triple-negative FMCs (67%). Overall, peritumoral Tregs were 344 times more numerous than intratumoral Tregs in the FMCs studied.
Associations between Treg numbers and clinicopathological parameters
High numbers of intratumoral Tregs were associated with tumor-associated inflammation and squamous differentiation (Table 1). FMCs with squamous differentiation contained in average 8 ± 14 ITcontact Tregs/mm2 and 9 ± 12 ITstroma Tregs/mm2, whereas FMCs without squamous differentiation contained on average only 4 ± 7 ITcontact Tregs/mm2 and 5 ± 6 ITstroma Tregs/mm2. There was also a positive association between ITstroma Tregs and LVI (Table 1).
Associations between Treg numbers and clinicopathological features of the 180 feline mammary carcinomas.
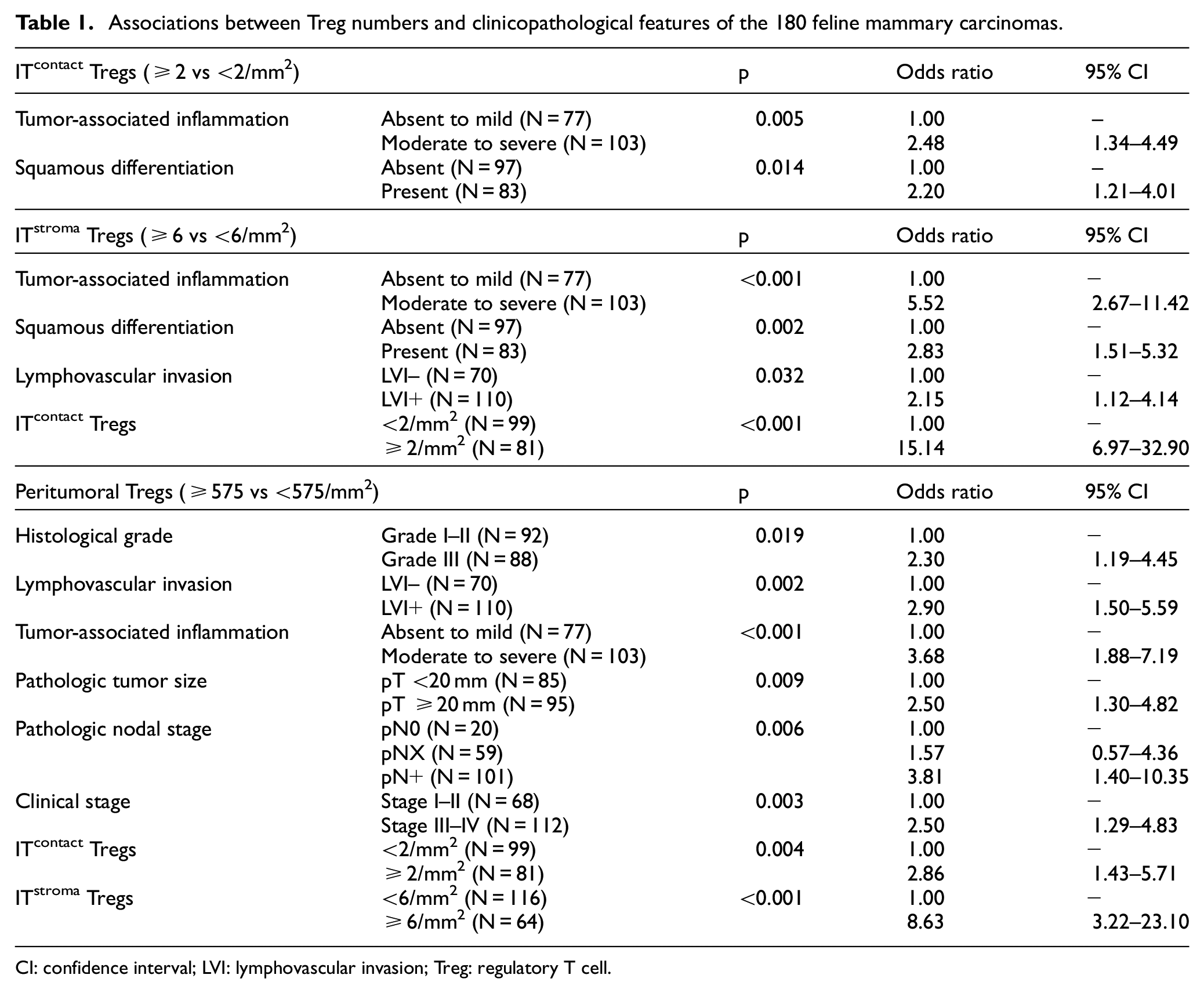
CI: confidence interval; LVI: lymphovascular invasion; Treg: regulatory T cell.
High numbers of peritumoral Tregs were positively correlated with (1) the histological grade (Table 1); Grade I–II FMCs were surrounded by 906 ± 719 peritumoral Tregs/mm2, compared to 1237 ± 848 around Grade III FMCs; (2) LVI; LVI+ FMCs were surrounded by 1193 ± 805 peritumoral Tregs/mm2, compared to 877 ± 748 around LVI– FMCs; (3) tumor-associated inflammation; FMCs with moderate to severe tumor-associated inflammation had 1237 ± 834 peritumoral Tregs/mm2, compared to only 834 ± 691 around FMCs with absent to mild inflammation; (4) the pT size; FMCs ≥ 20 mm in diameter had 1179 ± 791 peritumoral Tregs/mm2, compared to 935 ± 791 around smaller FMCs; (5) the pN stage; pN+ FMCs were surrounded by 1222 ± 877 peritumoral Tregs/mm2, compared to 933 ± 656 around pNX FMCs, and 782 ± 680 around pN0 FMCs; and (6) the clinical stages; there were 863 ± 647 peritumoral Tregs/mm2 around Stage I–II FMCs, versus 1193 ± 863 around Stage III–IV FMCs.
Tregs in different localizations were also correlated with each other (Table 1 for Tregs considered as categorical variables). There was a positive correlation between peritumoral Tregs and ITcontact Tregs (R2 = 0.031, p = 0.0179), between peritumoral Tregs and ITstroma Tregs (R2 = 0.087, p < 0.001), and between ITcontact and ITstroma Tregs (R2 = 0.262, p < 0.0001).
Prognostic value of ITcontact Tregs
By multivariate survival analysis, high numbers of ITcontact Tregs were associated with a shorter disease-free interval (HR = 1.45, p = 0.049) and overall survival (HR = 1.41, p = 0.035), independently of tumor size and distant metastasis, and a shorter cancer-specific survival (HR = 1.44, p = 0.049) with pT, distant metastasis and PR as the covariates (Table 2).
Prognostic value of intratumoral Tregs in direct contact with tumor cells (ITcontact Tregs).
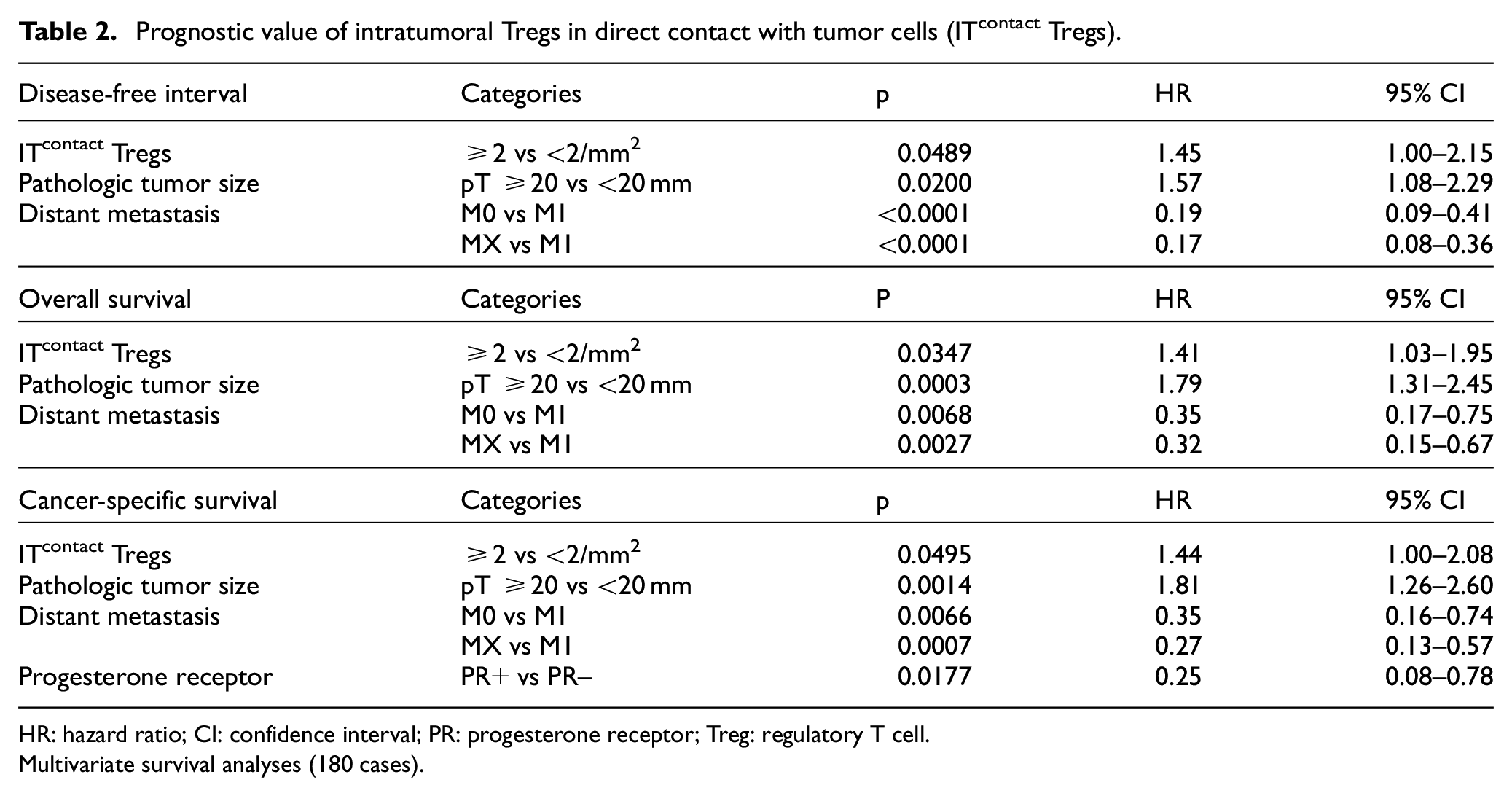
HR: hazard ratio; CI: confidence interval; PR: progesterone receptor; Treg: regulatory T cell.
Multivariate survival analyses (180 cases).
The prognostic value of ITcontact Tregs was not observed in luminal FMCs, but was significant in triple-negative FMCs (Table 3).
Treg prognostic value in luminal and triple-negative FMCs.

FMC: feline invasive mammary carcinoma; NS: not significant; HR: hazard ratio; CI: confidence interval.
Univariate survival analyses; Treg: regulatory T cell.
Prognostic value of ITstroma Tregs
By multivariate survival analysis, ITstroma Tregs were associated with a shorter disease-free interval (HR = 1.49, p = 0.045), overall survival (HR = 1.42, p = 0.031), and specific survival (HR = 1.45, p = 0.044), independently of tumor size and distant metastasis (Table 4).
Prognostic value of Tregs of the intratumoral stroma (ITstroma Tregs).

HR: hazard ratio; CI: confidence interval; Treg: regulatory T cell.
Multivariate survival analyses (180 cases).
ITstroma Tregs were also associated with an unfavorable outcome in the luminal subcohort, however not in triple-negative FMCs (Table 3).
Prognostic value of peritumoral Tregs
High numbers of peritumoral Tregs were associated with an unfavorable outcome in the 180 cats, in terms of disease-free interval (HR = 1.77, 95% CI: 1.22–2.58; p = 0.005; Figure 2(a)), overall survival (HR = 1.82, 95% CI: 1.33–2.48; p = 0.0004; Figure 2(b)), and cancer-specific survival (HR = 1.90, 95% CI: 1.33–2.72; p = 0.001; Figure 2(c)).

Kaplan–Meier survival curves of 180 cats according to peritumoral FoxP3+ regulatory T cells in their invasive mammary carcinomas: (a) disease-free interval, (b) overall survival, and (c) cancer-specific survival.
By multivariate survival analysis, peritumoral Tregs were associated with a shorter disease-free interval (HR = 1.71, p = 0.013) independently of pN (Table 5), poorer overall survival (HR = 1.58, p = 0.013) independently of pT, pN, and distant metastasis, and shorter specific survival (HR = 1.57, p = 0.034) after adjustment for pT, pN, M, and PR (Table 5).
Prognostic value of peritumoral Tregs.

HR: hazard ratio; CI: confidence interval; PR: progesterone receptor; Treg: regulatory T cell.
Multivariate survival analyses (180 cases).
Peritumoral Treg prognostic value was confirmed in both luminal and triple-negative FMCs (Table 3).
Peritumoral and intratumoral Tregs in triple-negative basal-like (TNBL) FMCs
Among the 93 TNBL FMCs, 39 (42%) were rich in ITcontact Tregs, 32 (34%) were rich in ITstroma Tregs, and 63 (68%) were rich in peritumoral Tregs. In TNBL FMCs, ITstroma Tregs were not significantly associated with outcomes, whereas ITcontact Tregs were associated with shorter disease-free interval and poorer overall survival, independently of clinical stages (Table 6), and poorer cancer-specific survival after adjustment for pT, pN, and M (Table 6, Model 1).
Treg prognostic value in triple-negative basal-like FMCs.

FMC: feline invasive mammary carcinomas; HR: hazard ratio; CI: confidence interval.
Multivariate survival analyses (93 cases).
Peritumoral Tregs surrounding TNBL FMCs were also associated with shorter specific survival by multivariate analysis, independently of distant metastasis (Table 6, Model 2), indicating that local immune suppression played a significant role in TNBL FMCs.
Discussion
The first objective of our study was to assess the numbers of FMC-associated Tregs. In this study, 148 FMCs (82%) contained at least 1 ITcontact Treg, and 165 FMCs (92%) contained at least 1 ITstroma Treg; in human breast cancer, Liu et al. 50 reported that 45% (1475 out of 3277) of breast cancers contained at least 1 ITcontact Treg, and 75% (2458 out of 3277) contained at least 1 ITstroma Treg, identified by FoxP3 positivity: there are more feline than human mammary carcinomas that contain tumor-infiltrating Tregs.
The mean intratumoral Treg numbers within FMCs were 6 ± 11 ITcontact Tregs/mm2, and 7 ± 9 ITstroma Tregs/mm2, for a mean total of 12 ± 18 intratumoral Tregs/mm2. By comparison, Sun et al. reported a mean density of 72 ITcontact Tregs/mm2 in 208 breast cancers: 51 ITcontact Tregs appear more numerous in human than in feline mammary cancers. Expressed per HPF (400×), there were 1.7 ± 3.3 ITcontact Tregs/HPF and 2.1 ± 2.9 ITstroma Tregs/HPF for a mean total of 3.9 ± 2.9 intratumoral Tregs/HPF within FMCs. By comparison in breast cancer, intratumoral Treg numbers are in the range of 5.552 to 13.753 per HPF: human breast cancers contain more intratumoral Tregs than FMCs.
However, the mean Treg numbers around FMCs were 1064 ± 800/mm2, corresponding to 318 ± 241 peritumoral Tregs/HPF. By comparison in breast cancer, peritumoral Treg numbers are in the range of 6.454 to 43.353 per HPF: peritumoral Tregs are much more abundant in FMCs than in breast cancers.
In this study, the ratio of intratumoral to peritumoral Tregs was 1:344, compared to 1:1.754 and 1:3.253 in breast cancers: in the cat as in human breast cancer, peritumoral Tregs are more abundant than intratumoral Tregs, but FMCs contain much more peritumoral Tregs than intratumoral Tregs.
The second objective of this study was to correlate Treg numbers with the clinicopathological characteristics of FMCs. Higher peritumoral Treg infiltration was associated with larger pT size, as reported for intratumoral Treg numbers in breast cancer, 27 and with nodal metastasis, as described in breast cancer for intratumoral Tregs.27,50,55 Thus, in both feline and human mammary carcinomas, increasing numbers of Tregs were associated with advanced clinical stage.50,51 In this study, peritumoral Tregs around FMCs were positively associated with a higher histological grade, as reported in breast cancer, for intratumoral50,51,53–58 and peritumoral Tregs.54,56 Finally, in FMCs, Treg numbers were positively correlated with tumor-associated inflammation, visible on HES-stained sections, which was associated with poor prognosis in cats of the present study and in the veterinary literature. 59 Taking into account the fact that larger and/or higher stage carcinomas contained higher Treg numbers, our results suggest that tumor enlargement in cats is accompanied by an increase in Treg-enriched inflammation. This would suggest that larger FMCs were diagnosed in the “immune escape” phase of cancer immunoediting,60–62 in which local immune suppression surpasses antitumoral immunity.
The third objective of this study was to assess Treg prognostic significance in FMCs. Intratumoral Tregs were associated with a worse outcome in terms of disease-free interval, overall survival and cancer-specific survival by multivariate survival analyses in the FMCs studied. ITstroma Tregs were similarly associated with unfavorable outcome in the subcohort of luminal FMCs. In human breast cancer, the presence of intratumoral Tregs, especially in luminal breast cancers, is also associated with poorer prognosis.53,55,56,63
However, peritumoral Tregs had a stronger prognostic value than intratumoral Tregs in cats of this study. Peritumoral Tregs were associated with shorter disease-free interval, overall survival, and cancer-specific survival by multivariate analyses, including in luminal FMCs considered alone, as reported in breast cancer, and in ER+ breast cancer in particular. 56
Specifically, in triple-negative FMCs, our results showed that both peritumoral and ITcontact Tregs were associated with poorer disease-free interval and cancer-specific survival, whereas in human breast cancer, higher numbers of FoxP3+ Tregs have been associated with a prolonged relapse-free interval 64 and better overall survival. 65 Two hypotheses might contribute to explain this discrepancy. First, women with triple-negative breast cancer receive chemotherapy, contrary to the present cats, and Tregs in breast cancer have been associated with better responses to neoadjuvant chemotherapy.66,67 Second, the immune microenvironment of triple-negative mammary carcinomas might not be identical between queens and women. In cats, there are no screening programs: all mammary carcinomas were diagnosed because they were symptomatic (visible or palpable), thus at a late stage. Later stages were associated with higher Treg numbers, suggesting that most FMCs might be diagnosed in the “immune escape” phase of cancer immunoediting. In women, breast cancer screening programs allow for early detection, and it is likely that most breast cancers are diagnosed in the “equilibrium” phase of cancer immunoediting, as high numbers of Tregs have been associated with high numbers of CD8+ cytotoxic T cells, 64 which have favorable prognostic influence in breast cancer.68–70 We attempted to identify CD8+ cytotoxic T cells in FMCs, but the commercially available monoclonal antibodies that we tested (clones vpg9, C8/1779R, and SP57) either do not cross-react with feline CD8, or do not work on formalin-fixed, paraffin-embedded tissues.
The last objective of this study was to investigate the prognostic significance of Tregs in the subgroup of TNBL FMCs, because TNBL breast cancers have been subdivided into BLIS and BLIA carcinomas or equivalent subtypes by gene expression analysis.18–22 We found that TNBL FMCs enriched in either peritumoral or ITcontact Tregs were associated with poorer prognosis, a finding in agreement with the shorter disease-free survival and cancer-specific survival reported for BLIS breast cancers compared to BLIA breast cancers. 18 In this study, most TNBL FMCs (63 out of 93, 68%) were enriched in peritumoral Tregs: most TNBL FMCs seem to be diagnosed in the “immune escape” phase of cancer immunoediting, and these carcinomas might correspond to BLIS breast cancers, in which the immune microenvironment is protumoral, and in which immunosuppressive Tregs may play a major role.
Conclusion
FoxP3+ Tregs were abundant in feline invasive mammary carcinomas, especially in peritumoral areas, were associated with advanced stage at diagnosis, and had negative prognostic value, especially in the triple-negative basal-like subgroup, which may contain a Treg-enriched “BLIS” subtype. Female cats with FMCs may thus be interesting spontaneous animal models to investigate new strategies of cancer immunotherapy that target Tregs in an immune-suppressed tumor microenvironment.
Supplemental Material
Supplementary_methods_1 – Supplemental material for Identification of an immune-suppressed subtype of feline triple-negative basal-like invasive mammary carcinomas, spontaneous models of breast cancer
Supplemental material, Supplementary_methods_1 for Identification of an immune-suppressed subtype of feline triple-negative basal-like invasive mammary carcinomas, spontaneous models of breast cancer by Elie Dagher, Laura Simbault, Jérôme Abadie, Delphine Loussouarn, Mario Campone and Frédérique Nguyen in Tumor Biology
Footnotes
Acknowledgements
The authors thank Dr Mélanie Pohu, Dr Floriane Morio, and Dr Clotilde de Brito, who helped in collecting the clinical and follow-up data, as well as the veterinary pathologists Dr Jean-Loïc Le Net, Dr Virginie Théau, Dr Pierre Lagourette, Dr Olivier Albaric, and Dr Sophie Labrut who performed the initial diagnoses of FMCs, and the technicians in histopathology, Mr Bernard Fernandez, Mrs Florence Lezin, and Mrs Catherine Guéreaud who made the slides. The authors are very grateful to Prof. Dominique Fanuel (DVM), for critical review of this work. Finally, the authors thank the referring veterinarians and the owners of the cats included in this study, who gave us the clinical and follow-up data.
Author contributions
Conceptualization, project administration, and supervision: J.A., M.C., and F.N. Format analysis, investigation, methodology: E.D., L.S., D.L., and F.N. Funding acquisition: J.A., M.C., and F.N. Writing, original draft preparation: E.D. and F.N. Writing, review, and editing: L.S., J.A., D.L., M.C., and F.N.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the French National Cancer Institute (INCa, Institut National du Cancer) with a grant for translational research (grant number INCa-DHOS 2010) (M.C.); by a grant for PhD students from the Ministry of Education and Higher Education of Lebanon (E.D.); and by Roche Diagnostics GmbH, Germany, which provided financial and technical support for the immunohistochemical characterization of the carcinomas. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Availability of data and materials
The datasets used and analyzed during this study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
