Abstract
Type 1 collagen is an important part of the extracellular matrix and changes in its metabolism and distribution are essential in breast cancer induction and progression. Serum concentrations of type 1 collagen synthesis (aminoterminal propeptide (PINP)) and degradation markers (carboxyterminal telopeptide (ICTP)) have previously been studied in early and metastatic breast cancer, but no data are available on specific breast cancer subtypes. We assayed 662 preoperative serum samples for PINP and ICTP and 109 postoperative serum samples for ICTP. The results were linked to prospectively collected clinical data and the cases were divided into breast cancer subtypes for survival analyses. The concentrations of both pre- and postoperative ICTP serum levels increased linearly from ductal in situ carcinoma to stage I–II tumors, stage III tumors, and finally to those with concomitant primary metastases (preoperative ICTP, p = 0.009; postoperative ICTP, p = 0.016). High-preoperative ICTP levels were associated with better breast cancer-specific survival in connection with luminal-B-like (HER2-negative) tumors (p = 0.017), which was confirmed in Cox regression analysis (relative risk = 3.127; 95% confidence interval = 1.081–9.049, p = 0.035), when T-class (relative risk = 4.049; 95% confidence interval = 1.263–12.981; p = 0.019) and nodal status (relative risk = 3.896; 95% confidence interval = 1.088–13.959; p = 0.037) were included in the analysis. In patients with triple-negative breast cancer, a high-preoperative ICTP level was a significant predictor of local relapse-free survival in univariate (p = 0.0020) and multivariate analyses (relative risk = 13.04; 95% confidence interval = 1.354–125.5; p = 0.026; for T-class, relative risk = 2.128 and 95% confidence interval = 0.297–15.23; p = 0.452; for N-class, relative risk = 0.332 and 95% confidence interval = 0.033–3.307; p = 0.347). A preoperatively elevated serum ICTP level appears to be an important marker of better prognosis in triple-negative breast cancer and luminal-B-like (HER2-negative) subtypes.
Introduction
Breast cancer is the most common cancer among women worldwide, with 90% 5-year overall survival (OS) in Finland, for instance.1,2 Because of the increasing rates of morbidity and mortality associated with this disease, patient-tailored therapy strategies, identification of new prognostic markers, therapeutic targets, and new therapeutic approaches are still needed.
The extracellular matrix (ECM) of the tumor microenvironment (TME) controls various aspects of tumor-cell behavior such as proliferation, apoptosis, migration, and invasion. 3 Type I collagen is the most abundant ECM protein, playing an essential role in maintaining tissue integrity. This protein is able to interact with several cell-surface receptors and it regulates intracellular signaling in pathological conditions. It downregulates cell proliferation, induces apoptosis, promotes survival, and protects cancer cells against chemotherapy. 4 Breast cancer is characterized by a dense reactive stroma associated with extensive collagen deposits. 5 Alterations in the structural organization of type I collagen occur during the first phases of breast cancer development, promoting local invasion. 6 Changes in the ECM in breast cancer tissue can be observed by using serum markers of type 1 collagen metabolism. 7 Two markers used for this purpose are the aminoterminal propeptide of type I procollagen (PINP), which reflects human type I collagen synthesis and the crosslinked carboxyterminal telopeptide of type 1 collagen (ICTP), which reflects type I collagen degradation. 8 Serum concentrations of both ICTP and PINP have previously been shown to be increased in samples collected from breast cancer patients.7,9,10 PINP and ICTP are widely studied markers of bone turnover and their clinical use is wide, being investigated in non-malignant diseases such as osteoporosis and rheumatoid arthritis, and malignant conditions such as metastatic breast and prostate cancer.9,11–14
Breast cancers can be divided into five intrinsic subtypes. 15 Surrogate characterization of breast cancer intrinsic subtypes following the European Society for Medical Oncology (ESMO) Clinical Practice Guidelines results in the following five molecular subtypes: (1) luminal-A-like (estrogen- and progesterone-receptor-positive, HER2-negative, low proliferation); (2) luminal-B-like, HER2-negative (estrogen-receptor-positive, HER2-negative and either high proliferation or progesterone-receptor-negative); (3) luminal-B-like, HER2 positive (estrogen-receptor-positive, HER2-positive); (4) HER2 overexpression (estrogen- or progesterone-receptor-negative, HER2-positive); and (5) triple-negative (estrogen- or progesterone-receptor-negative, HER2-negative). 16 Within these categories, there are many heterogeneous groups. Alternatively to immunohistochemical surrogates, the exact subtype can be determined by gene expression profiling, which, however, is much more resource-consuming.
An important aspect of treating breast cancer is detecting the tumors that will locally relapse or metastasize despite treatment and early diagnosis. As breast cancer subtypes differ in their tendency to develop metastases, earlier evidence encourages examination of how changes in the ECM differ among subtypes. Reflecting these changes, a new clinical tool could be use of biological serum markers such as PINP and ICTP, levels of which have been shown to be elevated in patients with aggressive breast cancer. 9
The purpose of this study was to investigate changes of type I collagen metabolism in the connective tissue of breast cancer patients and assess them as potential prognostic factors of breast cancer subtypes. For the first time, these type I collagen metabolism markers were linked to breast cancer subtypes in a prospective setting.
Materials and methods
Patients
The study population consisted of 662 breast cancer patients who were treated at Oulu University Hospital in 2003–2013 (Table 1). The patient data were prospectively collected from the archives of Oulu University Hospital. Follow-up time was defined as being from the day of operation to the last day of updating the data (15 July 2017). Median follow-up time was 72 months. Patients and tumors were classed using TNM classification and histopathological classification according to the World Health Organization (WHO) (Table 2).17,18
Patient characteristics.

DCIS: ductal carcinoma in situ; PR: progesterone receptor; ER: estrogen receptor.
Treatments.

Immunohistochemistry
Subtypes were confirmed by using immunohistochemistry (IHC) on surgically removed tumors at the Department of Pathology, Oulu University Hospital. Immunohistochemical scoring was recorded in connection with postoperative pathoanatomical diagnosis. Tumors were paraffin-embedded, cut, and stained with hematoxylin and eosin for histological analysis. Expression levels of nuclear estrogen and progesterone receptors (ERs and PRs) and Ki-67 were analyzed as described previously by Karihtala et al. 19 Tumor samples expressing nuclear ER or PR in more than 9% of invasive tumor cells were considered to be steroid receptor-positive prior to 2010. The clinical standard for defining ER and PR receptor status by IHC changed at the Department of Pathology around 2010. Tumor samples obtained in 2010 and later on and expressing nuclear ER or PR receptors in more than 1% of invasive tumor cells were considered receptor-positive. Eight (1.2%) of the surgically removed tumors had invasive tumor cells expressing low-grade (1%–9%) nuclear ER. Tumor cells in the triple-negative breast cancer (TNBC) group did not express any ER or PR positivity according to the definition stated above. If a specimen expressed a membranous HER2-positive result in IHC (1+ to 3+ on a scale of 0 to 3+), HER2 gene amplification status was determined by using chromogenic in situ hybridization. Specimens with six or more gene copies of HER2 in the cells were considered HER2-positive. 20 Of the 662 cases, 41 (6.2%) were classified in the ductal carcinoma in situ (DCIS) subgroup and 621 patients (93.8%) were diagnosed with invasive breast cancer. Of 621 patients, 612 patients (92.4%) were diagnosed with invasive breast cancer with no primary metastases and 9 patients (1.4%) had radiologically confirmed metastases at the time of diagnosis (Table 1).
Clinicopathological characteristics
Clinicopathological breast cancer surrogate definitions of intrinsic subtypes followed ESMO Guidelines. 21 Luminal-A-like carcinomas showed ER and PR expression, Ki-67 <15%, and no HER2 overexpression (n = 287, 43.4%). Luminal-B-like (HER2-negative) carcinomas were ER-positive and HER2-negative, and, in addition, showed either Ki-67 >15% or were PR-negative (n = 203, 30.7%). Luminal-B-like (HER2-positive) tumors expressed ER and overexpressed HER2 (n = 35, 5.3%). Triple-negative breast carcinomas were defined as tumors with negative ER, PR, and HER2 expression (n = 68, 10.3%). HER2-positive (non-luminal) cases showed HER2 overexpression without ER or PR positivity (n = 26, 3.9%). Two cases (0.3%) could not be included in any of the above subgroups according to the stated characteristics (Table 1).
Serum samples
Preoperative serum samples were collected from all study participants on the day of their operation or the day before, and postoperative serum samples were collected during follow-up visits at Oulu University Hospital. The timeframe for postoperative samples ranged from 2 days up to 1430 days after surgery. The median time between pre- and postoperative samples was 40.5 days. The samples were stored at ‒20°C until use. All 662 preoperative chemiluminescence assays (CLIAs) succeeded, resulting in defined PINP concentrations. With preoperative ICTP assays, the success rate was a little lower, that is, a defined concentration in 644 (97.3 %) serum samples. Of the 662 preoperative samples, 41 (6.4%) showed an increased ICTP serum concentration in comparison with the established reference values of 2.1–5.6 μg/L for women (over the age of 18 years). In these cases, the postoperative ICTP concentration was defined according to that in the first follow-up sample. We randomly selected 68 controls for postoperative ICTP analysis among the sera that did not show an increased ICTP concentration in the respective preoperative serum sample. Assay of all 109 postoperative serum samples resulted in a defined ICTP concentration. The median preoperative concentration of PINP was 40.21 ng/mL and pre- and postoperative ICTP concentrations were 4.471 and 4.749 μg/L, respectively. Invasive cases with no distant metastases at the time of diagnosis were included. In the analysis performed to compare serum ICTP levels at different stages of breast cancer, we included DCIS cases as well as those with distant metastases at the time of diagnosis.
PINP automated assay
PINP concentrations in serum samples were assessed by using the IDS-iSYS Intact PINP assay Multi-Discipline Automated System analyzer (IDS iSYS Intact PINP, Immunodiagnostics Systems, Boldon, UK). It is an automated assay based on CLIA technology and designed for quantitative determination of intact PINP in human serum. The method has been previously described in detail. 22 Serum samples of 20 μL were used in each analysis that we carefully conducted according to the manufacturer’s instructions. The CLIA involves the use of two monoclonal antibodies, a biotinylated anti-PINP monoclonal antibody and an acridinium-labeled monoclonal antibody. These are combined with streptavidin-labeled magnetic particles in assay buffer. Light is emitted by the acridinium label which is directly proportional to the concentration of intact PINP in the original sample. For calculating the concentrations of intact PINP a four-parameter logistic curve is used. The calibrators are measured in triplicate and the controls in duplicate. 22 PINP automated assays were performed in February 2017 and May 2017.
ICTP radioimmunoassay
Serum ICTP concentrations were measured by using quantitative radioimmunoassays (RIAs)—commercially available kits (UniQ® ICTP RIA, Orion Diagnostica, Espoo, Finland). The assays were conducted in the laboratory of the Department of Clinical Chemistry (Oulu University Hospital). The method is based on the competitive RIA technique using I125-labeled human ICTP antigens (tracer), non-labeled human ICTP antigens (sample) and polyclonal rabbit antibodies. Both labeled and non-labeled ICTP antigens compete for binding to the antibody (antiserum), which has a limited amount of binding sites. After separating the remaining free antigen, the amount of labeled antigen is inversely proportional to the amount of actual ICTP in the sample. The principals of this assay have been described previously by Risteli et al. 23 Duplicate serum samples of 100 μL were used in this assay, which was conducted according to the manufacturer’s instructions. The radioactivity of each sample is measured by using a gamma counter and the results determined via use of the calibration curve created in each assay run. ICTP RIAs of preoperative serum samples were performed in February 2017. The postoperative ICTP RIAs were performed in May 2017.
Statistical analysis
Statistical analysis was carried out by using IBM SPSS Statistics software, v. 23.0.0.0 (IBM Corporation, Armonk, NY, USA). The Mann–Whitney U test and the Kruskal–Wallis test were used to assess the significance of associations. The relationships between PINP and ICTP serum concentrations were assessed by using Spearman’s correlation coefficient. Survival was analyzed by using Kaplan–Meier curves and the log-rank test. Median serum levels were used as cut-off values in survival analysis and patients with distant metastases at the time of diagnosis were excluded from these analyses. Disease-free survival (DFS) was calculated from the date of diagnosis to the date of the first confirmed relapse, either local or distant. Relapse-free survival (RFS) was defined as the time from diagnosis to relapse in resected breast, chest wall, or in the ipsilateral axilla. Breast cancer-specific survival (BCSS) was calculated from the date of diagnosis to the time of death due to breast cancer. For multivariate analyses, we used Cox multivariate regression analysis (co-variates T-class and N-class). Probability values less than 0.05 were considered significant.
Ethics approval
The study was approved by The Regional Ethics Committee of the Northern Ostrobothnia Hospital District (123/2016). The principles of the Declaration of Helsinki were followed in this study.
Results
ICTP and tumor invasiveness
Patient characteristics are described in Table 1. Most of the patients were diagnosed with ductal carcinoma (69.3%) and T1 tumors (64.3%), and only 37.7% were node-positive at the time of diagnosis. ER status was positive in most of the tumors (84.6%). Ten percent of the patients were diagnosed with HER2-positive disease. The serum concentrations of both pre- and postoperative ICTP increased linearly from cases of in situ carcinoma to those patients with metastases at the time of diagnosis (Figure 1). Preoperative mean ICTP concentrations rose as follows: stage 0, 3.43 μg/L (range = 2.21–7.56 μg/L); stages I–II, 3.81 μg/L (range = 1.55–25.4 μg/L); stage III, 4.15 μg/L (range = 1.56–28.6 μg/L); and stage IV, 4.75 μg/L (range = 2.23–12.9 μg/L) (p = 0.009). Respectively, postoperative mean ICTP concentrations were as follows: DCIS, 4.19 μg/L (range = 3.71–4.48 μg/L); stages I–II, 5.23 μg/L (range = 2.60–17.0 μg/L); stages II–III, 6.15 μg/L (range = 3.20–4.7 μg/L); stage IV, 14.42 μg/L (p = 0.016). There was only one postoperative sample in the stage IV group.

Correlations between tumor stage and (a) preoperative ICTP and (b) postoperative ICTP concentrations. Lines represent 95% confidence intervals.
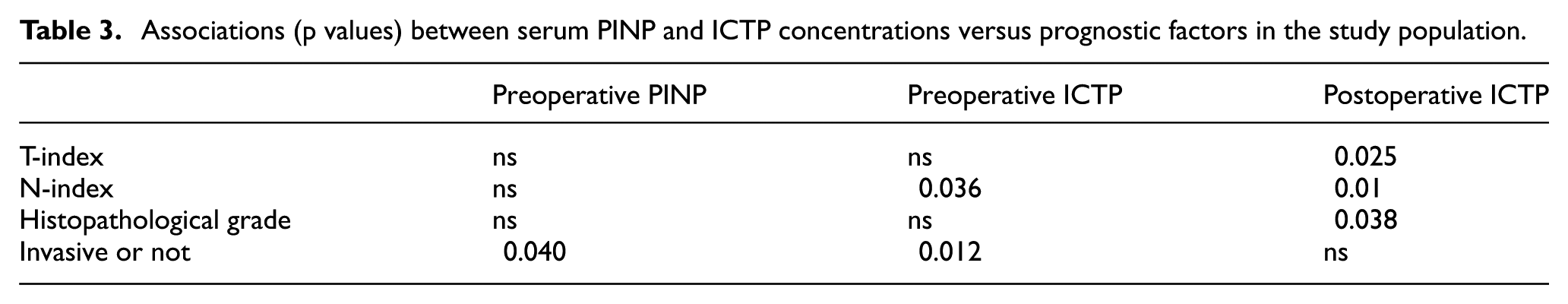
Traditional prognostic factors were re-formatted as two-class variables for the analyses. T-class was divided into T1 or T2–4 classes, and nodal status to either nodal negativity or positivity. Histopathological grade was divided to either grades 1–2 or grade 3. Elevated postoperative ICTP concentrations were associated with increased tumor size (p = 0.025), nodal involvement (p = 0.01), and a higher histopathological grade (p = 0.038) (Table 3). Elevated preoperative ICTP concentrations were associated with nodal involvement (p = 0.036). A high preoperative ICTP concentration was associated with invasive breast cancer (p = 0.012) compared with in situ carcinoma, whereas higher preoperative PINP concentrations were found in DCIS compared with invasive carcinomas (p = 0.040).
Associations (p values) between serum PINP and ICTP concentrations versus prognostic factors in the study population.
Correlations between type I collagen markers in different breast cancer subtypes
None of the studied markers were associated with a specific breast-cancer subtype. Preoperative serum PINP concentrations were correlated (Spearman’s correlation) with pre- and postoperative serum ICTP concentrations (Table 4). In addition, preoperative ICTP concentrations were correlated with postoperative ICTP concentrations. Correlations were carried out separately in the population of 612 invasive cases with no primary metastases and in different breast cancer subtypes.
Correlations between preoperative PINP and pre- and postoperative ICTP concentrations in different breast cancer subgroups (stages 0 and IV excluded).

TNBC: triple-negative breast cancer.
Survival analysis
A high preoperative ICTP level was associated with better BCSS, but only in the patients with luminal-B-like (HER2-negative) tumors (in univariate analysis p = 0.017) (Figure 1). This was confirmed in Cox regression analysis (relative risk (RR) = 3.127; 95% confidence interval (CI) = 1.081–9.049; p = 0.035), when T-class (RR = 4.049; 95% CI = 1.263–12.981; p = 0.019) and nodal status (RR = 3.896; 95% CI = 1.088–13.959; p = 0.037) were included in the analysis. A high preoperative ICTP level was also a significant predictor of local RFS in univariate (p = 0.0020) and multivariate analyses (RR = 13.04, 95% CI = 1.354–125.5, p = 0.026; for T-class, RR = 2.128, 95% CI = 0.297–15.23, p = 0.452; for N-class, RR = 0.332, 95% CI = 0.033–3.307, p = 0.347), but only in the patients with TNBC. Preoperative serum PINP concentrations were not associated with BCSS, DFS, or RFS (Figure 2).
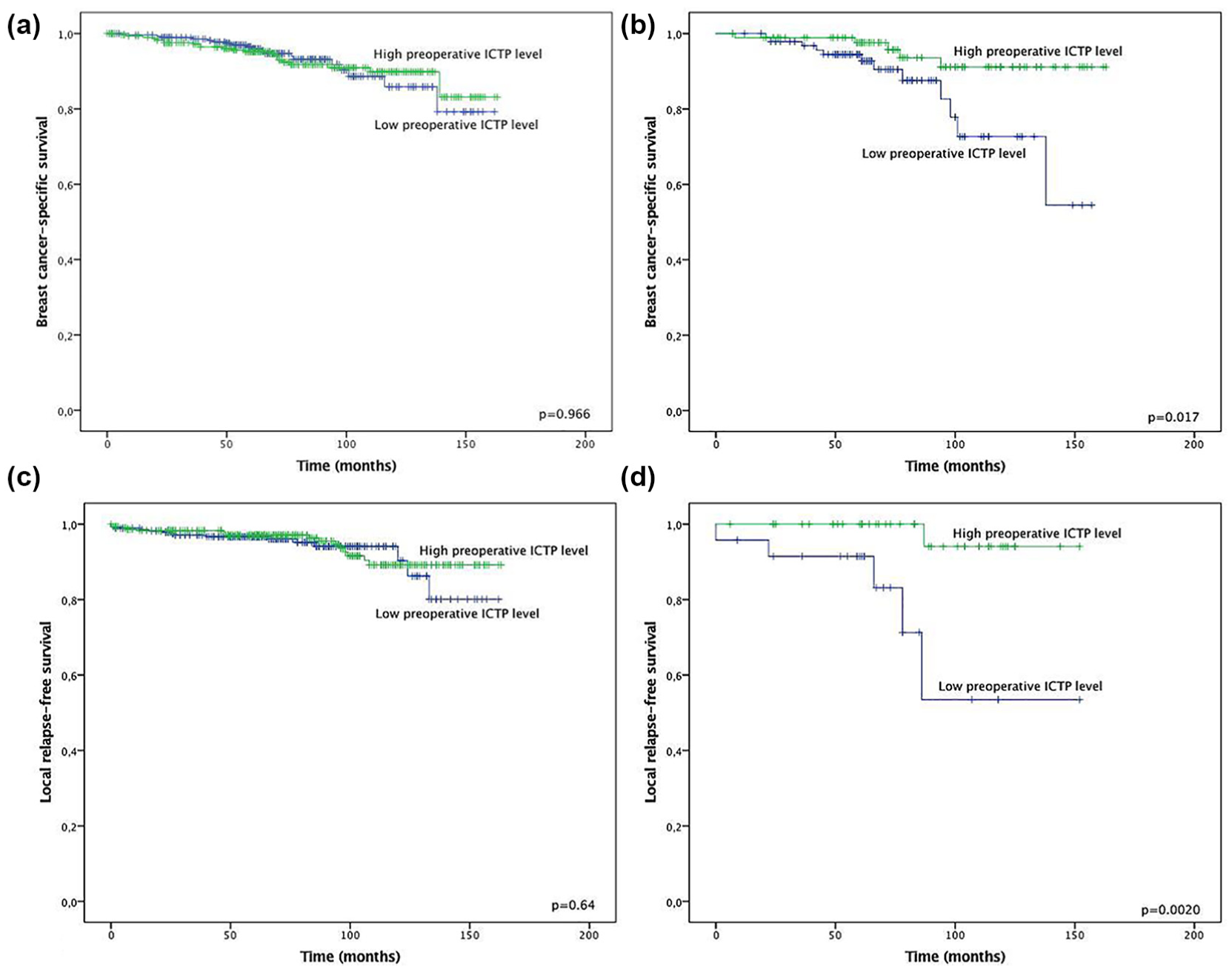
Kaplan–Meier curves showing BCSS in the (a) whole population and (b) separately in the patients with the luminal-B-like (HER2-negative) subtype. RFS in the (c) whole study cohort and (d) separately in cases of triple-negative breast cancer. Patients with distant metastases at the time of diagnosis were excluded from the survival analyses.
Discussion
This is the first prospective study in which early-stage breast cancer patients have been placed in subgroups on the basis of breast cancer subtypes and concentrations of their serum type I collagen synthesis and degradation markers compared. As the main finding, elevated preoperative serum ICTP levels were found to be associated with better prognosis in luminal-B-like (HER2-negative) early breast cancer. In addition, exceptionally dismal local RFS was observed with TNBC patients with low preoperative ICTP levels. Furthermore, our results indicate that in higher stages of breast cancer, preoperative and postoperative serum ICTP concentrations are elevated.
Elevated preoperative serum ICTP levels have previously been shown to be associated with poor outcomes in breast cancer. Imamura et al. 10 noticed worse RFS, but only in a subgroup of postmenopausal women. Keskikuru et al. 24 reported that higher preoperative ICTP levels were associated with poor DFS and OS. However, that study was a relatively small retrospective study and up to 35% of the patients developed recurrence during the 62-month follow-up time. Also, Imamura et al. noted that 10.7% of their patients developed metastatic disease during 23.6 months of follow up. On the basis of AZURE trial data, high preoperative ICTP levels predicted bone recurrence, but were not associated with other endpoints. 25 The contrast between the results of earlier studies and ours could be, at least to some extent, explained by the selection of patients. Our study is prospective, concerning 612 patients with non-metastatic early-stage breast cancer, with long-term follow-up. Serum ICTP levels were not associated with survival in the study population as a whole, but only within certain, preselected subgroups. Only 9.5% of our patients developed distant metastases during a 72-month follow-up period, which is well in line with modern treatment results.
High preoperative ICTP serum concentrations have also previously been connected to poor prognosis in other cancers. Simojoki et al. 26 presented evidence that a higher preoperative ICTP serum concentration predicts a poor outcome in epithelial ovarian cancer. Nurmenniemi et al. 27 found that a higher serum ICTP concentration predicted worse survival in cases of head and neck squamous cell carcinoma (HNSSC), and ICTP concentrations were continuously elevated from stage I to stage IV disease. This finding parallels ours, since we observed that both preoperative and postoperative ICTP levels increased along with stage (stages 0–IV).
The reason that a high ICTP level predicts a better outcome in TNBC and luminal-B-like (HER2-negative) subgroups could be linked to collagen I metabolism and organization of the ECM. Cancer cells invade and migrate through the ECM on their metastatic journey. Hypoxia and hypoxia-inducible factors (HIFs) can facilitate invasion and metastasis by upregulation of degradative enzymes and remodeling of the ECM. 28 Collagen I (Col1) fibers represent the main component of the ECM. In breast cancers, a high collagen density has been associated with increased malignancy and also tumor aggressiveness. 6 Col1 degradation is mostly due to matrix metalloproteinases MMP-1 and MMP-14. 29 In a recent paper, Goggins et al. 30 suggested that a decrease of MMP-1 and MMP-14 in HIF-silenced TNBC cells would prevent Col1 degradation and increase the amount of type I collagen fibers. Also, a reduction in lysyl oxidase (LOX) protein expression in HIF-downregulated tumors suggested that more non-crosslinked collagen I fibers are present despite an overall increase in fiber density. Lysyl oxidases are enzymes that catalyze the cross-linking of Col1. LOX expression was found to induce hypoxia and is regulated by HIF-1 in TNBC. 30 Usually, in tumors that are not HIF-silenced, type I collagen metabolism is active, with non-reduced amounts of MMPs, causing increased collagen I degradation reflected in an elevated serum level of ICTP. Along with the absolute amount of collagen in breast cancer tissue, collagen fiber structures are important in different stages of tumor-cell invasion. 6 As cancer cells have been shown to move along radially aligned collagen fibers, an increase of Col1 fibers does not automatically result in increased metastatic activity or aggressiveness of tumor cells.6,30 Molecular mechanisms within cancer cells do not solely define cancer behavior, and cellular and molecular components of the ECM and its three-dimensional structure play an essential role.31,32 This reinforces the importance of ECM organization in different breast cancer subtypes, as Col1 fiber alignment is a determinant of cancer progression. 6 Earlier evidence concerning TACS (patterns of collagen 1 fiber bundles at the tumor–host interface) and their association with cancer progression and prognosis encourages further study of ECM organization in different breast cancer subtypes.6,33 To summarize, it is possible that in some subgroups of breast cancer (TNBC, luminal-B-like (HER2-negative)), the collagen matrix can be protective and in those tumors increased degradation of collagen can be a marker of good prognosis, as demonstrated in our study.
The luminal B (HER2-negative) subgroup is heterogeneous and may also have a heterogeneous prognosis.34–36 It is known that the microenvironment is closely related to the development of these tumors. 37 As far as we know, there is no previous research concerning the relationship between the collagen metabolism markers PINP and ICTP and the luminal B (HER2-negative) subtype. However, Zhu et al. 38 have described the interactions between cancer cells, cytokines, and ECM collagen IV (as a prognostic marker) in the luminal B (HER2-negative) subtype.
Our study obviously has some weaknesses. We were not able to define exact biological subtypes of breast cancer by genotyping and we relied on immunohistochemical surrogates of these subtypes. Neither did we know the menopausal status of the patients, which potentially may have affected the results. The cut-off point for ER positivity changed in international guidelines from 10% to 1% during collection of the cohort, which resulted in different definitions of subgroups in some rare cases.
In conclusion, we were able to determine, for the first time, with this prospectively collected data and long-term follow-up, that a high preoperative serum ICTP concentration is associated with better local-RFS (in the TNBC group) and better BCSS (in the luminal-B-like, HER2- negative subgroup) by way of a partly unclear mechanism. Data from our study yield important information for the first time in different breast-cancer subtypes that the collagen I degradation marker ICTP is an important marker of better prognosis.
Footnotes
Acknowledgements
We gratefully acknowledge and thank Mr Kari Mononen for helping in patient data collection, Mrs Katja Koukkula for execution of the RIAs and CLIAs and advice on technical details, and Mrs Anne Bisi for help with sorting and managing the serum samples.
Author contributions
All authors contributed to the study design and its conception. Anniina Jääskeläinen and Arja Jukkola collected the data on patients. Peeter Karihtala and Anniina Jääskeläinen were responsible for assessing statistical analyses. Anniina Jääskeläinen, Arja Jukkola, and Peeter Karihtala were the major contributors in writing the manuscript. All authors provided comments on drafts of the manuscript and read and approved the final manuscript.
Availability of data and materials
The datasets analyzed during this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The local ethics committee of the Hospital District of Northern Ostrobothnia (144/2011, amendment 23.2.2015) and the Finnish National Supervisory Authority for Welfare and Health (1339/05.01.00.06/2009) approved the study design. With approval from the ethics committee of the Hospital District of Northern Ostrobothnia, written informed consent was not obtained from the patients at the time of sample donation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
