Abstract
Lung cancer stem cells are supposed to be the main drivers of tumor initiation, maintenance, drug resistance, and relapse of the disease. Hence, identification of the cellular and molecular aspects of these cells is a prerequisite for targeted therapy of lung cancer. Currently, analysis of circulating tumor cells has the potential to become the main diagnostic technique to monitor disease progression or therapeutic response as it is non-invasive. However, accurate detection of circulating tumor cells has remained a challenge, as epithelial cell markers used so far are not always trustworthy for detecting circulating tumor cells, especially during epithelial–mesenchymal transition. As cancer stem cells are the only culprit to initiate metastatic tumors, our aim was to isolate and characterize circulating tumor stem cells rather than circulating tumor cells from the peripheral blood of NSCLC adenocarcinoma as limited data are available addressing the gene expression profiling of lung cancer stem cells. Here, we reveal that CD44(+)/CD24(−) population in circulation not only exhibit stem cell–related genes but also possess epithelial–mesenchymal transition characteristics. In conclusion, the use of one or more cancer stem cell markers along with epithelial, mesenchymal and epithelial mesenchymal transition markers will prospectively provide the most precise assessment of the threat for recurrence and metastatic disease and has a great potential for forthcoming applications in harvesting circulating tumor stem cells and their downstream applications. Our results will aid in developing diagnostic and prognostic modalities and personalized treatment regimens like dendritic cell–based immunotherapy that can be utilized for targeting and eliminating circulating tumor stem cells, to significantly reduce the possibility of relapse and improve clinical outcomes.
Keywords
Introduction
Despite the recent advances in treatment modalities, non–small cell lung cancer remains to be one of the leading causes of mortality with a 5-year survival rate of 15%. 1 This is owed to the increasingly emergent concept of cancer stem cells (CSCs), a subpopulation of the tumor mass, alleged to be impervious to radiation and chemotherapeutic treatments, making them responsible for recurrence of the disease in cancer patients. 2 Current theories have recognized that a substantial number of tumor cells, that is, circulating tumor cells (CTCs), can be discharged into the blood circulation much sooner than tumor metastasis happens and even at the very early stage of cancer development. 3 Various non-invasive assays have been established to assess the prognosis of patients and diagnose them at the early onset of metastatic spread. One of the major confounders is CTCs isolation technique which holds great promise for early detection of minimal residual disease and recurrence of cancer in treated patients. Therefore, CTCs are conversed to be ambulatory as predictive biomarkers guiding personalized treatment approaches.4–6 Hence, sensitive isolation and insightful molecular characterization of CTCs could assist as a “liquid biopsy” and aid to improve individual treatment regimens for cancer patients; however, reproducible and accurate methods for identifying and segregating CTCs have remained a challenge.7–9
Till date, epithelial cell surface markers are being used as a sole modality for isolation of rare population of CTCs by various systems.10–14 Utilizing these markers for the effective isolation of CTCs may provide erroneous results as they result in the down-regulation of epithelial cell markers during the epithelial–mesenchymal transition (EMT). Increasing evidences suggest that an apparently small subset of CTCs also bears CSC characteristics based on their ability to give rise to tumors and thus could be considered circulating tumor stem cells (CTSCs).15–17 CTSCs represent a subset of exclusively tumorigenic CSCs having invasive characteristics and are known to be a potential therapeutic target for preventing disease progression, 18 suggesting that CTSCs rather than CTCs may become new biomarkers to fill the diagnostic gap. Thus, a sensitive bioassay is required for accurately detecting and isolating all CTSCs. To the best of our knowledge, this study is among the preliminary approaches for identifying CTSCs in the blood of lung cancer patients, using a combination of epithelial cell surface marker cytokeratin (CK), epithelial cell adhesion membrane protein EpCAM, and CSC markers CD44, ALDH, and CD24; combining surface markers has been shown to increase the accuracy of detecting CTSCs. Apart from this, EMT markers such as vimentin, E-cadherin, and N-cadherin were evaluated to check the epithelial/mesenchymal origin of the isolated CTSCs.
In this study, we aimed to develop a comparatively simple assay for detecting CTSCs from the blood of non small cell lung carcinoma (NSCLC) adenocarcinoma patients. These CTSCs were capable of forming pleurospheres (PS) in vitro, confirming that the isolated CTSCs (CD44(+)/CD24(−&x41;) represent tumor-initiating properties. In order to improve clinical outcomes for the patients, as an approach in the direction of personalized medicine, this can be used in the future for frequent monitoring of treatment response and eliminating CTSCs by targeting them with cell-based immunotherapy at the early onset of the metastatic condition.
Materials and methods
Sample collection and cell culture
Peripheral blood samples were obtained from patients with NSCLC adenocarcinoma (Table 1) with prior signed consent forms. Peripheral blood mononuclear cells (PBMNCs) containing CTSCs were obtained by the density gradient method. These PBMNCs were then further cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS) containing 1% antibiotic at 37°C in an atmosphere of 5% CO2. All samples were processed within 1 h after collection. This study was approved by the Ethics Committee of Gujarat Cancer & Research Institute (Ahmedabad, India).
Case description and PS-forming ability of NSCLC patients.

PS: pleurosphere; NSCLC: non small cell lung carcinoma.
+ sign indicates PS-forming ability at the density of 100 cells/well.
PS-forming assay from PBMNCs
CSCs are heterogeneous 19 in nature, and thus, we tried to isolate CTSCs from the whole population of PBMNCs by growing them in serum-free media instead of isolating by any single marker. Isolated PBMNCs containing CTSCs were transferred to six-well ultra-low attachment plates at the cell density of 100 cells per well and allowed to grow in appropriate medium to form PS. The medium was changed every 2 days. Wells that contained spheres were counted using inverted phase-contrast microscopy. Proliferative potential was evaluated in terms of doubling time by counting cells on days 2, 4, 6, 8, and 10 after seeding in multiwall plates using an automated cell counter. Growth curve was generated using the following formula. Hayflick’s formula was used to calculate the doubling time of cells during the logarithmic phase

where T is the population doubling time, t is the appointed time after subculture, Nt is the number of cells at the appointed time, and N0 is the number of cells at the beginning of subculture.
Chemotherapeutic resistance assay
Cells were seeded in 96-well plates to check the effect of chemotherapeutic agent gemcitabine commonly used in the treatment of lung cancer patients. Cells were exposed to this drug in the range of 10 nM–100 µM for 24 and 72 h to check their IC50 values as described previously by MTT assay to evaluate chemotherapy resistance of these sphere-forming cells. 20
Flow cytometry
A total of 1 × 105 cells were re-suspended in 100 µL of phosphate-buffered saline (PBS) incubated with 10-µL anti-CK-FITC, anti-EpCAM-PE, anti-CD24-PE, and anti-CD44-PE conjugated antibody for 15 min at 4°C. After washing twice with PBS, cells were again re-suspended in 500 µL and characterized using BD FACSCalibur™. CD45 was used as a gating marker. We have gated CD45-negative cells for the characterization of aforementioned markers.
Immunofluorescence and immunocytochemistry assay
Slides containing oncospheres were immersed in 2% paraformaldehyde (PFA) for 5 min for fixation, permeabilized in 0.25% Triton X-100 for 15 min, and washed with TBST (Tris-buffered saline (TBS) with 0.1% Tween-20) three times for 15 min. After blocking with 5% goat serum for 30 min at room temperature, the slides were incubated with mouse polyclonal anti-human vimentin, E-cadherin, and rabbit polyclonal anti-human n-cadherin (1:200 dilution) overnight at 4°C. Subsequently, the cells were washed with TBST before incubating with respective secondary antibody (1:250 dilution) for 45 min at 37°C. For fluorescent labeled secondary antibody, 4′,6-diamidino-2-phenylindole (DAPI) was used to stain nuclei, and then, slides were mounted in DABCO solution. Images were visualized using fluorescence microscopy. For horseradish peroxidase (HRP)-labeled antibodies, cells were incubated in 3,3’-Diaminobenzidine (DAB) and washed with TBST. Cells were incubated in Gill’s hematoxylin solution for 30 min for counterstaining. To remove excess stain, slides were dipped in acid alcohol solution and rinsed with ammonia water. Then, slides were dipped in 70% alcohol for 5 min and then in xylene for 5 min. Slides were mounted in DPX, and images were visualized using a light microscope.
Western blot analysis
Cells from PS, after 0–21 days of culture, were collected in RIPA lysis buffer (150-mM NaCl, 0.5% Nonidet P-40, 1% Triton X-100, 0.5-mM ethylenediaminetetraacetic acid (EDTA), 0.1% sodium dodecyl sulfate (SDS), 50-mM Tris-HCl, pH 7.6), containing protease inhibitor cocktail (Sigma-Aldrich) after washing with ice-cold PBS. Protein extracts were quantified by Qubit® Protein Assay (Life Technologies). For western blot analysis, 50 mg of total proteins was resolved on 12% BIS–TRIS gel and further transferred to a nitrocellulose membrane (Bio-Rad) with Bicine transfer buffer by electroblotting. After electrophoresis, the membrane was stained with Sypro Ruby and blocked with TBST containing 1% BSA for 15 min and then incubated overnight with primary antibodies. After washing with TBST, membranes were probed 1 h at room temperature with secondary antibodies. Finally, the results were evaluated by Gel Doc.
Real-time PCR analysis
Sphere-forming cells were taken, and total endogenous messenger RNA (mRNA) from these cells was isolated using a NucleoSpin RNA kit (MACHEREY-NAGEL) according to the manufacturer’s instructions to detect the expression of various CSC markers like ALDH, Nanog, BMI-1, and EpCAM and drug resistance–related genes such as ABCG2, Pgp, BCRP, and LRP. The quantity of RNA was determined by the Qubit 2.0 Fluorometer (Invitrogen). A total of 1 µg of RNA was reverse transcribed to complementary DNA (cDNA) by incubating the reaction mixture at 42°C for 30 min and 85°C for 5 min using the iScriptTM cDNA Synthesis Kit (Bio-Rad), and the resulting first-strand cDNA was used as a template in the quantitative polymerase chain reaction (qPCR). Reaction for PCR was prepared using Green I PCR reagents (KAPA Biosystems), and the primers for respective genes and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the endogenous control (Table 2). The reverse transcription polymerase chain reaction (RT-PCR) was performed using AriaMx PCR system (Agilent Technologies) under the following thermal profile: 95°C for 3 min, 40 cycles of 95°C for 3 s, 60°C for 20 s, and 72°C for 30 s. The comparative cycle threshold Ct method was used for the quantification. Results were compared with non-dividing cells.
List of primers used in qRT-PCR analysis.

qRT-PCR: quantitative reverse transcription polymerase chain reaction.
Statistical analysis
All data were expressed as the mean ± standard error of the mean (SEM) from triplicate samples of three independent experiments. Differences in measured variables between experimental and control groups were assessed by Student’s t test; p < 0.05 was considered statistically significant.
Results
Isolation and enrichment of circulating lung cancer stem cells in serum-free media and their self-renewal potential
Conferring to the CSC theory, only a small subset of cells in CTCs exhibit CSC characteristics, and they have potential to grow in serum-free condition. Mononuclear cells (MNCs) obtained from the peripheral blood of NSCLC adenocarcinoma patients were seeded in six-well ultra low-attachment plates and cultured in serum-free medium to grow as spheroids, to test the stemness of the isolated CTSCs as an indirect indicator of tumor-initiating potential. CTSCs started growing as distinct PS, while non-dividing cells rather cells without stemness property died over a period of time. Self-renewal potential is one of the characteristics of the CSCs. The results from the sphere formation efficiency assay depicted that putative CTSCs isolated from peripheral blood were able to form PS (Note: Spheres termed as PS were derived from lung cancer patients). Sphere-forming ability was associated with the metastatic condition as only patients positive for any metastasis were able to form PS, suggesting their potential in disease progression (Tables 1 and 3). However, cells isolated from healthy controls did not form any spheroidal structures, suggesting the absence of CTSCs. After being cultured in serum-free condition supplemented with various growth factors, putative CTSCs started growing in colonies of different size and numbers. PS were getting bigger in size over a period of time, suggesting that these cells have prominent and higher proliferative efficiency which is responsible for disease progression (Figure 1(a)), while non-dividing cells did not form spheres over a period of time (Figure 1(b)). Differences between the clonogenic potential of dividing and non-dividing cells at days 3 and 14 were described in Figure 1(c). The doubling time of generated PS cultured in their appropriate medium, determined from growth curve, was about 72 h initially. The doubling time of these PS from CTSCs was shown to be decreased on increasing passage numbers. The doubling time of these cells was around 72 h at a low passage (third) and around 36 h at a high passage (seventh).
Number and size of spheres in primary cultured NSCLC cells.

NSCLC: non small cell lung carcinoma.

Self-renewal potential of cultured pleurospheres from CTSCs of the blood of NSCLC patients. (a) Representative phase-contrast microscopy analysis of PS on days 0, 3, 7, and 14 after seeding the cells. (b) Images of non-dividing cells at days 0 and 14. Images of day 0 were taken as reference. (c) Graphical representation of clonogenic potential (sphere-forming capability) of PS and non-dividing cells with percentage of spheres (clones) formed on days 3 and 14. Day 0 was taken as reference for calculation. Data were represented as mean ± SD and statistical significance with their p values (***p < 0.0001).
Enhanced chemotherapeutic resistance of CTSCs
Quiescent stem cell population have the characteristic to frequently evade drug or chemo-/radiotherapy due to their ability to pump out the excess drug with the help of ABC transporters like Pgp or ABCG2 (BCRP). Thus, we assessed the cytotoxic effect of gemcitabine (most commonly used chemotherapeutic drug in the treatment of lung cancer) on CTSCs, in order to check whether CTSCs were resistant to this chemotherapeutic agent by MTT assay in a time- and dose-dependent manner, and also multi drug resistance (MDR) markers like Pgp, MRP1, BCRP, and LRP by real-time PCR (see section “Characterization of cancer stem-like cells from NSCLC adenocarcinoma”). Results confirmed that existence of CSC-like features is responsible for the presence of intrinsic drug resistance in CTSCs. MTT assay in the presence of different concentrations of gemcitabine revealed that these cells were chemo-resistant as there was no significant cytotoxic effect on CTSCs at 100-µM higher concentration as compared to non-dividing cells. Moreover, gemcitabine treatment of the bulk cells headed to the enrichment of cancer stem-like cell population, suggesting endurance of the resistant CSC population. Nevertheless, we tried to determine IC50 values of gemcitabine treatment for 72 h and found that CTSCs to be inherently resistant as the difference in the percentage viability of the cells was quite low (Figure 2).

Chemotherapeutic resistance study: Time and dose response curves demonstrated that pleurospheres were highly resistant toward gemcitabine (IC50 ≥ 100 µM) as compared to non-dividing cells (IC50 = 44.66 µM and IC50 = 7.41 µM) at 24 and 72 h. X-axis indicates logarithmic value of concentration of gemcitabine used in the study, and Y-axis indicates viability of the cells. ***Indicates level of significance in terms of p value (p < 0.0001).
Characterization of cancer stem-like cells from NSCLC adenocarcinoma
A number of reports suggest that cultured human cancer cells contain various cellular subpopulations including CSCs, suggestive of the fact that CSCs are heterogeneous in nature. Thus, in this study, we have isolated CTSCs from the peripheral blood of NSCLC adenocarcinoma patients without using any single-marker-based technique. Furthermore, we have characterized these CTSCs to check the purity of these cells by analyzing the presence of CSC and epithelial markers like CD44, CD24, EpCAM, and CK using flow cytometry. CTSCs demonstrated a strong expression of CD44 with 75.15% ± 2.45% positive cells, while cells were negative for CD24 expression, suggesting the presence of CD44(+)/CD24(−) phenotype. Moreover, results suggested that only 5.6% ± 0.3% and 0.5% ± 0.4% of the cells were positive for CK and EpCAM, respectively, which are markers associated with the epithelial phenotype (Figure 3(a)). Real-time PCR analysis showed significant up-regulation of other stem cell markers like ALDH, BMI-1, and Nanog in the CD44(+)/CD24(−) population, emphasizing enrichment of CSC population, although EpCAM expression was down-regulated. In addition, EMT drivers like ATP-binding cassette (ABC) transporters like ABCG2 (BCRP), Pgp, MRP1, and LRP were also remarkably higher in the CD44(+)/CD24(−) cell populations (Figure 3(b)). These results suggest that CD44(+)/CD24(−) subpopulation in NSCLC adenocarcinoma probably possess the CSC-like property. Collectively, results revealed that CTSCs should be given more importance over CTCs to determine the early onset of disease progression and therapeutic response.
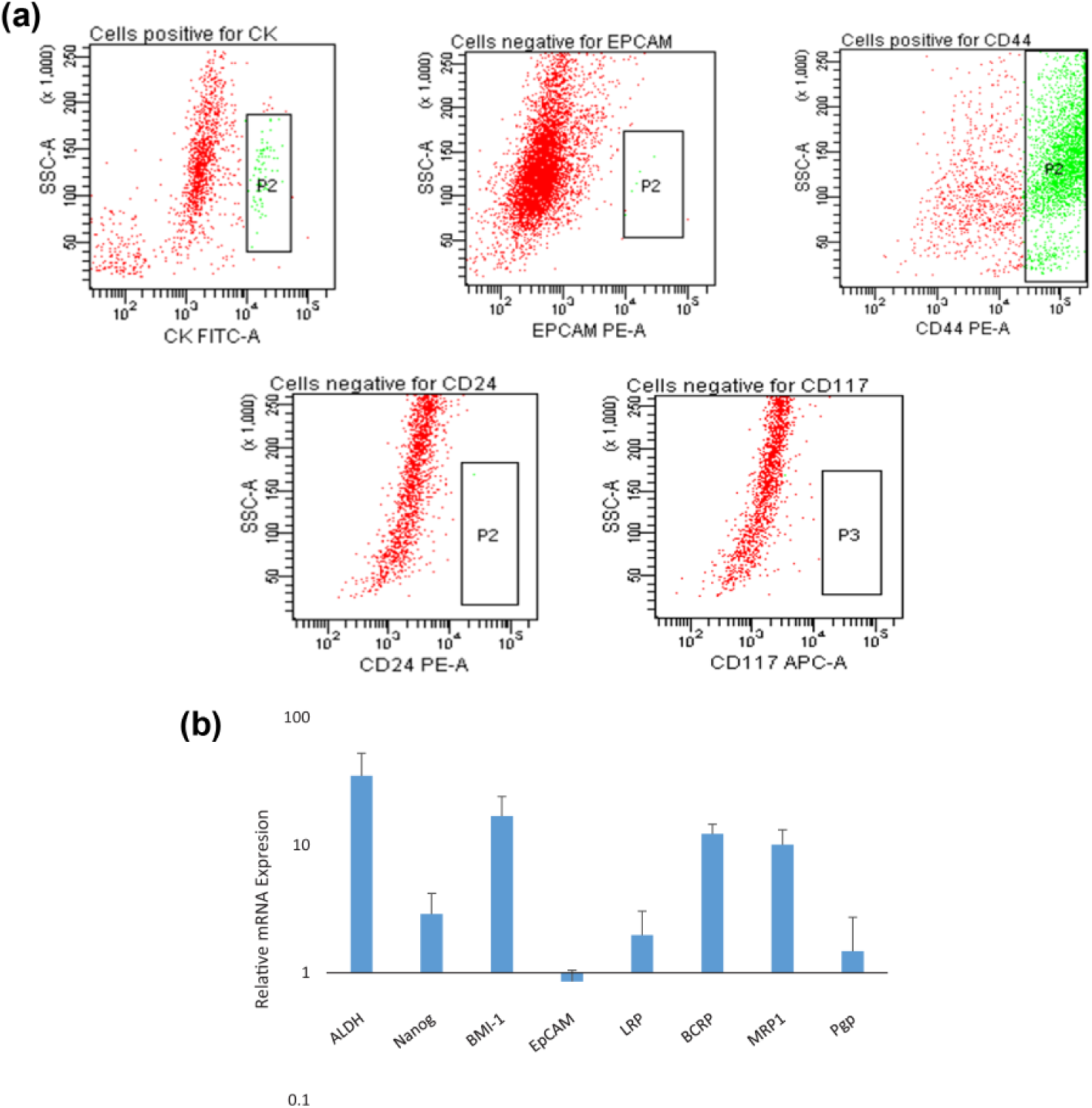
Characterization of stemness property of putative CTSCs. (a) CTSCs were analyzed by flow cytometry for the analysis of epithelial and CSC markers: cytokeratin (CK), epithelial cell adhesion molecule (EpCAM), CD44, CD24, and CD117. (b) qRT-PCR quantification of stem cells as well as drug resistance–related genes in CD44(+)/CD24(−) population of CTSCs using specific primers. Relative mRNA expression was normalized to GAPDH. Fold change >2 and <0.5 was considered as statistically significant. Healthy individuals (n = 5) were used as control.
Acquisition of EMT phenotype in CD44(+)/CD24(−) population
EMT is the underlying mechanism of tumor invasion and metastasis which can trigger conversion to CSC phenotype. Activation of EMT in cells results in a decreased expression of epithelial markers mainly E-cadherin and EpCAM and increased expression of mesenchymal markers, vimentin and N-cadherin. Thus, EMT markers like E-cadherin, vimentin, and N-cadherin were analyzed by immunofluorescence assay to evaluate epithelial/mesenchymal status or origin of the isolated CTSCs. This immunostaining assay depicted an extensive expression of vimentin in these PS, while lower levels of E-cadherin expression were distinguished in the same cells. However, N-cadherin expression was happened to be negative (Figure 4(a) and (c)). This might be attributed to the concordance of increased stemness on the occurrence of EMT.

Expression of EMT markers (vimentin, E-cadherin, and N-cadherin) in CD44(+)/CD24(−) population. (a) Representative immunofluorescence images of vimentin (green, anti-mouse, 1:200 dilution) and E-cadherin (green, anti-mouse, 1:200 dilution) staining and N-cadherin (red, anti-rabbit, 1:200 dilution). Blue color represents nucleus staining (DAPI). Secondary antibody (1:250 dilution). Scale bar = 100 µm. Ten random fields were observed for data analysis. (b) Representative images of immunocytochemistry staining of EMT markers. Brown color indicates positive reaction, whereas blue color indicates negative reaction. (c) Figure depicts percentage (%) of the cells positive for the expression of EMT markers in immunofluorescence (IF) and immunocytochemistry (ICC). Results are expressed as mean ± SD with their p value (***p < 0.0001) in the graph. Data were compared between mesenchymal marker (vimentin) and epithelial marker (E-cadherin). Experiments were performed in triplicates.
Immunocytochemistry and western blotting analysis
To check the consistency of the results obtained by immunofluorescence, same EMT markers were also characterized by immunocytochemistry and western blot analysis. The results were consistent with these two methods (Figures 4(b) and 5(a)). For immunocytochemistry, PS were stained with Gill’s hematoxylin for nucleus, E-cadherin for epithelial cells, and vimentin and N-cadherin for mesenchymal cells to evaluate their expression. In western blot assay, PS were collected and processed for analysis. β-actin was used as the loading control for western blot analysis. Collectively, PS from NSCLC adenocarcinoma patients were positive for vimentin and E-cadherin expression, whereas E-cadherin expression was lower as compared to vimentin (Figure 5(b)). Results were compared to healthy individuals, suggesting normal blood lacks CTSCs, whereas PS from NSCLC adenocarcinomas have the potential to initiate metastatic growth.

Western blot analysis of EMT markers. (a) Representative patterns of western blotting analysis of vimentin (anti-mouse, 1:5000 dilution), E-cadherin (anti-mouse, 1:5000 dilution), and N-cadherin (anti-rabbit, 1:5000 dilution). Secondary antibody (1:10,000 dilution). Data have been normalized with β-actin. (b) Relative expression of EMT markers in CD44(+)/CD24(−) cells as compared to healthy individuals (n = 5). Data are expressed as mean ± SD (***p < 0.0001).
Discussion
CSC theory denotes that a subpopulation within the tumor mass with characteristics including self-renewal efficacy, inherent resistance toward chemo- and radiotherapy, and high metastasis potential are happened to be the main drivers of initiation and progression of cancer, 21 including lung cancer. Although CSCs are identified by a most widely used method, that is, surface marker–based approach, the specificity and reliability of these markers for isolation of CSCs is not well established. In addition, for lung cancer, there is no widespread consensus regarding the best CSC markers. 22 Consequently, “liquid biopsy” has been used for isolation and characterization of CSCs in almost all cancers as it has the potential to be used for frequent genetic analysis of tumor diagnosis/prognosis, in personalized therapy selection and provides considerable leverage of being detected at any tumor stage.7–9,23 CTCs hold much more genetic/molecular information than free-circulating nucleic acid and proteins, thus revealing the importance of inter-cell heterogeneity in correlating cancer prognosis and treatment resistance. 24 However, CTCs are rare in the peripheral blood and tend to form aggregates, and because of their low concentration, there is limited study on the biology of CTCs. Due to these characteristics, CTC-detecting technologies confront many challenges with respect to the specificity, sensitivity, and reproducibility of the findings. It is difficult to capture all types of CTCs by EpCAM-based methods as they are heterogeneous in nature. However, there is growing interest in a more reasonable strategy to isolate CTSCs and ex vivo expansion of them as they have the self-renewal capability which is responsible for disease progression.24,25 In this study, we isolated and cultivated CTSCs by sphere formation assay to get heterogeneous population rather than isolating them by a single-marker-based approach which might be highly effective and have a tremendous potential in tracking the treatment response. Although information about molecular changes in cancer cells can also be obtained from circulating DNA fragments, the capture of CTSCs rather than CTCs could allow a more in-depth characterization including functional in vitro and in vivo studies. Thus, it is becoming increasingly important to track and eliminate the rare population of CTSCs, considered to be the main driver of tumor progression and metastatic spread. In addition, lung adenocarcinoma is heterogeneous with respect to histological and biological characteristics, and due to the heterogeneity of tumor cells, the reliability of single-marker-based detection of CTCs is limited. Moreover, the presence of CTCs is rare in the peripheral blood, but in spite of CTSCs being fewer, their ability to self-renew could be utilized as an effective in vitro method for future research. Therefore, we developed an in vitro model by generating PS which can be used as a surrogate model to extend our knowledge about the tumor cell dissemination biology in NSCLC with potential clinical implications for better prognosis.
EpCAM and members of CKs have been used for the positive selection of CTCs and have become the “gold standard” method for the validation of CTCs with epithelial phenotype in cancer patients. To date, “CellSearch System” is the only Food and Drug Administration (FDA)-approved technique used to detect CTCs, which uses epithelial markers like EpCAM and CKs.3,10–14,23 Epithelial cells undergo EMT to increase the capacity of invasiveness and metastasis, resulting in loss of expression of epithelial cell markers such as EpCAM and/or CKs, and thus, EpCAM-based techniques may not efficiently capture CTCs with mesenchymal characteristic following EMT. Mani et al. 26 revealed that cells acquire stem-cell-like properties during EMT by up-regulation of CD44 and down-regulation of CD24 as well as increased expression of other stem cell phenotypic markers. In this study, we have done an elaborate molecular and phenotypic characterization of the isolated CTSCs from NSCLC adenocarcinoma, and majority of the cells were characterized as CD44(+)/CD24(−). Moreover, these cells demonstrated a relatively high expression level of other CSC markers like ALDH, Nanog, and BMI-1 at mRNA level by real-time PCR. Although CD44(+)/CD24(−) cell population is firmly established as stem-like cells in breast cancer, studies in NSCLC have not been conclusive.
CD44(+)/CD24(−) cells were found to be of more on mesenchymal side, as reflected by loss of E-cadherin (epithelial marker) and gain of vimentin (mesenchymal marker) at protein level. In this context, it must be mentioned that Vesuna et al. 27 demonstrated that Twist (known EMT factor) down-regulates the expression of CD24 to enhance the expansion of breast CSCs. In NSCLC also, it may be speculated that CD24 depletion is a prerequisite for the induction of EMT. Nevertheless, CD24(+) cells have been observed as the definite stem cell marker in colorectal and nasopharyngeal epithelial malignancies,28,29 and CD44highCD24high cells population have been described as CSCs in head and neck squamous cell cancer (HNSCC). 30 Jaggupilli and Elkord 31 depicted no expression of both potential markers CD44 and CD24 in the COR-L23 NSCLC cell line; however, the CD44/CD24 phenotype has not been previously investigated in lung cancer clinical samples. Moreover, Roudi et al. showed indicated loss of CD44 expression and gain of CD24 expression (CD44(−&x41;/CD24(+)) in spheroid cells obtained from A549 cell line. 22 However, these studies were based on tissue samples or cell line, while our study is based on the CTSCs isolated from the peripheral blood of NSCLC adenocarcinoma patients. Conversely, we have found CD44(+)/CD24(−) cells to be able to form larger spheres within a few days, indicative of their ability to undergo cell division due to enhanced self-renewal efficiency which could be perpetuated for many generations. Although there are several reports suggesting the role of CD24 in promoting invasion and metastasis, 32 in our case, CD44(+)/CD24(−) cells were indeed more tumorigenic under in vitro conditions.
“Side population” cells, those that effectively efflux out drugs, have always been shown to have CSC property.33–35 In this study too, we found this characteristic in isolated CTSCs (CD44(+)/CD24(−)) which not only exemplifies stemness characteristic but also adds to the fact that these cells contribute to intrinsic chemo-resistance and hence difficult to eliminate. In addition to this, results showed up-regulation in the expression of ABC transporter genes like ABCG2, Pgp, and MRP1; and LRP at mRNA level. Thus, it eventually becomes clear with the chemo-resistant assays and mRNA expression assay whereby CD44(+)/CD24(−) cells exhibit enhanced survival upon drug treatment owing to their inherent chemo-resistant potential. This substantiates our finding that CTSCs should be given more importance in diagnosis over CTCs, which is the conventional way. Taken together, this finding makes a strong case for instituting further studies using cocktail of combination of markers for increased CTSC capture rate, including EMT cells.
In conclusion, the CD44(+)/CD24(−) cell population from isolated CTSCs of NSCLC adenocarcinoma displayed not only increased CSC and EMT properties but also enhanced self-renewal potential and intrinsic drug resistance which was demonstrated by their gene expression profile. Taken together, utilization of more than one CSC and EMT markers would not only provide the most accurate assessment to predict the risk of recurrence and metastasis but also have a great potential in improvising the harvesting of CTSCs in the most efficient and cost-effective manner. Our findings reinforce the foundation of CTSC assay which will aid in developing diagnostic/prognostic modalities and personalized treatment regimens like dendritic cell–based immunotherapy that can be utilized for targeting and eliminating circulating CSCs, to significantly reduce the possibility of relapse and improve clinical outcomes.
Footnotes
Acknowledgements
The authors thank the Gujarat Cancer & Research Institute for providing the administrative support to carry out this study. They also thank Dr Hemangini Vora and Dr Birva Raiya for flow cytometry analysis. The authors acknowledge Dr Kaid Johar for providing antibodies to perform immunofluorescence (IF), immunocytochemistry (ICC), and western blotting (WB) and his team for technical support. Sheefa Mirza is thankful to University Grants Commission (UGC; New Delhi) for the financial assistance in the form of MANF Senior Research fellowship (MANF-2013-14-GUJ-MUS-27104).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by University Grant Commission (MANF-2013-14-MUS-GUJ-27104).
Human and animal rights
This study was approved by the Institutional Review Board of Gujarat Cancer & Research Institute. All procedures performed in this study involving human participants were in accordance with the ethical standards of the Institutional Research Committee.
Informed consent
Informed consent was obtained from all individual participants included in the study.
