Abstract
Combining chemotherapy with radiotherapy potentiates the outcome of cancer treatment for the more comprehensive attack. In the current study, we continued to assess the therapeutic efficaciousness of the newly synthesized gallium nanoparticles (GaNPs) combined with low level of gamma radiation (IR), on the incidence of diethylnitrosamine (DEN)–induced hepatocellular carcinoma (HCC) in rats. Oral administration of GaNPs (1 mg/Kg b.wt.) 5 times per week for 6 weeks combined with IR to rats treated with DEN (20 mg/Kg b.wt. 5 times per week for 6 weeks) significantly reduced serum levels of alpha-fetoprotein (AFP), aspartate transferase (AST), alanine transferase (ALT), and gamma-glutamyltransferase (GGT). In addition, the immunoblotting results of matrix metalloproteinase-9 (MM-9) showed a marked downregulation of protein expression along with a significant decrease in the hepatic level of transforming growth factor β (TGF-β). Furthermore, GaNPs and/or low dose of radiation significantly elevated the level of caspase-3 gene transcript accompanied with evoked DNA fragmentation in rats treated with DEN. The ameliorative effect of GaNPs and IR well appreciated with the histopathological alteration finding in DEN groups. It can be concluded that the combination of GaNPs and/or IR can serve as a good therapeutic agent for the treatment of HCC, which ought to attract more studies.
Introduction
Diethylnitrosamine (DEN), a hepatocarcinogen and hepatotoxin, is synthesized endogenously and located in work area, processed meats, tobacco smoke, soybean, cheese, and extensive type of foods and is also made from metabolism of some medication. 1 It’s pronounced that DEN causes oxidative stress in the course of the metabolism, which leads to cytotoxicity, mutagenicity, and carcinogenicity. 2 Through the portal venous system, nitrosamine is absorbed in the gut, enters the liver and hepatocytes, and hampers the detoxing system of the liver; therefore, the liver gently proceeds to cirrhosis and hepatocellular carcinoma (HCC). 3 The traditional therapy of hepatocarcinoma such as chemotherapy, radiation, surgical resection, and ablation was unsuccessful for the restoration of health owing to the difficulty in diagnosis and serious side effects. Therefore, developing more effective and fewer virulent anticancer agents, including nanoparticles of metals to prevent or retard the process of hepatocarcinogenesis, was required. 4
Gallium nitrate is a first generation of gallium compounds that had anticancer property in human beings. 5 In phase II medical trials accomplished to evaluate the spectrum of its antineoplastic activity, gallium nitrate turned to administer patients with diverse kinds of cancers. In these malignancies, vast responses to gallium nitrate have been seen in patients whose tumors had relapsed or failed to reply to conventional chemotherapy. Other gallium forms combined with therapeutic strategies is a strategy to improve the results of the treatment. 6 On the other hand, high continuous infusion doses of gallium nitrate (300 mg/m2/day for 5–7 days) induced some side effects such as hypercalcemia, visual and auditory toxicities, also lymphoma, diarrhea rather than renal toxicity was dose-limiting. 7 However, universally targeting cells within a tumor is not always feasible because some drugs cannot diffuse efficiently and may induce multiple drug resistance—a situation where chemotherapy is failed. 8 Nanotechnology is defined as the introduction, manipulation, and use of materials on the nanometer size scale (1–100 nm). Even though nanoscale materials may be made by the use of variable traditional physical and chemical techniques, it’s presently feasible to biologically synthesize substances through surroundings pleasant green chemistry–based techniques. Nanoparticles are of chemical and physical differences in their properties compared to the bulk of the same chemical composition. 9 Metal nanoparticle–based therapeutic systems not only provide simultaneous diagnostic and therapy but also allow controlled and targeted drug release which helps to revolutionize cancer treatment and management. Recently, there is a great interest in the use of metal nanoparticles against tumor formation, development, and progression due to their intrinsic antitumor effects. The application of external stimuli has an extrinsic effect such as in hyperthermia where metal nanoparticles, acting as coadjuvants, are stimulated by external radiations such as IR or X-rays to produce free radicals to kill cancer cells and also increase the cytotoxic effect of ionizing radiations.10,11 Among these, the potential therapeutic application of noble metal nanoparticles represents an attractive platform for cancer therapy in a wide variety of targets and clinical settings.10,12
Radiation hormesis is often taken to imply some element of physiologic benefit from of low-dose radiation in the range of 0.1–0.5 Gy. The low dose of radiation has been observed to stimulate the radical detoxing system and enhancement of DNA repair rates and result in immune competence that enhances the increase in a wide variety of cytotoxic lymphocytes, resulting in a depletion of the prevalence of metastatic cancer. 13 Earlier our study has pronounced that gallium nanoparticles (GaNPs) combined with low-level gamma radiation (IR) exhibit suppressive induction of cytotoxic effects on cancer cells. 14 Considering the above-mentioned information on the possibility of using GaNPs as an antitumor agent, the present study was suggested to explore its potential therapeutic effect separately or combined with low dose of gamma radiation against hepatocellular carcinogenesis.
Materials and methods
Chemicals and reagents
DEN was obtained from Sigma Chemical Co. (St. Louis, MO, USA). All other chemicals and reagents used were of analytical grade.
Extracellular synthesis of gallium nanoparticles
The probiotic strain of Bacillus helveticus was used for GaNPs synthesis according to Kandil et al. 14 Lactobacillus helveticus ATCC 7995 used in this study was obtained from the Microbiological Resources Center (Cairo, Egypt) cultured in De Man, Rogosa and Sharpe (MRS) broth/ agar. Briefly, Lactobacillus helveticus was inoculated into flasks of sterile 100 mL of MRS broth. The culture flasks were incubated in a rotating shaker set at 200 r/min for 48 h at room temperature and then centrifuged at 6000 r/min for 10 min. The biomass and supernatant were separated and the supernatant was used for the production of GaNPs by mixing it with a filter-sterilized GaNO3 solution at 1 mM final concentration and incubated on rotating shaker (200 r/min) at room temperature for a period of 72 h. The resulting aqueous solution was filtered through a 0.22-mm Millipore filter before use. Further characterization was conducted for nanoparticles generated in our earlier study. 14 Synthesized GaNPs were characterized previously by transmission electron microscope (JEOL model 1200EX), ultraviolet (UV)–visible spectroscopy (Jenway UV spectrophotometer model 6505 in visible light at range of 200–600 nm), and Fourier-transform infrared spectroscopy (VERTEX 70/70v FTIR spectrometers (Bruker, India)). GaNPs obtained were of a diameter ranging from 8 to 20 nm with a relatively narrow particle size distribution. The absorption spectrum of GaNPs exhibited a peak at 265 nm assigned to surface plasmon resonance of the nanoparticles. 14
Cytotoxicity test
Human hepatocellular carcinoma (HepG2) cell line was obtained from the Egyptian National Cancer Institute, Cairo University. The antitumor effect and inhibitory concentration 50 (IC50) of GaNPs were investigated on the viability of the hepatocellular carcinoma cell line (HepG2) using sulforhodamine-B assay for cytotoxicity. 15
Radiation process
Whole-body gamma irradiation of rats was performed using a Canadian gamma cell-40 (137Cs) at the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt. Rats were exposed to a single dose of gamma radiation (0.5 Gy) at a dose rate 0.912 rad/s.
Experimental design
Sixty Sprague Dawley male albino rats (180 ± 20 g) obtained from Nile pharmaceutical Company housed in accordance with international animal care policies and were approved by the ethical committee for Animal Experimentation of NCRRT. They were kept in plastic cages, maintained under controlled conditions of light and temperature (25° ± 2°C), and provided with standard laboratory pellet diet and distilled water ad libitum for 1 week before starting the experiment for accommodation at NCRRT animal house.
Animals were divided randomly into six groups, each of 10 rats. The groups of animals were categorized as follows:
After 12 weeks, rats of each group were fasted overnight and then euthanized. Blood samples were obtained by heart puncture and collected in sterile heparinized and non-heparinized tubes. In each group, the liver of animals was quickly removed, washed with ice-cold saline, and divided into two portions: the first portions were fixed in 10% formalin and embedded in paraffin for histological examination, whereas the second portion was snap-frozen directly in liquid nitrogen and stored at −80°C prior to RNA isolation for gene expression analysis.
Biochemical assays
Detection of caspase-3 by quantitative real-time polymerase chain reaction
RNA was isolated from the livers of all groups after homogenization in Trizol® (Invitrogen) according to the protocol of the manufacturers. For real-time polymerase chain reaction (PCR), reverse transcription of the extracted RNA samples was performed using a SuperScript kit from Invitrogen. Real-time PCR was performed using a StepOnePlus™ sequence detection system from Applied Biosystems (Germany), with primer detecting
Determination of transforming growth factor-β level in liver tissue
Liver transforming growth factor beta (TGF-β) was estimated by rat enzyme-linked immunosorbent assay (ELISA) using a kit purchased from Glory Science Co., Ltd (USA).
Western blot analysis for matrix metalloproteinase-9 in liver tissue
Liver was washed, harvested, and then lyzed in lysis buffer (150 mM NaCl, 1% NP-40, 1% SDS, 1 mM phenylmethylsulfonyl fluoride (PMSF), 10 µg/mL leupeptin, 1 mM aprotinin, 50 mM Tris-HCl, pH 7.4) on ice for 30 min and centrifuged at 12,000 g at 4°C for 10 min. By The bicinchoninic acid (BCA) assay, the obtained supernatant was quantified for matrix metalloproteinase 9 (MMP-9) concentration. Equal amounts of protein were loaded (50 μg per lane) and separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred to polyvinylidene fluoride or polyvinylidene difluoride (PVDF) membrane. The membrane was blocked with 5% non-fat milk for 2 h, incubated with a specific antibody (1:1000 dilution) for 3 h, and then stained with appropriate secondary antibody conjugated with horseradish peroxidase (HRP) (1:2000 dilution) for 30 min at room temperature. The membrane was developed using enhanced chemiluminescence (ECL) reagent (Pierce, France) after final washing. The level of MMP-9 expression was normalized to β-actin.
Determination of alpha-fetoprotein concentration in serum
Serum alpha-fetoprotein (AFP) was estimated by ELISA using a rat AFP ELISA kit purchased from Glory Science Co., Ltd (USA).
Determination of serum iron concentration
Iron concentration was determined in serum by colorimetric assay according to Dreux. 17
Determination of liver enzymes in serum
Activities of serum alanine transferase (ALT) and aspartate transferase (AST) were determined colorimetrically according to Reitman and Frankel. 18 Serum gamma-glutamyltransferase (GGT) activity was measured kinetically according to Szasz. 19
Histopathological study
Samples were taken from the liver of rats of different groups and fixed in 10% formalin saline for 24 hours. After washing in tap water, serial dilutions of alcohol (methyl, ethyl, and absolute ethyl) were used for dehydration. Xylene was used to clear the samples and then samples were embedded in paraffin for 24 hours in a hot air oven at 56°. Tissue blocks were prepared for sectioning at 4 microns thickness by slide microtome from paraffin beeswax. The tissue sections were collected on glass slides, deparaffinized, and stained by hematoxylin and eosin stain for routine examination through the light electric microscope. 20
Statistical analysis
All statistical analyses were done by using the statistical package for Windows Version 15.0 (SPSS Software, Chicago, IL). The results for continuous variables were expressed as a mean ± standard error. Values were compared by one-way analysis of variance (ANOVA). Post hoc testing was performed for intergroup comparisons using the least significant difference test, and significance of p values, p <0.05, was considered statistically significant.
Results
Cytotoxicity test
The 50% growth inhibitory concentration (IC50) is shown in Figure 1. It is observed that GaNPs exhibited a cytotoxic effect against human hepatocellular carcinoma cell line (HepG2), which recorded IC50 value (8.0 μg/mL).
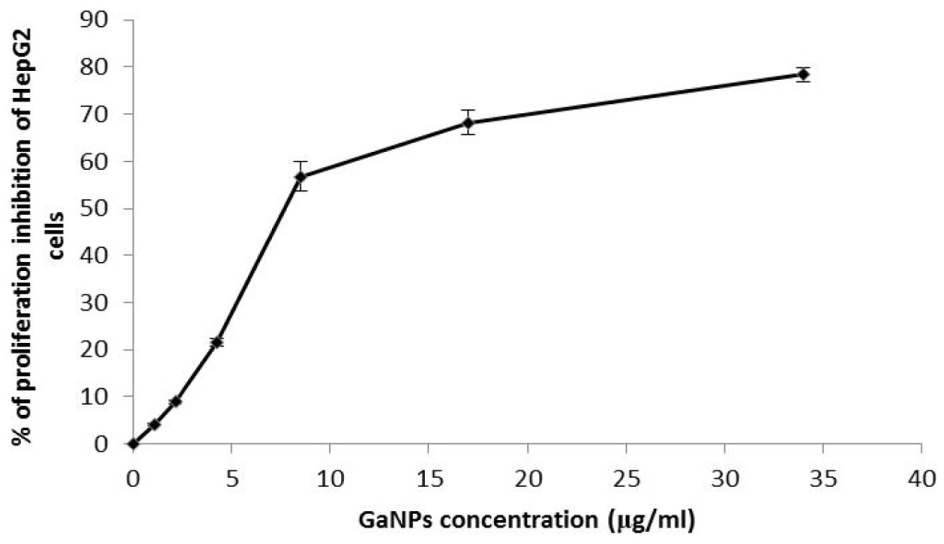
Evaluation of cytotoxicity gallium nanoparticles (GaNPs) against HepG2 cell line.
As shown in Table 1, liver caspase-3 level showed a significant decrease in the DEN group compared with the control group. Treatment with GaNPs, IR, and GaNPs + IR of DEN-treated rats normalized the liver caspase-3 mRNA level. On the other hand, TGF-β level significantly increased due to the administration of rats with DEN, while the level significantly increased in DEN + IR, DEN + GaNPs, and DEN + GaNPs + IR group compared with the control group.
The effect of GaNPs and/or IR on hepatic caspase-3 and TGF-β of DEN-treated rats.
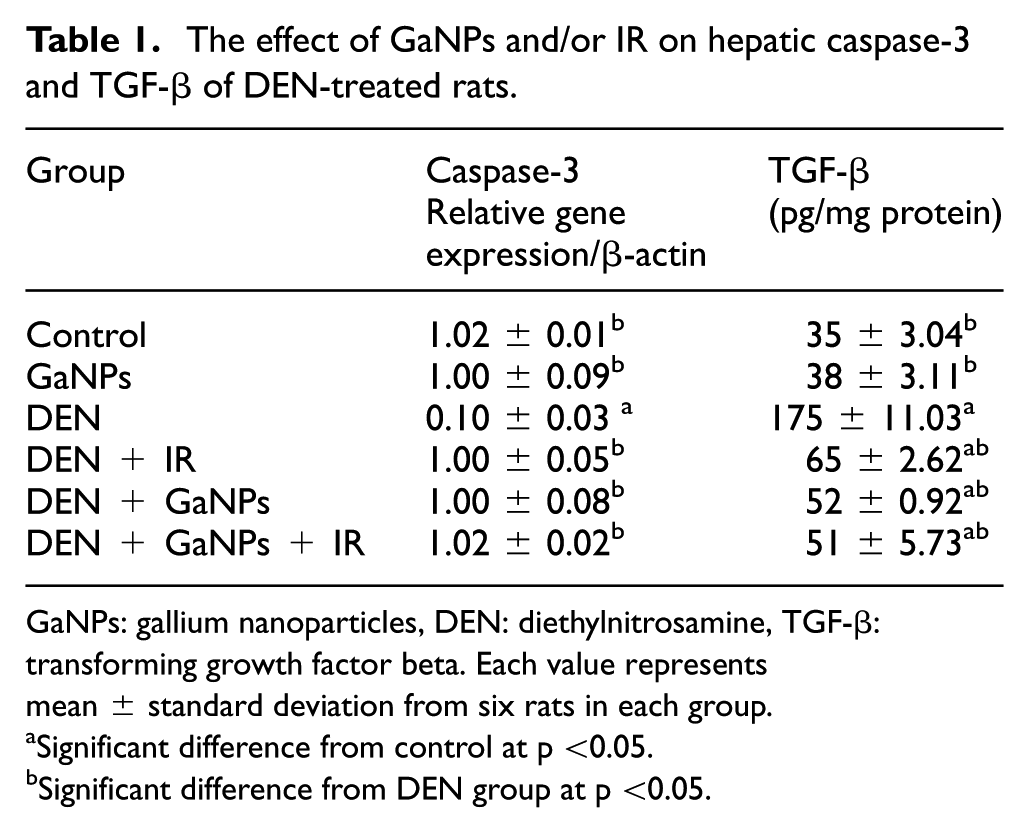
GaNPs: gallium nanoparticles, DEN: diethylnitrosamine, TGF-β: transforming growth factor beta. Each value represents mean ± standard deviation from six rats in each group.
Significant difference from control at p <0.05.
Significant difference from DEN group at p <0.05.
The hepatic activity of MMP-9 in Figure 2 exhibited a significant increase in rats treated with DEN compared with the control group. However, the activity of MMP-9 was restored in DEN + IR, DEN + GaNPs, and DEN + GaNPs + IR groups.

Representative Western blot analysis shows protein expression of matrix metalloproteinase 9 (MMP-9) (a) and β-actin (b) in different studied groups. L1: control; L 2: GaNPs; L3: DEN; L4: DEN + IR; L5: DEN + GaNPs; L6: DEN + GaNPs + IR. GaNPs: gallium nanoparticles, DEN: diethylnitrosamine.
As shown in the Table 2, animals injected with DEN exhibited a highly significant increase in serum activities of liver enzymes (AST, ALT, and GGT) and levels of AFP and iron compared with their corresponding control values. Administration of GaNPs and/or IR ameliorated the damage induced by DEN compared with values of DEN rats. The administration of DEN to the experimental groups caused deteriorative changes in liver function. Treatment of animals received DEN with GaNPs and/or IR significantly lowered the activities of the liver enzymes, AFP, and iron levels.
The effect of GaNPs and/or IR on the serum activities of AST, ALT, and GGT and levels of AFP and Fe of DEN-treated rats.

AFP: alpha-fetoprotein, ALT: alanine transferase, AST: aspartate transferase, DEN: diethylnitrosamine, GaNPs: gallium nanoparticles, GGT: gamma-glutamyltransferase
Each value represents mean ± standard deviation from six rats in each group.
Significant difference from control at p <0.05.
Significant difference from DEN group at p <0.05.
Histopathological findings
Histopathological examination of liver sections of control rats showed normal hepatic architecture (Figure 3(a)). In GaNP-treated rats, congestion in the central veins and sinusoids associated with diffuse Kupffer cells proliferation in between the hepatocytes (Figure 3(b)). On the other hand, animals treated with DEN showed a focal area of anaplastic hepatocytes, while the other cells forming acini were observed associated with fibroblastic cells proliferation dividing the degenerated and necrosed hepatic parenchyma into nodules (Figure 3(c)). Liver sections from rats treated with DEN and exposed to 0.5 Gy of gamma irradiation exhibited focal nodular area in the hepatic parenchyma showed dysplastic hyperplastic hepatocytes. There was congestion in the portal vein and inflammatory cells with dilated bile ducts in the portal area (Figure 3(d)). Rats treated with DEN and administrated with GaNPs diffuse ballooning degeneration and focal necrosis in the hepatocytes associated with inflammatory cells infiltration and fibrosis in the portal area besides congestion in both central and portal veins (Figure 3(e)). Rats received DEN and then administrated with the GaNPs and exposed to single dose of gamma irradiation infiltrated with focal inflammatory cells in the hepatic parenchyma associated with diffuse Kupffer cells proliferation in between the hepatocytes (Figure 3(f)).

Histopathological examination of liver (a–f). (a) Normal liver histology of a rat fed the standard diet. (b) Rats administrated gallium nanoparticles (GaNPs) showing congestion in central vein (cv) and sinusoids (s) with diffuse Kupffer cells proliferation(➘). (c) Rats administrated diethylnitrosamine (DEN). (d) Rats gavaged the DEN and then exposed to 0.5 Gy of gamma radiation showing area focal in hepatic parenchyma of dysplastic hepatocytes forming a nodule (a). (e) Rats gavage the DEN, and GaNPs showed degeneration in the hepatocytes (d) with focal necrosis in hepatic parenchyma (n) and inflammatory cells in portal area and congestion in central and portal veins (pv)). (f) Rats received DEN and GaNPs and exposed to 0.5 Gy of radiation showing few inflammatory cells infiltration (m) in hepatic parenchyma with diffuse Kupffer cells proliferation(➘).
Discussion
The result of our previous study on GaNPs exhibits antitumor properties in both in vitro and in vivo studies. 14 Therefore, GaNPs might be a potential alternative agent for cancer treatment and a sensitizing agent for radiotherapy. In the current work, we continue to evaluate the therapeutic efficacy of newly synthesized GaNPs combined with low level of gamma radiation (0.5 Gy) against DEN-induced hepatocellular carcinogenesis. The administration of DEN led to a marked elevation in serum levels of ALT, AST, and GGT indicating hepatocellular damage as previously reported. Due to their intracellular location inside the cytosol, the activity of serum aminotransferase enzymes was increased, 21 so toxicity affecting the liver with a subsequent breakdown in membrane architecture of the cells leads to their spillage into serum where their concentration rises. In addition, the overactivation of GGT attributed to its liberation from the plasma membrane into the circulation as it is located on the outer membrane of the hepatic cells, indicating damage of the cell membrane as a result of carcinogenesis. 22 Fascinatingly, GaNPs supplementation (1 mg/kg b.wt.) and/or IR brought back these enzymes in DEN-treated groups to near routine level by possibly preserving the functional integrity of the hepatocytes in DEN-treated groups which correlated with the histopathological finding. This improvement in hepatic injury suggesting that GaNPs separately or combined to radiation may have a potential ameliorative effect against DEN-induced liver damage.
In agreement with Song et al., 23 the current study shows a significant increase in the serum level of AFP in the DEN group. The possible explanations for the reinitiation of AFP synthesis by neoplastic hepatocytes are either increased transcription of AFP gene or posttranslational modification affecting AFP production. In rats which have been exposed to chemical carcinogens or HCC, AFP production is roughly proportional to the amount of transplantable mRNA present. 24 Administration of GaNPs and its combination with the low dose of gamma radiation significantly limited the elevation of serum AFP level as compared with the DEN-intoxicated group, suggesting that GaNPs and low-dose radiation might delay the hepatocarcinogenesis.
GaNPs inhibited the growth of tumor cells and induced morphological changes typical of apoptosis. The incidence of DNA fragmentation of tumor cells confirmed such morphological findings. Treatment with GaNPs induced ladder-like DNA fragmentation in tumor cells, which is a characteristic of DNA damage. Increased levels of DNA damage could cause the synthesis of a variety of incorrect proteins and therefore impaired cellular function. Most anticancer drugs either natural or synthetic have been known to cause DNA damage or suppress its replication, not necessarily killing the cells directly but inducing apoptosis. 14
The primary mechanism of antineoplastic action of gallium is its capacity to target and disrupt tumor cell iron homeostasis. 25 The present study observed a significant rise in the serum level of iron, which was more pronounced in the treated group with DEN compared with control group. As the cancer cells undergo massive cell division, a need for a high amount of iron for their development occurs. 26 Iron activates the proinflammatory cytokines and in the case of their excess leads to intracellular hypoxia. 27 Thus, it is very probable that by affecting iron metabolism and disturbing the process of its accumulation in malignant tissues, it may be possible to prevent a number of different cancers. Tissues having gallium concentrated with transferrin (TF), transferrin receptor (TFR), or ferritin in tissues; 28 therefore, gallium competed with iron when it reaches these sites and interfere with its absorption, metabolism, and activity. 29 Cancer cells that take up gallium become iron-deprived, which causes upregulation of TFR1. Increased TFR1 promotes increased Ga-TF uptake, leading to increased iron deprivation, and this cycle continues until the apoptosis of the cell takes place. 30
Regarding the apoptotic marker, the gene expression of caspase-3 was downregulated by administration of DEN, while the administration of GaNPs and/or radiation activated the expression of caspase-3. Therefore, GaNPs and radiation may induce apoptosis by upregulation of caspase-3. It was reported that the exposure to low dose of gamma radiation suppressed neoplastic transformation from a high challenge dose by stimulating intra- and intercellular signaling, leading to activated natural protection (ANP) against genomic instability–associated diseases, such as cancer.30,31 The activation ANP may include induced p53-dependent high-fidelity DNA repair along with normal apoptosis activation of an epigenetic protective apoptosis-mediated process, which selectively removes premalignant transformed cells,29,30 while also enhancing immune functions.32,33
The pathogenesis of HCC is characterized by an in-depth accumulation of extracellular matrix protein. The MMPs such as MMP-9 and TGF-β1 played important roles in the progression of HCC. 34 In the current study, the hepatic cell homogenates of DEN-treated rats showed increased levels of TGF-β and MMP-9. The increase in TGF-β expression in DEN-treated rats is expected due to the known role of this factor as an initiator of a signaling cascade that is closely linked to liver fibrosis, cirrhosis, and subsequent progression to HCC. The indicated profibrogenic role for TGF-β together with its role as an antiapoptotic factor provides a reasonable explanation for results observed in our study. The study of Wu et al. 35 suggested that TGF-β in cirrhotic liver promotes the neoplastic transformation of liver progenitor cells to hepatic tumor–initiating cells and facilitates hepatocarcinogenesis.
It was reported that MMP-9 can degrade type IV collagen in the basement membrane, which plays an important role in maintaining normal function of the hepatic cell. Thus, the action of MMP-9 activates the hepatic stellate cells, which promote the development of hepatic fibrosis. 35 On the other hand, the exposure to GaNPs and/or radiation strongly reduced the expression of MMP-9 and TGF-β, which suggests that GaNPs and/or radiation plays an important role against DEN-induced hepatocarcinogenesis by maintaining TGF-β and MMP-9 status.
The histopathological picture of the liver of rats received DEN, showed aberrant hepatocellular phenotype with variation in nuclear size and focal area of anaplastic cellular hepatocytes with other cells forming acini with fibroblastic cells proliferation. Sreepriya and Bali 36 reported that the presence of atypical nuclei is a marker of hepatocellular carcinogenesis. On the other hand, animals received GaNPs separately or combined with radiation, showed degeneration as well as focal necrosis in the hepatocytes associated with inflammatory cells infiltration. Also, hepatic parenchyma showed a diffused Kupffer cells proliferation in between the hepatocytes.
From the obtained results, it can be proposed that the combination of GaNPs and low-dose gamma radiation may have antineoplastic activity against hepatocarcinogenesis by stimulating apoptotic protein accompanied with modulation of iron homeostasis, TGF-β, and MMP-9. Further studies are required to reveal the mechanistic aspect of GaNPs and the low dose of radiation.
Footnotes
Acknowledgements
We appreciate the contribution of Dr. Adel Bakeer, Professor of Pathology, Faculty of Veterinary Medicine, Cairo University, for assistance in setting up histopathological study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
