Abstract
Breast cancer is the most commonly diagnosed cancer in Polish women. The expression of transcription nuclear factor kappa B, a key inducer of inflammatory response promoting carcinogenesis and cancer progression in breast cancer, is not well-established. We assessed the nuclear factor kappa B expression in a total of 119 invasive breast carcinomas and 25 healthy control samples and correlated this expression pattern with several clinical and pathologic parameters including histologic type and grade, tumor size, lymph node status, estrogen receptor status, and progesterone receptor status. The data used for the analysis were derived from medical records. An immunohistochemical analysis of nuclear factor kappa B, estrogen receptor, and progesterone receptor was carried out and evaluation of stainings was performed. The expression of nuclear factor kappa B was significantly higher than that in the corresponding healthy control samples. No statistical difference was demonstrated in nuclear factor kappa B expression in relation to age, menopausal status, lymph node status, tumor size and location, grade and histologic type of tumor, and hormonal status (estrogen receptor and progesterone receptor). Nuclear factor kappa B is significantly overexpressed in invasive breast cancer tissues. Although nuclear factor kappa B status does not correlate with clinicopathological findings, it might provide important additional information on prognosis and become a promising object for targeted therapy.
Keywords
Background
Inflammatory reaction plays a pivotal role as a protective mechanism against the invasion of harmful stimuli or injury as long as the process remains self-limiting. Unfortunately, pathological processes such as chronic inflammation may lead to cancer development. The transcription nuclear factor kappa B (NF-κB) is a key inducer of inflammatory response promoting carcinogenesis and cancer progression. 1
NF-κB/Rel is a family of transcription factors consisting of five members: RelA (p65), RelB, c-Rel, NF-κB1 (p50), and NF-κB2 (p52).2,3 All NF-κB proteins share highly conservative N-terminal Rel homology domain (RHD) which mediates dimers formation, interaction with specific inhibitors (inhibitor of kappa B (IκB)), translocation to the nucleus, and DNA binding. The majority of NF-κB dimers remain in the cytoplasm due to its association with IκB proteins (IκBα, IκBβ, and IκBε). Release of activated NF-κB requires proteasomal degradation of IκB upon phosphorylation induced by IκB kinase (IKK) complexes and subsequent ubiquitin conjugation. Transcriptionally active NF-κB undergoes nuclear translocation, binds to the specific promoters, and further regulates expression of target genes.2–4
There are several known different pathways triggering NF-κB activation altered by a variety of factors such as pro-inflammatory cytokines and growth stimuli. Other signaling pathways including phosphoinositide 3-kinase (PI3K)/Akt and Ras/mitogen-activated protein kinase (MAPK) may also be involved in NF-κB activation. 4
NF-κB as a potential link between chronic inflammation and cancer favors cell cycle progression, migration, and proliferation resulting from enhanced expression of target genes encoding proteins regulating cell cycle (e.g. cyclins D1, D2, D3, and c-myc) and antiapoptotic factors (e.g. bcl-2).1,4 Expression of cell adhesion molecules (e.g. E-selectin and intercellular adhesion molecule 1 (ICAM-1)) and angiogenic stimuli (e.g. vascular endothelial growth factor (VEGF)) tends to also be induced by NF-κB. 4
Besides all mentioned implications, deregulation of NF-κB pathway results in adjustment of steroid receptors’ expression as well. Levels of biomarkers such as estrogen receptors (ER) and progesterone receptors (PR) in breast cancer (BC) tissue are commonly used in clinical practice to predict prognosis and as a response to hormonal treatment.2,5 ER and NF-κB interact with each other which may result in mutually altered activity. 6
Abnormal expression of NF-κB has been reported in many malignant tumors including BC.2,4 Our study was planned to explore clinicopathological aspects which may be linked to this aberration. We evaluated the expression of NF-κB proteins in 119 BC patients by immunohistochemical staining. The results were correlated with age, histological subtype of BC, menopausal status, location and size of the tumor, and the presence of lymph node metastases. We also investigated the association between NF-κB and ER and PR expression in obtained BC tissue samples.
Patients and methods
Patients
Patients with primary breast carcinoma treated surgically at the 2nd Department of General and Oncological Surgery, Wroclaw Medical University from January 2008 to April 2014 were enrolled in the study. A total of 119 BC cases and 25 healthy control samples were analyzed. Data used for analysis were derived from medical records. The following information was obtained from all patients’ medical records: age, menopausal status, lymph node status, tumor size and location, grade and histologic type of tumor, and hormonal status (ER and PR).
Methods
All patients underwent Patey’s conservative radical mastectomy. Postoperative adjuvant therapy was performed based on the recommendations of the Polish Union of Oncology. Immunohistochemistry analysis was performed on formalin-fixed and paraffin-embedded BC tissues. Blocks were cut into 4 mm sections, deparaffinized in two changes of xylene, rehydrated in alcohols (96%, 80%, and 70% for 1 min each), washed in distilled water, stained in hematoxylin (Sigma-Aldrich, Inc., Steinheim, Germany), washed in tap water for 5 min, and then counterstained with eosin (Sigma-Aldrich, Inc.). Sections were then washed in distilled water, dehydrated through alcohols, and mounted in mounting medium (Dako Denmark A/S, Glostrup, Denmark). Sections stained with hematoxylin and eosin were evaluated with regard to the histopathologic diagnosis. Following the histopathologic analysis of the hematoxylin and eosin–stained sections, the most representative area of the tumor was marked and paraffin blocks were cut again into 4-mm-thick slices and were stained using immunohistochemistry. Immunohistochemical staining was performed using the labeled streptavidin biotin (LSAB) method (LSAB+System horseradish peroxidase (HRP); Dako Denmark A/S) with the following reagents: peroxidase blocking reagent, protein blocking reagent, antibody diluent with background-reducing components, biotinylated-conjugated antibody and streptavidin-HRP, and chromogen solution. Slides were deparaffinized in two changes of xylene for 10 min and then rehydrated in a series of graded alcohols (96%, 80%, and 70%) for 3 min each. Next, the specimens were washed twice for 4 min in distilled water and were microwaved in a citric buffer (0.1 M citric acid, 0.05% Tween 20 (pH 6.0); Sigma-Aldrich, Inc.) for 8 min for heat-induced epitope retrieval. Following two washes in distilled water for 4 min, the specimens were incubated for 10 min with peroxidase blocking reagent and rinsed twice for 5 min with phosphate-buffered saline. Next, incubation with protein block reagent was performed for 10 min, after which specimens were incubated with primary antibodies and stored overnight at 41°C (NFκB p50 (NLS): sc-114, dilution 1/100; Santa Cruz Biotechnology, Inc., Dallas, TX, USA). Following overnight incubation, the slides were incubated for 15 min with biotinylated-conjugated antibodies and streptavidin-HRP, rinsing twice with phosphate-buffered saline during and following the incubation. The reaction was detected and visualized using 3,3′-diaminobenzidine (DAB) in chromogen solution (Sigma-Aldrich, Inc.). Finally, the samples were counterstained with hematoxylin, dehydrated using the aforementioned alcohols for 3 min each, cleared in two changes of xylene for 5 min, and mounted with xylene-based mounting medium (Dako Denmark A/S). NF-κB expression was based on the percentage of tumor cells exhibiting a positive reaction. Evaluation of histologic slides was performed by two independent pathologists by means of light microscopes (Olympus BX51; Olympus America, Inc., Melville, NY). Discrepancies in results between two pathologists were resolved by a third pathologist.
Statistics
Statistical analyses were performed using the Statistica version 12 software (StatSoft, Inc., Tulsa, OK, USA). Comparison of expression values between normal (control) and cancer groups was performed by the non-parametric Mann–Whitney U test and comparison between three or more groups was performed by the Kruskal–Wallis test followed by Dunn’s multiple comparison test, and p values <0.05 were considered as statistically significant.
Results
Clinicopathological characteristics
Clinicopathological features of all 119 patients diagnosed with primary invasive BC are listed in Table 1. The age of enrolled female patients ranged between 29 and 84 years with an average of 59.0 years (standard deviation (SD) = 12.9 years). The majority of the group (70.6%) consisted of patients over 50 years of age. Most common recognition (99/119) was ductal carcinoma. Postmenopausal women (89/119) predominated over premenopausal. More than half of a total of 119 BC patients (59.7%) showed no lymph node metastasis, and none had distant metastases.
Clinical characteristics of female patients.

M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; Max: maximum value.
NF-κB expression
The mean percentage value of NF-κB expression in BC tissue (90.1 ± 21.8) was higher than in the corresponding healthy control samples (83.2 ± 23.4). Characteristics of NF-κB expression are shown in Table 2. The difference between experimental and control groups was statistically significant (p = 0.025; Mann–Whitney U test; Figure 1).
Expansion characteristics of tested markers.

NF-κB: nuclear factor kappa B; ER: estrogen receptors; PR: progesterone receptors; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.

Comparison of NF-κB expression in experimental and control groups—Mann–Whitney U test result.
NF-κB and clinicopathological characteristics
Tables 3–9 and Figures 2–8 show the relationship between NF-κB expression and clinicopathological factors including patients age (Table 3 and Figure 2), histological subtype of BC (Table 4 and Figure 3), menopausal status (Table 5 and Figure 4), side of the body affected by the tumor (Table 6 and Figure 5), tumor location (Table 7 and Figure 6), tumor size (Table 8 and Figure 7), and lymph node metastases (Table 9 and Figure 8). NF-κB activity showed no significant correlation with above-mentioned features (p > 0.05; Kruskal–Wallis/Mann–Whitney U tests).
Primary expression statistics of NF-κB marker in different age groups—Kruskal–Wallis test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.
Basic statistics of NF-κB marker expression in groups of patients with a different diagnosis—Kruskal–Wallis test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.
Basic statistics of NF-κB marker expression in groups of patients with different menopausal status—Mann–Whitney U test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.
Basic statistics of NF-κB marker expression in groups of patients with different sides of the body affected—Mann–Whitney U test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.
Basic statistics of NF-κB marker expression in groups of patients with a different tumor location—Kruskal–Wallis test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; Max: maximum value.
Kruskal–Wallis test: p = 0.371.
Basic statistics of NF-κB marker expression in groups of patients with a different tumor size—Kruskal–Wallis test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.
Basic statistics of NF-κB marker expression in groups of patients with different lymph node metastases—Kruskal–Wallis test result.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.

Expression comparison of NF-κB in different age groups—Kruskal–Wallis test result.

Comparison of NF-κB expression in groups of patients with different diagnosis—Kruskal–Wallis test result.

Comparison of NF-κB expression in groups of patients with different menopausal status—Mann–Whitney U test result.
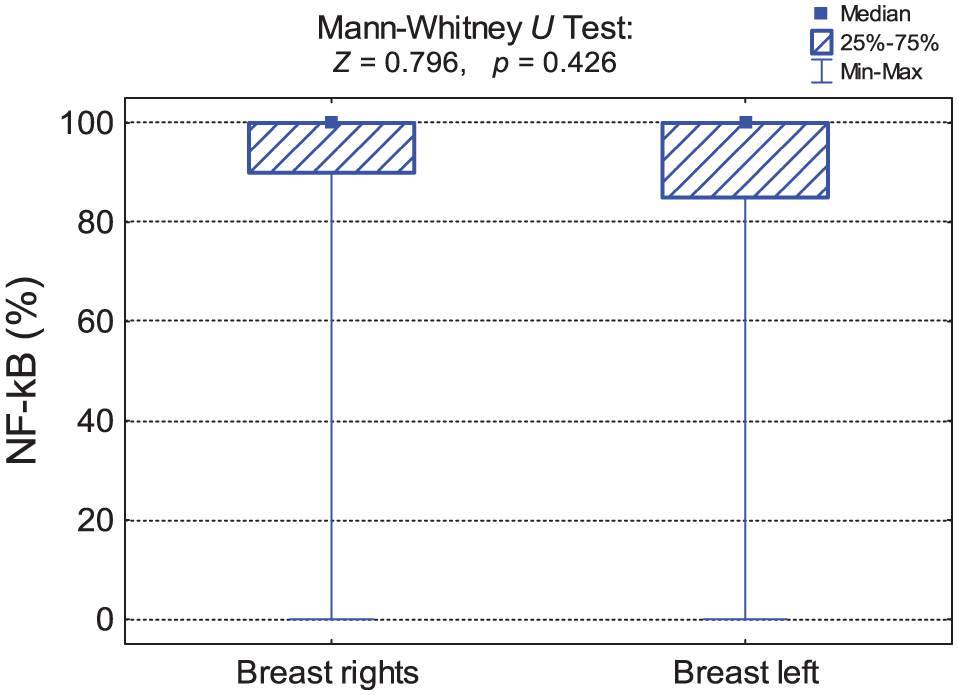
Comparison of NF-κB expression in groups of patients with different sides of the body affected—Mann–Whitney U test result.

Comparison of NF-κB expression in groups of patients with a different tumor location—Kruskal–Wallis test result.

Comparison of NF-κB expression in groups of patients with different tumor sizes—Kruskal–Wallis test result.

Comparison of NF-κB expression in groups of patients with different lymph node metastases—Kruskal–Wallis test result.
The relationship between NF-κB and ER and PR expression
In our study, we found no relevant correlation between ER/PR and NF-κB expression (Table 10 and Figure 9). NF-κB activity within ER-positive and ER-negative as well as PR-positive and PR-negative groups showed no significant differences (p > 0.05; Mann–Whitney U test; Figure 9).
Basic statistics of NF-κB marker expression in groups of patients with a different ER and PR expression.

NF-κB: nuclear factor kappa B; M: mean; SD: standard deviation; Me: median; Q1: lower quartile (25%); Q3: upper quartile (75%); Min: minimum value; max: maximum value.

Comparison of NF-κB expression in groups of patients with a different ER and PR expression—Mann–Whitney U test result.
Discussion
NF-κB is critical for organ development in an early post-natal stage, although activation may become abberant during aging, chronic inflammatory process, carcinogenesis, and cancer progression.4,7,8 Wide range of malignancies including hamatopoietic proliferations as well as solid tumors shows NF-κB alterations.4,9 Mammary glands also require NF-κB expression to develop. 10 Cyclin D1 activation induced by NF-κB tends to be crucial for normal mammary gland growth and BC development. 4
Accumulated data provide evidence for promoting the role of NF-κB hyperactivity in cell transformation. A study using an animal model in vivo and human mammary epithelial cells in vitro showed that increased NF-κB expression is an early event resulting from carcinogen treatment. NF-κB activation preceded malignant transformation. 11
In our study, the specimens obtained from BC patients were analyzed. We assessed NF-κB expression detected by immunohistochemical staining (Figure 10). In this report, we show that NF-κB is expressed at a higher level in breast tumor samples than in the corresponding healthy control. Consistent with these findings, increased activity of NF-κB has been demonstrated in many BC cell lines and breast carcinoma.12,13

Positive nuclear and cytoplasmic immunohistochemical staining for NF-κB in cells of invasive ductal breast carcinoma; DAB was used as a reaction substrate and hematoxylin for counterstaining 400× magnification.
NF-κB and clinicopathological aspects
It is suggested that inflammation within the microenvironment of the tumor increases its aggressiveness. 14 Potential association of augmented NF-κB expression with carcinogenesis, invasion, and metastasis has been reported.15,16 However, the correlation between NF-κB expression and clinicopathological aspects has been rarely investigated. Our research group analyzed factors which may correspond with NF-κB hyperactivation. Obtained results revealed no significant correlation between clinicopathological features and NF-κB expression.
According to Zhou et al., 5 NF-κB expression in BC was significantly correlated with size of the tumor, tumor grade, and tumor–node–metastasis (TNM) stage. Other clinicopathological factors such as age, histological subtype, or menopause showed no association.
NF-κB and ER and PR expression
Up to 75% of BC express ER. 17 ER remains as a useful biomarker to predict the patient’s outcome and response to hormonal therapy. Although endocrine treatment is likely to be effective in ER-positive tumors, nearly half of them fail and develop metastatic recurrence. It is suggested that hormonal therapy failure may be connected with many mechanisms which are influenced by NF-κB signaling pathway. Aberrant or constitutive activation of NF-κB can lead to hormone-independent growth (loss of ER expression) and failure of adjuvant treatment. Additionally, hyperactive NF-κB may be associated with the development of endocrine resistance in ER-positive tumors.
Interestingly, there are many hypotheses explaining positive as well as negative correlation between NF-κB and steroid receptors such as ER and PR. However, in contrast, here, we report a lack of correlation between NF-κB activity and ER/PR expression.
Generally, there is an inverse correlation between NF-κB and ER. NF-κB represses an expression of ER in BC tissue and cell lines.18,19 Besides inhibitory crosstalk, NF-κB and ER may mutually interact in a positive manner. According to Frasor et al.,6,20 synergistically elevated messenger RNA (mRNA) transcription results from stabilizing the binding of ER/p65 complex to its response elements.
Nearly 50% of BC show PR expression. 21 As reported in the literature, ER activates PR expression.22,23 Kalkhoven et al. demonstrated that RelA (p65)—member of NF-κB family—and PR repress each other’s activity as a result of direct interaction. Inhibitory crosstalk can also involve tumor necrosis factor alpha (TNF-α). TNF-α-induced NF-κB expression may be repressed by PR, and PR repression may follow NF-κB activation triggered by TNF-α. 24
Footnotes
Acknowledgements
Anil Kumar Agrawal, Siddarth Agrawal, Ewa Pielka, Artur Lipinski, and Michal Jelen participated in study design and coordination, carried out the molecular studies, and prepared draft of the manuscript; Wojciech Kielan also participated in the study design and coordination. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by Bioethics Committee of the Wroclaw Medical University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Polish National Science Center (grant no. 2015/19/N/NZ5/00001) and Wroclaw Medical University.
