Abstract
Guided treatments with nanoparticles and radiotherapy are a new approach in cancer therapy. This study evaluated the beneficial antitumor effects of γ-radiation together with gallium nanoparticles against solid Ehrlich carcinoma in female mice. Gallium nanoparticles were biologically synthesized using Lactobacillus helveticus cells. Transmission electron microscopy showed gallium nanoparticles with size range of 8−20 nm. In vitro study of gallium nanoparticles on MCF-7 revealed IC50 of 8.0 μg. Gallium nanoparticles (0.1 mg/kg body weight) were injected intraperitoneally daily on the seventh day of Ehrlich carcinoma cells inoculation. Whole-body γ-radiation was carried out at a single dose of 0.25 Gy on eighth day after tumor inoculation. Biochemical analysis showed that solid Ehrlich carcinoma induced a significant increase in alanine aminotransferase activity and creatinine level in serum, calcium, and iron concentrations in liver tissue compared to normal control. Treatment of Ehrlich carcinoma–bearing mice with gallium nanoparticles and/or low dose of γ-radiation exposure significantly reduced tumor volume, decreased alanine aminotransferase and creatinine levels in serum, increased lipid peroxidation, and decreased glutathione content as well as calcium and iron concentrations in liver and tumor tissues with intense DNA fragmentation accompanied compared to untreated tumor cells. Moreover, mitochondria in the treated groups displayed a significant increase in Na+/K+-ATPase, complexes II and III with significant reduction in CYP450 gene expression, which may indicate a synergistic effect of gallium nanoparticles and/or low dose of γ-radiation combination against Ehrlich carcinoma injury, and this results were well appreciated with the histopathological findings in the tumor tissue. We conclude that combined treatment of gallium nanoparticles and low dose of gamma-radiation resulted in suppressive induction of cytotoxic effects on cancer cells.
Keywords
Introduction
Experimental tumors have great importance for the purposes of modeling, and Ehrlich ascites carcinoma (EAC) is one of the most common tumors which resembles human tumors that are more sensitive to chemotherapy treatment because they are undifferentiated and they have a rapid growth rate. 1 Cancer has become a hot topic in medicine since it is a major cause of death in both the developed and developing countries, and it is now only secondary to myocardial infarction. 2 Chemotherapeutic agents that abrogate tumor cells, but can also cause dangerous side effects as they not only affect the proliferated cells but also the normal cells, 3 currently achieve the most effective antitumor treatment. With this in mind, novel strategies for treating malignancies are being developed on the experimental level in order to be applied in clinical setting. Nanoparticles produced by a biogenic enzymatic process could be the answer, which provide new approach to synthesize new metal-containing agents. The metallic nanoparticles prepared by chemical and physical methods are the most promising as they show good antimicrobial and antitumor properties. 4 Despite that the latter methods are able to produce large quantities of nanoparticles with a defined size and shape in a relatively short time, they are complicated, outdated, costly, inefficient, and produce hazardous toxic wastes to the environment and human health. An enzymatic process or called “green” route is energy saving and environment friendly. The particles generated by these processes have higher catalytic reactivity, greater specific surface area, and an improved contact between the enzyme and metal salt under study due to the bacterial carrier matrix.5,6
Gallium (Ga) comes after platinum as the second metal ion to be used in cancer treatment. Its activities are numerous and various. It modifies three-dimensional structure of DNA and inhibits its synthesis and modulates protein synthesis; also, it inhibits the activity of a number of enzymes, such as ATPases, DNA polymerases, ribonucleotide reductase, and tyrosine-specific protein phosphatase. Ga alters plasma membrane permeability and mitochondrial functions. 7 Moreover, scientists tended to combine gallium salts with other therapeutic strategies hoping that this will improve the results of the treatment. 8
Radiotherapy is one of the most important modalities for tumor regression. Exposure to high doses of ionizing radiation increases the formation of reactive oxygen species (ROS) in high quantity, which is harmful to normal organs, while exposure to low dose of ionizing radiation is normal phenomena and found to have some beneficial effects. Total body radiation to low dose of gamma radiation (0.02–0.25 Gy) has been reported to have antitumor effects and induces tumor growth delay and antimetastatic effects. 9 In some parts of the globe, the level of natural background radiation is significantly higher than the world average with no adverse health effects. 10 Ionizing radiation is considered a natural resource affecting human life, as well as an important therapeutic and diagnostic tool in medicine. There is an increasing interest in the biological effects of low-dose radiation from natural and environmental exposure. Thus, low-dose irradiation induces diverse effects on biological functions, such as alterations in genomic DNA and the cell cycle.11 –13 Low dose of gamma radiation modified apoptotic gene expressions such as upregulation of Bcl-2, an anti-apoptotic molecule, and downregulation of BAX, a pro-apoptotic gene. 14 Accordingly, this study aimed to evaluate the antitumor activity of biologically synthesized gallium nanoparticles (GaNPs) and the synergistic effect of GaNPs together with low-dose gamma radiation against Ehrlich carcinoma (EC) in female mice.
Materials and methods
Chemicals
Gallium nitrite (Sigma-Aldrich Company, Sigma-Aldrich, USA) dissolved in deionized water was used in the preparation of GaNPs.
Microorganism and culture conditions
Lactobacillus helveticus ATCC 7995 used in this study was obtained from the Microbiological Resources Center (Cairo, Egypt) cultured in de man, rogosa and sharpe (MRS) broth/agar following ATCC product sheet with the standard microbiological procedures in the laboratory with shaking at 150 r/min for 12 h at 30°C. The culture was stored at −25°C in 20% sterile glycerol and liquid MRS (Oxoid Ltd, Basingstoke, UK).
Cell line
The EAC cell line
The EAC cell line used in this study was supplied by The Egyptian National Cancer Institute (NCI), Cairo University (Giza, Egypt). The cell line was maintained by weekly intraperitoneal injection of 2.5 million cells in a volume of 0.2 mL per mouse in female Swiss albino mice. The EAC cells were diluted using physiological sterile saline solution, and cells were counted using the Bright-LineTM hemacytometer.
Human breast cancer (MCF-7) cell line
Human breast cancer (MCF-7) cell line was obtained from the Egyptian National Cancer Institute, Cairo University.
Animals
Female Swiss albino mice weighing 22 ± 3 g purchased from the animal house breeding unit of the Nile Company for Pharmaceuticals and Chemical Industries (Cairo, Egypt) were used in this study. The animals were maintained on a commercial standard pellet diet and tap water ad libitum. Animal maintenance and treatments were conducted in accordance with the National Institute of Health Guide for Animal, as approved by Institutional Animal Care and Use Committee (IACUC).
Tumor transplantation
To produce Ehrlich solid tumor in female Swiss albino mice, 0.1 mL of EAC cells (2 × 106 cells/mouse) was inoculated intramuscularly in the right thigh of the lower limb of female mouse.
Radiation facility
Mice whole-body gamma irradiation was performed at The National Center for Radiation Research and Technology (NCRRT), Atomic Energy Authority (Cairo, Egypt) using the 137Cesium biological irradiator source (gamma-cell-40), Atomic Energy of Canada Limited (Chalk River, ON, Canada). After tumor inoculation, mice were exposed to a single dose of 0.25 Gy at a dose rate of 0.423 Gy/min.
Methods (work flow design)
Chemical studies
Biosynthesis of GaNPs
Lactobacillus helveticus was inoculated into sterile MRS 250-mL Erlenmeyer flask containing 100 mL sterile nutrient broth. The cultured flasks were incubated in a rotating shaker set at 200 r/min for 48 h at room temperature. The culture was centrifuged at 6000 r/min for 10 min. The biomass and supernatant were separated and the supernatant was used for studying extracellular production of GaNPs by mixing it with filter-sterilized GaNO3 solution at 1 mM final concentration. All the reaction mixtures were incubated on rotating shaker (200 r/min) at room temperature for a period of 72 h. Visual observation was conducted periodically to check for the nanoparticle formation. Further characterization was conducted for nanoparticle generated through extracellular methods. 15 The resulting aqueous solution was filtered through a 0.22 μm Millipore filter before use.
Characterization of GaNPs
Transmission electron microscopy
GaNPs’ size and shape were analyzed by transmission electron microscopy (TEM). TEM sample was prepared by placing a drop of GaNPs suspension on carbon-coated copper grids and allowing water to evaporate. The size of nanoparticles was determined from TEM micrographs of JEOL model 1200EX. The software (Advanced Microscopy Techniques, Danvers, MA, USA) for the digital TEM camera was calibrated for size measurement of the nanoparticles. 16
Ultraviolet-visible absorption
GaNPs spectrum absorbance was analyzed by ultraviolet-visible (UV-VIS) spectrophotometer using Jenway UV spectrophotometer model 6505 in visible light at range of 200–600 nm. 17
Fourier-transform infrared spectroscopy
Samples of GaNPs and the biological extract used in the nanoparticle synthesis were analyzed by Fourier-transform infrared spectroscopy (FTIR). Infrared (IR) absorption of the functional groups may vary over a wide range. The device used was VERTEX 70/70v FTIR spectrometers (Bruker, India). 18
Biochemical studies
Evaluation of therapeutic antitumor efficacy of a new compound GaNPs
This study was designed to comprise a series of in vitro and in vivo investigations as follows.
In vitro study
GaNPs toxicity on MCF-7 cell line: The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was employed to determine the cytotoxicity effect of GaNPs on the human breast cancer (MCF-7) cell line based on the mitochondrial dehydrogenase conversion of the MTT to a blue formazan product in the viable cells by an enzyme present in the mitochondria of viable cells. The colorimetric changes of blue formazan dissolved in dimethyl sulfoxide (DMSO) were measured spectrophotometrically at 595 nm using enzyme-linked immunosorbent assay (ELISA) plate reader DV990BV4 microplate reader from Gio. de Vita E C. S.r.l (Rome, Italy) and data were analyzed using 990win6 software. 19
In vivo studies
Acute toxicity studies
Determination of GaNPs LD50 in female mice: Determination of the LD50 of GaNPs as a new compound is an essential initial step in evaluation of the toxic characteristics of the compound in vivo and provides information on health hazards likely to arise from short-term exposure to the drugs. A total of 40 female Swiss albino mice were divided into four groups and administrated with GaNPs in elevated doses (0.1, 5, 10, 30, 70, 90, and 100 mg/kg body weight) through intraperitoneal injection. Mice were observed for 1–7 days after administration, and LD50 was determined. 20 The optimum safe dose of GaNPs to be used is 1/10 of the determined LD50 value.
2. Investigation of the antitumor activity
Experimental design: A total of 80 female Swiss albino mice were randomly divided into eight equal groups, each of 10 mice as follows:
Group 1 (control): Mice were intraperitoneally injected with 0.2 mL of sterile saline.
Group 2 (Ehrlich solid carcinoma (EC)): Mice were subcutaneously injected at the right thigh of the lower limb with 0.1 mL (2 × 106 cells) of EAC cells.
Group 3 (GaNPs): Mice were intraperitoneally injected with 0.2 mL (1 mg/kg body weight) of GaNPs started from the seventh day after EAC inoculation for 21 days.
Group 4 (irradiation): Mice whole body were exposed to a single dose of (0.25 Gy) gamma irradiation at the eighth day after EAC inoculation.
Group 5 (GaNPs + irradiation): Mice were intraperitoneally injected with GaNPs as described in group 3 and exposed to gamma irradiation as mentioned in group 4.
Group 6 (EC + GaNPs): Ehrlich solid tumor–bearing mice were intraperitoneally injected with GaNPs as described in group 3.
Group 7 (EC + irradiation): EC-bearing mice were exposed to gamma radiation as described in group 4.
Group 8 (EC + GaNPs + irradiation): EC-bearing mice were injected intraperitoneally with GaNPs as described in group 3 and exposed to gamma radiation as described in group 4.
At the end of 4 weeks, all animals were sacrificed 24 h after the last treatment under urethane anesthesia. Blood samples were collected, sera were separated, and tumors were harvested, perfused with cold isotonic saline, dried, and freezed at −20°C until biochemical analysis.
Tumor volume monitoring
The volume of solid tumor was measured by the Vernier caliper after inoculation with EAC cells. The tumor volume was calculated by the following equation: tumor volume = 1/2 (length × width2), where length is the greatest longitudinal diameter and the width is the greatest transverse diameter. 21
Biochemical analysis
The activity of alanine transaminase (ALT) was measured colorimetrically in serum 22 using assay kit (QCA, Amposta, Spain). The level of creatinine in serum was determined using colorimetric assay. 23 Total iron and calcium concentrations were measured colorimetrically in liver and tumor tissues,24,25 respectively. Reduced glutathione (GSH) was determined in tumor tissue according to the method of Beutler et al. 26 Lipid peroxide level was evaluated by determination of malondialdehyde (MDA) concentration in tumor tissue according to the method of Yoshioka et al. 27 Na+/K+-ATPase activity was measured in tumor tissue. 28 The reaction mixture for Na+/K+-ATPase assay contained 5.0 mM MgCl2, 80.0 mM NaCl, 20.0 mM KCl, and 40.0 mM Tris-HCl, pH 7.4, in a final volume of 200 μL. After 10 min of pre-incubation at 37°C, the reaction was initiated by addition of adenosine triphosphate (ATP) to a final concentration of 3.0 mM and was incubated for 20 min. Controls were carried out under the same conditions with the addition of 1.0 mM ouabain. Na+/K+-ATPase activity was calculated by the difference between the two assays. Released inorganic phosphate (Pi) was measured as described by Chan et al. 29 Specific activity of the enzyme was expressed as nanomole of Pi released per minute per milligram of protein.
Gene expression of complexes II and III and cytochrome P450 2E1
Isolation of mitochondria
The mitochondria of tumor tissue cells were isolated by differential centrifugation using a buffer composed of 210 mM mannitol, 70 mM sucrose, and 5 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES; MSH) buffer, pH 7.5. 30 The level of mouse CYP450 (CYP2E1) was evaluated by ELISA using a kit from Elabscience, Inc (Hubei, China).
RNA isolation and reverse transcriptase–polymerase chain reaction
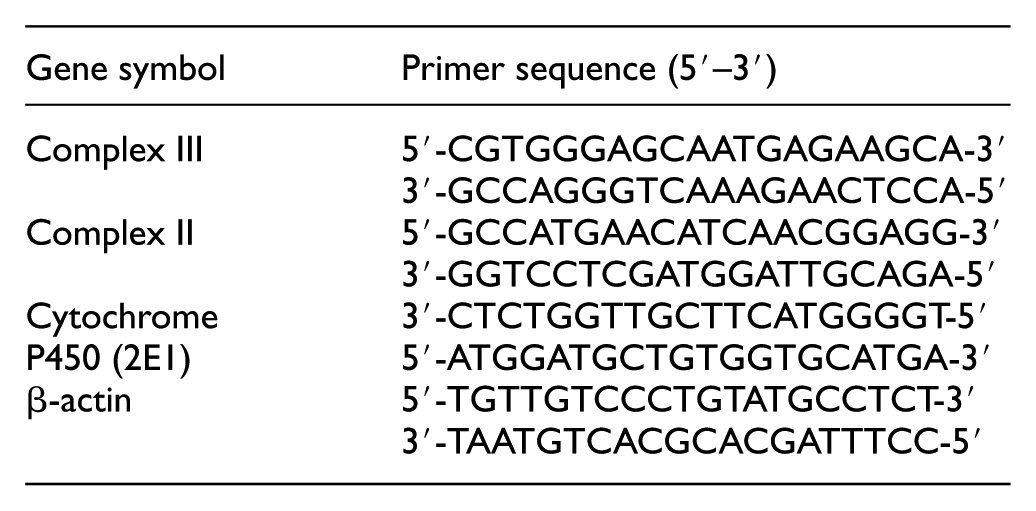
To investigate the changes in messenger RNA (mRNA) expression of complexes II and III and cytochrome P450 (2E1), 12 mg of isolated mitochondria was lysed in 1 mL of TRIzol reagent (Gibco BRL, Life Technologies, Grand Island, NY, USA). Total RNA was isolated in accordance to the manufacturer’s instructions, and its integrity was confirmed by agarose gel electrophoresis. Total RNA concentration was quantified by spectrophotometry at 260 nm in a SmartSpec 300 Spectrophotometer (Bio-Rad, Hercules, CA, USA). Total RNA was converted to complementary DNA (cDNA) using the following sense and antisense primers; the polymerase chain reaction (PCR) primers used in this study were purchased from Bioneer (Daejeon, South Korea).
Sequence of the primers used for real-time PCR is as follows:
DNA synthesis using oligo-dT was performed in total volume of 10 μL at 70°C for 10 min and chilled on ice. Then, reaction mix was added (first-strand buffer, 10 mM dithiothreitol (DTT), 250 mM of deoxyribonucleotide triphosphates (dNTPs), and 200U M-MLV Reverse Transcriptase (Gibco BRL)). The reaction mix was incubated at 37°C for 60 min and at 70°C for 10 min. Quantitative reverse transcriptase (RT)-PCR was performed in duplicate in a 25 μL reaction volume consisting of 2× SYBR Green PCR Master Mix (Applied Biosystems, Waltham, MA, USA; 900 nM of each primer and 2–3 μL of cDNA). Amplification conditions were 2 min at 50°C, 10 min at 95°C, and 40 cycles of denaturation for 15 s, and annealing/extension at 60°C for 10 min. Data from real-time assays were calculated using the v1.7 sequence detection software from PE Biosystems (Foster City, CA, USA). Relative expression of target mRNA was calculated using the comparative CT method. All values were normalized to the β-actin gene (R). 31
DNA fragmentation
DNA samples were analyzed on 2% agarose gel electrophoresis. DNA samples were loaded with loading buffer and electrophoresed on agarose gel in 40 mM Tris, 20 mM Boric acid, and 1 mM ethylenediaminetetraacetic acid (EDTA; TBE) at 100 V for 45 min and visualized by ethidium bromide staining. 32
Histopathological examination
At the time of sacrifice, the tumors were excised from the animals. Samples from each tumor tissues were fixed in 10% formalin and embedded in paraffin. Sections of 5 µm thickness were cut and stained with hematoxylin and eosin 33 and examined by light microscope for histopathological investigation.
Statistical analysis
Data are reported as mean ± standard error (SE). Data were analyzed using one-way analysis of variance (ANOVA) followed by least significant difference (LSD) as a post hoc test. The level of significance between mean values was set at p < 0.05. All statistical analyses were performed using SPSS software (Version 20.0).
Results
Chemical studies
Characterization of new compound GaNPs
Transmission electron microscope of GaNPs
Transmission electron microscope (TEM) photos clearly shows the formed GaNPs morphology and size distribution. Size distribution of GaNPs was of diameter ranging from 8 to 20 nm with a relatively narrow particle size distribution (Figure 1).

TEM image analysis of gallium nanoparticles synthesized by L. helveticus.
UV-VIS spectroscopy
GaNPs absorbance was scanned using the visible range of UV-VIS spectrophotometer. The absorption spectrum of GaNPs exhibited a peak at 265 nm assigned to surface plasmon resonance of the nanoparticles (Figure 2).

Ultraviolet-visible absorption spectrum of gallium nanoparticles synthesized by L. helveticus.
FTIR
The FTIR analysis of GaNPs and the extracellular extract of L. helveticus used for GaNPs green synthesis showed different stretches of bonds at different peaks as presented in Figure 3(a) and (b). FTIR spectra of the broth containing bacterial extracellular metabolites showed major strong peaks and some weak peaks, a strong signal at 3318.77 cm−1 (represents hydroxyl group), strong signal at 1636.93 cm−1 (represents amide I bond of proteins), and a strong peak at 602.31 cm−1 (represents signal bond of C–H and C–N bonds). The FTIR analysis of GaNPs showed shifting in wave number representing –OH group at 3321.45 cm−1 also in the signal of amide I of proteins at 1637.29 cm−1 and at 595.64 cm−1 signal of C–H and C–N bonds.

(a) FTIR spectrum of L. helveticus extracellular metabolites. (b) FTIR spectrum of GaNPs synthesized by L. helveticus extracellular metabolites.
Biochemical studies
In vitro studies
GaNPs cytotoxicity on MCF-7 cell line
GaNPs when incubated with MCF-7 cell line exerts cytotoxic effect in a dose-dependent manner resulted in a significant reduction of cell proliferation after 24 and 48 h of incubation. Data showed that 8 μg of GaNPs caused 50% decrease in the viable cells count compared to the corresponding control.
In vivo studies
Acute toxicity studies
The LD50 of GaNPs was found to be 10 mg/kg body weight. Accordingly, the safe dose was calculated by dividing LD50 by 10 to be used in the in vivo study of GaNPs against solid Ehrlich tumor–bearing mice.
Tumor volume monitoring
Ehrlich solid tumor size monitoring cleared that GaNPs have anti-proliferation effect on solid tumor (Figure 4). The inoculation of EC cells in the thigh region of female mice produced a tumor with a mean size of 830 ± 11.8 mm3 on the 10th day after inoculation of 2.5 million of EC. Tumor size proceeds with time reaching 2940 ± 32 mm3 at the 20th of inoculation. GaNPs and/or exposure to low dose of γ-radiation (at 17th day) caused a marked suppression of tumor growth compared to corresponding EC group.

Effects of gallium nanoparticles (GaNPs) and/or γ-radiation exposure 0.25 Gy (R) on the tumor volume (mm3) of mice bearing solid tumor (EC).
Biochemical analysis
EC-bearing mice (EC group) showed a significant elevation of serum ALT and creatinine levels as well as calcium and iron concentrations in liver and tumor tissues as presented in Table 1. Administration of GaNPs and/or exposure to single low dose of gamma radiation (0.25 Gy; R) to EC-bearing mice (EC + GaNPs, EC + R, and EC + GaNPs + R groups) induced significant reduction (p < 0.05) in ALT and creatinine levels in serum accompanied with a significant decrease in total iron and calcium concentrations in liver and tumor tissues upon comparison with EC group.
Effect of gallium nanoparticles (GaNPs) and/or low dose of γ-radiation exposure (0.25 Gy; R) on ALT and creatinine levels in serum as well as total iron and calcium content in the liver and tumor tissues.
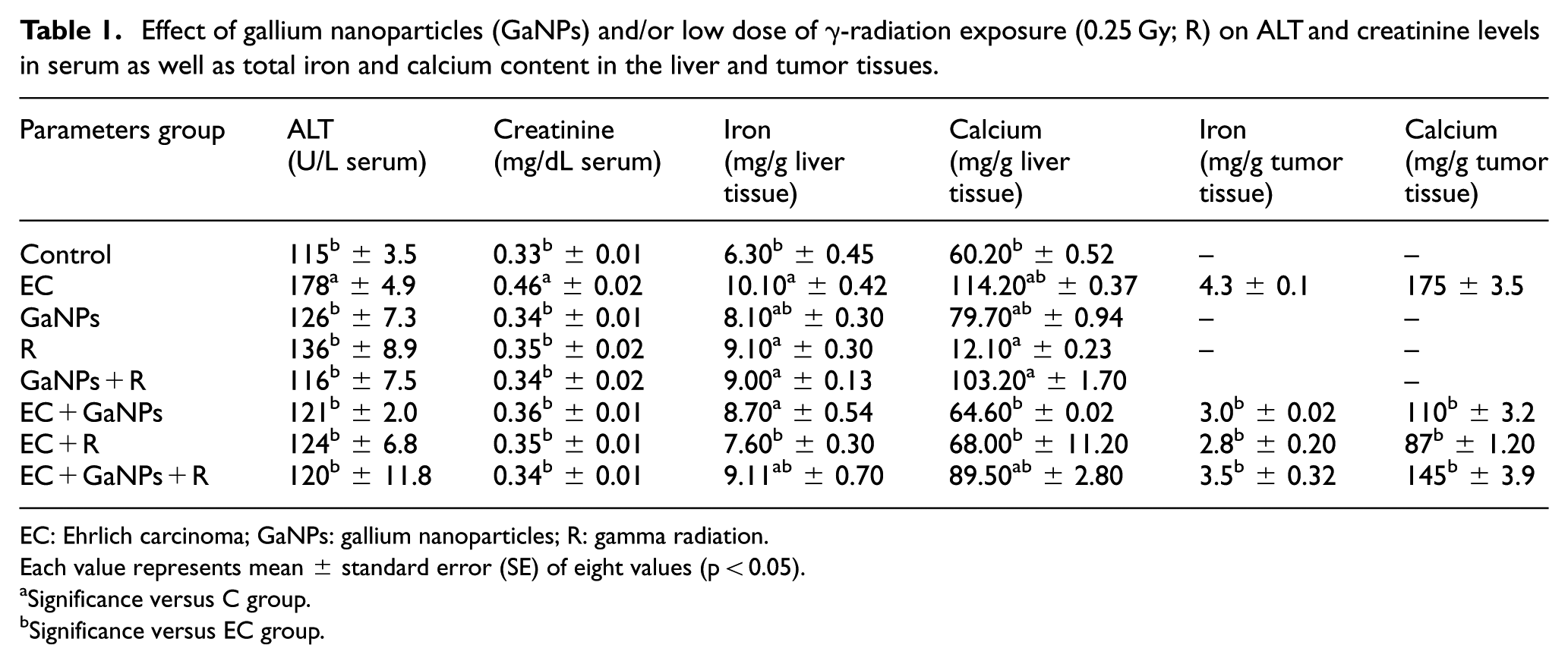
EC: Ehrlich carcinoma; GaNPs: gallium nanoparticles; R: gamma radiation.
Each value represents mean ± standard error (SE) of eight values (p < 0.05).
Significance versus C group.
Significance versus EC group.
The results presented in Table 2 and Figure 5 revealed that the treatment of EC-bearing mice with GaNPs and/or exposure to single low dose of gamma radiation (0.25 Gy) caused a significant increase in the level of MDA level, Complex II, Complex III (EC + GaNPs, EC + R, and EC + GaNPs + R groups), and Na+/K+-ATPase (EC + GaNPs + R and EC + R groups). While the activity of GSH, CYP450 (EC + GaNPs + R, EC + R, and EC + GaNPs groups) and Na+/K+-ATPase (EC + GaNPs group) in the tumor tissue was significantly decreased (p < 0.05) upon comparison with EC tumor group.
Effect of gallium nanoparticles (GaNPs) and/or low dose of γ-radiation exposure (0.25 Gy; R) on CYP450 messenger RNA (mRNA) expression and Complex II and Complex III levels in the tumor tissue.

mRNA: messenger RNA; EC: Ehrlich carcinoma; GaNPs: gallium nanoparticles; R: gamma radiation.
Each value represents mean ± SE of eight values (p < 0.05).
Significance versus C group.
Significance versus EC group.

Effect of gallium nanoparticles (GaNPs) and/or low dose of γ-radiation (0.25 Gy) on MDA, reduced glutathione, and Na+/K+-ATPase activity in tumor tissue of EC-bearing mice.
DNA fragmentation results
Gel electrophoresis for extracted DNA from Ehrlich solid tumor tissue represented in Figure 6 revealed that DNA of solid tumor of EC group showed intact non-fragmented DNA. DNA of solid tumor of EC mice treated with GaNPs and/or gamma radiation of EC + GaNPs, EC + R, and EC + GaNPs + R groups was fragmented into discrete bands which is a characteristic feature of apoptosis cell death mode.
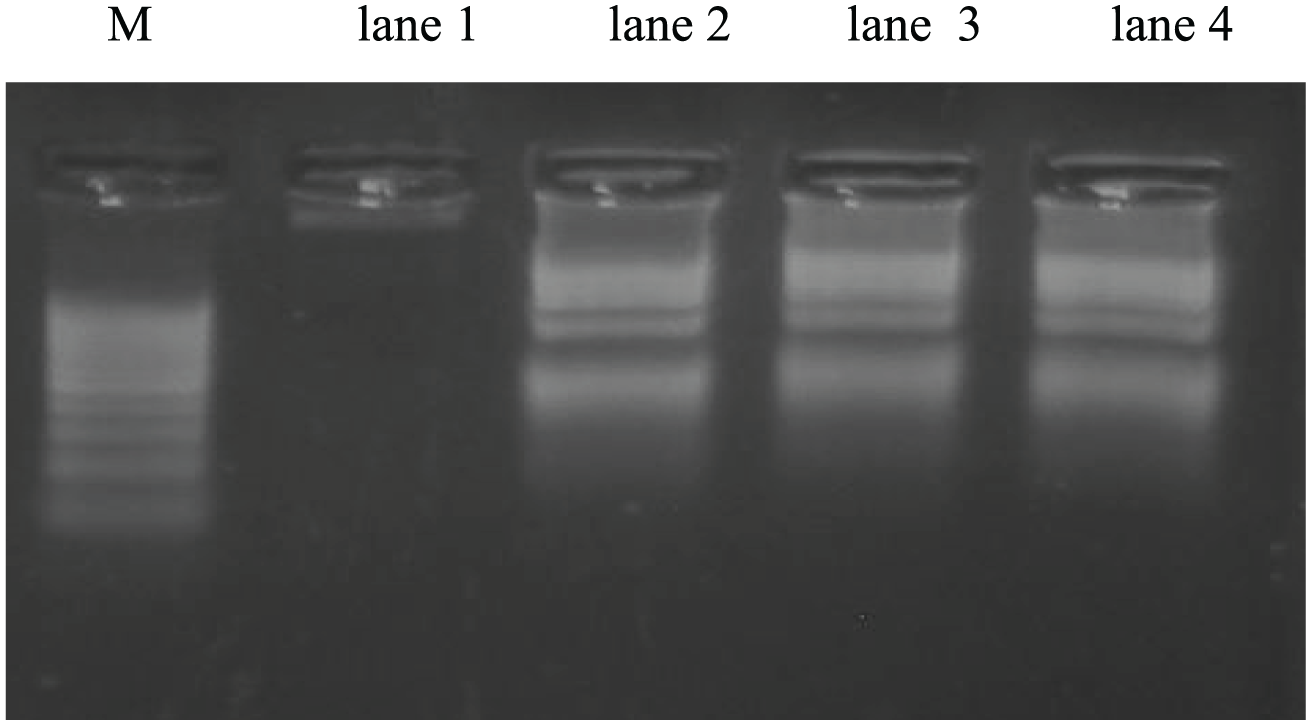
Gel electrophoresis of DNA extracted from tumor tissue. Lane M: DNA marker, lane 1: Ehrlich solid tumor (EC), lane 2: EC treated with GaNPs, lane 3: EC group treated with gamma irradiation, and lane 4: EC group treated with GaNPs and gamma irradiation.
Histopathological findings
Histopathological examination of solid EC under the light microscope showed massive aggregation of tumor cells spread within the thigh muscular tissue, with muscle fibers intermingled with tumor areas. Tumor cells (EC group) are intact in between the skeletal bundles (Figure 7(a)). Treatment of female mice bearing EC with GaNPs (EC + GaNPs group) showed moderate form of necrosis found in all over the tumor cells (Figure 7(b)). Treatment of animals bearing EC with gamma radiation (EC + R) recorded that there is necrosis found in mild form all over the tumor cells (Figure 7(c)). Treatment of animals bearing EC with GaNPs and gamma radiation (EC + GaNPs + R group) revealed that there was a severe form of necrosis in the tumor cells (Figure 7(d)).

Photomicrographs of sections in solid Ehrlich carcinoma (EC) stained by H&E (hematoxylin and eosin). (a) Section in muscular tissue of EC-bearing mice. (b) Section in muscular tissue of EC treated with GaNPs. (c) Section in muscular tissue of EC treated with gamma irradiation. (d) Section in muscular tissue of EC treated with GaNPs and gamma irradiation.
Discussion
Combining chemotherapy with radiotherapy improves cancer treatment results for more comprehensive attack. Chemoradiotherapy is able to enhance efficacy of chemotherapy and radiotherapy.
Guided treatments with nanoparticles and radiotherapy are a new approach in cancer therapy. Nanoparticles possess the ability to permeabilize the cells efficiently due to their small size, which facilitates activity and in vivo distribution. Smaller nanoparticles show higher accumulation at tumor sites and prolong in vivo half-life due to their avoidable capture by the reticuloendothelial system.4,5 Previous study has shown that green biologically based methods using microorganisms and plants to synthesize nanoparticles are safe, inexpensive, and an environment-friendly alternative. 34
This study aimed to evaluate the antitumor effect of biologically synthesized GaNPs with or without low dose of gamma radiation (0.25 Gy) in vitro and in vivo against EC solid tumor in female mice. The efficiency of GaNPs as anticancer agent and/or low dose of gamma radiation as combined treatment was evaluated in vitro on MCF-7 viable cell count and in vivo measuring tumor size, biochemical parameters, and histopathological studies.
In the current investigation, the biologically synthesized GaNPs were characterized via many analysis methods such as TEM, UV/VIS absorbance spectroscopy, and FTIR. Biologically synthesized GaNPs revealed nanoparticles of diameter size ranging from 8 to 20 nm. In addition, UV/VIS shows that the absorption spectra for GaNPs were scanned at 265 nm in agreement with others 35 who found that gallium is sensitive to UV radiation below 365 nm wavelength.
Nanoparticles less than 100 nm is useful for biological applications. Particle size is an important property, which may influence the biological activity of nanoparticles and has been suggested as a key factor through the interaction with charged surfaces. Nanoparticles with different particle size may have different mechanisms of inhibition. As results show, 8 µg of GaNPs in in vitro study challenging was able to reduce the initial viable cell count of MCF-7 breast cell line cancer cells to 50%. This low concentration of IC50 compared to other gallium salts indicated that nanoparticles form of gallium is more cytotoxic and effective against tumor cell growth. Several researches have reported that gallium is taken up by various malignant cells in vitro through cell surface transferrin receptor (TFR)-mediated endocytosis of Ga-transferrin complexes. 36
In addition, as data show, a delay in tumor volume growth was observed in the female mice treated with GaNPs or low dose of gamma radiation and combined treatment of GaNPs with low dose of gamma radiation exposure markedly reduced tumor volume compared to the corresponding controls that pronounced a synergistic effect of such combined treatment. Gallium is capable to inhibit tumor growth, mainly because of its competition to ferric ion. It affects cellular acquisition of iron by binding to transferrin besides its interaction with the iron-dependent enzyme ribonucleotide reductase, deactivating the enzyme through a conformational change resulting in reduced dNTP pools and inhibition of DNA synthesis. Upregulation of ribonucleotide reductase and the abundance of TFRs render tumor cells susceptible to the cytotoxicity of gallium. 37 Gallium may interact with DNA by acting as a competitor with magnesium for DNA binding because Ga’s affinity to DNA is 100 times higher than that of magnesium. 38 Ga may bind to DNA phosphate group and nucleic bases causing DNA structural modifications, which destabilizes DNA helix. Previous investigations showed that gallium activates caspases and induces apoptosis through the mitochondrial pathway. 39 It has also been shown in in vitro study that Ga induces chromatin condensation, which is an early step of apoptosis. 7 In this study, GaNPs inhibited the growth of tumor cells and induced morphological changes typical of apoptosis. The incidence of DNA fragmentation of tumor cells confirmed such morphological findings. Treatment with GaNPs induced ladder-like DNA fragmentation in tumor cells, which is a characteristic of DNA damage. Increased levels of DNA damage could cause the synthesis of a variety of incorrect proteins and therefore impaired cellular function. Most anticancer drugs either natural or synthetic have been known to cause DNA damage or suppress its replication, not necessarily killing the cells directly but inducing apoptosis. During apoptosis, a specific nuclease cuts the genomic DNA between nucleosomes and generates apoptotic chromatin condensation and DNA fragments. 40 However, general histopathological observations agreed to potency of GaNPs in DNA fragmentation of tumor cells. Skeletal muscle is the tissue with the largest mass in the body and consists of post-mitotic cells, which are more prone to accumulate oxidative damage.37,41 Thus, it is highly plausible that the muscle damage associated with muscle soreness causes oxidative damage by enhancement of free radical generation. The cytotoxic effects of free radicals include the oxidative damage of lipid of cell membrane. 41 In this study, examination of tumor tissue sections under the light microscope revealed that administration of GaNPs to female mice bearing tumor (EC + GaNPs group) showed a necrosis in severe form all over the tumor tissue in histopathological findings. This finding is consistent with others 42 who reported that gallium induces cell death via oxidative stress and block the proliferation of bovine retinal endothelial cells (BRECs) which are essential for angiogenesis in endothelial cells. Antiangiogenic effect of GaNPs could be due to its direct inhibitory effect on epithelial cells viability during vascular endothelial growth factor–induced angiogenesis in endothelial cells, thereby inhibiting the development of angiogenic disorders.
Results in this study of mice bearing tumor tissue that received GaNPs showed a marked decrease in GSH level accompanied with increase in MDA levels. The decrease in GSH level could be a result of extreme oxidative damage caused by gallium or due to its utilization during detoxification of the free radicals generated by GaNPs and enhances lipid peroxidation.43,44 The decrease in the level of GSH content is in close relationship with the induction of lipid peroxidation. The antitumor activity of nanoparticles and gallium is suggested to induce their toxicity through oxidative stress and inflammation by generating ROS involved in a variety of different cellular processes ranging from apoptosis and necrosis to cell proliferation and carcinogenesis. 45 It can be concluded that the newly synthesized GaNPs had shown low cytotoxicity to normal cells. This study provides a preliminary insight into the mode of action of the usage of GaNPs in vivo as a therapy of solid carcinoma. Therefore, GaNPs may constitute a promising tool for anticancer therapeutics in the near future.
In this study, significant increase in ALT activity and creatinine level of tumor-bearing mice (EC group) in serum compared to normal healthy control indicates hepatic and renal damage as a result of Ehrlich solid tumor formation. These increase attributed to cancer cell invasion in the vital organs, which causes overload in the liver and renal damage as degenerative changes in the epithelial lining of renal tubules. 46 Administration of GaNPs and/or R to tumor-bearing mice displayed a significant decrease in ALT activity and creatinine level upon its comparison with EC group. The decline in ALT activity and creatinine level may be attributed to the effect of GaNPs against solid tumor that consequently decreases the secreted toxins from tumor. 47
Earlier researches reported that exposure to low dose of gamma radiation (0.25 Gy) modulates ALT activity 48 and suppress high level of creatinine.49,50 They owed that the enhancement effect of low dose of gamma radiation on antioxidant system may modulate damage effect of tumor induction on the liver and kidney.
Hypercalcemia usually happens in advanced cancer and is rare in people with earlier stages of cancer. In this study, high calcium concentration in the liver tissue in EC group compared to normal control group attributed to calcium leak out into the bloodstream from bones, so the level in the blood gets too high. Administration of GaNPs and/or R exposure to tumor-bearing mice significantly decreased calcium concentration in liver and tumor tissues in the treated groups (EC + R, EC + GaNPs, and EC + GaNPs + R) compared to EC group, and this result is consistent with others.51,52 Gallium appears to be a uniquely acting agent for treatment of bone-resorptive conditions reducing accelerated bone loss in patients with cancer. In contrast to other calcium-saving drugs, gallium enhances calcium and phosphorus content of the bone and has direct, non-cytotoxic effects on osteoclasts at markedly low doses, and it is well established that the mechanism involves gallium insertion into the hydroxyapatite matrix protecting it from resorption and improving biomechanical properties of the skeletal system. 52 Low-dose ionizing radiation suppressed intracellular Ca2+ influx. It was found that vRBL-2H3 cell line exposed to ionizing radiation in the range of 0.01–0.5 Gy showed decrease in the levels of calcium influx in a dose-dependent manner. 53
Previous studies showed increased iron metabolism in malignant cells because they undergo intensive cell division. 8 In this regard, our data concerning iron level in the liver tissue showed a significant increase in the level of hepatic iron in EC group in comparison with the control. This finding is consistent with the study of others 54 who found that increase in the oxidative stress due to tumor induction decreases the rate of erythropoiesis in the bone marrow compared to eryptosis in the liver causing imbalance in RBC’s count equilibrium and anemia and increases iron level in the liver. In the current investigation, treatment with GaNPs/and or exposure to R revealed a significant reduction in the iron concentration in the liver and tumor tissues in the EC-treated groups compared to EC-bearing mice group.
Cancer cells undergo intensive cell division and need a considerable amount of iron for their development. Thus, it is very probable that by affecting iron metabolism and disturbing the process of its accumulation in malignant tissues, it may be possible to prevent a number of different cancers. Some researchers hold that the lowering of iron content may be a way to treat cancer, in which the proliferation of tumors is limited. 55 Gallium is found to concentrate in proliferating tissue (including most tumors), where large amounts of TFR are expressed. When gallium reaches these sites, it will compete with ferric ion and will interfere with its absorption, metabolism, and activity.56,57
The mechanisms of antineoplastic activity of gallium are complex and can be viewed as a two-step process in which the first step involves the targeting of gallium (as transferrin-gallium) to TFR-bearing tumor cells. In the second step, gallium perturbs cellular iron metabolism by interfering with TFR-mediated uptake of iron and with the endosomal release of iron from transferrin to the cytoplasm. This leads to a condition of relative cellular iron deprivation and inhibition of the iron-dependent function of ribonucleotide reductase, the enzyme essential for deoxyribonucleotide synthesis, thus blocking DNA synthesis and transferrin-mediated endocytosis, ultimately resulting in apoptosis.36,37,45
More studies to investigate the mechanistic anticancer effect of GaNPs are required
Cancer cells exhibit fundamentally different metabolic pathways for maintenance of intracellular ion homeostasis concerning Na+/K+-ATPase activity markedly depends on tumor cell or cancer type. 58
In this regard, the obtained data showed a significant decrease of Na+/K+-ATPase activity in cancer-bearing mice (EC group) in comparison with other treated groups (EC + R and EC + GaNPs + R). This finding is consistent with others59,60 who reported that hepatic Na+/K+-ATPase activity concentration was significantly decreased in tumor-bearing mice when compared to control. Therefore, the decreased activity of hepatic Na+/K+-ATPase in mice inoculated with EAC may be resulted from some changes such as the altered molecular structure of the Na+/K+-ATPase change in membrane fluidity and/or inhibition of its activity by specific protein.
Exposure to relatively low doses of radiation at 0.24 Gy showed increased levels of ATPase activity as reported in previous study. 61 Similarly, in this work, administration of GaNPs and R exposure to tumor-bearing mice showed a significant elevation in Na+/K+-ATPase in the tumor tissue upon comparison with EC group. The role of Gallium in the modulation of cell membrane permeability could be related to several mechanisms: alteration of the cell membrane potential, modification of electric charges at the surface of the cell membrane, and modification of ATPase activity. 7
Mitochondria are central to the intrinsic pathway of apoptosis and therefore are targets of choice for cancer therapy. The mitochondria pathway of apoptosis is characterized by mitochondrial membrane permeability and release of pro-apoptotic proteins (cytochrome c) from the intermembrane space to the cytosol. These events launch activation of the initiator caspase 9, which in turn triggers the caspases’ cascade leading to DNA condensation/fragmentation and cell death. 62
Intracellular ROS may act as signaling molecules for carcinogenesis and cell death induction, a sudden intracellular raise in ROS accounting for a pro-death situation. 63 They are produced by various enzymatic complexes including NADPH oxidase and mitochondrial electron transport chain, the latter being considered as the most important source of ROS. 64
Deficiencies in the respiratory chain (RC) are frequently found in cancer cells. Among the various causes are numerous mutations in the mitochondrial genome (mtDNA) affecting subunits in RC complexes (RCC) I, III, and IV 65 and in nuclear-coded components of complex II 66 whose assembly factors are likewise mutated. 67 These alterations lead to specific defects of the RC.
In this study, our complexes II and III data showed a significant increase in tumor tissue in treated group either lonely or with low-dose irradiation compared to mice bearing solid tumor. Chitambar and Antholine 8 reported that the iron–sulfur cluster (Fe-S) proteins in the citric acid cycle and mitochondrial complexes are potential targets for the cytotoxic action of gallium compounds. Gallium nitrate displays an increase in ROS within 4 h of gallium exposure, and this is followed by an increase in the expression of metallothionein and heme oxygenase-1. Gallium induced upregulation of heme oxygenase-1 gene expression. This suggests that the upregulation of metallothionein and heme oxygenase-1 by gallium nitrate results from gallium-induced oxidant stress in cells. 43
Regarding the mitochondrial enzymes, another key regulatory enzyme determined was Cytochrome P450 2E1 (CYP2E1). CYP2E1 is a potential regulator of intracellular ROS levels in breast cancer cells, and it plays a role in ROS generation and also might regulate migration of tumor cells (metastasis). CYP2E1 gene expression is affected and regulated under cancer-related transcription factor p53.68,69 In this regard, our CYP450 (2E1) data showed significant decrease in the level of gene transcript of CYP2E1 in tumor tissue homogenate of EC mice upon exposure to GaNPs and/or R compared with EC group. Similarly, Vaclavikova et al. 70 suggested that CYP2E1 enzyme might serve important functions in breast carcinogenesis and play a link between oxidative stress and tumor growth. The detected changes of CYP2E1 expression are depending on the dose and intensity of ionizing radiation or oxidative stress source that would explain diversity of CYP2E1 gene expression response in different tissues. 71
Thus, the result of this study demonstrated that aqueous dispersions of GaNPs exhibit antitumor properties in in vitro and in vivo studies. In conclusion, GaNPs might be a potential alternative agent for cancer treatment and a sensitizing agent for radiotherapy. However, more studies are needed to elucidate the mechanism of GaNPs action for the treatment of cancer and other illness, with lower cost and effectiveness.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
