Abstract
The length of proximal margin for gastrectomy remains controversial. The proximal margin and its relationships with clinicopathological variables and overall survival of 922 gastric cancers were retrospectively analyzed. Proximal margin was divided into four groups (0–2.0, 2.1–4.0, 4.1–6.0, and >6.0 cm). It indicated that the overall survival was improved with the increase of proximal margin. The proximal margin of 2.1–4.0 cm was associated with a better overall survival for gastric cancers with solitary-type (T1 and T2 stages, N0 stage, tumor–node–metastasis stages I and II, tumor size <5 cm, histological G1 and G2, and Bormann type I and II). Futhermore, proximal margin of 4.1–6.0 cm was associated with a better overall survival for gastric cancers with infiltrative-type (T3 and T4 stages, N1 stage, tumor–node–metastasis stage III, tumor size ⩾5 cm, histological G3 and G4, and Bormann type III and IV). Univariate analysis revealed that T stage, N stage, tumor–node–metastasis stage, histological grade, Bormann type, carcinoembryonic antigen, carbohydrate antigen 199, extent of gastrectomy, tumor location, and proximal margin were significantly associated with overall survival. Multivariate analysis revealed that tumor–node–metastasis stage, histological grade, Bormann type, carcinoembryonic antigen, carbohydrate antigen 199, extent of gastrectomy, and proximal margin were independent prognostic factors for gastric cancers with radical gastrectomy. In conclusion, the proximal margin was an independent prognostic factor for gastric cancer and should be decided individually. Proximal margin of 2.1–4.0 cm and 4.1–6.0 cm were needed for patients with solitary-type and infiltrative-type, respectively.
Introduction
Although the incidence and mortality are decreasing, gastric cancer (GC) is still the second most leading cause of cancer-related deaths worldwide.1,2 Radical resection is the only curative treatment for patients with GC, and the length of resection margin which determined the volume of stump, digest reconstruction, and quality of postoperative life is very important for gastrectomy.
It is well studied that the length of tumor invasion was determined by tumor characteristics (T classification, N classification, tumor site, size, histological type, and macroscopic type).3,4 However, the optimal length of proximal margin (PM) remains controversial, and there is still no robust evidence to define a safe length of PM for GC.5,6
In the past decades, several studies focused on the length of tumor invasion and prognostic value of lengths of resection margin on overall survival (OS) of patients with early GC.7,8 But there are seldom reports on patients with advanced stage.
In this study, we retrospectively analyzed the relationships of PM with clinicopathological variables and evaluated the prognostic value of PM on outcomes of GC patients with radical gastrectomy.
Materials and methods
Clinical data
From January 2007 to December 2011, 922 consecutive GC patients with radical gastrectomy at Tianjin Medical University Cancer Institute and Hospital were enrolled for this study. Patients histologically confirmed with GC underwent radical gastrectomy and D2 lymphadenectomy, and the tumor was located in middle or lower third of stomach. Patients with positive distal or PM, history of gastrectomy or other malignancy, distant metastasis, peritoneal dissemination, previous adjuvant chemotherapy, immunotherapy or radiotherapy, and incomplete clinicopathological date were excluded.
The preoperative examination included upper gastrointestinal (GI) series, abdominal ultrasonography (US), computed tomography (CT), endoscopic US, serum carbohydrate antigen 199 (CA199), and carcinoembryonic antigen (CEA).
The choice of the PM and reconstruction method was based on the Japanese Gastric Cancer Association (JGCA), 9 tumor characteristics (gross type, T stage, N stage, tumor site, size, histological grade), 10 and experience of surgeons with the aim of radical gastrectomy and improving patients’ quality of life. The intraoperative frozen-section was routinely performed in all patients, and the results were defined according to the final histological examination.
The PM was measured surgically before formalin fixation and defined as the minimum length from the gross proximal edge of tumor to the proximal resection line. Tumor sites were equally divided into upper, middle, and lower third section of the stomach. Histological grade were classified as G1 (well differentiated), G2 (moderate differentiated), G3 (poorly differentiated), and G4 (undifferentiated, signet ring cell carcinoma, and mucinous adenocarcinoma). Recurrences were classified as locoregional recurrence (remnant gastric and anastomotic recurrence, regional lymph node recurrence), peritoneal dissemination, and distant recurrence.
Tumor grade and stage were classified according to the seventh edition of the American Joint Committee on Cancer/International Union Against Cancer (AJCC/UICC) tumor–node–metastasis (TNM) classification. 11 This study was approved by the Medical Ethics Committee of the Tianjin Medical University Cancer Institute and Hospital.
Follow-up
The follow-up was performed every 3 months for the first 2 years and then every 6 months with physical examination, blood tests, chest radiographs, abdominal US, CT, serum CEA, and CA199 levels. The follow-up lasted until death or the cutoff date of 31 May 2016.
Statistical analysis
Values were tested by means of Student’s t test and presented as mean ± standard definition (SD). Categorical variables were compared by Pearson chi-square test or Fisher’s exact test. OS was evaluated using the Kaplan–Meier method and log-rank test. The Cox proportional hazards model was employed for multivariate analysis. A two-sided p < 0.05 was considered to be statistically significant, and all statistical analyses were performed using the SPSS 16.0 software (SPSS, Chicago, IL, USA).
Results
Demographic characteristics of patients with GC
The clinicopathological variables of patients are shown in Table 1. The median age was 57 years old (range: 21–85 years), and most of patients were male (72.3%), G3 (67.0%), advanced stage (stage III: 58.6%), and underwent distal gastrectomy (74.3%). The mean PM was 5.29 ± 1.67 cm. The median follow-up time was 54 months (range: 4–106 months).
The clinicopathological features of gastric cancers.

CA199: carbohydrate antigen 199; TNM: tumor–node–metastasis; CEA: carcinoembryonic antigen.
Survival analysis basing on discrepancies of PM
The discrepancies of PM are shown in Figure 1. The PM was divided into four subgroups (0–2.0, 2.1–4.0, 4.1–6.0, and >6.0 cm). Basing on analysis of the entire cohort, the OS was improved with the increasing PM (p = 0.001). However, there was no significant difference of survival (p = 0.637) between patients with 4.1–6.0 and >6.0 cm (Figure 2).
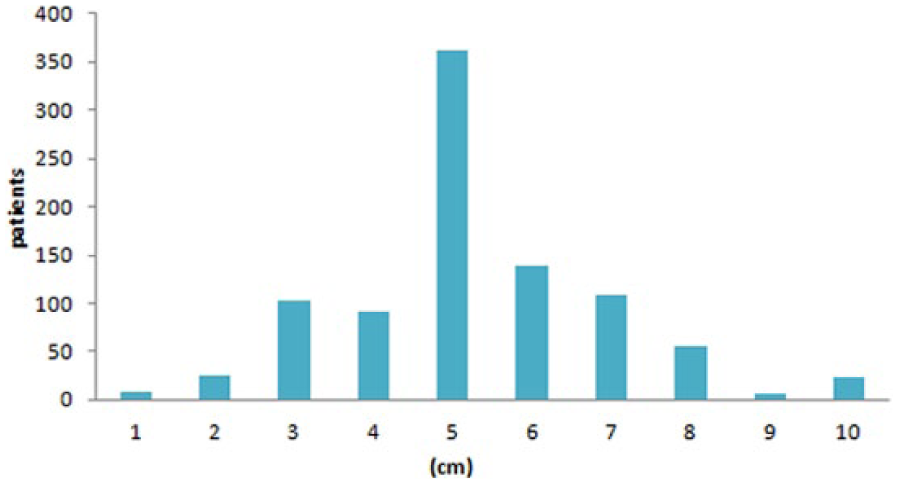
The discrepancies of PM in GC patients with distal or total gastrectomy.

The overall survival of all patients according to the length of PM (0–2.0, 2.1–4.0, 4.1–6.0, and 6.0 cm).
Univariate analysis revealed that T classification (p = 0.001), N classification (p < 0.001), TNM classification (p < 0.001), histological grade (p < 0.001), gross type (p < 0.001), CEA (p < 0.001), CA199 (p < 0.001), extent of gastrectomy (p < 0.001), tumor location (p = 0.025), and PM (p = 0.001) were significantly associated with OS. Multivariate analysis demonstrated that TNM classification (p < 0.001), histological grade (p = 0.010), gross type (p < 0.001), CEA (p < 0.001), CA199 (p = 0.009), extent of gastrectomy (p < 0.001), and PM (p = 0.048) were independent prognostic factors for OS in GC patients with radical gastrectomy (Table 2).
Univariable and Multivariable analysis of patients with gastric cancer.

HR: hazard ratio; CI: confidence interval; CEA: carcinoembryonic antigen; CA199: carbohydrate antigen 199; TNM: tumor–node–metastasis; PM: proximal margin.
Evaluation of the OS of patients with subgroups of PM among different clinicopathological variables
Among patients with TNM stages I and II (Figure 3(a)), the PM was not significantly associated with OS (p = 0.068). However, there was significant difference of survival between patients with 0–2.0 and 2.1–4.0 cm PMs (p = 0.030), and the OS of patients with more than 2 cm PM (2.1–4.0, 4.1–6.0, and >6.0 cm) was similar (p = 0.892). In patients with TNM stage III (Figure 3(b)), the PM significantly correlated with OS (p = 0.001). But the OS of patients with 4.1–1.0 and >6.0 cm PMs were not significantly different (p = 0.745).
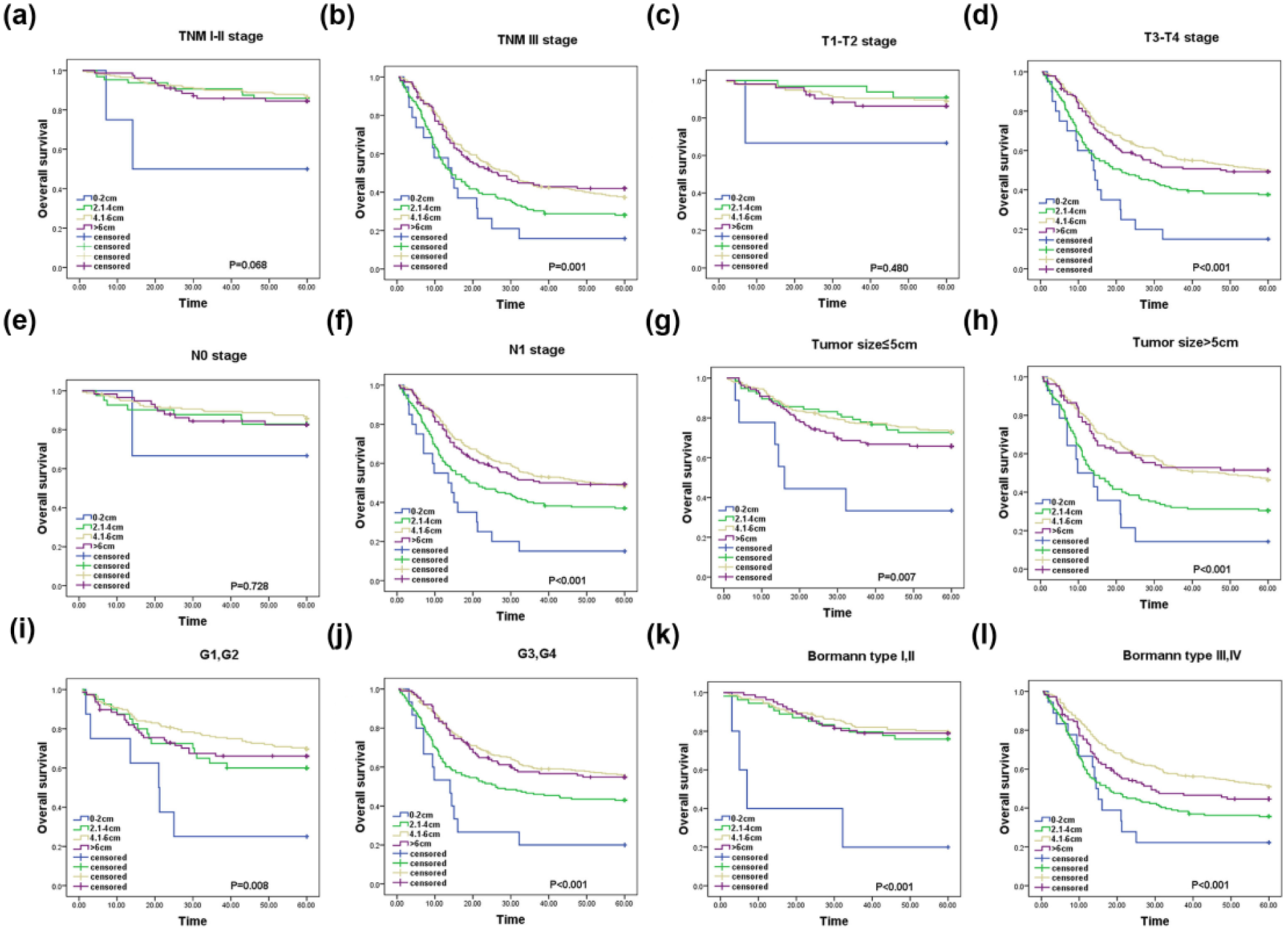
The overall survival (OS) of patients according to the length of PM among different clinicopathological variables. (a) OS of patients with TNM stages I and II according to the length of PM. (b) OS of patients with TNM stages III and IV according to the length of PM. (c) OS of patients with T1 and T2 stages according to the length of PM. (d) OS of patients with T3 and T4 stages according to the length of PM. (e) OS of patients with N0 stage according to the length of PM. (f) OS of patients with N1 stage according to the length of PM. (g) OS of patients with tumor size ⩽5 cm according to the length of PM. (h) OS of patients with tumor size >5 cm according to the length of PM. (i) OS of patients with G1 and G2 according to the length of PM. (j) OS of patients with G3 and G4 according to the length of PM. (k) OS of patients with Bormann types I and II according to the length of PM. (l) OS of patients with Bormann types III and IV according to the length of PM.
In patients with T1 and T2 stages (Figure 3(c)), the PM was not significantly associated with OS (p = 0.480). Although the OS of patients with 0–2.0 cm PM was shorter than those with 2.1–4.0 cm PM, there were no statistically significant differences between them (p = 0.485). In patients with T3 and T4 stages (Figure 3(d)), the PM was strongly associated with OS (p < 0.001), and there were no significant differences of OS between patients with 4.1–1.0 and those with >6.0 cm (p = 0.689).
Among patients with N0 stage (Figure 3(e)), the PM was not significantly associated with OS (p = 0.728), but in patients with N1 stage (Figure 3(f)), the PM was significantly associated with OS (p < 0.001). However, there were no significant differences of OS between patients with 4.1–1.0 cm PM and those with >6.0 cm PM (p = 0.851).
The PM strongly correlated with OS in patients with any subgroups of tumor size, histological grade, and Bormann type (Figure 3(g)–(l)). Among patients with tumor size ⩽5 cm, G1 and G2, and Bormann type I and II, the OS of patients with 2.1–4.0 cm PM was much better than those with 0–2.0 cm PM (p = 0.033, p = 0.003, and p < 0.001, respectively), but the OS among patients with 2.1–4.0, 4.1–1.0, and >6.0 cm PMs was not significantly different (p = 0.419, p = 0.318, and p < 0.842, respectively). Among patients with tumor size >5 cm, G3 and G4, and Bormann Bormann type III and IV, the OS of patients with 4.1–6.0 cm PM was much better than those with 2.1–4.0 cm PM (p < 0.001, p < 0.001, and p < 0.001, respectively). But the OS among patients with 4.1–1.0 and >6.0 cm PMs was not significantly different (p = 0.860, p = 0.708, and p = 0.113, respectively).
Discussion
The margin status is an independent prognostic factor in patients with GC.12,13 According to the JGCA guidelines, the length of the resection margin should be increased with tumor progression (2 cm for T1 stage, 3 cm for T2–T4 stages with solitary-type, and 5 cm for T2–T4 stages with infiltrative-type). 9 However, it often has troubles to secure the recommended lengths of resection margin. 14
In clinical practice, the resection margin was usually judged by surgeons through palpation and gross inspection according to the intraoperative findings. This mainly depends on surgeon’s experience and sometimes it is an unreliable method. As for lengths of resection margin, a shorter PM would increase the hazard of reccurence, a larger PM would increase the extent of gastrectomy, impact the reconstruction method, impair the postoperative quality of life, and even have profound implications on postoperative morbidity.15,16 So the excellent outcome of resection margin should be considered not only according to the curability but also the safety and quality of postoperative life. 17
With the popular frozen-section examination for assessing the resection margin and preoperative endoscopic clipping or straining for guiding the tumor borderline, less invasive surgery (endoscopic submucosal dissection (ESD), endoscopic mucosal resection (EMR), and local resection with or without adjacent lymphadenectomy) was performed carefully in selected patients, and the quality of postoperative life was also improved.18,19
It is reported that the lengths of resection margin should be determined by tumor characteristics. 4 Moreover, when an adequate resection margin is achieved, the operative approach is not associated with an alteration in OS even with longer margin or extended resection.15,19,20
In this study, the PM was an independent prognostic factor in patients with GC, and the OS was improved with the increase of margin lengths. The OS of patients with 2.1–4.0 cm PM was significantly longer than patients with 0–2.0 cm PM in TNM stages I and II and solitary-type (tumor size ⩽5, histological G1 and G2, and Bormann type I and II), and the differences of OS in patients with 2.1–4.0, 4.1–1.0, and >6.0 cm PM groups were not statistically significant. The OS of patients with 4.1–6.0 cm PM was significantly longer than patients with 2.0–4.0 cm PM in TNM stage III and infiltrative-type (tumor size >5, histological G3 and G4, and Bormann types III and IV), and the differences of OS in patients with 4.1–6.0 and >6.0 cm PM groups were not statistically significant. It suggested that the 2.1–4.0 cm PM was associated with an optimal OS in patients with TNM stages I and II and solitary-type, and the 4.1–6.0 cm PM was associated with an optimal OS in patients with TNM stage III and infiltrative-type. These indicated that the PM should be increased with tumor progression, and when a certain PM was achieved, the OS was not improved by the increase of PM.
The PM should also be taken a considerate evaluation of tumor character such as extent of disease, tumor site, tumor grade, gross type, patients’ general health, and feasibility of resection which can individually determine the length of PM.10,21 In addition, if there is any uncertainty on tumor borderline, the frozen-section examination should be performed.
In conclusion, the PM is significantly associated with OS and is an independent prognostic factor for OS in GC patients with radical gastrectomy. The PM should be decided individually, and a 2.1–4.0 cm PM is needed for patients with TNM stages I and II and solitary-type GC, and a 4.1–6.0 cm PM is needed for patients with TNM stage III and infiltrative-type GC. When an adequate PM was achieved, the longer PM did not associate with additional survival benefits.
Footnotes
Acknowledgements
The authors thank all the people who had participated in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
