Abstract
Evidence is sparse regarding the clinical performance of luminescent oxygen channeling immunoassays–based tumor marker assays in gynecological cancer. Analyzing serum samples of 336 patients with Dimension™Vista1500, we investigated the diagnostic power of carbohydrate antigen 15-3, carbohydrate antigen 125, carcinoembryonic antigen, carbohydrate antigen 19-9, and alpha-fetoprotein in patients suffering from different types of gynecological cancer and precancerous gynecological diseases and compared findings to appropriate control groups. The cohort comprised 177 female patients with gynecological cancers (73 breast, 22 cervical, 16 endometrial, 17 vulva, and 49 ovarian cancers), 26 patients with precancerous gynecological diseases (11 vulva, 4 cervical, and 10 breast), 109 patients with benign gynecological diseases, and 24 healthy controls. Discriminative power was assessed by areas under the curve in receiver operating characteristic curves, and sensitivities were determined at a fixed specificity of 95%. Levels of biomarkers in healthy controls were in the expected ranges and a discriminative power between gynecological cancers and healthy controls was observed for several tumor markers. Established tumor type–associated markers were elevated in specific gynecological cancers and benign controls as well as within precancerous gynecological diseases and healthy control group. In ovarian cancer, carbohydrate antigen 125 and carbohydrate antigen 15-3 were significantly elevated compared to the respective benign diseases. Carbohydrate antigen 125 was the most conclusive marker (area under the curve = 0.86% and 77.6% sensitivity at 95% specificity). In breast cancer, carcinoembryonic antigen and carbohydrate antigen 15-3 were significantly higher than in the respective benign diseases. Carcinoembryonic antigen achieved the most conclusive area under the curve (0.65) with 31.5% sensitivity at 95% specificity. None of the investigated markers was found to be of value in discriminating benign and malignant cervical diseases. Carcinoembryonic antigen and alpha-fetoprotein distinguished precancerous breast and vulva diseases from healthy controls. These findings show that luminescent oxygen channeling immunoassays–based tumor marker assays provide reliable results in routine diagnostics.
Keywords
Introduction
Of the 14 million estimated new cases of cancer diseases worldwide and with over eight million cancer-related deaths in 2012, a large number of these were gynecological tumors. 1 In descending order of incidence, these include an estimated 1.7 million new cases of breast cancer, 320,000 new cases of endometrial cancer, 266,000 new cases of cervical cancer, and 239,000 new cases of ovarian cancer. Breast cancer is the most common cancer, while cervical cancer is the fourth most common cause of cancer-related deaths in women. Certain tumor markers are cornerstones of the workflow in diagnosis, therapy control, and monitoring of advanced gynecological diseases. 2 Of all ovarian tumors, 90% are of epithelial origin and carbohydrate antigen 125 (CA 125) and human epididymis protein 4 (HE4) are the most relevant tumor markers. Elevated levels depend on histological type and disease stage. 3 While sensitive in early stages, CA 125 has limited specificity in early detection of ovarian cancer, particularly in premenopausal women. 4 Furthermore, several factors can also cause elevated CA 125 levels, such as ethnicity, pregnancy, age, premenopausal versus postmenopausal stage, and menstrual cycle.5–8 In addition, gynecological and non-gynecological benign diseases can cause elevated serum levels of CA 125. 4 In breast cancer, a combination of carcinoembryonic antigen (CEA) and CA 15-3 has prognostic power in a preoperative setting. While CEA and CA 15-3 are recommended for therapy monitoring and early detection of recurrence, 2 they are not recommended for early diagnosis or screening due to their low sensitivity. 2 In cervical cancer, National Academy of Clinical Biochemistry (NACB) guidelines discuss (among other tumor markers such as squamous cell carcinoma antigen (SSCA)) CEA and CA 125 for pretreatment prediction of prognosis and preoperative prediction of metastasis presence. 9 CEA has prognostic relevance in colorectal cancer. 10 In cases where the origin of a uterine tumor is unclear, tumor marker panels including CEA are recommended.11,12
All of the important guidelines recommend clinical evaluation and method comparison when new methods for the measurement of tumor markers are established.
Luminescent oxygen channeling immunoassays (LOCITM)-based assays are homogeneous immunoassays that skip one washing step in comparison with heterogeneous assays and therefore would enable a faster detection. In earlier studies, good general correlations for the tumor markers CEA, CA 15-3, CA 19-9, CA 125, and prostate-specific antigen (PSA) were observed when comparing chemiluminescent assays with LOCI™-based tumor marker assays. Nevertheless, higher variations were detected for CA 15-3 and CA 19-9.13,14 Apart from methodical evaluation, assessment of the clinical performance of LOCI™-based tumor marker assays is a precondition for their use in clinical practice. As there is no conclusive data available so far with regard to gynecological cancers, this study was conducted.
Aim
Aim of this study was to investigate the diagnostic power of LOCI™-based tumor marker assays CA 15-3, CA 125, CEA, CA 19-9, and alpha-fetoprotein (AFP) in patients with gynecological cancer in comparison to healthy controls, precancerous gynecological diseases, and benign gynecological diseases as relevant control groups for differential diagnosis.
Patients and methods
Patients
Overall, 336 female patients/individuals were included, comprising 24 healthy individuals, 26 patients with precancerous gynecological diseases (11 vulva, 5 cervical, and 10 breast diseases), 109 patients with benign gynecological diseases (e.g. myoma, endometriosis, condylomata, mastopathy), and 177 patients with gynecological cancer (73 breast, 22 cervical, 16 endometrial, 17 vulva, and 49 ovarian cancers). Median age and range are shown in Table 1. The study was approved by the local ethics committee and all participants gave their written informed consent. Blood samples were collected after vein puncture and prior to any therapy. Samples were handled according to the standard operating procedure (SOP) of the Biofluid Biobank at the Institute for Clinical Chemistry and Clinical Pharmacology of the University Hospital Bonn in cooperation with the Center for Integrated Oncology Cologne–Bonn (CIO). SOP stipulates, after immediate delivery of the samples to the Central Laboratory, a centrifugation at 4000 r/min for 10 min, storage at 4°C until further aliquoting, and archiving at −80°C in the Biobank storage bank on the same day.
Patient characteristics.

ECV: endometrial/cervical/vulva.
Absolute numbers, median age and range of healthy controls, benign gynecological diseases, precancerous diseases, and patients with breast, cervical, endometrial, vulva, and ovarian cancer are given.
Methods
Measurements were performed with LOCI™-based tumor marker assays for Dimension™ Vista 1500 analyzer (Siemens Healthcare Diagnostics, Eschborn, Germany). Internal and external quality control specifications were observed according to the guidelines of the German Federal Medical Society (RiliBÄK). 15 The tumor marker assays are part of the routine methods at the Central Laboratory of the University Clinics Bonn. According to the manufacturer’s instructions, LOCI™-based assays are heterogeneous sandwich chemiluminescent immunoassays. These assays work with chemibeads, which contain a chemiluminescent dye and sensibeads, which contain a photosensitizer dye. Sandwiches are generated via biotinylated antibodies and chemibeads. After addition of sensibeads, they generate immunocomplexes. Finally, a chemiluminescent reaction is triggered at a wavelength of 680 nm before the final signal will be detected at 612 nm.
Statistics
Data were statistically analyzed (IBM SPSS Statistics, Ehningen, Germany, Version 20) and p < 0.05 was considered statistically significant. Median, range, and 95th percentiles were calculated in defined groups. Wilcoxon tests were used to calculate the differences between the collectives. Value distributions of the different tumor markers are represented by box plots. The diagnostic power of CEA, CA 19-9, CA 15-3, CA 125, and AFP is shown by receiver operating characteristic (ROC) curves. Furthermore, areas under the curve (AUCs) and sensitivities at 95% specificity versus healthy controls and versus benign patients with gynecological diseases were calculated.
Results
Numbers of patients in different cohorts, median age, and range are displayed in Table 1 (Table 1). Healthy individuals were significantly younger than cancer patients in total as well as for the single cancer entities (p < 0.001). In addition, significant differences were seen between age of patients with ovarian, endometrial, vulva, and cervical cancers when compared with respective benign diseases.
Median, range, and 95th percentiles for AFP, CA 12-5, CA 15-3, CA 19-9, and CEA in healthy female individuals, patients with benign gynecological diseases, patients with precancerous gynecological diseases, and in the various subgroups of gynecological cancers are shown in Table 2 and Figure 1. Levels of tumor markers in healthy controls were in the expected ranges: AFP (median = 2.0 ng/mL, 95th percentile = 7.2 ng/mL), CA 125 (median = 9.8 U/mL, 95th percentile = 15.9 U/mL), CA 15-3 (median = 14.3 ng/mL, 95th percentile = 23.7 ng/mL), CA 19-9 (median = 4.1 U/mL, 95th percentile = 14.1 U/mL), and CEA (median = 0.8 ng/mL, 95th percentile = 1.9 ng/mL).
Value distribution in diagnostic groups.
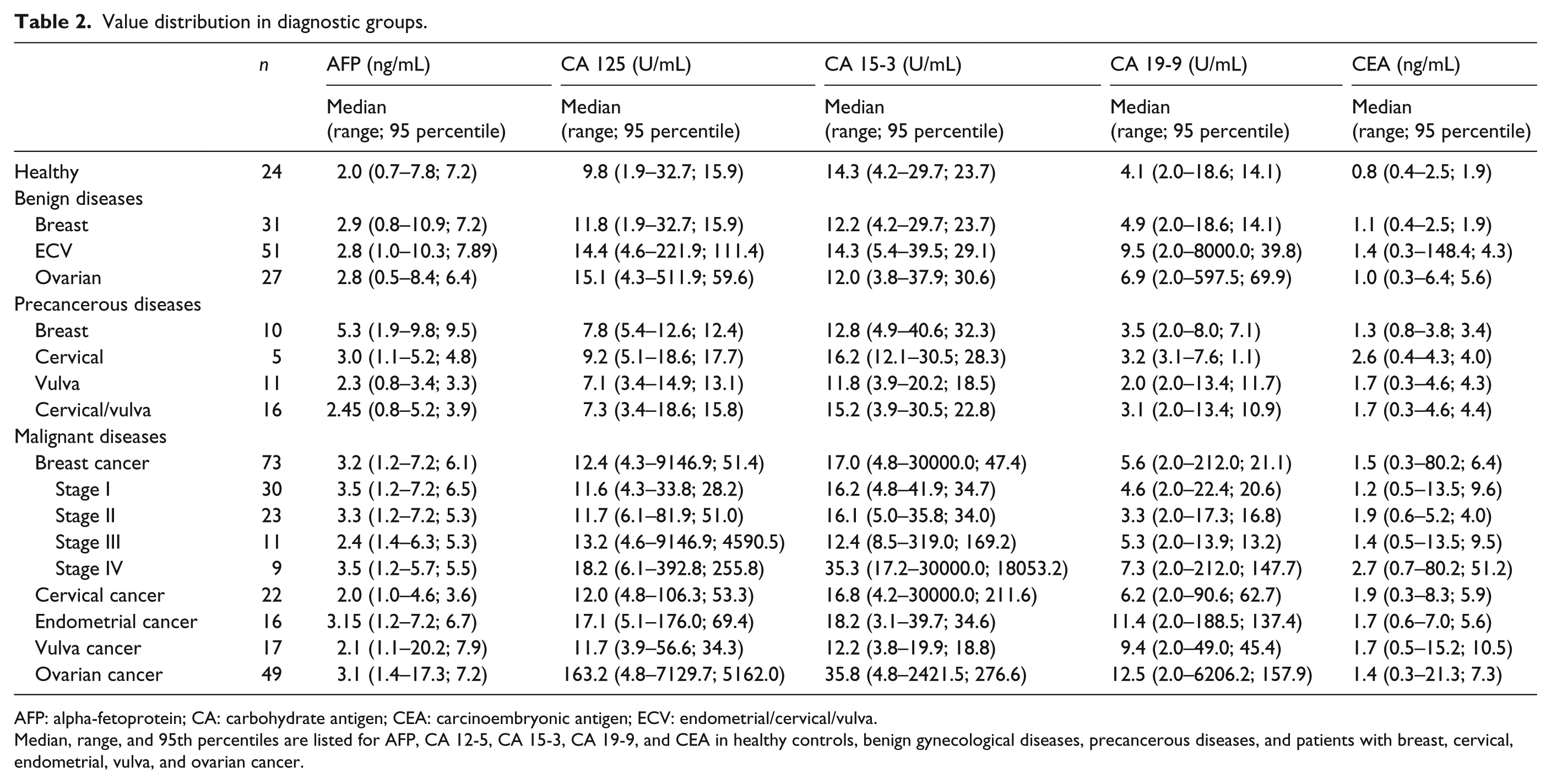
AFP: alpha-fetoprotein; CA: carbohydrate antigen; CEA: carcinoembryonic antigen; ECV: endometrial/cervical/vulva.
Median, range, and 95th percentiles are listed for AFP, CA 12-5, CA 15-3, CA 19-9, and CEA in healthy controls, benign gynecological diseases, precancerous diseases, and patients with breast, cervical, endometrial, vulva, and ovarian cancer.
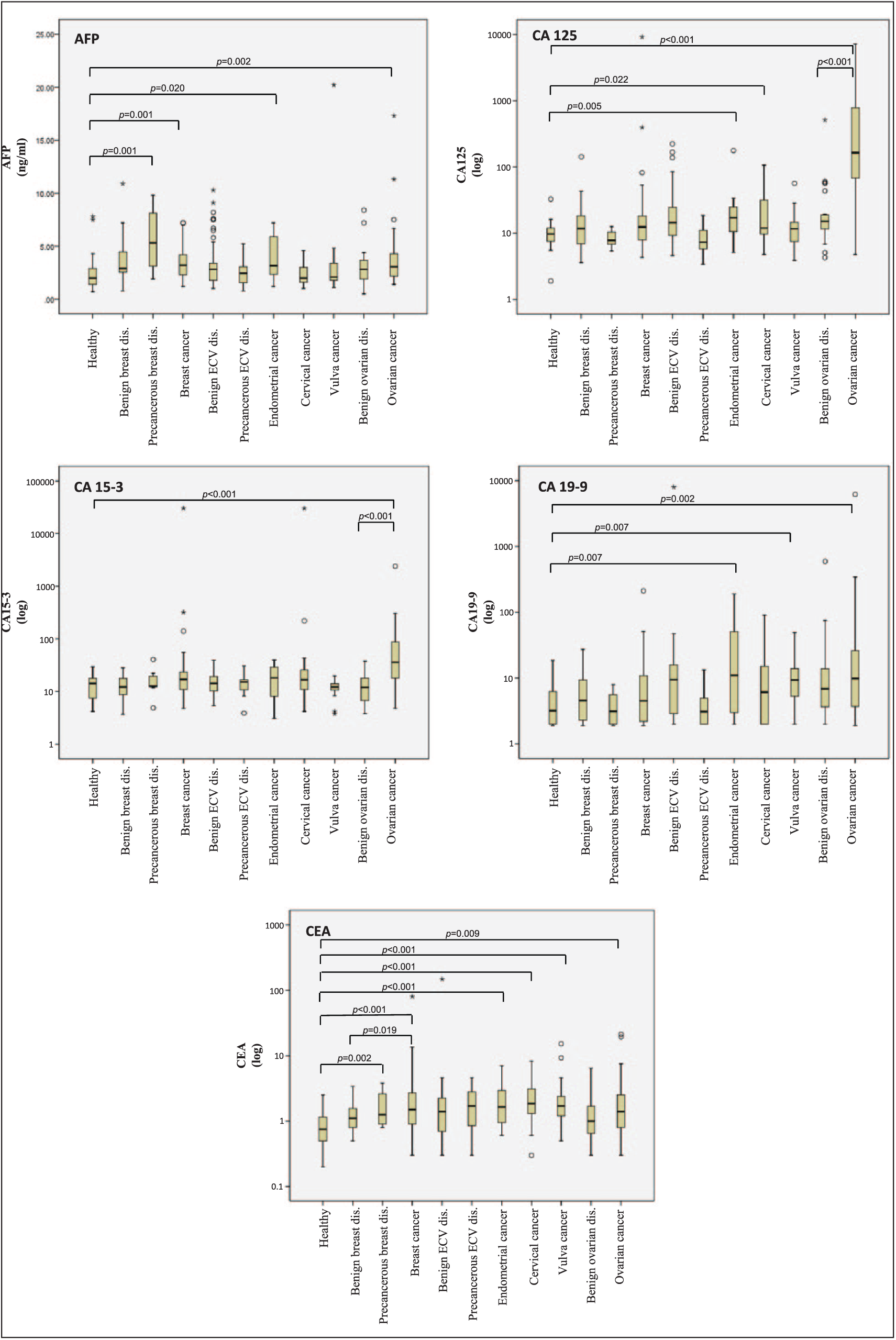
Value distributions in diagnostic groups. Displayed are value distributions of AFP, CA 125, CA 15-3, CA 19-9, and CEA in gynecological cancers. Bars represent the median, box plots show interquartile range and whiskers. p values are presented for significant differences between groups. Values for CA 125, CA 15-3, CA 19-9, and CEA were log transformed.
Breast cancer
In comparison to healthy controls, patients with breast cancer had significantly higher values for CEA (median = 1.5 ng/mL, 95th percentile = 6.4 ng/mL; p < 0.001) and also for CA 15-3 (median = 17.0 ng/mL, 95th percentile = 47.4 ng/mL; p = 0.030) and AFP (median = 3.2 ng/mL, 95th percentile = 6.1 ng/mL; p = 0.001). In addition, significantly higher levels were found for CEA (p = 0.019) and CA 15-3 (p = 0.022) in breast cancer as compared with benign breast diseases. Furthermore, there were significant differences between precancerous breast diseases and healthy controls with higher values for AFP (p = 0.001) and CEA (p = 0.002). Regarding discrimination of breast cancer patients from control groups, best diagnostic performance was achieved for CEA (vs healthy controls: AUC = 0.79, 37.0% sensitivity at 95% specificity; vs. benign breast diseases: AUC = 0.65, 31.5% sensitivity at 95% specificity) (see Tables 2 and 3, Figure 2, and Supplementary 1).
Discriminative power of tumor markers.
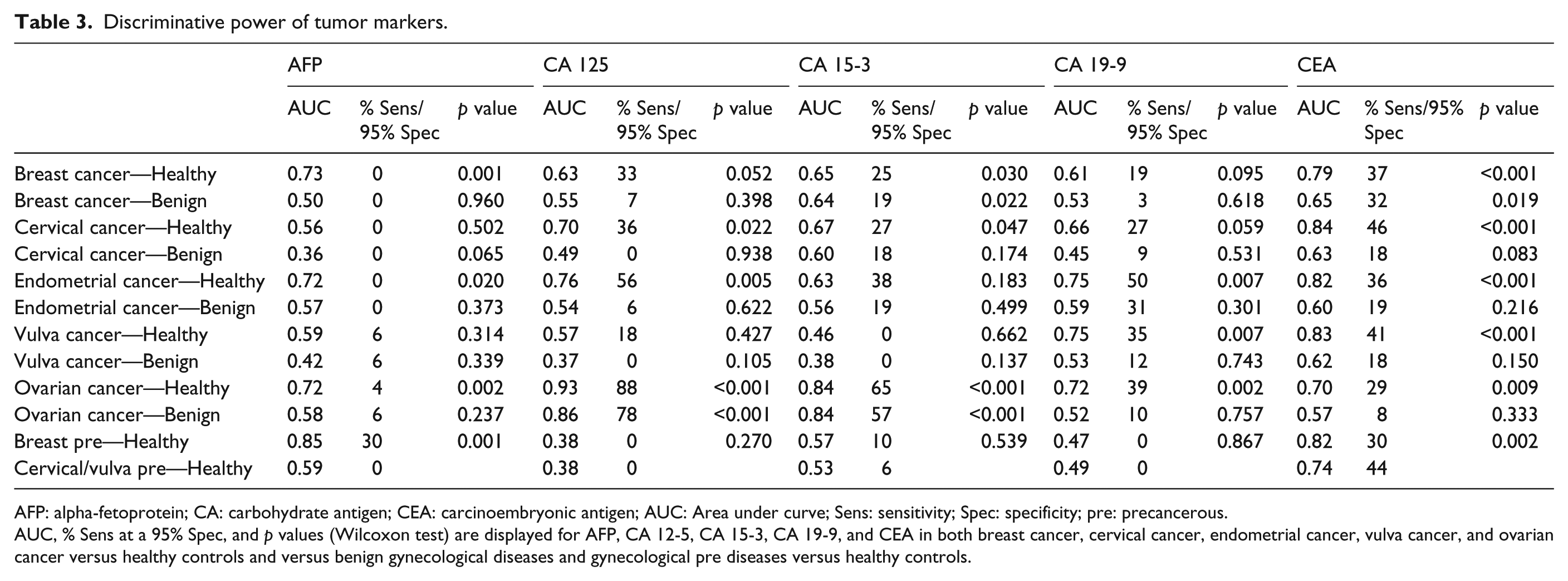
AFP: alpha-fetoprotein; CA: carbohydrate antigen; CEA: carcinoembryonic antigen; AUC: Area under curve; Sens: sensitivity; Spec: specificity; pre: precancerous.
AUC, % Sens at a 95% Spec, and p values (Wilcoxon test) are displayed for AFP, CA 12-5, CA 15-3, CA 19-9, and CEA in both breast cancer, cervical cancer, endometrial cancer, vulva cancer, and ovarian cancer versus healthy controls and versus benign gynecological diseases and gynecological pre diseases versus healthy controls.

ROC curves for cancer detection versus controls. Displayed are receiver operating characteristic (ROC) curves and area under the curve (AUC) for the discrimination between patients with gynecological cancers versus healthy controls, versus benign gynecological diseases, and, if available, healthy versus precancerous gynecological diseases using AFP CA 12-5, CA 15-3, CA 19-9, and CEA.
Cervical cancer
In comparison to healthy controls, patients with cervical cancer had significantly higher values for CEA (median = 1.9 ng/mL, 95th percentile = 8.3 ng/mL; p < 0.001) and also for CA 125 (median = 12.0 U/mL, 95th percentile = 106.3 U/mL; p = 0.022) and CA 15-3 (median = 16.7 U/mL, 95th percentile = 211.6 U/mL; p = 0.047). There were no significant differences between cervical cancer and benign cervical diseases nor between precancerous cervical diseases versus healthy controls and benign cervical diseases. Regarding discrimination of cervical cancer patients from control groups, best diagnostic performance was achieved for CEA (vs. healthy controls: AUC = 0.84, 45.5% sensitivity at 95% specificity; vs. benign diseases: AUC = 0.63, 18.2% sensitivity at 95% specificity) (see Tables 2 and 3, Figure 2, and Supplementary 1).
Endometrial cancer
In comparison to healthy controls, patients with endometrial cancer had significantly higher values for CEA (median = 1.7 ng/mL, 95th percentile = 7.0 ng/mL; p < 0.001) and also for CA 125 (median = 17.1 U/mL, 95th percentile = 176.0 U/mL; p = 0.005), CA 19-9 (median = 11.4 U/mL, 95th percentile = 188.5 U/mL; p = 0.007), and AFP (median = 3.2 U/mL, 95th percentile = 7.2 U/mL; p = 0.020). There were no significant differences between endometrial cancer and benign endometrial diseases, while levels were slightly elevated in cancer patients. Regarding discrimination of endometrial cancer patients from control groups, best diagnostic performance was achieved for CEA (vs. healthy controls: AUC = 0.82, 37.5% sensitivity at 95% specificity; vs. benign diseases: AUC = 0.60, 18.8% sensitivity at 95% specificity) (see Tables 2 and 3, Figure 2, and Supplementary 1).
Vulva cancer
In comparison to healthy controls, patients with vulva cancer had significantly higher values for CEA (median = 1.7 ng/mL, 95th percentile = 15.2 ng/mL; p < 0.001) and also for CA 19-9 (median = 9.4 U/mL, 95th percentile = 49.0 U/mL; p = 0.007), CA 125 (median = 11.4 U/mL, 95th percentile = 188.5 U/mL; p = 0.007), and AFP (median = 3.2 ng/mL, 95th percentile = 7.2 ng/mL; p = 0.020). There were no significant differences between vulva cancer and benign vulva diseases but between precancerous vulva diseases and healthy controls (p = 0.030). Regarding discrimination of vulva cancer patients from control groups, best diagnostic performance was achieved for CEA (vs. healthy controls: AUC = 0.83, 41.2% sensitivity at 95% specificity; vs. benign diseases: AUC = 0.62, 17.6% sensitivity at 95% specificity) (see Tables 2 and 3, Figure 2, and Supplementary 1).
Ovarian cancer
In comparison to healthy controls, patients with ovarian cancer had significantly higher values for CA 125 (median = 163.2 U/mL, 95th percentile = 5162.0 U/mL; p < 0.001) and CA 15-3 (median = 35.8 U/mL, 95th percentile = 2421.5 U/mL; p < 0.001) but also for CA 19-9 (median = 12.5 U/mL, 95th percentile = 6206.2 U/mL; p = 0.002), AFP (median = 3.1 ng/mL, 95th percentile = 7.22 ng/mL; p = 0.002), and CEA (median = 1.4 ng/mL, 95th percentile = 21.3 ng/mL; p = 0.009). In addition, significant differences were found between ovarian cancer and benign ovarian diseases with higher values for CA 125 (p < 0.001) and for CA 15-3 (p < 0.001) in ovarian cancer. Regarding discrimination of ovarian cancer patients from control groups, best diagnostic performance was achieved for CA 125 (vs. healthy controls: AUC = 0.93, 87.8% sensitivity at 95% specificity; vs. benign ovarian diseases: AUC = 0.86, 77.6% sensitivity at 95% specificity) and CA 15-3 (vs. healthy controls: AUC = 0.84, 65.3% sensitivity at 95% specificity, vs. benign diseases: AUC = 0.84, 57.1% sensitivity at 95% specificity) (see Tables 2 and 3, Figure 2, and Supplementary 1).
Discussion
Accurate determination of serum tumor marker levels is crucial as their impact for diagnosis, prognosis, and therapy monitoring has been shown for many solid tumors.16,17 When measurement methods for tumor markers are changed in a laboratory, a thoroughful methodical comparison of the old and new method as well as an assessment of the clinical performance of the new assay is recommended. This is due to the well-known method-dependency of tumor marker results which is caused by different antibody combinations used and different assay compositions. As a consequence, guidelines also recommend comparative and parallel measurements of both methods when tumor marker courses should be interpreted in single patients. 9 Recently, tumor marker assays were released that are based on LOCI™ technology. Major advantages of this technology are insensitivity to particle interference and real-time monitoring of particle reactions 18 as well as rapid quantification due to the saving of one washing step. 19 Potential limitations, however, are potential sensitivity issues in the low-value range.
In a method comparison with other chemiluminescent technologies, we recently showed good general correlations for tumor markers AFP, CEA, CA 15-3, PSA, CA 19-9, and CA 125. Nevertheless, we revealed considerable variations for CA 15-3 and CA 19-9.13,14 In addition, good inter- and intra-day precisions were observed when comparing LOCI™-based tumor marker assays for Dimension™ VISTA and chemiluminescent immunoassays for Cobas™ e411. 20 Clinically relevant may be the finding that within the decision range of marker values, higher levels were obtained with the LOCI™-based tumor marker assay than with other technologies, particularly for AFP.13,14 This shows the necessity to assess the performance of tumor markers for clinical questions, as we recently did for gastrointestinal cancer. 20
For the proper investigation of tumor markers in gynecological cancers, we considered several control groups such as healthy women and patients with respective benign diseases—a group that is highly relevant for differential diagnostic purposes—and had also a look on premalignant diseases of the breast, the cervix, and the vulva. Furthermore, we tested not only the tumor-related antigens such as CA 125 for ovarian cancer but also all other LOCI™-based markers. Even if they are not relevant for differential diagnosis of a specific tumor, it is important to know whether and how much they are influenced by a given malignant or non-malignant condition.
Our findings showed that LOCI™-based tumor marker assays provide reliable results in different gynecological cancers for routine diagnostics. While LOCI™-based tumor marker assays detected the healthy women correctly within specified marker ranges, tumor type–associated markers discriminated well between gynecological cancer patients and healthy controls. In breast cancer patients, best diagnostic performance was achieved for CEA and CA 15-3, and in ovarian cancer, CA 125 and CA 15-3, while none of the markers was relevant for the detection of cervical cancer. Interestingly, CEA and AFP distinguished precancerous breast and vulva diseases from healthy controls, a finding which will have to be further explored. Importantly, overall AUCs in ROC curves as well as sensitivities at specified cutoffs were comparable to other methods already used in routine diagnostics. 21
Our findings correspond further with current guidelines such as from the NACB that recommend CA 15-3 in breast cancer—though not for diagnosis but for monitoring of advanced disease and postoperative surveillance. 22 Although the application of CEA is still controversially discussed, 22 several studies have shown its importance in, for example, prediction and early detection of disease progression and metastasis.23,24 While we did not focus on breast cancer stage analysis, our results generally demonstrate the high clinical performance of the CA 15-3 LOCI™-based tumor marker assay for Dimension Vista in breast cancer disease. Other studies investigated subgroups with different disease stages and, using a different immunoassay, found AUCs of 0.62 and 0.90 in breast cancer patients with loco-regional and metastatic breast cancer versus benign diseases. 25 In ovarian cancer, best diagnostic performance was achieved for CA 125. These results are in line with current recommendations, suggesting that CA 125 is of great importance in therapy monitoring, differential diagnosis for pelvic masses, recurrence, and prognosis.4,22
Studies comparing different immunoassays reported a high correlation with minimal slope and intercept. 21 For CA 125, the International Federation of Gynecology and Obstetrics (FIGO) stage-dependent values have been published. 26 Unfortunately, HE4 as another highly relevant marker for ovarian cancer is not available with LOCI™ technology and thus could not be investigated here. In particular, the combination of CA 125 and HE4 enables calculation of the Risk of Ovarian Malignancy Algorithm (ROMA score)4,27 that is used for risk assessment if suspicious pelvic masses are present. As reported earlier, 25 CA 15-3 was confirmed to be a valuable marker for ovarian cancer as well.
Furthermore, our results confirm the clinical relevance of the pan marker CEA not only in colorectal cancer but also in gynecological cancers such as breast and ovarian cancer9,22 as well as for the detection of precancerous breast and vulva disease. While SCCA is known to be another sensitive marker for squamous cell cancers of the cervix and vulva, this marker is not available with LOCI™ technology so far and was not investigated in this study. Obviously, it would be valuable to do a clinical performance evaluation with two different methods to show their consistency. As LOCI™ technology was the only one in our routine application and as we had done several method comparisons before we performed this part of the evaluation only with the LOCI™-based assays. As two markers were meaningful for the differential diagnosis of breast and ovarian cancer, respectively, a combination could lead to additive sensitivity and specificity. However, as the patient numbers in these specific cancer cohorts and their control groups were limited, we resigned to do marker combinations in order not to overestimate accidental results.
Beyond the use of appropriate patients groups, we adhered to the requirements for clinical tumor marker evaluation suggested by the European Group on Tumor Markers (EGTM). Hence, in our study, a high diagnostic quality was obtained because samples were measured under real-time conditions with internal and external quality controls. We performed sample analyses in batches and due to the standardized procedures of our Biofluid Biobank, a high quality in preanalytic handling was reached. As our numbers were limited, no subgroup analyses were performed.
Conclusion
In summary, LOCI™-based tumor marker assays, CA 15-3, CA 125, and CEA are valuable biomarkers for the differential diagnosis of patients with breast and ovarian cancer providing their well-known profiles of sensitivity and specificity and thus demonstrated their reliability for routine laboratory diagnostics.
Footnotes
Acknowledgements
The authors thank all coworkers of the Biofluid Biobank of the University Hospital Bonn for sample handling and Mrs Dentler as native speaker for her assistance in English writing. Ramona C Dolscheid-Pommerich and Stefan Holdenrieder designed the study; Mignon Keyver-Paik, Thomas Hecking, and Walther Kuhn were involved in sample collection and clinical documentation; Ramona C Dolscheid-Pommerich, Gunther Hartmann, Birgit Stoffel-Wagner, and Stefan Holdenrieder in sample definition and tumor marker measurement; Ramona C Dolscheid-Pommerich and Stefan Holdenrieder did the evaluation of the data; and all authors were included in discussing the results, writing, and approving the paper. Reagents were provided by Siemens Healthineers. Stefan Holdenrieder is the guarantor of this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was ethically approved by local institutional review board (IRB; No. 319-12).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ramona C Dolscheid-Pommerich received honoraria for lectures by Siemens Healthineers.
Supplement material
Supplementary material is available for this article online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
