Abstract
A repertoire of monoclonal antibodies was generated by immunization of mice with cancer-associated glycoprotein CA19.9, and two of them were selected as optimal capture and detecting counterparts for sandwich test system for detection of CA19.9. Fine epitope specificity of the antibodies was determined using printed glycan array, enzyme-linked immunosorbent assay, and inhibitory enzyme-linked immunosorbent assay. Unexpectedly, both immunoglobulins did not bind key epitope of CA19.9 glycoprotein, tetrasaccharide SiaLeA, as well as its defucosylated form sialyl LeC (known as CA-50 epitope). The antibodies were found to have different glycan-binding profiles; however, they recognized similar glycotopes with common motif Galβ1-3GlcNAcβ (LeC), thus resembling specificity of human natural cancer-associated anti-LeC antibodies. We propose that cancer-specific glycopeptide epitope includes Galβ1-3GlcNAcβ fragment of a glycoprotein O-chain in combination with proximal hydrophobic amino acid(s) of the polypeptide chain.
Introduction
The concept of tumor-associated carbohydrate antigens (TACAs) 1 proved to be productive in the search of new cancer-associated glycans, studies of their biosynthesis, and understanding of cancer-promoting functions. However, numerous attempts of its application in cancer immunodiagnostics and construction of synthetic cancer vaccines had no visible practical yield. For example, monoclonal antibodies (mAbs) generated against synthetic antigen, tetrasaccharide LeY, 2 demonstrated good affinity, excellent glycan-specificity, but failed to discriminate LeY-enriched tumor cells from normal tissue. The presence of an aberrant chemical structure on a cell appears to be insufficient for recognition by the immune system without consideration of the ambient molecular context.
An idea of cancer-associated Abs capable of recognition of specifically presented glycans, or glycan-containing molecular patterns, is believed to be more fruitful. Particularly, the level of Abs against Thomsen–Friedenreich (TF) 3 and LeC antigens 4 is reduced in blood of patients with gastrointestinal (TF) and breast (LeC) cancers, suggesting their tumor-surveillance function. 4 Human natural anti-LeC Abs demonstrate unusual epitope specificity that allows to ignore the widespread LeC-terminated polylactosamine-type glycans. 5 In this work, we describe two mice mAbs (both IgG1a) with epitope specificity resembling human natural anti-LeC, both generated by immunization with glycoprotein CA19.9 isolated from ascitic fluid of cancer patients.
Results
Data of printed glycan array
Printed glycan array (PGA)6,7 consisted of ~400 synthetic glycans, mostly fragments of glycoprotein and glycolipid carbohydrate chains. The array included the main fragment of the glycan of CA19.9 antigen, SiaLeA, in the form of tetrasaccharide Neu5Acα2–3Galβ1–3(Fucα1–4) GlcNAc and pentasaccharide Neu5Acα2–3Galβ1–3(Fucα1–4) GlcNAcβ1–3Galβ. Natural glycoprotein CA19.9 (isolation and characterization are presented in Supplementary Data) was not printed because the difference (uncontrollable) in degree of immobilization could distort real ratio in affinities of low molecular weight (MW) glycans versus high MW glycoprotein. Concentration of the Abs was preselected to observe maximum signal close to possible upper relative fluorescence unit (RFU) value (~70,000). Data are shown in Figures 1 and 2. Notably, both mAbs do not bind SiaLeA glycans. mAb 58/1 binds desialylated form, LeA, whereas mAb 58/2 does not. mAb 58/2 was found to have more restricted epitope specificity. It binds only two related glycans (both are normal components of breast milk), whereas 58/1 is capable of binding to five glycans with rather different structure. However, all glycans contain the same common motif—Galβ1–3GlcNAcβ (LeC), marked in bold in the figures. Notably, neither LeC disaccharide as is, without substituents, nor most common naturally occurring LeC-terminated tetrasaccharide Galβ1–3GlcNAcβ1–3Galβ1–3Glc (LNT) are positive in the PGA. Taking into account all the mentioned features, one can reconstruct glycotope for the Abs like “Galβ1–3GlcNAcβ obligatory containing an additional Ab-contacting fragment,” apparently hydrophobic, like benzyl group, or methyl group of fucose or N-acetylglucosamine residues. One ligand, Galβ1–3GlcNAcβ1–3Galβ1–4Glc, drops out of this regularity, so another “reconstruction” of real epitope could be done, namely, “Galβ1–3GlcNAcβ with distorted/constrained conformation due to a bulky substituent neighboring to Gal residue.” Although both reconstructions are well compatible to each other.
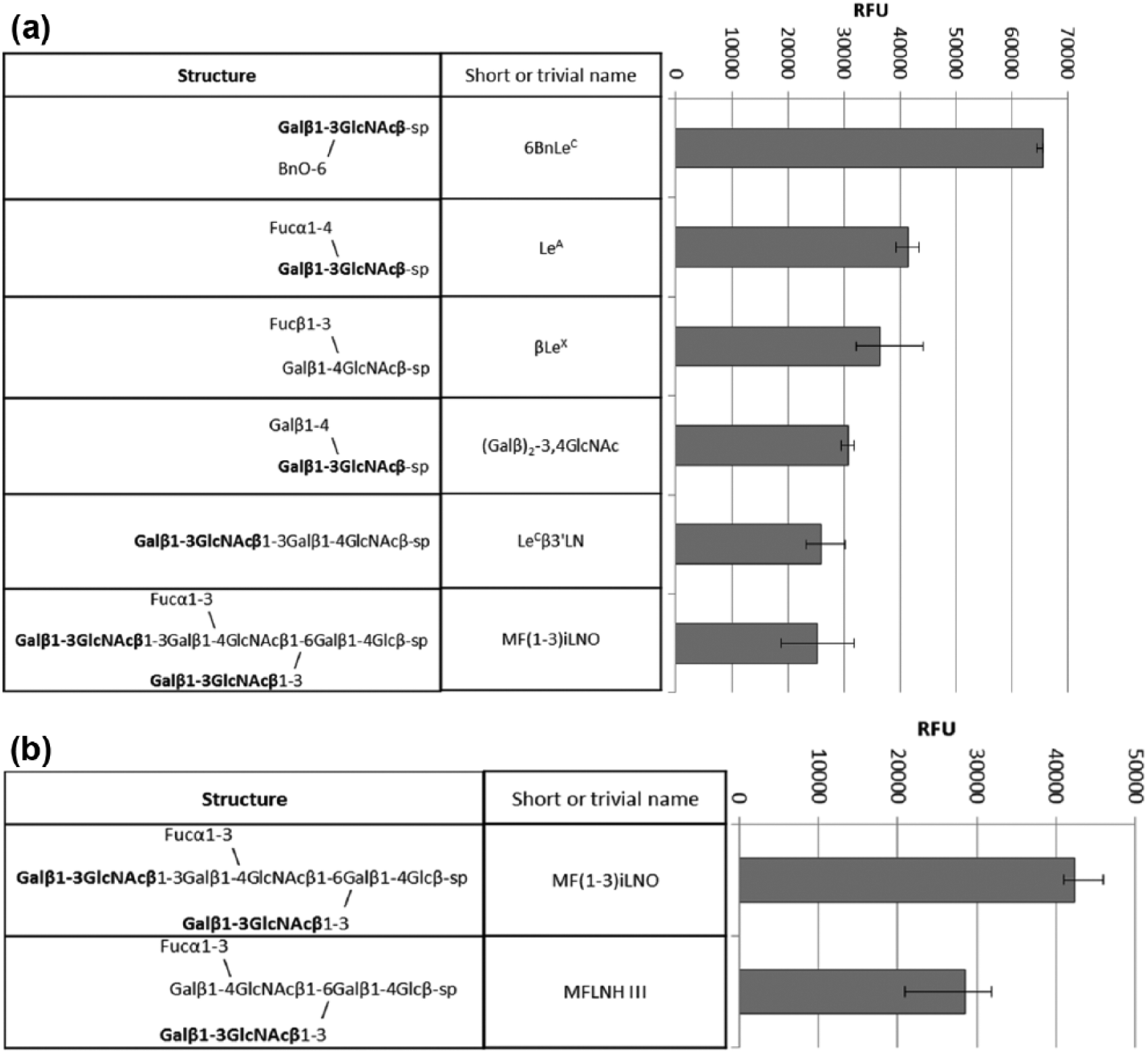
Top rank glycan ligands for (a) mAb 58/1, conc. 6 µg/mL and (b) mAb 58/2, conc. 4 µg/mL in PGA (full data of the PGA assay are presented in S6).
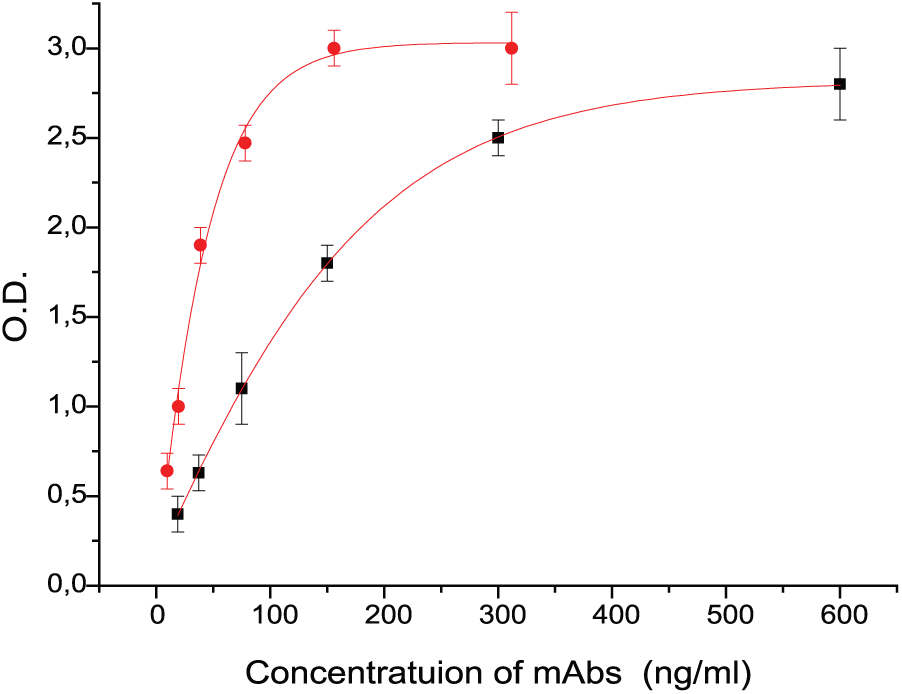
Direct binding of mAbs to 19-9 glycoprotein: 58/1 (black square), 58/2 (red circle).
Data of binding to PAA-conjugated glycans (enzyme-linked immunosorbent assay)
To make sure that all the observed mAbs–glycan interactions were specific, additional experiments on concentration dependence were done. Since PGA analysis is not convenient for the study of concentration dependence, we further confirmed PGA data with regular enzyme-linked immunosorbent assay (ELISA), where coating antigens were soluble polyacrylamide (PAA) conjugates of the glycans.8,9 We selected glycans demonstrated affinity in PGA, as well as related molecules, for ELISA experiments. One of them, Galβ1–3GlcNAcβ1–3GalNAcα (LeC-Tn), absent in PGA, was synthesized anticipating its unusual conformational properties comparing to disaccharide LeC, and its possible tackle to cancer-associated glycans. Glycoprotein CA19.9 was also included in the list of antigens for ELISA. Data are shown in Figure 2. The results of direct binding tests for the total set of PAA are presented as histograms in Figure 3. For convenience of data comparison, absorbance values characterizing binding of mAbs with PAA glycoconjugates are given only for one of the series of consecutive dilutions of Abs used in the experiment: 1:1.6 × 104 for 58/1 mAbs and 8 × 103 for 58/2 mAbs.
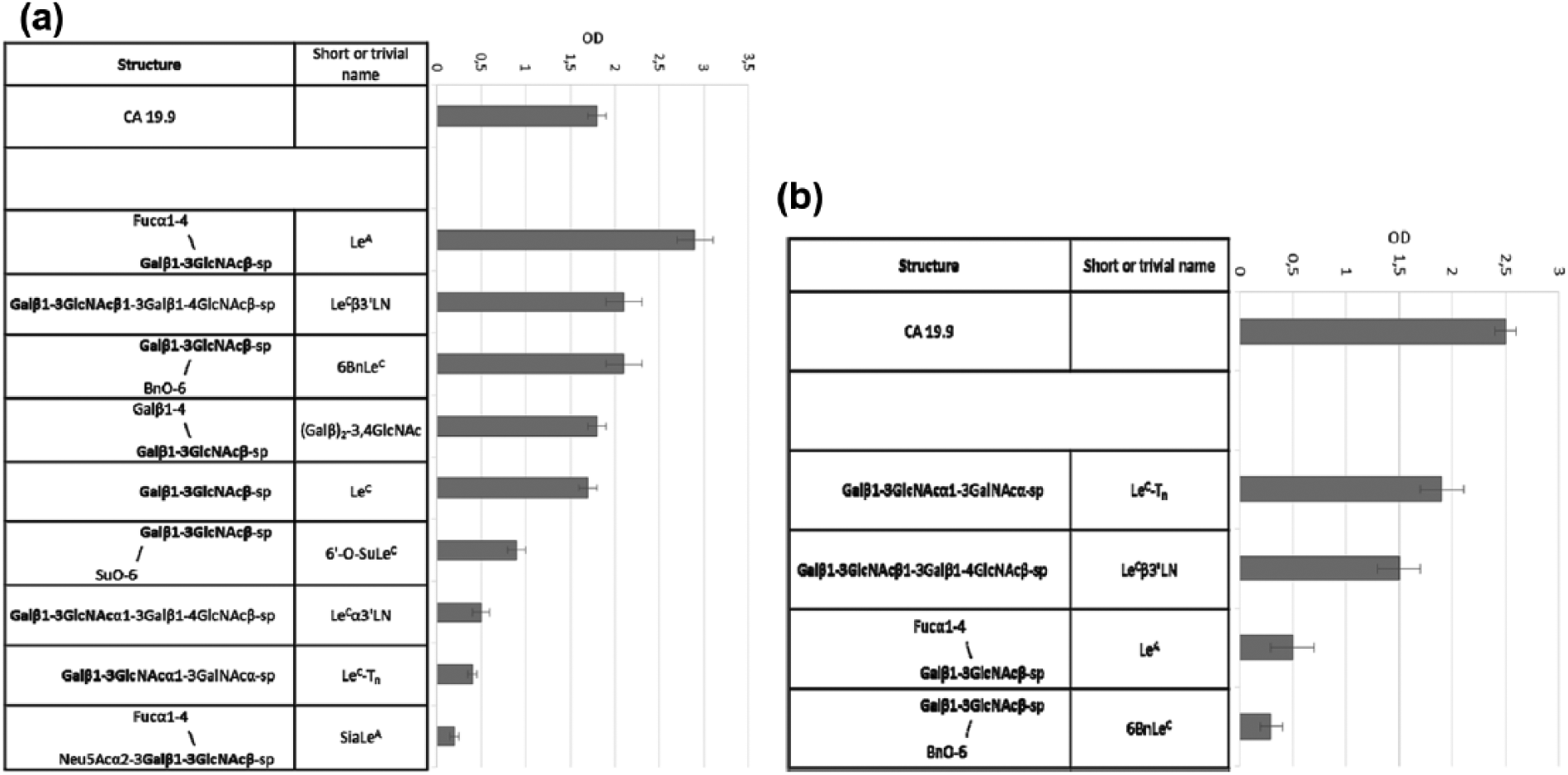
Direct binding of mAbs to PAA glycoconjugates: (a) 58/1 mAbs (150 ng/mL), no binding to Galα1–3GlcNAcβ-PAA, β Fuc isomer of LeA-PAA, 3’SiaLeC (b) 58/2 mAbs (156 ng/mL), no binding to Galα1–3GlcNAcβ-PAA, LeC3′αLN-PAA, LeC-PAA, SiaLeC-PAA, 3′SiaLeC-PAA, 6′-O-SuLeC-PAA, (Galβ)2-3,4GlcNAc-PAA, β Fuc isomer of LeA-PAA.
As in PGA assay, Ab 58/2 was found to be more narrowed, with top rank antigen found to be natural glycoprotein CA19.9, which demonstrated concentration dependence. The dependence curve for mAb 58/1 was more close to classical shape possessing well-expressed plateau. As in PGA, no significant binding was observed with SiaLeA tetrasaccharide. Generally, the data of ELISA confirmed the results of PGA, although the coincidence was not complete. The most pronounced seems to be rather good binding of mAb 58/1 (but not of mAb 58/2) to disaccharide LeC devoid of any additional substituents. We did not include both ligands that demonstrated the highest affinity toward 58/2, that is, nonasaccharide MF(1–3)iLNO and heptasaccharide MFLNH III, and therefore, LeC3′LN (low binder in PGA) and LeC-Tn (absent in PGA) apparently exhibited maximum reactivity.
Inhibitory ELISA
To confirm specificity of monoclonal binding, especially to unusual Bn-substituted glycan, we performed ELISA in an inhibitory version, using CA19.9 as coating antigen. The obtained data (Figure 4) are in accordance with direct binding, particularly high binder for mAbs 58/1 antigen 6BnLeC appears to be a good inhibitor; similarly high binder for mAbs 58/2 antigen LeC-Tn appears to be a good inhibitor in inhibitory ELISA. The same range of 50% inhibition concentration for 6BnLeC and LeCβ3′LN (mAbs 58/1) also argues in favor of specific binding rather than just hydrophobic interaction of benzyl-substituted molecule. At the same time, rather high concentration of 50% inhibition for the top rank antigens says that true cognate antigens for both Abs are more complicated/extended than all the probes used by us in our assays.
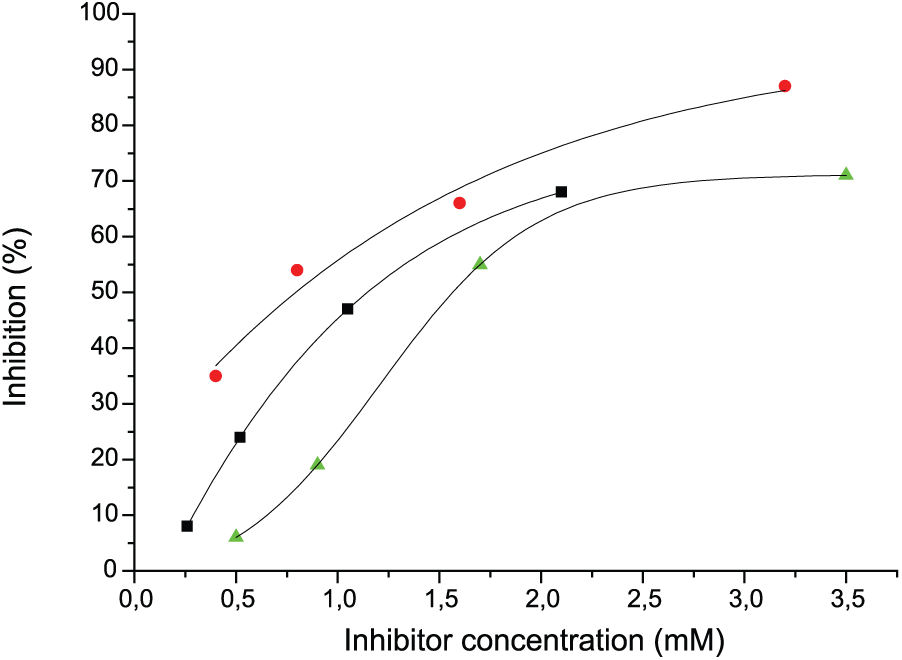
Inhibition of mAbs (1:104) binding to 19-9 by free (unconjugated) glycans: 58/1 mAbs and 6BnLeC (red circle), LeCβ3’LN (green triangle); 58/2 mAbs and LeC-Tn (black square). No inhibition in case 58/1mAbs and LeC-Tn (3.5 mM); 58/2 mAbs and 6BnLeC (2.1 mM).
Histochemistry
Immunohistochemical staining (Supplementary Material 8) of breast cancer tissue was performed with 58/1 Abs for five patients. The reactivity pattern was not tumor-specific; staining was observed in both normal and malignant cells; the only difference was found to be constant apical membranous expression in normal duct epithelial cells. The intensity of the staining in all cases was moderate, with no significant difference from case to case. The key difference between 58/1 and natural Abs against LeC is reactivity of 58/1 with normal breast ducts’ epithelium (moderate intensity, membranous cytoplasmic, with accentuation of apical membrane) and smooth myocytes of breast areola.
Discussion
mAb LU-BCRU-G7 (referred hereinafter as G7) against breast cancer–specific glycoprotein 230 kDa was generated in 1989, and analysis of antigen-binding specificity revealed
In this work, two mAbs capable of being a good capture/tracer pair for the specific detection of CA19.9 glycoprotein were obtained by isolation from ascitic fluid of cancer patients. It is known from literature that Abs against CA19.9 (http://www.glycoepitope.jp/epitopes?utf8=%E2%9C%93&keywords=SLeA) as well as against its Fuc free form CA-50 (http://www.glycoepitope.jp/epitopes?utf8=%E2%9C%93&keywords=SLeA) ultimately require the presence of sialic acid residue. In contrast to the above-mentioned canonical anti-CA19.9 mAbs, both 58/1 and 58/2 Abs did not bind specific carbohydrate epitope of CA19.9, tetrasaccharide SiaLeA; moreover, sialic acid abolished the binding. Glycan-binding profiling revealed specific asialo-motif Galβ1-3GlcNAc (LeC) as recognition glycotope both for 58/1 and 58/2. Thus, all three mAbs G7, 58/1, and 58/2 demonstrate similar epitope specificity, although not identical glycan-binding profile, particularly mAb 58/1 lacks unusual inability of three other Abs to recognize LeC disaccharide as a part of Galβ1–3GlcNAcβ1–3Galβ1–4Glc(NAc) chain. Interestingly, human natural Abs (in all individuals, both healthy donors and with breast cancer), isolated with the help of LeC hapten-specific affinity chromatography5,7 demonstrated similar inability to recognize LeC disaccharide located at terminal position of long carbohydrate chain and ability to bind LeC motif located at reducing end (Figure 5).

Unusual epitope specificity of human natural anti-LeC; mAbs G7 and 58/2 were found to have similar specificity: (a) conformational formulas of tetrasaccharide
Carbohydrate chain
Materials and methods
Reagents
Horseradish peroxidase (HRPO) labeled anti-Igs were obtained from Southern Biotechnology Associates Inc. (USA). Biotin-labeled goat-anti-mouse IgG + M were obtained from Thermo Fisher (USA), and Alexa555-labeled streptavidin were obtained from Life Science (USA). Tween 20, phosphate-buffered saline (PBS; tablets), and bovine serum albumin (BSA) were obtained from Sigma (USA). All other chemicals were analytical grade from Fluka (Switzerland) or Merсk (Germany). MaxiSorp 96-well microtiter immunoplates were obtained from Nunc (Germany). PAA-conjugated glycans and their spacered forms were obtained from Lectinity (Russia). Microchips were produced by Semiotik LLC (Russia).
Study of mAbs’ specificity by ELISA
Direct binding
Plates were coated with PAA-conjugated glycans or 19-9 glycoprotein, 10 µg/mL in 0.05 M Na-carbonate buffer, pH 9.6, for 1 h at 37°C, followed by blocking with 3% BSA in PBS (0.01 М Na2HPO4, 0.01 M NaH2PO4, 0.138 M NaCl, 0.0027 M KCl, pH 7.4) for 1 h at 37°C. Plates were washed three times with PBS containing 0.1% Tween-20 (PBS-0.1%). mAbs 58/1 and 58/2 (in 2-fold dilution starting from 1:103; 100 µL per well) were added and the plates were incubated for 1 h at 37°C and washed three times with PBS-0.1%. Anti-mouse Ig-HRPO conjugate (1:2000 dilution in PBS containing 0.3% BSA) were added and the plates were incubated for 1 h at 37°C. After washing with PBS-0.1%, the color was developed by 30-min incubation with 0.1 M sodium phosphate/0.1 M citric acid buffer, containing 0.04% o-phenylenediamine and 0.03% H2O2, and the reaction was stopped with the addition of 50 µL 1 M H2SO4. Absorbance was recorded at 492 nm using a microtiter plate reader (Multiskan MCC/340; Labsystems, Finland). The results were presented as mean value with standard deviation.
Inhibition assay
Plates were coated and blocked as described. In the next step, inhibitors in 2-fold dilution (from 3.5 to 0.25 mM) were added simultaneously with Abs. Plates were incubated for 1 h at 37°C and then developed as above. The percent of inhibition was calculated as (ODA − ODI) × 100/ODA, where ODA is the mean value of optical density in the absence of inhibitor and ODI is the mean value of optical density in the presence of inhibitor.
Study of mAbs’ specificity by microchip
Microarrays on glass microscope slides containing 380 glycans (50 mM solutions, at eight replicates each) were produced by Semiotik LLC (Moscow, Russia); 6 µg/mL solution of 58/1 and 4 µg/mL solution of 58/2 Abs in PBS-1% containing 1% BSA were applied to array (pretreated with PBS-0.1% for 15 min) and incubated in a humidified chamber for 1.5 h at 37°C. Then the microarrays were washed with PBS-0.1% and incubated with the 20 µg/mL solution of biotin-labeled secondary goat-anti-mouse IgG + M in PBS-0.1% under the same conditions. After washing with PBS-0.001%, the 1 µg/mL solution of Alexa555-labeled streptavidin in PBS-0.1% was added and arrays were incubated in a humidified chamber for 45 min at 23°C. After washing with PBS-0.05% and deionized water, the arrays were scanned on a InnoScan AL 1100 scanner (Innopsys, France) using the excitation wavelength of 543 nm. The obtained data were processed using ScanArray Express 4.0 software and the fixed 80-µm-diameter circle method as well as Microsoft Excel software. Data are reported as median RFUs of replicates. Median deviation was measured as interquartile range. A signal the fluorescence intensity of which exceeded the background (i.e. the signal from the surface which is not containing a ligand) value by a factor of 5 was considered as significant.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Russian Science Foundation grant #14-14-00579.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
