Abstract
Neuron-glial antigen 2 (NG2, also known as CSPG4) and hyaluronic acid receptor CD44 are chondroitin sulphate proteoglycans actively involved in brain development and its malignant transformation. Here, we aimed to compare prognostic significances of NG2, CD44 and Ki-67 expression in glioblastoma multiforme patients. Totally, 45 tissue samples and 83 paraffin-embedded tissues for 75 patients were analysed. The prognostic values of the genes were analysed using Kaplan–Meier survival curves. Grade III gliomas showed 2-fold difference in NG2 expression between anaplastic astrocytoma and oligoastrocytoma (10.1 ± 3.5 and 25.5 ± 14.5, respectively). For grade IV gliomas, upregulated NG2 expression (21.0 ± 6.8) was associated with poor glioblastoma multiforme prognosis (overall survival < 12 months) compared with glioblastoma multiforme patients with good prognosis (4.4 ± 3.2; overall survival > 12 months). Multivariate survival analysis using Cox proportional hazards model confirmed that high NG2 expression was associated with low survival of the patients (hazard ratio: 3.43; 95% confidence interval: 1.18–9.93; p = 0.02), whereas age (hazard ratio: 1.02; 95% confidence interval: 0.96–1.09; p = 0.42), tumour resection (hazard ratio: 1.03; 95% confidence interval: 0.98–1.08; p = 0.25) and sex (hazard ratio: 0.62; 95% confidence interval: 0.21–1.86; p = 0.40) did not show significant association with prognosis. Although the positive correlation was shown for NG2 and CD44 expression in the glioblastomas (Pearson coefficient = 0.954), Kaplan–Meier and multivariate survival analyses did not revealed a significant association of the increased CD44 expression (hazard ratio: 2.18; 95% confidence interval: 0.50–9.43; p = 0.30) or high Ki-67 proliferation index (hazard ratio: 1.10; 95% confidence interval: 1.02–1.20; p = 0.02) with the disease prognosis. The results suggest that upregulation of NG2/CSPG4 rather than changes in CD44 or Ki-67 expression is associated with low overall survival in glioblastoma multiforme patients, supporting NG2/CSPG4 as a potential prognostic marker in glioblastoma.
Introduction
For a long time, glioblastoma multiforme (GBM) treatment remains a challenge for oncologists, and survival has not been significantly improved over the last decades.1,2 Molecular markers for more precise glioma classification and prognosis could significantly contribute to the diagnostics and treatment of malignant brain tumours.3,4 However, despite the significant progress in this field, identification of molecular subtypes and search for new prognostic biomarkers for glioblastoma remain very actual. 5 During the last decade, a number of different potential biomarkers for glioblastoma progression-like cell invasion–related molecules NG2/CSPG4 and CD44 were suggested.6,7
Neuro-glial proteoglycan 2 (NG2), also known as chondroitin sulphate proteoglycan 4 (CSPG4), is a type-1 membrane protein expressed by a range of cell types within and outside the mammalian nervous system. In central nervous system (CNS), NG2+ cells (also known as polydendrocytes) are a population of CNS cells that are distinct from neurons, mature oligodendrocytes, astrocytes and microglia and usually are generally defined as oligodendrocyte precursor cells,8,9 although the cells are capable of neuronal differentiation in response to environmental stimuli as well. 10 In glioblastoma, NG2 expression identifies an actively proliferating cell population with an aggressive molecular signature.11,12 Possibly, NG2-positive cells may be potential cells of origin in the genesis of oligodendroglioma-like tumours as it was shown in a rat model. 13 The NG2+ cells demonstrate elevated resistance to radiotherapy and chemotherapy14,15 and represent potential target for cancer molecular therapy.16–18
CD44 is a cell adhesion molecule that represents glycoprotein/proteoglycan with facultative chondroitin sulphate chain(s) on the alternatively spliced core protein variants. It functions as a receptor for hyaluronan and many other extracellular matrix (ECM) components and plays an important role in tumour progression and metastasis. 19 CD44 belongs to glioblastoma cancer stem cell (CSC) markers and might promote cancer cell stemness by interacting with ECM components, growth factors and cytokines.20,21 CD44 protein marker expression correlates with molecular subtype of GBM 22 and positively associated with radioresistance of glioblastoma. 23 Data on the prognostic significance of CD44 in glioblastoma are controversial – it was suggested that enhanced CD44 expression is associated with poor survival rates in glioblastoma although this finding was not statistically significant; 24 lower levels of CD44 expression surprisingly correlated with lower survival for GBM patients. 25
Ki-67 is an established marker of cell proliferation, and Ki-67 index correlates with the clinical course of several cancer types. In brain cancer, current meta-analysis revealed that Ki-67 expression might be a predictive factor for poor prognosis in glioma patients. 26 In fact, the bioinformatical analysis has been applied to a common pool of glioma patients. However, an important point is that Ki-67 is one of the main molecular markers during diagnosis and stratification of patients into different grades, and low or high Ki-67 expression levels are directly associated with grade II–III or grade IV gliomas. In this term, Ki-67 expression really is a predictive factor for poor prognosis of glioma grade II–III patients,27,28 but the point about its prognostic value for glioblastoma (grade IV) patients remains unclear.
The aim of this study was to investigate and perform a comparative analysis of the prognostic significance of NG2/CSPG4, CD44 and Ki-67 in glioblastoma progression.
Patients and methods
Patients and tissue samples
All tissue samples were obtained from brain tumours during radical surgery at the Neurosurgical Centre of Research Institute of Circulation Pathology, Novosibirsk, Russia. Tissues were collected in RNAlater (Thermo Fisher Scientific, USA) and stored at −20°C or fixed in buffered 4% formalin solution and embedded in paraffin. H&E-stained sections were prepared to define representative tumour regions, and GBM diagnosis was confirmed according to the World Health Organization (WHO) classification by qualified pathologists. Control brain tissue samples were obtained from peritumoral area. In total, 75 patients with diagnosis of high-grade glioma (61 cases of glioblastoma (WHO Grade IV), 7 cases of anaplastic astrocytoma and 7 cases of anaplastic oligoastrocytoma (WHO Grade III)) were used in this study. The patient sample group had a median survival of 12 months and mean and median age of 48 and 51 years, respectively. All the glioblastoma patients received temozolomide in combination with radiotherapy and 8.3% patients received lomustin, mustophoran or avestin in addition. Informed consent was obtained from all individual participants included in the study. The study protocol was approved by the Local Ethics Committee in accordance with the Helsinki Declaration of 1975. Clinical information is presented in Table 1.
List of patients used in this study.

Reverse transcription polymerase chain reaction analysis
Total RNA was extracted from the clinical samples using the TRIzol® Plus RNA Purification Kit (Thermo Fisher Scientific) according to the manufacturer’s instructions. Complementary DNA (cDNA) was synthesised from 1 to 2 μg of total RNA using a First-Strand cDNA Synthesis Kit (Fermentas, USA) and 1/10th of the product was subjected to polymerase chain reaction (PCR) analysis. Quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) was performed using the CFX96™ Real-Time PCR Detection System (Bio-Rad, USA) and the Taq-pol (IMCB, Russia) Maxima SYBR Green/RO master mix (Thermo Fisher Scientific) under the following conditions: 95°C for 3 min, followed by 40 cycles at 95°C for 15 s, 59°C for 30 s and 72°C for 1 min. The total reaction volume was 25 µL. The relative amount of messenger RNA (mRNA) was normalised against glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA, and the fold change for each mRNA was calculated by the 2−ΔCt method. Primer sequences were as follows: NG2-F 5′-TATGTTGGCCAGACTTGCAT-3′, NG2-R 5′-TGCAGGTCTATGTCGGTCAG-3′; CD44-F 5′-GACAAGTTTTGGTGGCACG-3′, CD44-R 5′-CACGTGGAATACACCTGCAA-3′; and GAPDH-F 5′-AAGGTGAAGGTCGGAGTCAA-3′, GAPDH-R 5′-AATGAAGGGGTCATTGATGG-3′.
Immunostaining
For immunohistochemistry (IHC), 3- to 3.5-μm sections of formalin-fixed paraffin-embedded tissue sections were treated using Lab Vision™ Autostainer 720-2D (Thermo Fisher Scientific), primary anti-NG2 rabbit polyclonal antibody (1:200; Abcam, USA), anti-Ki-67 rabbit monoclonal antibody (mAb; Thermo Fisher Scientific) and UltraVision Quanto Detection System HRP DAB (Thermo Fisher Scientific) according to the manufacturer’s instructions. Staining patterns were counterstained with haematoxylin and observed by light microscopy with magnification of 400× (Axiostar Plus; Carl Zeiss, Jena, Germany).
Statistical analysis
Statistical analyses were performed using OriginPro 8.1 software; a value of p < 0.05 was considered to indicate a statistically significant difference. Data are expressed as the mean ± standard error of mean (SEM). Pearson correlation coefficient was acquired using OriginPro 8.1 software. Patient survival was analysed using the Kaplan–Meier with MedCalc 12.5.0.0 software.
Results
High NG2/CSPG4 expression is associated with poor prognosis in patients with glioblastoma
The expression levels of NG2 in different-grade human gliomas (anaplastic astrocytoma and oligoastrocytoma (grade III) and glioblastoma (grade IV)) were determined by real-time RT-PCR and normalised to that of GAPDH. RT-PCR analysis revealed significant (up to 3-fold) difference in NG2 expression between anaplastic astrocytoma and anaplastic oligoastrocytoma tumours (10.1 ± 3.5 and 25.5 ± 14.5, respectively; Figure 1). Interestingly, the average expression level of NG2 in glioblastoma tumours was more similar to that of anaplastic astrocytoma (11.8 ± 15.9) but high heterogeneity of NG2 expression in these tumours supposed their possible association with clinical characteristics of the patients.
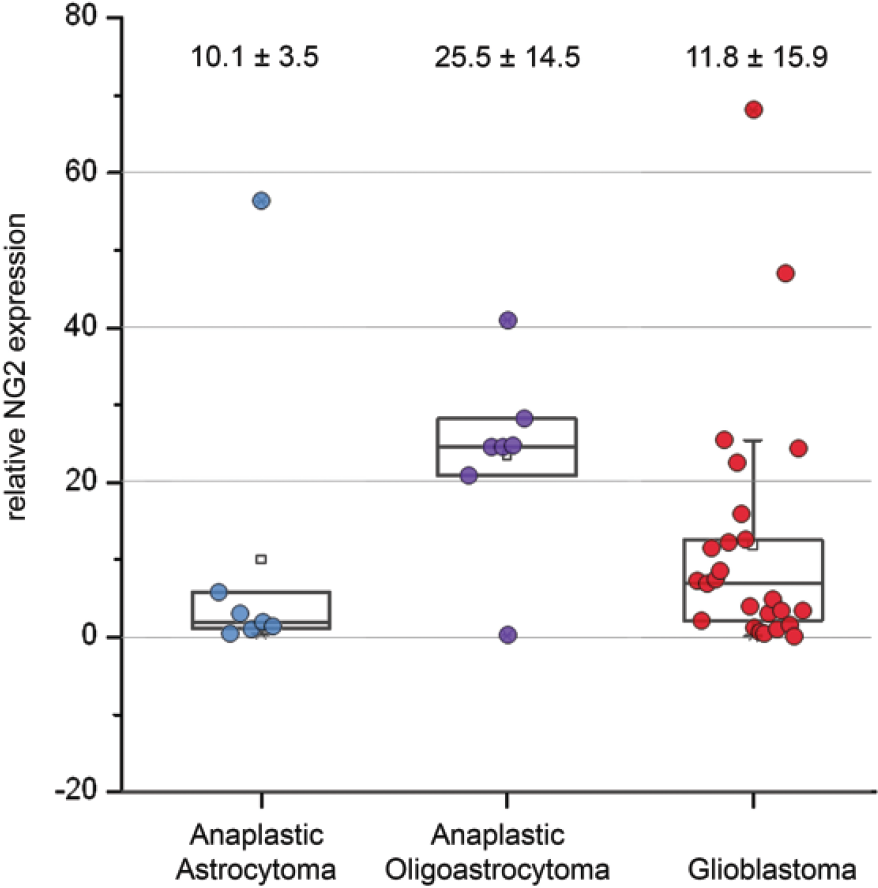
Expression of NG2/CSPG4 in human gliomas. The patients are grouped according to clinical diagnosis. Real-time RT-PCR analysis: intensity of the amplified DNA fragments normalised to that of GAPDH. The mean ± SD is presented for each group (OriginPro 8.1).
Sub-division of the glioblastoma patients into two clinical sub-groups with good prognosis (overall survival (OS) more than 12 months) and poor prognosis (OS less than 12 months) revealed a positive association of high NG2 expression levels with poor disease prognosis (Figure 2(a)). On protein level, the effect was confirmed using immunohistochemical staining with anti-NG2 antibody (Figure 2(b)). According to the Kaplan–Meier analysis, patients with high NG2 expression had a significantly shorter median survival of 8 months compared with 13 months in the patients with low NG2 expression. Survival probability for patients with low NG2 expression was significantly higher (p < 0.05) than for those with high NG2 expression.

CSPG4/NG2 expression in human glioblastoma with high and low overall survival (OS). (a) Real-time RT-PCR analysis: intensity of the amplified DNA fragments normalised to that of GAPDH, and the graphs show the mean expression levels ± SD (OriginPro 8.1). The patients (17–88) are grouped according to high and low survival. (b) Immunohistochemical analysis of CSPG2/NG2 in (1) normal brain tissue and glioblastoma specimens with (2) good and (3) poor disease prognosis. (c) Kaplan–Meier curves for NG2-low and NG2-high patient groups.
The obtained data demonstrate that NG2 expression is associated with OS of patients with glioblastoma, supporting NG2 as the clinically perspective prognostic marker for glioblastoma.
NG2 and CD44 expression levels are highly correlated in glioma patients
Cell-surface glycoprotein CD44 is another (facultative) proteoglycan bearing chondroitin sulphate chain(s) at specific CD44 isoforms. It is known as a CSC marker in different tumours including breast, lung, pancreas, prostate, colorectal, renal, and ovarian. CD44 expression was also suggested as a useful prognostic indicator for GBM. 25
For the comparative analysis, CD44 expression was determined in the same glioma tissue samples analysed for NG2 expression (Figure 2). It was shown that the NG2 and CD44 expression levels were very similar within the good- or bad-prognosis patient sub-groups (Pearson coefficient = 0.954) hypothesising a coexpression of NG2 and CD44 in the same cancer cell subtype (Figure 3(a)). However, Kaplan–Meier survival analysis for high- and low-CD44 glioblastomas demonstrated no significant difference (p > 0.4) between these groups, unlike to high- and low-NG2 tumours (Figure 3(b)). The results suppose a different molecular mechanism for dysregulation of NG2 and CD44 transcription in glioblastoma, even if their expression levels in the tumours are similar.

NG2 and CD44 expression in glioblastoma specimens. (a) RT-PCR expression analysis for normal brain tissue and different prognostic groups of glioblastoma. (b) Kaplan–Meier survival analysis for high- or low-CD44 tumours.
Ki-67 expression is not associated with prognosis for glioblastoma patients
Ki-67 is a nuclear proliferation marker used in clinic for diagnostics and differentiation of high- and low-grade gliomas. It was suggested as a prognostic factor for low-grade gliomas but its use as prognostic indicator of cerebral high-grade glioblastoma is still debated.
All glioblastoma tumours (stage IV) are characterised by high Ki-67 expression; so, in our case, all patients under the study were subdivided into two sub-groups according to the clinical information on good or poor OS (OS > 12 months or OS < 12 months) as it was done before for NG2 analysis. Ki-67 expression levels in the same glioblastoma tissue samples as for NG2 and CD44 were determined by immunohistochemical staining during the routine clinical diagnostics (Figure 4(a)). It was shown that patient groups with the good prognosis and poor prognosis demonstrated almost no difference in median Ki-67 expression levels (18% and 16%, respectively; Figure 4(a)). Grouping of the glioblastoma tumours according to the relatively ‘low’ or ‘high’ Ki-67 expression levels with the subsequent Kaplan–Meier survival analysis also did not reveal the significant differences between the low- and high-Ki-67 tumours (Figure 4(b)). Overall, the results do not demonstrate an association of the Ki-67 expression levels with the patients’ survival and thus cannot serve as a reliable prognostic marker in glioblastoma.

Ki-67 levels in glioblastoma tumours with different prognostic characteristics. (a) Ki-67 expression in different prognostic groups. IHC analysis and Ki-67 expression were rated as a percentage of cells expressing the marker. (b) Kaplan–Meier survival analysis for patients with high- and low-Ki-67 expression level.
In addition to the bilateral analysis of the prognostic significance of the studied biomarkers, multivariate survival analysis using Cox proportional hazards model was used to assess the impact of clinical metrics and NG2, CD44 and Ki-67 expression on patients’ survival. It was shown that only high NG2 expression was associated with low survival rate of the patients (hazard ratio (HR): 3.43; 95% confidence interval (CI): 1.18–9.93; p = 0.02), whereas age (HR: 1.02; 95% CI: 0.96–1.09; p = 0.42), tumour resection (HR: 1.03; 95% CI: 0.98–1.08; p = 0.25), sex (HR, 0.62; 95% CI: 0.21–1.86; p = 0.40), high CD44 expression (HR: 2.18; 95% CI 0.50–9.43; p = 0.30) and high Ki-67 proliferation index (HR: 1.10; 95% CI: 1.02–1.20; p = 0.02) did not demonstrate significant association with the prognosis (Table 2).
The impact of clinical metrics and NG2, CD44 and Ki-67 expression on patients’ survival by multivariate survival analysis using Cox proportional hazards model.

CI: confidence interval.
Discussion
This study for the first time presents a comparative analysis of the prognostic values of NG2, CD44 and Ki-67 expression levels in glioblastoma (grade IV glioma) tumours.
The obtained data support a suitability of NG2 proteoglycan as a potential prognostic biomarker for GBM patients. The results are in line with the published data on the potential prognostic value of NG2 in several tumour types 29 including chordoma, 30 hepatocellular carcinoma, 31 head and neck cancer 32 and GBM. 15 At the same time, the presented comparative analysis of NG2 and CD44 prognostic significance extends our knowledge on NG2 involvement into brain carcinogenesis.
It is known that high NG2 proteoglycan expression is a molecular marker of the specific NG2+ brain cell population representing oligodendrocyte progenitor cells (OPC).8–10 Activation of NG2 expression in glial cancer cells protects the NG2+ cells from oxidative stress, 14 facilitates vessel formation,33–35 contributes to high chemo- and radioresistance of glioma13–15 and imparts the NG2+ glioma cells stem cell–like phenotype. 11 The simultaneous upregulation of NG2 and CD44 expression in the GBM tumours with high positive Pearson coefficient (p = 0.954) seems support the idea; however, the Kaplan–Meier curves suppose more complex interrelation between NG2+ and CD44+ cell populations. Possibly, the simultaneous activation of NG2 and CD44 expression reveals a stem cell–like cell population inside the heterogeneous tumour mass, but this population possesses its own heterogeneity as well. OPC subpopulation possibly exists inside the common glioma stem cell population, and the NG2+ cells but not CD44+ cells characterise the most aggressive cell phenotype and are associated with poor prognosis in glioblastoma patients. This hypothesis can be indirectly supported by the fact that NG2 is frequently co-expressed with stem cell markers nestin and vimentin but rarely with CD133 in GBM biopsies from newly diagnosed and untreated patients. 15 Five human patient-derived primary glioma-initiating cell (GIC) lines coexpress NG2, nestin, platelet-derived growth factor alpha (PDGF-α), and glial fibrillary acidic protein (GFAP) and represent a unique type of cells reminiscent of an immature phenotype that closely resembles but is not identical to NG2 glia. 36 Comparative analysis of established glioma stem cell line SHG-139S with its parental conventional SHG-139 glioma cell line revealed different molecular marker patterns for these cells. SHG-139 cells were positive for A2B5, GalC, GFAP, S-100 and vimentin, while SHG-139S cells were positive for NG2, A2B5 and nestin and negative for Vimentin, CD133 and IDHR132H. 37
As to CD44 prognostic value in GBM, the question is still under investigation. It was shown that CD44 is one of the GBM CSC markers, 20 and high expression of CD44 identifies proneural subtype of cancer cells. 22 Only CD44 expression correlates positively with radioresistance of primary glioblastoma cell culture, while all other tested CSC markers (CD133, nestin, SOX2, GFAP, Musashi1 and L1 cell adhesion molecule) show an inverse correlation. 23 From the other side, in spite of elevated CD44 expression in GBM tissues, lower levels of CD44 expression surprisingly correlate with lower survival. 25 Association of enhanced CD44 expression levels with poor survival rates is not statistically significant to state it as a valid prognostic factor in GBM. 24
Overall, the data suggest that characterisation of NG2+ cells as CSC or GIC populations in GBM is somehow “terms-dependent” and still remains debatable. Possibly, the presence of OPC and their characteristic markers (CSPG4/NG2) could be better prognostic marker for glioblastoma than known stem cell biomarkers. 20
Another important moment is a high heterogeneity of NG2 molecule itself. 38 In mice model, co-existence of several sub-populations of NG2 glia is demonstrated, supporting the idea of a substantial regional and developmental heterogeneity in this subtype of microglia. 39 In GBM cell lines, 48 immunologically distinct NG2/CSPG4 isoforms are identified using a panel of 60 anti-NG2 mABs, and 14 mAbs among them identify specific NG2/CSPG4 isoforms common for foetal and neoplastic cerebral sections and absent in the adult brain. These results highlight a very complex pattern of NG2/CSPG4 isoform expression in foetal and neoplastic brain tissues. 40
Possibly, the appearance of different foetal-like NG2 core protein isoforms in GBM tissues could be accompanied also by changes in their polysaccharide chondroitin sulphate (CS) chains as well. It was shown that both NG2 and CD44 bear foetal-like CS chains in different tumours 41 although it was not demonstrated for brain tumours directly.
Taken together, these results assume the complex changes in NG2 molecule in glioblastoma tissues and cells both at NG2 core protein and CS chains levels.
For Ki-67, the obtained results indicate low prognostic value of this molecular marker in high-grade (grade IV, glioblastoma) glioma patients. It contradicts with previously published results, where proliferation marker Ki-67 was identified as an independent prognostic indicator in Chinese GBM patients. 42 Persson and Englund 43 also suggest possible prognostic use of immunohistochemical Ki-67 proliferation index in GBM but note that different methods of Ki-67 evaluation may give markedly dissimilar results pointing a necessity of standardisation of Ki-67 quantification methods.
In summary, the presented data suggest NG2/CSPG4 as more valuable prognostic factor for glioblastoma than CSC marker CD44 or proliferation marker Ki-67. Practical use of the potential NG2 marker during clinical IHC characterisation of glioblastoma tumours could be useful for choice of the treatment strategy. High NG2 expression in glioblastoma cells might predict a poor response to chemo- and radiotherapy and suppose a second-line therapy or NG2-targeted drugs as a better treatment strategy to these patients. The pilot experiments show that abrogating NG2 function with lentivirally encoded shRNAs targeting NG2 in two heterogeneous GBM xenografts significantly reduces tumour growth, oedema and angiogenesis and normalises vascular function, supporting NG2 as a perspective target for cancer molecular therapy. 18 Targeting glioblastoma with natural killer (NK) cells and mAb against NG2/CSPG4 in animal model in vivo prolongs animal survival and demonstrate a proof of concept that this antibody may hold potential for the treatment of GBM. 17 Further studies on diagnostic and therapeutic potential of NG2 for patient stratification and treatment are a perspective direction for future research.
Footnotes
Acknowledgements
A.Y.T. and G.M.K. have contributed equally.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by the research grant from Russian Scientific Foundation (grant RSF No. 16-15-10243).
