Abstract
Previously, we have shown that A549, a human lung adenocarcinoma, can be adapted to nitric oxide (NO●). NO● is a nitrogen-based free radical that is synthesized by a family of enzymes known as nitric oxide synthases. NO● has been shown to be overexpressed in patient populations of different cancers. In addition, it has been observed that patients who express high levels of nitric oxide synthases tend to have poorer clinical outcomes than those with low levels of expression. The original cell line A549 (parent) and the adapted A549-HNO (high nitric oxide) cell line serve as a useful model system to investigate the role of NO● in tumor progression and prognosis. We have previously shown that the A549-HNO-adapted cells grow aggressively when compared to A549-parent cells. Furthermore, we have shown that the A549-HNO-adapted cells exhibit a higher percentage of cell viability when exposed to ultraviolet and X-ray radiation than the A549-parent cells. Cancer patients who develop resistance to one treatment often become resistant to other previously unencountered forms of treatment. This phenomenon is known as cross-tolerance. To determine whether NO● is a potential cross-tolerance causing agent, we have expanded our research by conducting parallel studies to a variety of other agents and conditions beyond radiation and ultraviolet exposure. We exposed both cell lines to varying levels of chemotherapeutic drugs (taxol and doxorubicin), temperature, pH, calcium chloride, cadmium chloride, copper chloride, sodium chloride, ferrous chloride, and sodium-R-lipoic acid. Our results show that the A549-HNO cells exhibit greater viability than the A549-parent cells when exposed to each of the various conditions. Therefore, NO● is one potential driving force that can make tumor cells exhibit cross-tolerance.
Introduction
In recent years, there have been significant breakthroughs in our knowledge of lung cancer. The discovery of driver oncogenes in adenocarcinomas of the lung has resulted in a change in the treatment of these tumors. Treatments utilizing biological agents targeting these genomic changes have shown to be successful in subsets of lung cancer patients. Despite all of these advances in molecular targeted therapies, DNA-damaging cytotoxic therapies remain the main treatment of lung cancers. Radiation and chemotherapeutics have been shown to be effective in individual lung cancer patients; however, the acquired resistance toward these two forms of treatment diminishes their effect. Perturbations in DNA repair mechanisms are known to alter the resistance of the affected tumors to chemotherapeutic agents and radiation. These perturbations in DNA repair arise due to gene mutations, epigenetic events, and changes in signal transduction pathways.1–4
Radiation therapy is one of the modalities used to treat cancer patients. Radiation generates free radicals which are lethal as they result in unrepairable DNA double-strand breaks (DSB). Like the free radicals produced by radiation, it is believed that NO● drives tumor cells to become more aggressive. With an increasing number of tumor stem cells for a given dose of radiation, the chance of causing a lethal event is reduced. Other factors affect the sensitivity of a tumor to radiation, such as the tumor environment (hypoxia), upregulation or downregulation of DNA repair pathways, and changes in cell survival pathways.5–8
Chemotherapy is another cytotoxic method used to treat lung cancers. Attempts to overcome chemoresistance, such as finding drugs with different mechanisms of action or figuring out by trial and error which drug to give to each patient, have not been very successful. Chemoresistance can be divided into two groups: (1) de novo resistance in untreated patients and (2) acquired resistance developed by exposure to another treatment agent.9–11
Both radiation and chemotherapy kill cancer cells by inducing DNA damage, such as DNA DSBs. Several studies have linked perturbations in DNA damage as a contributing factor in cancer cell therapeutic resistance.4,12–14
Nitric oxide (NO●) is a nitrogen-based free radical that is synthesized by a family of enzymes known as nitric oxide synthases (NOS). The NOS family consists of inducible NOS (iNOS), endothelial NOS (eNOS), and neuronal NOS (nNOS). Nitric oxide is involved in several normal biological processes including vasodilation, neurotransmission, and macrophage-mediated immunity. Vasodilation and neurotransmission are regulated by a low concentration of NO●, produced by eNOS and nNOS, while inflammation is regulated by a high concentration of NO●, produced by iNOS.15–19
Clinical studies have shown that all three isoforms (iNOS, eNOS, and nNOS) are overexpressed by tumor cells. 20 In cancer biology, NO● is known to have a dual effect. 21 Low levels of NO● promote tumor growth, while high levels of NO● are known to be cytotoxic for tumor cells. At low levels, NO● causes tumor growth in a cyclic guanosine monophosphate (cGMP)-dependent manner. NO● binds easily to the ferrous iron of the enzyme soluble guanylate cyclase (sGC), which results in an increase in cGMP production.20–25 At higher concentrations, NO● is involved in growth arrest and cell death pathways. Since NO● is a highly reactive molecule, it generates different NO● metabolites such as nitrites and nitrate, S-nitroso-thiols, and peroxynitrite that are involved in DNA damage. However, cancer cells are able to bypass cell death by modulating oncogenes and tumor suppressors, giving rise to NO●-resistant cell populations. This parallels the resistance of chemotherapy and/or radiation therapy of these tumors cells in a NO●-mediated cross-tolerance manner. 26 It has also been observed that patients who present or progress to high levels of NOS expression tend to have poorer clinical outcomes than those with low-level expression. However, the role NO● plays in tumor pathophysiology is still not completely understood.27–35
Previously, our lab has established an in vitro model system to determine whether the cells could adapt to increasing levels of NO●. This model would mimic the spectrum of NO expression found in cancer patient populations. The in vitro model system consisted of 10 cancer cell lines; of which, 5 were adenocarcinomas (1 lung cancer + 4 breast cancer) and 5 were squamous carcinomas (head and neck cancer). The original cell lines were called “parent” cell lines. These “parent” cells were adapted to increasing levels of NO● donor, giving rise to a new set of high nitric oxide (HNO) cell lines. 36 DETA-NONOate was chosen as the NO● donor for the adaptation process because of its high level of free radical donation and relatively long half-life. The adaptation threshold was 600 µM, a dose that was lethal when introduced directly to the parent cell lines; however, the NO●-adapted cells were able to survive in this environment.36–40 The adapted HNO cells were morphologically identical to the parent cells, but they exhibited major phenotypic changes. The HNO cells grew more aggressively than their corresponding parent cell lines under both normal and low-nutrient conditions. 41
Furthermore, the adapted A549-HNO cells were shown to survive better and thrive in hypoxic (oxygen deficient) conditions than A549-parent cells. To mimic the hypoxic conditions, the A549-parent and A549-HNO cells were exposed to varying concentrations of CoCl2 (cobalt chloride). The A549-HNO cells expressed higher levels of hypoxia-inducible factor 1 alpha (HIF-1α), a transcription factor commonly upregulated under hypoxic conditions (unpublished data).
In this study, we will focus on A549-parent and A549-HNO human lung adenocarcinoma cell lines. As mentioned previously, long term NO● exposure resulted in more aggressive, fast growing tumors. Previous studies have shown that the DNA damage response (DDR) is altered in A549-HNO cancer cells.42,43 The A549-HNO cells were more resistant to both ultraviolet (UV) and X-ray radiation than the corresponding A549-parent cells. This shows that A549 cells have acquired a NO●-mediated cross-tolerance to radiation and UV via NO●.42,43 This is relevant to clinical observations in cancer patients where resistance to one form of treatment (chemotherapy) can lead to resistance to other forms of treatment (radiotherapy) and vice versa. We have shown that this could be due to the induction of DNA repair pathways in the A549-HNO-adapted cells. The A549-HNO cells show enhanced anti-apoptotic signaling.42,43 Exposure to NO● resulted in the HNO cells to be more resistant to the oxygen-based free radical, H2O2. 41
In this study, we have looked at the cross-tolerant properties of A549-HNO in other environmental stressors. We exposed the A549-HNO and A549-parent cells to chemotherapeutic agents (taxol and doxorubicin), higher temperatures, a broad pH range, various concentrations of calcium chloride (CaCl2), heavy metal salts (cadmium chloride (CdCl2), copper chloride (CuCl2), ferrous chloride (FeCl2)), various concentrations of sodium chloride (NaCl), and sodium-R(+)-lipoic acid (NaRLA).
Materials and methods
Cell culture and cell lines
Except where noted, all media and supplements were purchased from Invitrogen (CA, USA). All other reagents were from Sigma-Aldrich (MO, USA). The HNO-adapted A549 (A549-HNO) human lung adenocarcinoma cell line was previously prepared.
37
In brief, incremental doses of the nitric oxide donor (DETA-NONOate) were given to the original tumor cells (i.e. the “parent” cell line, grown without the presence of NO● donor). Cells were adapted to 600 μM of DETA-NONOate. A549 cells were grown in RPMI-1640 media supplemented with 10% fetal calf serum inactivated at 56°C for 30 min, 100 U/mL of penicillin, 100 μg/mL of streptomycin, 2 mM of
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
A549-parent and A549-HNO-adapted cells were seeded (100 µL) onto 96-well plates in the RPMI media (HNO cell media contains 600 µM of NO● donor) and incubated for overnight. Then, 5 h before the end of the incubation time, 100 µL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution was added to each well containing the cells. The cells were incubated in a CO2 incubator at 37°C for 5 h. After the incubation time, the media was removed, and 100 µL of dimethyl sulfoxide (DMSO) was added in each well to dissolve the crystals. The plates were put in the incubator for 5 min. Cellular viability was measured by reading at 540 nm absorbance on SpectraMax® Plus 384 Plate Reader (Molecular Devices, CA, USA).
Taxol growth assays
A549-parent and A549-HNO-adapted cells were seeded (100 µL) onto 96-wells plates in the RPMI media (HNO cell media contains 600 µM of NO● donor) and incubated for overnight. A volume of 100 µL of 200 µM taxol stock solution (HNO cell stock solution contains 600 µM of NO● donor) was added to the first column of wells in a 96-well microtiter plate and serially diluted (1:1). Then the cells were incubated at 37°C for 48 h. MTT assays were used to measure the cell viability of both parent and HNO-adapted cell lines.
Doxorubicin growth assays
A549-parent and A549-HNO-adapted cells were seeded (100 µL per well) onto 96-well plates in the RPMI media (HNO cell media contains 600 µM of NO● donor) and incubated for overnight. A volume of 100 µL of 50 µM doxorubicin stock solution (HNO cell stock solution contains 600 µM of NO● donor) was added to the first column of wells in a 96-well microtiter plate and serially diluted (1:1). The cells were incubated at 37°C for 24 h. MTT assays were used to measure the cell viability of both parent and HNO-adapted cell lines.
Growth curves with varying temperature
Harvested cells (A549-parent and A549-HNO) were aliquoted into 10 sterile polymerase chain reaction (PCR) tubes and placed in a programmed thermocycler (Programmable Thermal Controller; MJ Research, Inc., Waltham, MA) set to increase at specific temperatures (30°C, 37°C, 40°C, 42°C, 44°C, 46°C, 48°C, 50°C, 55°C, and 60°C). The samples were held to temperature for 5-min periods. Then, treated cells were plated onto 96-well plates and incubated for 72 h at 37°C. Afterward, the MTT assay was carried out to determine the quantification of cellular viability.
Growth curves with varying pH
Harvested cells (A549-parent and A549-HNO) were plated onto 96-well plates in the RPMI media for overnight incubation. The standard RPMI media was split into tubes, then the media was adjusted using either HCl or NaOH on a microprocessor pH meter (Hanna Instruments, Woonsocket, Rhode Island) at specific pH (3, 4, 5, 6, 7, 8, 9, 10, 11, and 12). They were then sterile filtered through a 0.22 µm filter. After overnight incubation, the media was replaced with various pH media onto 96-well plates and incubated for 48 h at 37°C. Afterward, the MTT assay was carried out to determine cellular viability.
Growth curves with varying CaCl2 concentrations
The A549-HNO and A549-parent cells were plated onto 96-well plates in the RPMI media for overnight incubation. The next day, a serial dilution was performed with CaCl2, creating a range of different calcium concentrations in the 96-well plates. After 48 h of incubation, the MTT viability/proliferation assay was performed to quantify cell growth.
Growth curves with varying CdCl2, CuCl2, or NaCl concentrations
A549-parent and A549-HNO-adapted cells were plated (100 µL per well) onto 96-well plates using RPMI media for overnight incubation. The next day, a serial dilution was performed with CdCl2, CuCl2, and NaCl, creating a range of different concentrations. After 48 h of incubation, the MTT viability/proliferation assay was performed to quantify cell growth.
Growth curves of iron chloride
A549-parent and A549-HNO-adapted cells were plated (100 µL per well) onto 96-well using RPMI media for overnight incubation. Iron chloride was dissolved in water at a concentration of 1 M and sterile filtered. The next morning, various concentrations of FeCl2 solution were serially diluted from 100 to 0.19 mM. They were then incubated for 24 h. Afterward, the MTT assay was carried out to determine the quantification of cellular viability as reported above.
Growth curves of NaRLA
A549-HNO and A549-parent cell lines were plated (100 µL per well) onto 96-well plates using RPMI media for overnight incubation. The A549-HNO cells were incubated in media with NO● donor. The A549-HNO and A549-parent cells were exposed to various concentrations of NaRLA for a period of 24 h (both in media without NO● donor). Cell growth was measured using the MTT viability/proliferation assay.
Gene chip assay
In addition, we carried out gene chip assays comparing the gene expression profile of the A549-parent to the A549-HNO cells. Table 1 provides a list of these genes with their corresponding fold-change values. Gene expression studies of A549-parent and A549-HNO cells were performed using the Quant Array 3.0 system (PerkinElmer, MA, USA). A comparative analysis of the data collected was carried out using an R 2.9 Windows compatible platform and the statistical analytic package “Limma 2.16.” 44 Background correction was carried out as described, 45 using “Normexp” background correction protocol, with an “Offset” threshold value of 50. The weights of control spots were positioned at 0 for background correction. The data were normalized using “loess” and “scale” methods of “Normalize Within Arrays” and “Normalize Between Arrays” for within-array and between-array methodologies, respectively, as described in detail. 46 The normalized signals were used to determine the differential expression levels using linear modeling systems through “lmFit.” The Bayes statistics was carried out using “eBayes” and “topTable,” as described. 47
Gene chip results comparing the A549-parent cell line to the A549-HNO-adapted cell line.
Results
The A549 cell line which was adapted to NO● was used throughout this study as previously prepared. 36 Cells were gradually introduced to increasingly higher levels of the nitric oxide donor DETA-NONOate (at 50 µM increments) over the course of approximately 65 days, reaching a final concentration of 600 µM of DETA-NONOate. Due to the short half-life of DETA-NONOate (approximately 30 h at 37°C and pH 7.4), media was refreshed every 3–4 days. As in our previous studies, the analogous A549 “parent” (control) cell line was grown without additional NO● donor (i.e. the original, commercially-available cell line prior to the adaptation process), was also used in this study.
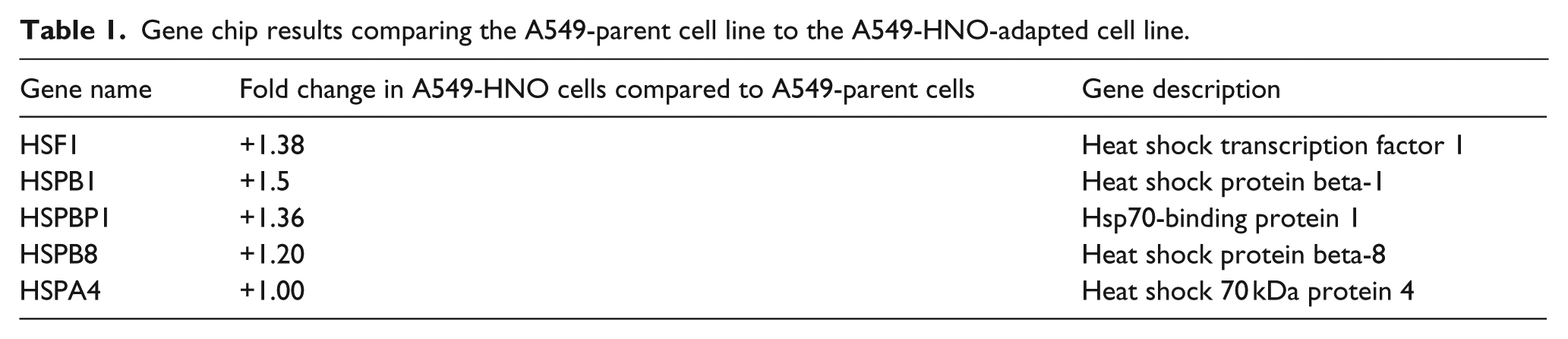
Studies were carried out to determine the resistance of the A549-parent and A549-HNO cells to taxol, a chemotherapeutic agent (Figure 1). As expected, cell viability decreased for both the parent and adapted cells as the concentration of taxol increased. However, at concentration up to 6.25 µM, the A549-HNO cells exhibited a measurably higher percentage of cell viability. Similarly, both A549-parent and A549-HNO cells were exposed to another chemotherapeutic agent, doxorubicin. Figure 2 shows concentration-dependent growth curves, as measured by the MTT assay, upon exposure to increasing levels of doxorubicin. The viability of both cell lines decreased as the doxorubicin concentration was increased, similar to the taxol results, and the A549-HNO cells exhibited greater cell viability than the parent cells at each concentration tested.
MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with taxol (chemotherapeutic agent). The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.

MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with doxorubicin (chemotherapeutic agent). The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
The ability of the A549-parent and A549-HNO cells to withstand various temperatures was also determined (Figure 3). Similar viability, as measured by the MTT assay, was found for both A549-parent and A549-HNO cells at 30°C. In general, both cell lines exhibited a decrease in viability as temperature was increased; however, within the range 40°C–48°C, the A549-HNO cells showed a measurable increase in viability relative to the parent cells. Both parent and A549-HNO cells exhibited similar viability at temperatures of 50°C and above. In addition, the gene chip data show that genes involved in the heat shock response pathway are upregulated in the A549-HNO cells when compared to their corresponding A549-parent cells (Table 1).

MTT proliferation/viability assays of A549-parent and A549-HNO cells treated at various temperatures. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
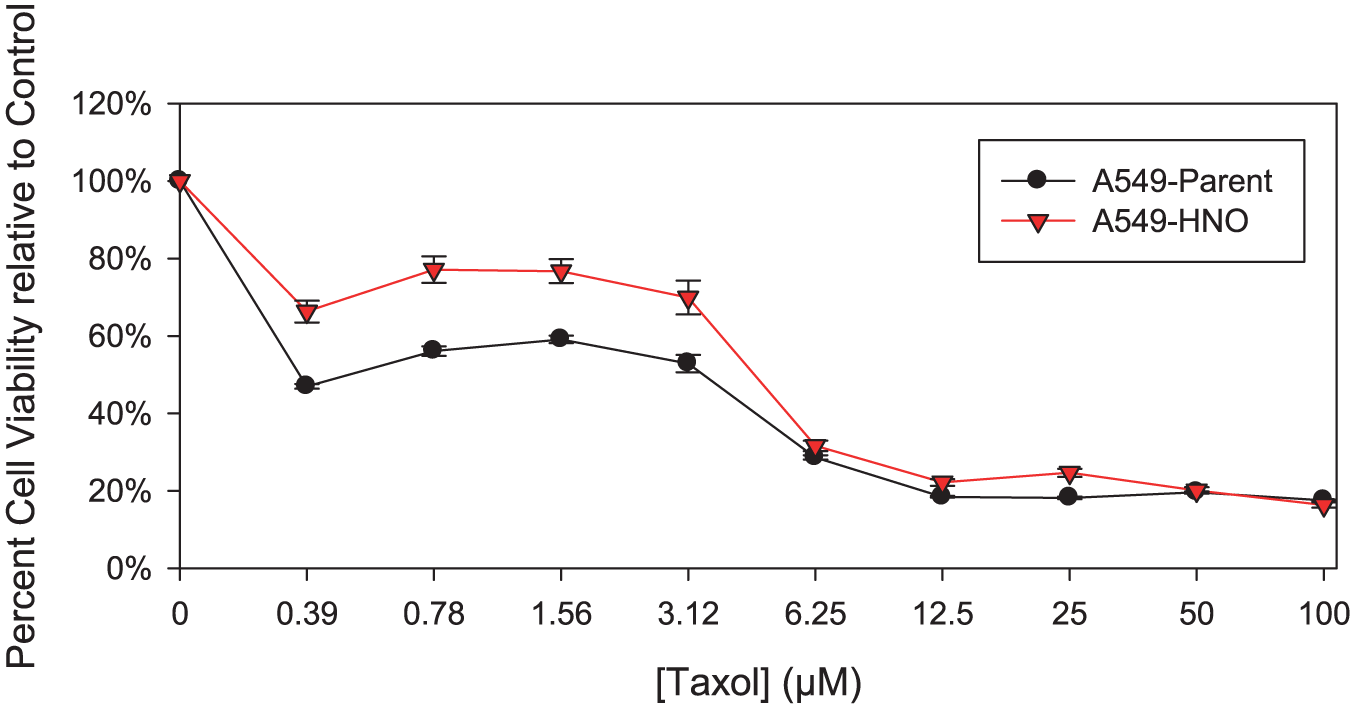
The A549-parent and A549-HNO cells were exposed to a range of pH. Cell viability measured by MTT assay shows that the A549-HNO cells are more resistant to pH values (5–8) than their corresponding A549-parent cells (Figure 4). In addition, the A549-HNO cells were more resistant than the A549-parent cells when exposed to different concentrations of CaCl2 (Figure 5).

MTT proliferation/viability assays of A549-parent and A549-HNO cells treated in pH media ranging from 3.0 to 12.0. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with calcium chloride (CaCl2) at different concentrations. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
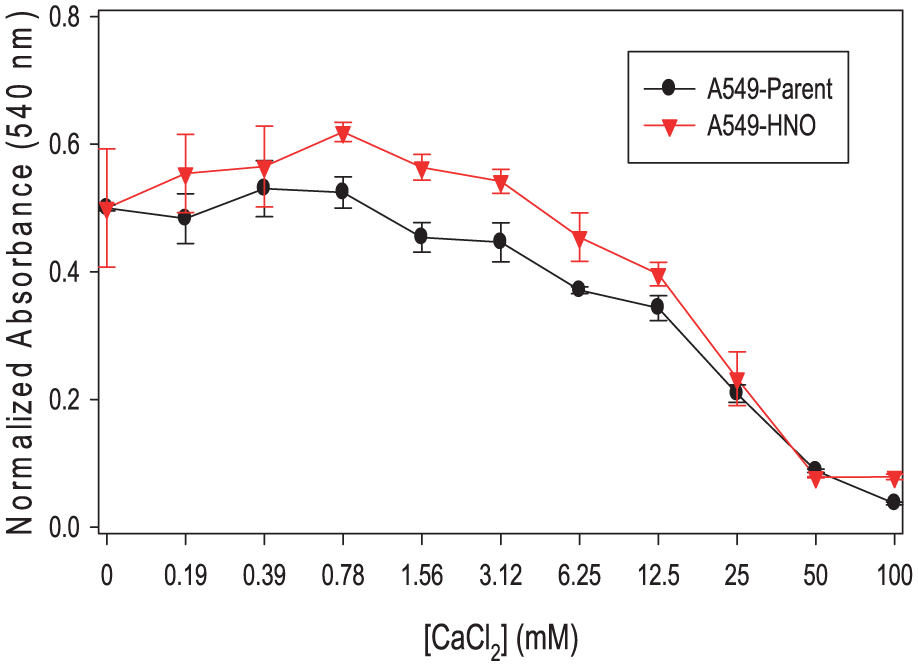
The A549-parent and A549-HNO cells were exposed to varying concentrations of metals such as CdCl2, CuCl2, and NaCl (Figures 6–8, respectively). Cell viability measured by MTT assay shows that the A549-HNO cells are more resistant to varying concentrations of CdCl2 and CuCl2 than their corresponding A549-parent cells. Only at the highest concentration of NaCl tested, there was a noted difference between the cell line pairs.

MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with cadmium chloride (CdCl2) at different concentrations. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with copper chloride (CuCl2) at different concentrations. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.

MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with sodium chloride (NaCl) at different concentrations. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
The ability of A549-HNO and A540-parent cells to withstand different concentrations of metal salts was also determined. Both A549-HNO and A549-parent cells were exposed to varying concentrations of FeCl2 (Figure 9). The A549-HNO cells showed higher viability when exposed to FeCl2 than their corresponding A549-parent cells.
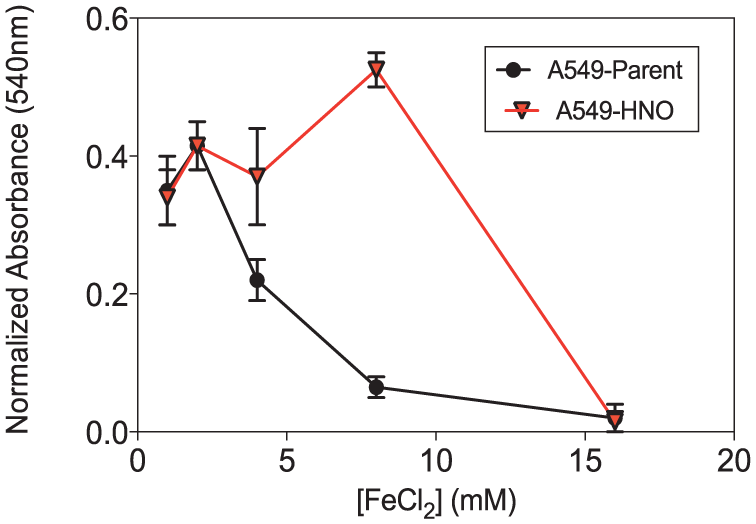
MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with ferrous chloride (FeCl2) at different concentrations. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
The A549-HNO and parent cells were treated with the antioxidant, NaRLA. The MTT assays of A549-HNO and A549-parent cells show that the A549-HNO cells grow better than the parent cells in the presence of NaRLA (Figure 10).

MTT proliferation/viability assays of A549-parent and A549-HNO cells treated with sodium-R-lipoic acid (NaRLA) at different concentrations. The media of the adapted cells was supplemented with 600 μM of DETA-NONOate for the A549-HNO cells. The standard error of the mean has been plotted on the graph.
Discussion
As previously reported, A549 lung adenocarcinoma cells were successfully adapted to high levels of NO● over a long period of time. 37 Although morphologically indistinguishable from the parent cells (which were grown without the presence of NO● donor), the adapted cells were able to survive and grow in comparatively high levels of NO●, a level that is lethal to the parent cells. Furthermore, the adapted cells exhibited stronger growth than the parent cells under both normal and low-nutrient growth conditions.36,37 These results suggested that prolonged NO● exposure results in biological changes to tumor cells, which enables them to survive in adverse, high free radical environments.
It is important to note that the tumor cells, both in patients and in tissue culture, produce their own NO●. This NO● acts as an autocrine factor to induce physiological changes in themselves and in the surrounding cells. We have previously shown that the A549-HNO cells are more resistant to UV and X-ray radiation.42,43 This implies that A549 cells acquired an NO●-mediated cross-tolerance to radiation along with NO● adaptation. This is similar to what is observed when cancer patients become resistant to one modality/drug of treatment (chemotherapy) and then are found to be resistant to another unrelated modality/drug (radiotherapy).9–11
Since UV and X-ray radiation kill cancer cells by causing DNA damage, we previously assessed DNA damage pathways in the A549-HNO and A549-parent cells. We have shown that the A549-HNO cells show an upregulation of genes involved in the DDR pathway. Furthermore, the A549-HNO cells exhibit an enhanced anti-apoptotic signaling.42,43
We hypothesized that the HNO-adapted cells would be better equipped to withstand other adverse growth situations including chemotherapeutic agents, extreme temperatures, different pH ranges, CaCl2, NaCl, and heavy metals (salts). Indeed, under each condition (Figures 1–9), the A549-HNO cells were found to be more resistant to the external stress and showed a higher level of cell viability and proliferation.
The A549-HNO cells were found to be more resistant to elevated temperatures than the parent cells (Figure 3). When cells are exposed to elevated temperatures, the expression of heat shock proteins (HSPs) increases. The heat shock response is universal and is used by all organisms. Elevated temperatures have deleterious effects on a variety of cellular functions and components such as protein unfolding, disruption of the cytoskeleton, loss of correct localization of organelles, and breakdown of intracellular transport processes. Elevated temperatures also affect nuclear processes. In the presence of stressful conditions, the expression of HSPs results in the stabilization of unfolded proteins. 48 HSPs are overexpressed in various human cancers and are implicated in carcinogenesis.49,50 In an effort to unravel possible molecular mechanisms involved in the cross-tolerance of the adapted cell, we compared the genome expression of HSPs in A549-parent and A549-HNO cells. This is noted in Table 1, which lists HSF1 (heat shock transcription factor 1), HSPB1 (heat shock protein beta-1), HSPBP1 (Hsp70-binding protein 1), HSPB8 (heat shock protein beta-8), and HSPA4 (heat shock 70 kDa protein 4). The HSPs were upregulated in the A549-HNO cells when compared to the A549-parent cells.
Intracellular levels of calcium influence gene expression and can be altered by both changes in pH and extracellular calcium. We therefore questioned whether the A549-HNO cells had built up a wider range of tolerance toward pH and calcium in their microenvironment. Viability over a broader range of pH is consistent with a more aggressive phenotype. Given that pH changes are known to cause changes in intracellular calcium levels, thereby altering gene expression, we also investigated the effect of different calcium concentrations in both A549-HNO and A549-parent cells. In vivo, the tumor mass grows and if the blood supply does not keep up with growth, the center of the mass will become necrotic (and acidic). Tumor cells abutting the necrotic field will be environmentally challenged. The A549-HNO cells have a greater tolerance to changes in pH and calcium than their corresponding A549-parent cells. This suggests that the A549-HNO cells have a change in gene expression. This change in gene expression in turn suggests that the HNO adaptation process alters the intracellular calcium signaling in the A549-HNO cells. These inferred differences in intracellular calcium signaling between the A549-parent and A549-HNO cells suggest a possible mechanism by which the A549-HNO cells are able to better survive in harsh pH environments than the A549-parent cells.
Chemotherapeutic drugs kill cancer cells using different modes of action. Sometimes they exert their effect through heavy metals. Carboplatin and cisplatin are two of the heavy metals containing chemotherapy drugs. Cisplatin has been shown to be very effective in treating cancers; however, some cancers are resistant to this treatment. 48 We began to look at the resistance of A549-HNO to metals. When exposed to increasing concentrations of NaCl, the A549-HNO cells were more resistant than the A549-parent cells at 130 mM (Figure 8). The A549-HNO cells showed higher resistance when exposed to CdCl2, CuCl2, and FeCl2 (Figures 6, 7, and 9).
Studies in the field of antioxidants have suggested the potential for the development of antioxidant therapy for patients. 51 Antioxidant compounds are often found in foods and remove free radical stress from the environment. NaRLA is one of the potent antioxidants found in many foods. Our studies showed that the HNO-adapted cells grew better when compared to the parent cells in the presence of NaRLA. Since tumor cells overproduce NO●, which serves as a growth promoting agent, we hypothesized that an environment in which we inhabited the availability of NO● that cell proliferation would be affected. Exposure to NO● resulted in the HNO cells to be more resistant to antioxidant treatment (Figure 10).
In clinical studies, we and others have shown that patients presenting HNO-expressing tumors show poor prognosis compared to those with low NO●-expressing tumors.27–35 Our research provides a link for this phenomenon whereby the A549-HNO cells exhibit higher cell viability in the presence of different environmental stressors and have developed cross-tolerant properties.
NO●, which is overexpressed in many solid tumors, is one potential driving force which makes tumor cells exhibit cross-tolerance to a wide range of environmental stressors. Despite the fact that patients have tumors that look histologically the same, they oftentimes respond differently to diverse modes of treatment. Once a patient exhibits resistance to one kind of treatment, they often exhibit resistance to other kinds of treatments. For example, if they become resistant to radiation therapy which works through free radicals, they are also more likely to become resistant to chemotherapeutics, many of which generate free radicals. Hence, we questioned whether NO● is a signaling molecule responsible for this cross-tolerance phenomenon. We tested commonly used free radical modes of treatments as well as the extent to which cross-tolerance properties are expressed. Proving that NO● is at least one naturally produced product by human tumors that can promote tumor cells to express cross-tolerance to a wide range of environmental stressors would make this agent a target in the treatment of human cancers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
