Abstract
Dysregulation of SLC34A2 (NaPi2b) in tumors has attracted wide attention, but its expression and function in non–small cell lung cancer remains unclear. By examining its expression in lung adenocarcinoma and correlation to patient outcome, we aimed to explore its prognostic and therapeutic values in this deadly disease. Overall, 175 cases of lung adenocarcinoma sample were included in this study. Histological subtyping of them was diagnosed according to standards of the International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society in 2011. Protein expression of SLC34A2 and anaplastic lymphoma kinase in these samples was determined by immunohistochemistry. Epidermal growth factor receptor mutations were examined using amplification refractory mutation system. Statistical analysis was performed using software of Pearson’s correlation coefficient. High expression of SLC34A2 was identified in about 2/3 patients and correlated with significantly better patient’s overall survival. Epidermal growth factor receptor mutations were detected in about 53% of patients with no statistically significant difference to patient’s overall survival. Anaplastic lymphoma kinase rearrangement was found in 8 out of 175 patients, harboring this abnormality leads to shorter overall survival. No correlation has been found between SLC34A2 expression and epidermal growth factor receptor mutation or anaplastic lymphoma kinase rearrangements in lung adenocarcinoma. High expression of SLC34A2 is present in about 3/4 lung adenocarcinoma samples and predicts better outcome. Since it is a membrane protein, antibody-based drugs targeting this marker might bring new resolution to this deadly disease.
Keywords
Background
Lung cancer is the most common cause of cancer-related deaths worldwide. Non–small cell lung cancer (NSCLC), which accounts for approximately 80% of all lung cancer cases, is composed of lung adenocarcinoma (LAC), squamous cell carcinoma (SCC), and large cell carcinoma (LCC) subtypes. 1 LAC accounts for more than half of NSCLC. 2 Increasing studies have focused on the histological subtyping of LAC following the novel classification proposed by the International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society (IASLC/ATS/ERS). Finding of association between the histological subtypes and gene mutation status in LAC patients is crucial. 3 Even some gene targeting drugs have been developed and brought great benefits to lung cancer patients, 5-year survival rates of NSCLC patients are currently still less than 15%. 4 Thus, finding new diagnostic and therapeutic molecular targets to lung cancer are still remaining challenges.
As a potential molecule of this kind of targets, SLC34A2 has attracted increasing attention in some tumors, including NSCLC. It is a member of the family of solute carrier series (SLC) which locates on chromosome 4p15.2, encoding the sodium-phosphate cotransporter protein type II (NaPi2b) which is responsible for transcellular inorganic phosphate absorption and maintenance of phosphate homeostasis. 5 It was first cloned from human small intestine and lung in 19996,7 and then revealed it is widely expressed in other organs too, such as ovary, thyroid, breast, and kidney. Its aberrant expression has been associated with several kinds of tumors,8–12 but its function still remains unclear. As in NSCLC, Yang et al., 13 reported that elevated expression of SLC34A2 inhibited viability and invasion of A549 cells, while Jiang et al., 14 showed that high expression of SLC34A2 is required for tumorigenicity and self-renewal of lung cancer stem cell–like cells. Thus, fully understanding its expression in LAC patients is critical. In this study, we fully investigated its expression and association with overall survival (OS) in 175 LAC patients. Moreover, epidermal growth factor receptor (EGFR) mutations and anaplastic lymphoma kinase (ALK) rearrangements are major existing therapeutic/diagnostic targets of NSCLC, thus we also examined their incidences and association with histological subtypes and OS in these patients.
Materials and methods
Patient tissue specimens
This project was consulted and approved by the research ethics committee of Anhui Medical University. Totally, 175 LAC tumor samples (biopsies or surgical sections) were collected between 2014 and 2015 at the pathology department of Anhui Provincial Hospital (Hefei, P.R. China). Follow-up of patients was made up to 31 October 2016 with the period of 10–39 months. General patient’s information is summarized in Table 1.
Clinical characteristics of 175 LAC patients.

LAC: lung adenocarcinoma; TNM: tumor–node–metastasis.
Overall, 11 patients had both bone and brain metastasis.
Other: multiple organ metastasis except bone and brain, such as liver and kidney.
Immunohistochemistry
Expression of ALK and SLC34A2 in patient samples was examined by immunohistochemistry (IHC). Polyclonal rabbit anti-SLC34A2 antibody (1:400; ab122431; Abcam, Cambridge, UK) and monoclonal rabbit anti-human ALK antibody (ready to use; D5F3; Roche/Ventana, USA) were used for detection of these two molecules. Immunostaining was performed on the Roche Benchmark XT autostainer system (Roche/Ventana) according to manufacturer’s instructions. In brief, sections of 4 μm thickness were dewaxed in EZprep Concentrate Buffer at 75°C for 4 min. Epitope retrieval was performed in Cell Conditioning Solution at 100°C for 30 min. Incubation of anti-SLC34A2 and anti-ALK was conducted at 37°C for 16 min. Horseradish peroxidase (HRP)-conjugated goat anti-mouse/anti-rabbit IgG/IgM secondary antibody reagent accompanying this immunohistochemical system was then incubated for 8 min. Antibodies were visualized with Optiview DAB IHC Detection Kit (Roche/Ventana) following manufacturer’s protocols.
For ALK staining, strong brown granular staining in cytoplasm of any percentage of tumor cells was diagnosed as positive according to the Food and Drug Administration (FDA). SLC34A2 expression level was determined as previously described, 13 positive staining in less than 30% cells was considered as low expression and in more than 30% of cells were considered as high expression.
Amplification refractory mutation system
EGFR mutations in all samples were detected using EGFR Mutation Test v1 kit (Roche/Ventana) which is designed to detect the following EGFR mutations—G719A/C/S in exon 18; 29 deletions in exon 19; S768I, T790M, and 5 insertions in exon 20; and L858R in exon 21 (approved by FDA to be used in clinic for EGFR mutation diagnosis)—following manufacturer’s instructions. Genomic DNA of each sample was extracted using the QIAamp DNA FFPE Tissue Kit (QIAGEN, Germany) following manufacturer’s protocol.
Statistical analyses
The associations of SLC34A2 expression, EGFR mutation, and ALK rearrangement with clinical factors (age at diagnosis, gender, clinical stages, lymph-node metastasis, distant metastases, pathological subtypes, and patient’s OS) were analyzed by Pearson’s correlational analysis software. Patient survival curve was obtained by Kaplan–Meier method and compared using the log-rank test. All statistical analysis was performed on SPSS version 21.0 (IBM SPSS, USA). p < 0.05 for the two-tailed test was considered as statistically significant difference.
Results
Tumor–node–metastasis and IASLC/ATS/ERS classification of LAC patients
Overall, 175 LAC patients were diagnosed according to the Eighth Edition of TNM Classification of Lung Cancer: 3 cases of stage IA, 4 cases of stage IB, 4 cases of stage IIA, 3 cases of stage IIB, 24 cases of stage IIIA, 20 cases of stage IIIB, 1 case of stage IIIC, 41 cases of stage IVA, and 75 cases of stage IVB. IA, IB, and IIA were classified as early stage; IIB, IIIA, and IIIB were classified as medium stage; and IIIC, IVA, and IVB were classified as end stage. Altogether, 164 out of 175 (about 95%) patients were at medium and end stages.
According to the IASLC/ATS/ERS classification, these patients were grouped into the following subtypes (Figure 1): 34 cases of lepidic predominant (19.57%), 45 cases of acinar predominant (25.71%), 10 cases of papillary predominant (5.71%), 4 cases of micropapillary predominant (2.29%), 63 cases of solid predominant with mucin production (36%), and 19 cases of invasive mucinous adenocarcinoma (10.86%). Subtype of solid predominant with mucin production was more common in male than female patients (p < 0.05) while subtype of acinar predominant was more common in female than male patients (p < 0.01). Brain metastasis is more common in papillary predominant subtype.

Pathological subtyping of LAC patient samples. All 175 samples were processed by hematoxylin and eosin (H&E) staining and diagnosed following IASLC/ATS/ERS classification standards (2011). The representative results of subtypes were as follows: (a) lepidic predominant, (b) acinar predominant, (c) papillary predominant, (d) micropapillary predominant, (e) solid predominant with mucin production, and (f) invasive mucinous adenocarcinoma (magnification: 100×).
EGFR mutation in LAC patients
EGFR mutations were identified within 93 out of 175 LAC patient samples in total (53.14%), major mutations were L858R in exon 21 (51/93, 54.83%) and exon 19 deletions (38/93, 40.86%), other mutations were rare, such as T790M (3/93, 3.23%) and 1 case in other exon (1/93, 1.08%, Figure 2). EGFR mutations were significantly higher in female than in male patients (p < 0.001).

EGFR mutations in LAC patient samples. EGFR mutations were identified in 93 out of 175 samples (54% patients). The majority of mutations were L858R and exon 19 deletions.
The association of EGFR mutations with subtypes of IASLC/ATS/ERS classification was as follows: 22 cases of lepidic predominant (22/34), 26 cases of acinar predominant (26/45), 7 cases of papillary predominant (7/10), 2 cases of micropapillary predominant (2/4), 25 cases of solid predominant with mucin production (25/63), and 11 cases of invasive mucinous adenocarcinoma (11/25). L858R mutation is more common in the lepidic predominant subtype (14/34, 41.18%, p < 0.001). EGFR mutations were less common in patients of solid predominant with mucin production subtype (25/63, 39.68%, p = 0.067). No significant association of EGFR mutations with patient’s OS has been identified (Figure 4(b)).
SLC34A2 expression in LAC patients
Aberrant expression of SLC34A2 has been associated with NSCLC and proposed as tumor therapeutic target. 10 In this study, we completely examined its expression in 175 LAC samples using IHC. Results showed that this gene was differentially expressed among these samples. In total, 130 out of 175 patients exhibited high expression (positive staining cells > 30%, Figure 3(b) and (c)) while other 45 patients presented low expression (positive staining cells < 30%, Figure 3(e) and (f)).
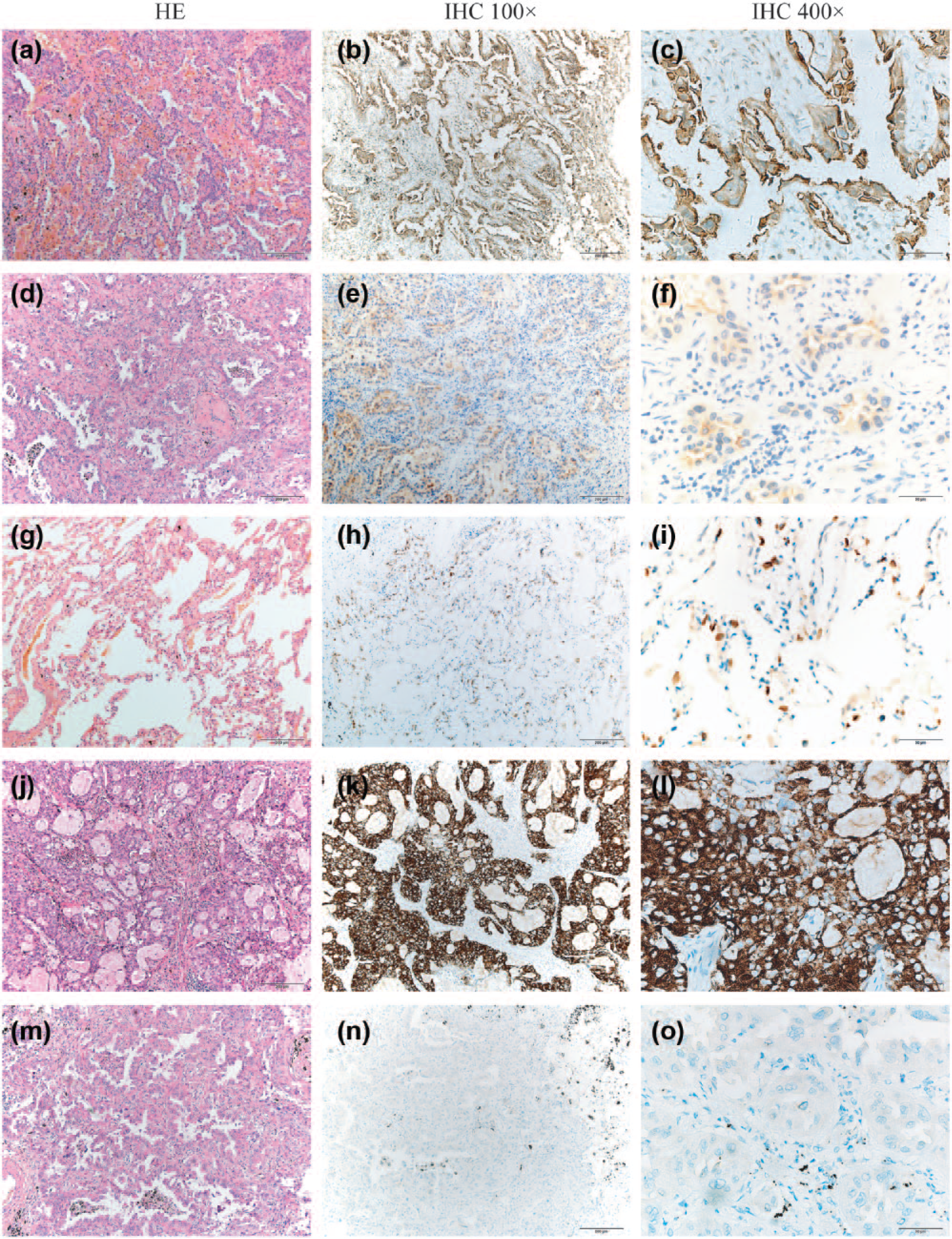
SLC34A2 and ALK expression in LAC samples. Expression of both SLC34A2 and ALK in all 175 samples was determined by IHC. (a, d, g, j, and m) Sections were proceeded with H&E staining to ensure tumor tissues be used in immunohistochemical staining. (b and c) Representative results of SLC34A2 high expression: it was mostly detected on the membrane of more than 30% cancer cells. (e and f) Representative results of SLC34A2 low expression: it was weakly expressed on the membrane of less than 30% cancer cells. (h and i) Representative results of SLC34A2 staining on adjacent normal lung tissues: it was moderately expressed. (k and l) Representative results of ALK positive staining: it was highly expressed in cytoplasm of tumor cells. (n and o) Representative results of ALK negative staining: no staining in cytoplasm was detected. (magnification—100×: a, b, d, e, g, h, j, k, m, and n; 400×: c, f, i, l, and o).
SLC34A2 expression was predominantly localized on the surface of epithelium. High expression of it had a significant favor in patient’s OS (Figure 4(a)) with no significant association with other clinical characteristics, such as tumor subtypes, metastasis, EGFR mutation, or ALK rearrangement.

Kaplan–Meier curves showing association of EGFR mutation, ALK rearrangement, and SLC34A2 expression with patient’s overall survival (OS). The median OS of all 175 patients was 14 months. (a) The group of SLC34A2 high expression had a significantly longer OS than the group of low expression (p < 0.01). (b) No significant OS difference was identified between EGFR wild-type and mutation patients. (c) Incidence of ALK rearrangement was low in these patients (8 out of 175 in totals). OS of patients harboring this abnormality had worse outcome at short-term comparison (p = 0.026).
ALK expression in LAC patients
EML4-ALK fusion is an important molecular abnormality in LAC which is suggested by World Health Organization (WHO) to be examined in all these kinds of patients beyond histological classification. Currently approved methods by FDA include VENTANA ALK (D5F3; Roche) immune-histological staining kit 15 and fluorescence in situ hybridization (FISH). In this study, we used VENTANA ALK (D5F3) kit to examine ALK expression. Overall, 8 out of 175 patient samples were identified to be positive (5.80%, Figure 3(k) and (l)), other 167 samples were negative (94.20%, Figure 3(l) and (n)). ALK positive is more common in younger patients (p < 0.05, r = −0.165). High ALK expression correlated with bad OS rate of patients (Figure 4(c)).
Discussion
Highest incidence and poorer outcome make lung cancer still to be the biggest threat to human beings; even many big progressions have been made in understanding molecular defects and gene targeting drug developments, such as tyrosine kinase inhibitors (TKIs) to EGFR mutations, in the last decade. Thus, indentifying new molecular targets to combat this disease still remains a big challenge. SLC34A2 has shown its potential to become this kind of target in NSCLC. This protein is expressed on cell membrane which will provide accessibility to antibody drugs. In fact, preclinical experiment of antibody–drug conjugates targeting SLC34A2 has shown promising therapeutic effects to ovarian and NSCLC tumor xenograft models with tolerated toxicity. 10 But the uncertainty of its expression in NSCLC samples needs to be clarified, as Lin et al., 10 showed its moderate to high expression in 77% of 31 LAC samples, while Wang et al., 16 demonstrated decreased expression in 14 out of 15 NSCLC samples. Here, we first confirm its high expression in most of LAC patients (130 out of 175 patients in total), which means 3/4 LAC patients will be benefited if this molecule can be used as tumor therapeutic target. Also, tumor stem cells have been convinced to be the major cause of tumor recurrence and metastasis; high expression of SLC34A2 has also been reported necessary to tumorigenicity and self-renewal of CD166 (+) lung cancer stem cell–like cells. 14 Meanwhile, our results showed no significant association between SLC34A2 high expression and EGFR mutations nor ALK rearrangement in LAC patients, so targeting SLC34A2 possesses possibility to treat TKI-resistant patients.
Function of SLC43A2 high expression in tumors is also controversial currently. Some studies showed that high expression of SLC34A2 can present a tumor-suppressor function, as Zhang et al., 9 demonstrated elevated miR-410 expression promoted NSCLC cell growth and metastasis in vivo but restoring its downstream target SLC34A2 expression effectively reversed this function; Yang et al., 13 showed that high SLC34A2 expression inhibits the viability and invasion of NSCLC A549 cells. But some experiments indicated that high expression of SLC34A2 was oncogenic, as Ye et al. 17 demonstrated that high expression of SLC34A2 was associated with large tumor size and poor survival of bladder cancer patients, and depletion of SLC34A2 inhibited xenograft bladder cancer tumor growth in vivo; Hong et al. 18 reported knockdown of SLC34A2 (NPT2b) suppresses lung tumorigenesis in mouse model. From our results, in a relatively large scale of LAC patients, elevated expression of SLC34A2 was significantly correlated with better patient’s OS. Mechanisms underlining this effect still remain to be clarified.
Mutant EGFRs are major tumor-driving gene in cancers, especially in NSCLC, with over 50% of incidence. Examining its mutation is not only important to understand tumorigenicity element in NSCLC but also crucial to patient treatment with TKI-targeting therapy. Even mostly consistent with previous reports,3,19 our data still provides some important information in this dominated abnormality of LAC. Our results showed that L858R in exon 21 (51/93, 54.83%) and exon 19 deletions (38/93, 40.86%) are the two major defects which account for more than 95% EGFR mutations in LAC. ALK rearrangements are another tumor initiating element to NSCLC. Altogether, 8 out of 175 patients were identified to be ALK rearrangement positive. Coexistence of EGFR mutations and ALK rearrangement is rare which was only identified in one patient. High expression of SLC34A2 has no association with both EGFR mutation and ALK rearrangement.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Grant Nos 81272800 and 81072057).
