Abstract
To investigate the effects of a novel synthetic epidermal growth factor receptor inhibitor, COMPOUND7809, on the inhibition of lung cancer growth in vitro and the underlying mechanisms, we treated three lung tumor cell lines (A549, SK-LU-1, and NCI-H23) with COMPOUND7809 and a Food and Drug Administration–approved epidermal growth factor receptor inhibitor gefitinib. Then, we examined cell growth in a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, cell survival in a Cell Counting Kit-8 assay, and cell apoptosis by Annexin V flow cytometry in the presence of fluorouracil. We found that compared to gefitinib, COMPOUND7809 inhibited cell growth more potentially and induced more cell death in the presence of fluorouracil. Thus, our study demonstrates that COMPOUND7809 may be a promising epidermal growth factor receptor inhibitor for human lung cancer therapy.
Introduction
Lung cancer is a commonly occurred malignant cancer in humans. Most types of lung cancer are not sensitive to either chemotherapy or radiotherapy.1–3 Fluorouracil (5-FU) treatment has been used as a supplementary treatment to surgical removal of the primary cancer, which improves the survival of the patients.4–8 However, some lung cancers have been shown to be resistant to 5-FU treatment, and the molecular mechanisms underlying this phenomenon are not completely understood.
Epidermal growth factor receptor (EGFR) signaling pathway plays a critical role in the growth and metastasis of lung cancer.9–12 Epidermal growth factor (EGF) is one of the major ligands that binds to EGFR leading to receptor dimerization, resulting in receptor phosphorylation at tyrosine residues, leading to the activation of their downstream signaling, of which the mitogen-activated protein kinase (MAPK) pathways, the Jun N-terminal kinase (JNK) pathway, or the phosphatidylinositol 3-kinase (PI-3K) pathway mediate signal transduction in different cell types. Since the discovery of protein kinase activity in 1954, many kinase inhibitors have been comprised and used in basic research and clinical practice. At present, first-line treatment with EGFR TKIs (gefitinib, erlotinib, and afatinib) has been approved for patients who harbor exon 19 deletions or exon 21 (Leu858Arg) substitution EGFR mutations. These agents have been shown to substantially improve response rates, time to progression, and overall survival. Unfortunately, patients develop resistance, limiting patient benefit and posing a challenge to oncologists. Thus, novel and more potent EGFR inhibitors are in great need. 13
High-throughput docking is used for drug discovery since 2000, which forecasts an optimized conformation for the protein and ligand molecule.14–16 Hence, development of novel therapeutic treatments, for example, specific pathway inhibitors for lung cancer, is highly urgent, which may substantially improve the patients’ 5-year survival ratio.
In this study, we investigated the effects of a novel synthetic EGFR inhibitor, COMPOUND7809, on the inhibition of lung cancer growth in vitro and the underlying mechanisms, using three lung tumor cell lines (A549, SK-LU-1, and NCI-H23). We examined cell growth in an 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, cell survival in a Cell Counting Kit-8 (CCK-8) assay, and cell apoptosis by Annexin V flow cytometry in the presence of 5-FU. Our study demonstrates that COMPOUND7809 may be a promising EGFR inhibitor for human lung cancer therapy.
Materials and methods
Study approval
All the experimental methods have been approved by the research committee at Union Hospital of Guangdong University of Pharmacy. All the experiments have been carried out in accordance with the guidelines from the research committee at Guangdong University of Pharmacy.
Cell line and reagents
Three human lung cancer cell lines A549 (origin from carcinoma), SK-LU-1 (origin from adenocarcinoma), and NCI-H23 (origin from non-small-cell lung cancer) were all purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). These three cell lines were used in this study, since they represent different types of lung cancer. Three cell lines were all cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Invitrogen, Carlsbad, CA, USA) supplemented with 15% fetal bovine serum (FBS; Sigma-Aldrich, St. Louis, MO, USA) in a humidified chamber with 5% CO2 at 37°C. 5-FU (Sigma-Aldrich) was prepared in a stock of 1 mmol/L and applied to the cultured cells at 10 µmol/L.
Cell viability assay
The CCK-8 detection kit (Sigma-Aldrich) was used to measure cell viability according to the manufacturer’s instructions. Briefly, cells were seeded in a 96-well microplate at a density of 5 × 104 mL−1. After 24 h, cells were treated with resveratrol. Subsequently, CCK-8 solution (20 mL/well) was added and the plate was incubated at 37°C for 2 h. The viable cells were counted by absorbance measurements with a monochromator microplate reader at a wavelength of 450 nm. The optical density value was reported as the percentage of cell viability in relation to the control group (set as 100%).
Cell growth assay
A MTT assay was performed to determine cell growth. An amount of 5000 cells per well was seeded in a 96-well plate to allow the cells to grow. Then, the media were removed and washed with phosphate-buffered saline (PBS), after which 5 g/L of thiazolyl tetrazolium (Amresco, Indianapolis, IN, USA) was added to each well. After 4 h, MTT was removed and 150 µL of dimethyl sulfoxide (Sigma-Aldrich) was added. The viability of the cells was calculated from the absorption at 570/630 nm with an enzyme-linked immunosorbent assay reader.
Apoptosis assay by flow cytometry
For analysis of cell apoptosis, the dissociated cultured cells were re-suspended at a density of 106 cells/mL in PBS. After double staining with fluorescein isothiocyanate (FITC)-Annexin V and propidium iodide (PI) from an FITC-Annexin V Apoptosis Detection Kit I (Becton-Dickinson Biosciences, San Jose, CA, USA), cells were analyzed using FACScan flow cytometer (Becton-Dickinson Biosciences) equipped with CellQuest software (Becton-Dickinson Biosciences) for determination of Annexin V+ PI− apoptotic cells.
Statistical analysis
All data were statistically analyzed using one-way analysis of variance (ANOVA) with a Bonferroni correction, followed by Fisher’s exact test for comparison of two groups (GraphPad Prism, GraphPad Software, Inc., La Jolla, CA, USA). All values are depicted as mean ± standard deviation and are considered significant if p < 0.05.
Results
Self-docking study
Self-docking was carried out on the X-ray structure using the Surflex-Dock in geom mode. After virtual screening, the top compound named COMPOUND7809 was chosen (Figure 1). Next, we compared the biological effects of COMPOUND7809 with a Food and Drug Administration (FDA)-approved EGFR inhibitor gefitinib on the growth and survival of lung cancer cells with and without the presence of 5-FU.

Self-docking study. Self-docking was carried out on the X-ray structure using the Surflex-Dock in geom mode. After virtual screening, the top compound named COMPOUND7809 was chosen.
Anti-proliferative potential of COMPOUND7809 in lung cancer cells
To investigate the effects of COMPOUND7809 on the inhibition of lung cancer growth in vitro and the underlying mechanisms, we treated three lung tumor cell lines (A549, SK-LU-1, and NCI-H23) with COMPOUND7809 and an FDA-approved EGFR inhibitor gefitinib. Then, we examined cell growth in an MTT assay. We found that compared to gefitinib, COMPOUND7809 inhibited cell growth more potentially in A549 (Figure 2(a)), SK-LU-1 (Figure 2(b)), and NCI-H23 cells (Figure 2(c)). These data demonstrate the cellular anti-proliferative activities of COMPOUND7809 in lung cancer cells.
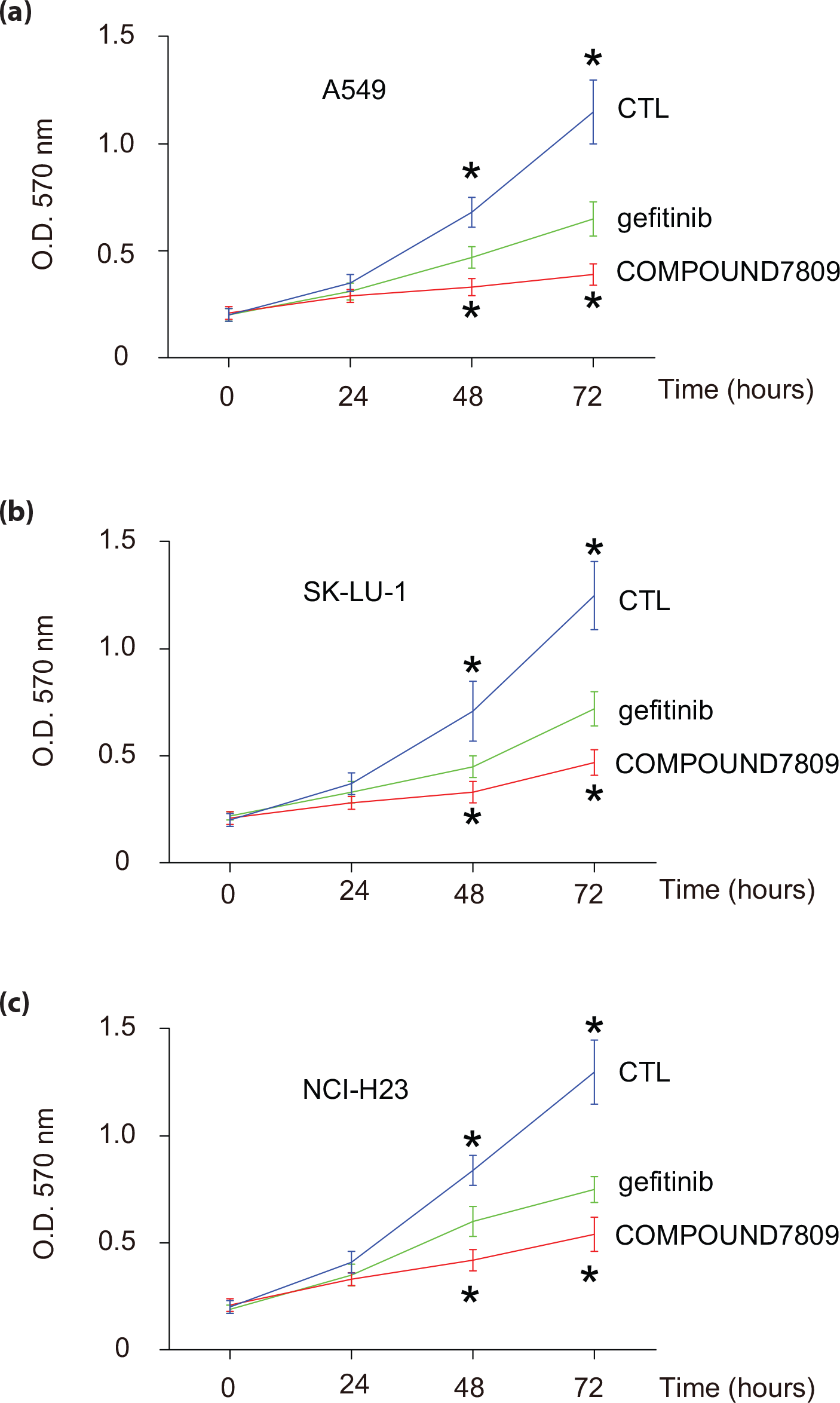
Anti-proliferative potential of COMPOUND7809 in lung cancer cells. (a–c) To investigate the effects of COMPOUND7809 on the inhibition of lung cancer growth in vitro and the underlying mechanisms, we treated three lung tumor cell lines (A549, SK-LU-1, and NCI-H23) with COMPOUND7809 and an FDA-approved EGFR inhibitor gefitinib. Then, we examined cell growth in an MTT assay using (a) A549, (b) SK-LU-1, and (c) NCI-H23 cells (*p < 0.05, N = 5).
COMPOUND7809 decreases lung cancer cell survival upon 5-FU treatment
Next, we examined lung cancer cell survival in a CCK-8 assay in the presence of 5-FU. We found that compared to gefitinib, COMPOUND7809 reduced cell survival more potentially in A549, SK-LU-1, and NCI-H23 cells (Figure 3). These data suggest that COMPOUND7809 may decrease lung cancer cell survival upon 5-FU treatment.

COMPOUND7809 decreases lung cancer cell survival upon 5-FU treatment. We examined lung cancer cell survival in a CCK-8 assay in the presence of 5-FU (*p < 0.05, N = 5).
COMPOUND7809 decreases lung cancer cell survival upon 5-FU treatment by increasing apoptosis
To understand the mechanisms underlying the effects of COMPOUND7809 on cell survival in the presence of 5-FU, we performed an Annexin V flow cytometry assay. We found that compared to gefitinib, COMPOUND7809 induced more apoptotic cell death in the presence of 5-FU, shown by quantification (Figure 4(a)) and by representative flowcharts (Figure 4(b)). Thus, our study demonstrates that COMPOUND7809 may be a promising EGFR inhibitor for human lung cancer therapy.

COMPOUND7809 decreases lung cancer cell survival upon 5-FU treatment by increasing apoptosis. To understand the mechanisms underlying the effects of COMPOUND7809 on cell survival in the presence of 5-FU, we performed an Annexin V flow cytometry assay. (a and b) We found that compared to gefitinib, COMPOUND7809 induced more apoptotic cell death in the presence of 5-FU, shown by (a) quantification and (b) representative flowcharts (*p < 0.05, N = 5).
Discussion
EGFR signaling pathway has been shown to be highly activated in lung cancers and plays a critical role in tumor proliferation and metastasis. Hence, selective Inhibition of EGFR signaling pathway appears to be a promising strategy for future tumor therapy. Thus, in our study, we identified COMPOUND7809 as a critical EGFR inhibitor that has a potent effect on lung cancer growth and susceptibility to chemotherapy, as treatment with kinase inhibitors has demonstrative anti-cancer effects including anti-proliferation, cell cycle arrest, and apoptosis. 17
Compared to an FDA-approved EGFR inhibitor gefitinib, COMPOUND7809 showed greater effects on cell growth arrest in an MTT assay, greater decreases in cell survival in an CCK-8 assay by 5-FU, possibly by augmentation of the cell apoptosis, and decrease in cell proliferation. The combination of these effects resulted in a significant enhancement of the anti-cancer effect. In future, the effects of this compound in vivo may be tested in a proper lung cancer animal model to evaluate whether it is similarly effective inside the body as in vitro.
Thus, we present a novel compound, which is a potential EGFR inhibitor, which appears to be a better choice than gefitinib in treating lung cancer. COMPOUND7809 may be a promising EGFR inhibitor for human lung cancer therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Guangdong Province Natural Scientific Fund (no. S2011010002100).
