Abstract
Epstein–Barr virus, a ubiquitous human herpes virus with oncogenic activity, can be found in 6%–16% of gastric carcinomas worldwide. In Epstein–Barr virus–associated gastric carcinoma, only a few latent genes of the virus are expressed. Ionizing irradiation was shown to induce lytic Epstein–Barr virus infection in lymphoblastoid cell lines with latent Epstein–Barr virus infection. In this study, we examined the effect of ionizing radiation on the Epstein–Barr virus reactivation in a gastric epithelial cancer cell line (SNU-719, an Epstein–Barr virus–associated gastric carcinoma cell line). Irradiation with X-ray (dose = 5 and 10 Gy; dose rate = 0.5398 Gy/min) killed approximately 25% and 50% of cultured SNU-719 cells, respectively, in 48 h. Ionizing radiation increased the messenger RNA expression of immediate early Epstein–Barr virus lytic genes (BZLF1 and BRLF1), determined by real-time reverse transcription polymerase chain reaction, in a dose-dependent manner at 48 h and, to a slightly lesser extent, at 72 h after irradiation. Similar findings were observed for other Epstein–Barr virus lytic genes (BMRF1, BLLF1, and BcLF1). After radiation, the expression of transforming growth factor beta 1 messenger RNA increased and reached a peak in 12–24 h, and the high-level expression of the Epstein–Barr virus immediate early genes can convert latent Epstein–Barr virus infection into the lytic form and result in the release of infectious Epstein–Barr virus. To conclude, Ionizing radiation activates lytic Epstein–Barr virus gene expression in the SNU-719 cell line mainly through nuclear factor kappaB activation. We made a brief review of literature to explore underlying mechanism involved in transforming growth factor beta–induced Epstein–Barr virus reactivation. A possible involvement of nuclear factor kappaB was hypothesized.
Background
Epstein–Barr virus (EBV), a ubiquitous human herpes virus with oncogenic activity, can be found in 6%–16% of gastric carcinomas worldwide.1,2 EBV genome is present in every carcinoma cell of an EBV-associated gastric carcinoma (EBVaGC). Only a few latent genes of EBV are expressed in EBVaGC. Although EBV is considered to play significant roles in the development of EBVaGC, LMP1, an important EBV oncoprotein, is only rarely expressed in such carcinomas. 3 On the contrary, EBV-encoded small RNAs (EBERs) are expressed in almost all EBVaGC cells, suggesting its importance for developing and maintaining the carcinoma. Several studies have shown that EBV might be reactivated in vivo before the development of these diseases.4–6 Levine et al. reported that EBVaGC patients have significantly high immunoglobulin G (IgG) and IgA antibody titers against EBV capsid antigen (VCA) more than 5 years preceding their diagnoses. 4 Similarly, a study in Japan also showed the presence of IgG antibody against VCA. 7 These observations suggest that EBV reactivation, producing lytic viral proteins rather than latent proteins, takes place before EBVaGC development. Indeed, spontaneous EBV reactivation in the tonsils is considered to occur regularly in healthy EBV carriers. 8 However, factors involved in EBV reactivation are yet unclear.
The reactivation of the replicative cycle of EBV induces the expression of EBV lytic transcripts, including immediate early lytic genes (such as BZLF1 and BRLF1), early lytic genes (such as BMRF1), and late lytic genes (such as BLLF1 and BcLF1), the expression of late lytic proteins (VCA and gp350/220), and the release of virus particles.9–14 The EBV late lytic proteins are produced during the late phase of the lytic cycle, and the majorities are structural proteins that are incorporated into the viral capsid and tegument as well as the envelope.
EBV reactivation can be induced in vitro experiments by chemicals, such as the phorbol ester—12-O-tetradecanoylphorbol-13-acetate and the histone deacetylase inhibitors—sodium butyrate, trichostatin A, and valproic acid. Anti-Ig can also induce EBV reactivation.10,15–18
Radiation also causes EBV reactivation in human and marmoset lymphoblastoid cell lines.10,19,20 A study has shown that transforming growth factor (TGF) beta is involved in spontaneous EBV lytic cycle reactivation in Burkitt Lymphoma cell lines (P3HR-I and Akata). 21
In lymphoblastoid cell lines with latent EBV infection, ionizing radiation also induced lytic EBV infection.10,19 Irradiation can activate TGF-beta signaling, 22 which is known to induce EBV reactivation in EBV-infected B cells. 23 In this study, we examined the effect of ionizing radiation on EBV genome expression in the SNU-719 cell line, a human gastric epithelial carcinoma cell line with natural EBV infection. 24 This cell line shows modified latency 1 EBV infection closely resembling EBVaGC and was reported to develop tumors when injected into nude mice and the developed tumor displayed similar characteristics with EBVaGC.24,25
Materials and methods
Cell lines and chemicals
The human gastric cancer cell lines SNU-719 (EBV-positive) and SNU-216 (EBV-negative) were received from Korean Cell Line Bank, Seoul, Korea. The SNU-216 and SNU-719 are moderately differentiated adenocarcinomas obtained from different patients. Both cell lines were cultured in RPMI-1640 medium supplemented with 10% heat-inactivated fetal bovine serum (Sigma-Aldrich, St. Louis, MO, USA), penicillin (10,000 U/mL), and streptomycin (10 mg/mL) at 37°C in a humidified 5% CO2 incubator. Antibody against TGF-beta1 (3C11) was purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Induction of lytic replication of EBV in cell culture (positive control)
For a stock solution, 1 mg/mL of O-tetradecanoylphorbol-13-acetate (TPA; Sigma-Aldrich, St. Louis, MO, USA) in dimethyl sulfoxide was prepared. The TPA-treated SNU-719 (EBV-positive) cells served as a positive control (chemically induced viral activation). SNU-216 (EBV-negative) cell line served as a negative control.
Irradiation of cells
The cells were exposed to radiation after 24 h of plating using the MBR-1505R X-ray unit (Hitachi Power Solutions Co., Ltd, Ibaraki, Japan). The machine was operated at 120 kVp and 3.8 mA with a filter of 0.5 mm of aluminium. The dose rate was 0.5398 Gy/min at a focus surface distance of 30 cm. The cells were irradiated with 1, 5, or 10 Gy. The cells post radiation were incubated at 37°C. The cells were then harvested at different time points.
Cell viability assays
Cell viability was determined using the WST-8 Assay Kit (Dojindo Molecular Laboratories, Kumamoto, Japan) according to the manufacturer’s instructions. EBV-positive SNU-719 and EBV-negative SNU-216 cell lines were cultured in 96-well plates, followed fixed dosages of radiation. At each time of incubation, the plates were washed with Hank’s Balanced Salt Solution and followed by WST-8 assay. Absorbance was measured using a microplate reader at 450 nm. Values represent the mean ± standard error of three experiments.
Transfection of RelB small interfering RNA by electroporation
SNU-719 cells were harvested by trypsinization, and then, the trypsinization reaction was neutralized with culture media containing serum and supplements and then the cells were washed with RPMI-1640 medium without serum and supplements. Briefly, 1 × 106 SNU-719 cells were resuspended with 10 μg of small interfering RNA (siRNA) in 100 μL of EP buffer and transfected using Nepa21 electroporator (Nepa Gene, Chiba, Japan) with an electric pulses of (1) poring pulse: 100 V, 2.5 ms of length with 50 ms interval and (2) transfer pulse: 20 V, 50 ms of length with 50 ms interval. After transfection, the cells were immediately mixed with pre-warmed 1.2 mL of RPMI-1640 medium with serum and supplements and transferred to a 12-well plate. Cells were irradiated after 24 h. At 48 and 72 h after radiation, the cells were harvested to check the RelB messenger RNA (mRNA) by real-time reverse transcription polymerase chain reaction (RT-PCR). siRNA to target RelB (sense: 5′-GGAUUUGCCGAAUUAACAA-dTdT-3′ and antisense: 5′-UUGUUAAUUCGGCAAAUCC-dTdT-3′) and a negative control siRNA (MISSION siRNA Universal Negative Control SIC-001) were provided by Sigma Genosys (Sigma-Aldrich, Japan). All experiments were performed in triplicate.
Real-time RT-PCR
For quantitative real-time RT-PCR, total RNA of the cells was isolated using RNeasy Mini kit (QIAGEN, Hilden, Germany), and complementary DNAs (cDNAs) were synthesized from total RNA using Quanti Tect Reverse Transcription Kit (QIAGEN). Expressed genes were detected quantitatively using the Light Cycler® 2.0 Instrument (Roche Diagnostics, Mannheim, Germany) with Light cycler Fast Start DNA MasterPLUS SYBR Green I (Roche Diagnostics) according to manufacturer’s instructions. The primers for the genes were purchased from FASMAC Co. Ltd (Japan). Primer sequences are listed in Table 1. PCR amplification was performed in a total volume of 20 µL containing cDNA and each primer (0.5 µM). The PCR cycling conditions were 95°C for 10 s, 60°C for 10 s, and 72°C for 15 s. The fluorescent product at the end of the 72°C was determined. All PCR assays were performed at least four times.
List of primers used.

The data obtained were analyzed using the Light Cycler analysis software. To confirm the amplification specificity, the PCR products were subjected to melting curve analysis. Threshold cycle values of the target genes were normalized to those of the internal control genes. Values represent the mean ± standard error of three experiments.
Results
Cell cytotoxicity induced by ionizing radiation
Cultured cells were irradiated with different doses of X-ray 24 h after cell culture when it reached confluence. At 48 h after irradiation, cytotoxicity was observed in the EBV-positive cell line (SNU-719) in a dose-dependent manner (Figure 1(a)). However, the EBV-negative cell line (SNU-216) was more resistant to radiation. Even 10 Gy of irradiation did not show any significant cytotoxicity in the SNU-216 cells at 48 h after irradiation. At 72 h after irradiation, the cell viability did not show any significant evident difference between those two cell lines (Figure 1(b)).

Cell viability of SNU-719 (EBV-positive) and SNU-216 (EBV-negative) cells at (a) 48 h and (b) 72 h after irradiation. Values represent the mean ± standard error of three experiments.
EBV reactivation induced by ionizing radiation
To understand the reactivation of the replicative cycle of EBV, cells were harvested at 12, 24, 48, and 72 h after irradiation, and the viral lytic gene expression was quantified (Figure 2(a)–(c)). The mRNA expression of BZLF1 and BRLF1, immediate early EBV lytic genes, was increased in a dose-dependent manner at 48 h (Figure 2(b)) and, to a slightly lesser extent, at 72 h (Figure 2(c)) after irradiation. The mRNA expression of BMRF1, BLLF1, and BcLF1 were also upregulated. Those EBV lytic genes were also reactivated by TPA in SNU-719 cell line (data not shown).

The EBV early gene expression in SNU-719 cells at (a) 24 h, (b) 48 h, and (c) 72 h after irradiation. Values represent the mean ± standard error of three experiments.
TGF-beta1 expression induced by ionizing radiation
Similarly, after irradiation of SNU-719 cells, the expression of TGF-beta1 mRNA was increased in a dose-dependent manner and reached a peak at around 12–24 h (Figure 3). The irradiated cells showed no expression of TGF-beta1 mRNA in the presence of antibody against TGF-beta1 (5 ng/mL), which was added to the culture medium 3 h before irradiation. In the presence of TGF-beta1 antibody in the culture medium, the radiation-induced mRNA expression of EBV lytic genes was decreased (data not shown).

TGF-beta1 expression in SNU-719 cells after irradiation. Values represent the mean ± standard error of three experiments.
RelA and RelB expression induced by ionizing radiation
To elucidate the induced activation of lytic pathway by ionizing radiation, irradiated SNU-719 cells were checked for the RelA and RelB expression by real-time PCR. RelB level showed a dose-dependent enhancement on 2nd and 4th day, whereas only a slight upregulation in RelA is noticed (Figure 4). No appreciable change was observed in RelB level of the irradiated cells as well as in untreated controls on day 1.
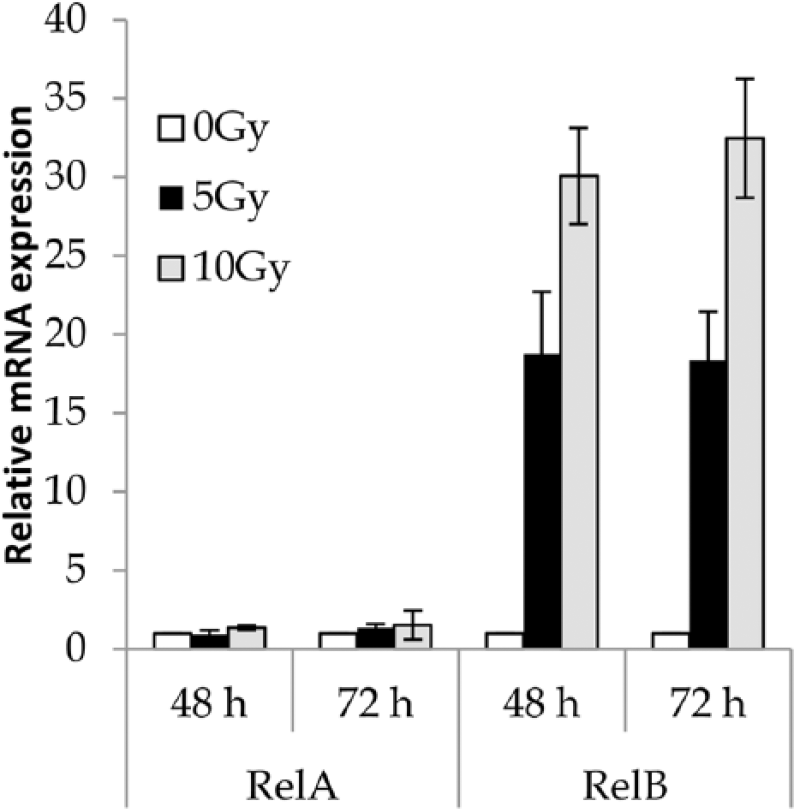
Relative expression of mRNA level of NF-kappaB subunits, RelA and RelB.
EBV reactivation induced by ionizing radiation with RelB knockdown
The results indicated that ionizing radiation induced EBV activation mainly through RelB upregulation. To justify the role of RelB, SNU-719 cells were treated with ionizing radiation after the RelB gene was knocked down using RelB siRNA and were examined for the presence of time- and dose-dependent regulation of EBV activation. Figure 5(a–d) shows the relative expression of immediate early lytic genes, BZLF1 and BRLF1, RelA, and RelB after the RelB gene was knocked down. BRLF1 expression was completely downregulated (Figure 5(b)). BZLF1 and RelA expression was also downregulated at 5 Gy of radiation but a slight upregulation was observed at 10 Gy of radiation (Figure 5(a) and (c)).

Relative expression levels of EBV lytic genes, (a) BZLF1 and (b) BRLF1, and NF-kappaB subunits, (c) RelA and (d) RelB, with/without siRNA treatment for RelB.
Discussion
In this study, we showed that the mRNA expression of EBV lytic genes was induced 48–72 h after irradiation in a dose-dependent manner in the SNU-719 cell line. Our findings are consistent with the results reported by Westphal et al. 10 They have shown that lower doses of gamma radiation can induce the protein level expression of BZLF1 and BMRF1, which indicates the EBV reactivation in various EBV-positive malignant cell lines including gastric carcinoma cell line (AGS-EBV). In vitro, high-level expression of either of the two EBV immediate early genes, BZLF1 and BRLF1, is sufficient to convert latent EBV infection into the lytic form. In addition, activation of the lytic form of EBV infection in tumors would result in release of infectious EBV, although it is uncertain that such a release would result in clinical consequences. 10
The SNU-216, an EBV-negative gastric cancer cell line, was more resistant to radiation when compared to SNU-719 (Figure 1). The difference may be explained by EBV lytic infection induced by irradiation in the SNU-719 cell line. However, we cannot deny the possibility that the observed difference is attributable to the factors other than EBV. It has to be noted that those cell lines are from different patients, and the SNU-719 and SNU-216 cell lines were established from the tumor of the stomach and lymph node, respectively. 26 For example, radiation sensitivity of the hosts of those carcinomas might have been different from each other.
In this study, the TGF-beta1 expression was also increased by irradiation, and its expression reached a peak at 12–24 h after irradiation. This result is consistent with Dancea et al. 22 and with the previous studies in murine mammary gland, in which active TGF-beta was apparent after radiation exposure and persisted for up to several days post irradiation.27–29 Our results of the experiments using TGF-beta1 antibody indicated that the induction of TGF-beta1 gene is necessary to induce the expression of lytic EBV genes.
TGF-beta is well known to upregulate nuclear factor (NF)-kappaB signaling, which reactivates EBV. A study reported that TGF-beta1 induced the expression of early antigens of EBV lytic cycle in Burkitt lymphoma cell lines (Akata and P3HR-1); however, this study did not investigate the downstream signaling pathway of TGF-beta1 involved in the EBV reactivation. 21 In a more recent study, which used another Burkitt lymphoma cell line (Raji), purified recombinant TGF-beta1, added to the culture medium, induced EBV reactivation through cFos and cJun which were upregulated by the mitogen-activated protein kinase (MAPK) pathway. 30 The study also showed that TGF-beta1 upregulated MAPK pathway through a transcription factor Smad3/4. A later study confirmed the involvement of this transcription factor in addition to cFos and cJun in the EBV reactivation induced by TGF-beta1. However, BZLF1, an immediate early gene of EBV lytic cycle, was induced by Smad3/4 even in the presence of a MAPK pathway inhibitor, indicating that MAPK is not involved in the Smad3/4-mediated EBV reactivation. 31 Another study revealed that the TGF-beta overexpression after the radiation exposure induced TGF-beta signaling through the induction of Smad7 and TGF-beta type II receptor. 22 It is also involved in the activation of p53 and ataxia telangiectasia mutated (ATM) signaling pathways thereby increasing apoptosis in irradiated cells. Low-dose radiation (50 mGy gamma-rays) exposure of normal cells showed some bystander effects on transformed cells leading to their apoptosis in a TGF-beta-dependent manner. 22
Although TGF-beta typically represses NF-kappaB signaling in normal cells, recent in vitro and in vivo evidence suggests an aberrant ability of TGF-beta signaling to activate NF-kappaB signaling in several different cancers as a means of promoting malignant tumor cell phenotypes.32,33 Aspirin was also shown to have such a dual nature in NF-kappaB regulation. In normal cells, aspirin is known to suppress NF-kappaB expression. However, in Burkitt lymphoma cells and gastric cancer cell line (SNU-719), aspirin induced NF-kappaB expression and EBV reactivation.34,35 It is to be noted that NF-kappaB is well known to be induced by cFos and cJun.36,37 Taking together, ionizing radiation reactivates EBV lytic cycle mainly through TGF-beta1, which upregulates cFos and cJun through Smad3/4. cFos and cJun may reactivate EBV lytic cycle through NF-kappaB upregulation.
The radiation doses used in this study are so large that it does not give any insight into the effect of low-dose radiation on the EBV reactivation. However, the combination of some cell stress and low-level radiation, such as natural radiation or medical X-ray, may be able to induce the EBV reactivation in the cells with latent EBV infection. Indeed, it was reported that the combination of clinically relevant doses of radiation and treatment with a glucocorticoid (the final effecter of the stress response) increased the expression of ZEBRA, the EBV protein coded by BZLF1, in B95-8 cells. 19
One of the important limitations of this study is the fact that the production of EBV virion or EBV proteins was not confirmed. For example, the presence of the immediate early protein ZEBRA is a hallmark of EBV reactivation. In this study, the mRNA expression of BZLF1, which encodes the ZEBRA protein, was upregulated. In addition, this study showed the mRNA expression of BLLF1, a late gene encoding the viral envelope glycoprotein gp350. Therefore, we can conclude that late stages of EBV reactivation took place by irradiation. Westphal et al. 10 demonstrated that lower doses of gamma radiation can induce BZLF1 and BMRF1 at protein levels in EBV-positive gastric carcinoma cell line (AGS-EBV). Although we did not examine these proteins, similar protein expression would be expected in our study as well because of the results of RNA expression. The second limitation is the use of only one set of EBV-positive and -negative GC cell lines. Thus, there is no guaranty that the same results can be obtained in other cell lines. The third one is a lack of determination of other potential transcriptional proteins. Only RelA and RelB were targeted in this study.
Conclusion
Ionizing radiation activates lytic EBV gene expression in SNU-719 cell line as a result of the NF-kappaB activation. Previous studies10,35 reported that anti-herpes treatment could be enhanced after the induction of EBV reactivation. Further ex vivo and in vivo studies are warranted to confirm these results and to evaluate clinical relevance of the utilizing ionizing radiation alone or in combination with anti-herpes agents as a therapeutic strategy for EBVaGC.
Footnotes
Acknowledgements
The authors thank Joint Research Laboratory of Kagoshima University Graduate School of Medical and Dental Sciences for providing the facilities and Professor Kim Woo Ho for his scientific advice.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Kodama Memorial Fund for Medical Research.
