Abstract
Labile iron pool is a cellular source of ions available for Fenton reactions resulting in oxidative stress. Living organisms avoid an excess of free irons by a tight control of iron homeostasis. We investigated the altered expression of iron regulatory proteins and iron discrimination in the development of liver fluke–associated cholangiocarcinoma. Additionally, the levels of labile iron pool and the functions of transferrin receptor-1 on cholangiocarcinoma development were also identified. Iron deposition was determined using the Prussian blue staining method in human cholangiocarcinoma tissues. We investigated the alteration of iron regulatory proteins including transferrin, transferrin receptor-1, ferritin, ferroportin, hepcidin, and divalent metal transporter-1 in cholangiocarcinoma tissues using immunohistochemistry. The clinicopathological data of cholangiocarcinoma patients and the expressions of proteins were analyzed. Moreover, the level of intracellular labile iron pool in cholangiocarcinoma cell lines was identified by the RhoNox-1 staining method. We further demonstrated transferrin receptor-1 functions on cell proliferation and migration upon small interfering RNA for human transferrin receptor 1 transfection. Results show that Iron was strongly stained in tumor tissues, whereas negative staining was observed in normal bile ducts of healthy donors. Interestingly, high iron accumulation was significantly correlated with poor prognosis of cholangiocarcinoma patients. The expressions of iron regulatory proteins in human cholangiocarcinoma tissues and normal liver from cadaveric donors revealed that transferrin receptor-1 expression was increased in the cancer cells of cholangiocarcinoma tissues when compared with the adjacent normal bile ducts and was significantly correlated with cholangiocarcinoma metastasis. Labile iron pool level and transferrin receptor-1 expression were significantly increased in KKU-214 and KKU-213 when compared with cholangiocyte cells (MMNK1). Additionally, the suppression of transferrin receptor-1 expression significantly decreased intracellular labile iron pool, cholangiocarcinoma migration, and cell proliferation when compared with control media and control small interfering RNA. In Conclusion, high expression of transferrin receptor-1 resulting in iron uptake contributes to increase in the labile iron pool which plays roles in cholangiocarcinoma progression with aggressive clinical outcomes.
Keywords
Introduction
Cholangiocarcinoma (CCA) is the malignant tumor of bile duct epithelial cells, which is the most common cancer in Thailand and the major public health problem of the northeast region. Results from epidemiological and experimental studies support that inflammation-related diseases are the etiology of CCA progression, such as the liver fluke (Opisthorchis viverrini) infection which is the etiology of CCA development in Thailand. Our previous studies strongly suggested that CCA carcinogenesis could be induced by oxidative/nitrative stress through O. viverrini infection–driven chronic inflammation. 1 It has also been shown that heme oxygenase 1 (HO-1)-derived iron accumulation and inducible nitric oxide synthase (iNOS)-derived nitric oxide (NO) could participate in the enhancement of oxidative DNA damage along with O. viverrini–associated CCA carcinogenesis. 2 Iron is an essential nutrient and the most abundant transition metal in the human body. Due to its ability to accept and donate electrons during conversion among ferric (Fe3+) and ferrous (Fe2+) oxidation states, iron is a critical component of sensor, transporter and storing molecules, and enzymes involved in energy production, intermediate metabolism, cell proliferation, cell cycle, and DNA damage sensing.3–6 Organisms try to avoid an excess of “free” iron by a tight control of iron homeostasis. However, iron overload could result from increase in the reactive oxygen species (ROS) production via Fenton reaction. Since it needs different proteins to carry out the uptake and storage of iron, there is a pool of accessible iron ions, called labile iron pool (LIP) that acts as crossroads of metabolic pathways of iron-containing compounds and it is a cellular source of iron ions available for Fenton reaction.
Cancer cells increase metabolically available iron by increasing iron uptake and decreasing iron storage, as well as by decreasing iron efflux. Previous studies on the role of iron and cancer have revealed that proteins involved in iron metabolism can be regulated. 7 Thanan et al. 8 demonstrated that the oxidative stress induced by inflammation causes the oxidation of transferrin (Tf), an iron (Fe3+) binding and transporting protein, leading to the accumulation and release of iron; therefore, the increased LIP may mediate Fenton reactions and generate ROS, which may contribute to all steps of CCA genesis. In addition, the increase in transferrin receptor-1 (TfR-1), which is responsible for transferrin-mediated iron uptake, occurs in many cancers. 9 Moreover, the non-transferrin-bound iron enters cells via a pathway mediated by divalent metal transporter-1 (DMT-1), which is another protein involved in iron uptake. Reduction of ferritin, which is an iron storage protein protecting cells against iron toxicity, can cause increase in labile pool; therefore, it may act as a potential source of LIP. 10 Cancer cells increase metabolically available iron by increasing iron uptake and decreasing iron storage, as well as by decreasing iron efflux. Moreover, one of the most important discoveries in iron regulation was the identification of low ferroportin and high hepcidin, which is correlated with poor prognosis in breast cancer. 11
In combination, these iron regulatory proteins (IRPs), which are aberrantly expressed or modified substantially contribute to the regulation of systemic iron levels and play key roles in cancer development including CCA genesis. Therefore, our aim was to identify whether the aberrant expression of key proteins involved in iron regulation including iron uptake (Tf, TfR-1, and DMT-1), iron storage (ferritin), and iron efflux (ferroportin and hepcidin) substantially contributed to the regulation of systemic iron levels and hence play key roles in CCA development.
Materials and methods
Human CCA tissues
The paraffin-embedded CCA tissues (n = 49) and cadaveric donors (n = 3) were obtained from the specimen bank of the Cholangiocarcinoma Research Institute, Khon Kaen University. The protocol of collection and study design were approved by the Ethics Committee for Human Research, Khon Kaen University (HE571283), and informed consent was obtained from each subject before surgery.
Cell culture
The immortalized human cholangiocyte MMNK1 (MMNK1) cell line was supplied by Professor Naoya Kobayashi (Okayama University, Japan). 12 CCA cell lines including KKU-214 and KKU-213 were established from the tumor of CCA patients at Srinagarind Hospital, Khon Kaen University by Dr. Banchob Sripa (Khon Kaen University), and a certificate of analysis was obtained from the Japanese Collection of Research Bioresources (JCRB) Cell Bank. Cells were cultured in phenol red Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen, USA) supplemented with 10% fetal calf serum, 100 U/mL penicillin, and 0.1 mg/mL streptomycin at 37°C in 5% CO2. The culture medium was changed every 48 h.
Prussian blue staining
Prussian blue was used for localization of ferric iron in CCA tissue sections. Human CCA sections were deparaffinized and rehydrated with graded alcohol. Slides were then immediately transferred into a working staining solution (equal volumes of 10% potassium ferrocyanide and 10% hydrochloric acid) for 20 min at room temperature. Any ferric ions (Fe3+) in the tissue combine with the ferrocyanide and result in the formation of a bright blue pigment. Subsequently, slides were rinsed in distilled water and then counterstained with nuclear-fast red. Finally, sections were dehydrated with stepwise increasing concentrations of ethanol, cleared with xylene, and mounted with permount solution. The stained sections were examined under a light microscope. The staining frequency of iron accumulation was semi-quantitatively scored based on the percentage of positive area as 0% = negative, 1%–25% = +1, 26%–50% = +2, and >50% = +3. The staining intensity in tumor tissue was scored as negative = 0, weak = 1, moderate = 2, and strong = 3. Qualitative iron accumulation patterns were calculated by multiplying the frequency score with intensity score and classified into three categories: 0, negative; 1–3, low accumulation; and 4–9, high accumulation.
Immunohistochemistry
An immunohistochemical method was performed to determine the expression patterns of Tf, TfR-1, ferritin, ferroportin, hepcidin, and DMT-1 in human CCA tissues. Sections of human liver CCA tissues were deparaffinized and rehydrated with stepwise decreasing concentrations of ethanol. Antigen retrieval was performed by microwave treatment in 10 mM citrate buffer (pH 6.0) at high power for 10 min, and then, sections were immersed for 30 min in 3% (v/v) hydrogen peroxide in phosphate-buffered saline (PBS) for endogenous hydrogen peroxide activity blocking. Nonspecific binding was blocked by 5% bovine serum albumin (BSA) in PBS for 30 min. Sections were incubated with mouse monoclonal anti-Tf (10 µg/mL; Abcam, UK), rabbit polyclonal anti-TfR-1 (0.5 µg/mL; Sigma, USA), goat polyclonal anti-ferritin (1 µg/mL; Abcam), mouse monoclonal anti-ferroportin (5 µg/mL; Abcam), rabbit polyclonal anti-hepcidin (4 µg/mL; Abcam), or rabbit polyclonal anti-DMT-1 (5 µg/mL; Abcam) at 4°C overnight. Subsequently, sections were washed in PBS and incubated with peroxidase-conjugated secondary antibody. After washing in PBS, the color was developed with 3,3′-diaminobenzidine tetrahydrochloride (DAB) substrate kit (Vector Laboratories, USA) for 10 min and then counterstained with Mayer’s hematoxylin. The sections were dehydrated with stepwise increasing concentrations of ethanol, cleared with xylene, and mounted with permount solution. The staining frequency of these proteins was semi-quantitatively scored based on the percentage of positive cells as follows: 0% = negative, 1%–25% = +1, 26%–50% = +2, and >50% = +3. The staining intensity in tumor cells was scored as negative = 0, weak = 1, moderate = 2, and strong = 3. Qualitative expression patterns were calculated by multiplying the frequency score with intensity score and classified into three categories: 0, negative; 1–3, low expression; and 4–9, high expression.
Transient knockdown of TfR-1 by small interfering RNA
The small interfering RNAs (siRNAs) of human TfR-1 and control siRNA were purchased from Dharmacon (GE Healthcare, UK) (TfR-1 siRNA ID: L-003941-00-0005 and control siRNA ID: D-001810-10-05). The KKU-214 cell line with high TfR-1 expression was used in this experiment. Briefly, 1.0 × 105 cells were transfected with a final concentration of 50 nM of siRNA duplexes using Lipofectamine transfection reagent (Invitrogen). Then, transfected cells were incubated at 37°C in 5% CO2 for 48 h. Silencing of TfR-1 expression was confirmed with western blot analysis.
Western blot analysis of transferrin receptor in cell lines
Protein solutions were extracted from cell pellets of MMNK1, KKU-213, KKU-214, and siTfR-1 KKU-214 cell lines using radioimmunoprecipitation assay (RIPA) buffer (containing the Protease K inhibitor cocktail, 1% NP-40, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS), and deionized water) and subjected to western blot analysis. SDS–polyacrylamide gel electrophoresis (PAGE) was used to separate 15 μg of proteins after which the separated protein(s) was transferred into the polyvinylidene difluoride (PVDF) membrane. Immunodetection of TfR-1 was detected using 0.5 μg/mL of rabbit anti-TfR-1 (Sigma) or 0.04 μg/mL of mouse anti-actin (Sigma) as a primary antibody and corresponding horseradish peroxidase (HRP)-conjugated anti-IgG antibody as a secondary antibody. The chemiluminescence band intensities were detected by enhanced chemiluminescence (ECL) solution (GE Healthcare, UK) and calculated by ImageJ free software.
Determination of LIP
LIP was analyzed using RhoNox-1 staining kit (Goryo, Japan). For Fe2+ uptake experiments, cells were treated with 100 μM ferrous ammonium sulfate (FAS, Fe(NH4)2(SO4)2 from 10 mM stock solution in water) in DMEM media without fetal bovine serum (FBS) at 37°C for 30 min. The cells were washed with Hank’s Balanced Salt Solution (HBSS; Gibco, USA) for three times. Then, 5 μM of RhoNox-1 was added and incubated for 1 h at 37°C. After that, the cells were washed with HBSS. For nuclear staining, 4′,6-diamidino-2-phenylindole (DAPI; 1:2000 in 50% glycerol) was added to the cells before imaging. The fluorescence intensity was measured using confocal microscopy with excitation wavelength at 546 nm and with emission wavelength at 575 nm. The different intensities were calculated by ImageJ free software.
Cell proliferation assay
The sulforhodamine B (SRB) assay was used for cell density determination, based on the measurement of cellular protein content. Cell monolayers were fixed with 10% (w/v) trichloroacetic acid (TCA) and stained for 45 min in dark. The excess dye was removed by washing repeatedly with 1% (v/v) acetic acid. The protein-bound dye was dissolved in 10 mM Tris base solution for optical density (OD) determination at 540 nM using microplate reader.
Clonogenic assay
The clonogenic cell survival assay determines the ability of cell proliferation, thereby retaining its reproductive ability to form a large colony. A colony was defined to consist of at least 200 cells cultured in DMEM media for 10 days. Media was removed and changed every 3 days. Cells were then removed from media and fixed with 5% paraformaldehyde for 20 min. Cells were washed with PBS before adding 0.5% crystal violet for 15 min and were counted using a light microscope.
Migration assay
A cell migration assay was performed using a Boyden Transwell chamber which consisted of a membrane filter inserted in 24-well plates (8-μm pore size; Corning, USA). The siTfR-1-transfected cells (20 × 104 cells) were seeded into the upper chamber with serum-free medium, and aliquots of DMEM complete media were placed in the lower chamber. After 24 h, non-migrating cells residing in the upper filter were removed. Migrating cells that were attached at the underside of the filter were fixed with methanol and stained with hematoxylin overnight at room temperature. The number of migratory cells was quantified by counting under a light microscope (10× magnifications). The experiments were performed in triplicates and two independent experiments were repeated.
Statistical analysis
Statistical analyses were performed using SPSS software. The associations of iron with Tf, TfR-1, ferritin, ferroportin, hepcidin, and DMT-1 expression in tissues and patients’ clinicopathological data were analyzed using the Chi-square test. Survival curves were calculated according to the method of Kaplan and Meier. A p value < 0.05 was considered statistically significant.
Results
Iron accumulation in human CCA tissues
Prussian blue staining demonstrated that iron was strongly stained both at the edges of tumors and in tumor tissues, whereas negative staining was seen in normal bile duct and hepatocytes from cadaveric donors as shown in Figure 1(a). Fe3+ was also strongly found in hepatocytes of the tumor adjacent tissues and Kupffer cells in both tumor adjacent and normal liver tissues. In addition, 51% (25/49) of CCA patients had high accumulation of Fe3+ in their tumor tissues. Interestingly, a correlation analysis showed that high iron accumulation was significantly associated with poor prognosis of CCA patients (p = 0.01) as shown in Figure 1(b).

(a) Iron deposition in the liver using Prussian blue staining. (b) The survival curves were calculated for iron according to Kaplan–Meier, with a log-rank test (p = 0.010; original magnification was 400×; BD: bile duct).
Expression of IRPs in CCA tissues
Figure 2 shows the expression patterns of IRPs including Tf, TfR-1, ferritin, ferroportin, and hepcidin. High expression of transferrin was detected in 41% (20/49) of CCA patients. It was intensively localized in cytoplasm of hyperplasia bile ducts and the bile duct cancer cells in CCA tumor tissues. Furthermore, 41% of CCA patients had high expression of TfR-1 in their tumor tissues, which was strongly detected in hyperplasia bile ducts and the cancer cells of CCA tissues, when compared to normal cholangiocytes from the individual normal bile duct cells.

Localization of Tf, TfR-1, ferritin, ferroportin, and hepcidin in normal, hyperplasia, and the cancer of bile duct epithelium cells in CCA tissues by immunohistochemistry method (original magnification was 400×; BD: bile duct).
In contrast, ferritin was strongly detected in the cytoplasm of hepatocytes in normal livers and the adjacent normal area of CCA tumor tissues. It was also weakly expressed in CCA cells when compared with the individual cholangiocytes in CCA tumor tissues. CCA patients (59%, 29/49) had low ferritin expression in the tumor tissues.
Ferroportin was strongly localized in the cytoplasm of hepatocytes, inflammatory cells, hyperplasia bile ducts, and CCA cancer cells. Moderate immunoreactivities of ferroportin were detected in normal cholangiocytes from normal liver and adjacent normal area of CCA tissues. Most of CCA patients (62%, 31/49) had low ferroportin expression.
Hepcidin was expressed in hyperplasia bile ducts and the cancer cells of CCA tumor tissues and slightly detected in normal bile duct epithelium cells. Most of CCA patients (84%, 41/49) had low hepcidin expression in the tumor tissues.
However, staining of DMT-1 was not seen in any cell types of CCA tissues, suggesting that DMT-1 may not be associated with iron accumulation in CCA (data not shown).
The association of IRP expressions and clinicopathological data of CCA patients
The expressions of Tf, TfR-1, ferritin, ferroportin, and hepcidin were then analyzed with clinicopathological data including sex, age, histology types, survival rate, and metastasis stages of patients. There were no correlations of the IRP with sex, age, tumor types, and/or survival rate. Only high expression of TfR-1 was significantly correlated with positive metastasis stage as shown in Table 1, suggesting its function in tumor progression of CCA carcinogenesis. Therefore, TfR-1 was then selected for further functional analysis in CCA progression.
The association between iron and iron regulatory protein expression and metastasis data of CCA patients.

CCA: cholangiocarcinoma.
p < 0.05 (Chi-square test).
Expression of TfR-1 and accumulation of intracellular LIP in CCA and cholangiocyte cell lines
The levels of TfR-1 expression and LIP accumulation were measured in CCA cell lines (KKU-213 and KKU-214) compared with the immortalized human cholangiocyte cell line (MMNK1). TfR-1 levels were measured by western blot analysis, while LIP was determined by RhoNox-1 staining. The results showed that TfR-1 was higher in KKU-214 than KKU-213 and MMNK1 as shown in Figure 3(a) and (b). In addition, the level of LIP was also significantly increased in KKU-214 cells (Figure 3(c) and (d)). These results demonstrated that increasing levels of TfR-1 were associated with LIP accumulation.
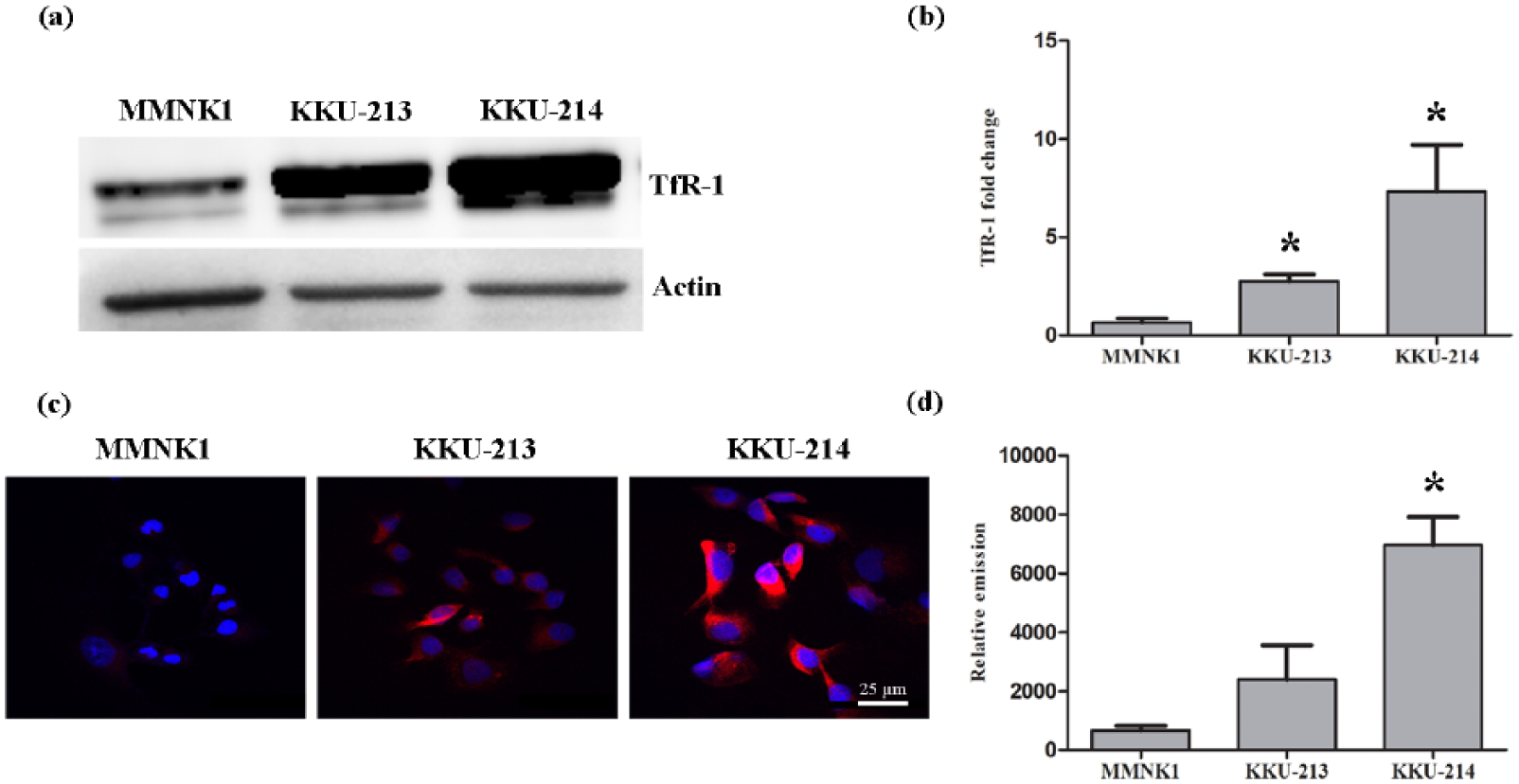
(a) Western blotting shows the expression of TfR-1 in MMNK1 and CCA cell lines (KKU-213 and KKU-214). (b) Level of TfR-1 was normalized to beta-actin. (c) Microscopic analysis of RhoNox-1 fluorescence in MMNK1, KKU-213, and KKU-214 cell lines. (d) Histograms represent the level of LIP in each group (*p < 0.05 compared to MMNK1 control).
Knockdown of TfR-1 reduced intracellular LIP, CCA migration, and cell proliferation ability
In this study, the KKU-214 cells were transiently transfected with siTfR-1 according to the methods in previous studies.13,14 Our result showed that siTfR-1 transfection achieved 85% suppression of TfR-1 protein level compared with control siRNA (non-targeting siRNA; Figure 4(a) and (b)). Consequently, the level of intracellular LIP upon knockdown of TfR-1 was tested using reactivity of the LIP with RhoNox-1. TfR-1 knockdown CCA cells had a significant reduction of LIP level compared with control siRNA and control media–transfected cells (Figure 4(c) and (d)).

Effect of siTfR-1 on KKU-214 cell function. (a and b) Western blot analysis shows a markedly decreased TfR-1 protein level after 48 h of transfection. (c and d) Level of intracellular LIP upon knockdown of TfR-1 was tested using reactivity of the LIP with RhoNox-1. (e) Transfection of siTfR-1 significantly decreased cell proliferation at 24, 48, and 72 h post transfection as determined by SRB assay, which was confirmed by (f) clonogenic assay. (g and h) Inhibition of CCA migration was demonstrated by Transwell migration assay (*p < 0.05).
We further investigated TfR-1 functions on cell proliferation and migration upon siTfR-1 transfection. As shown in Figure 4(e), we performed an SRB assay to determine cell proliferation after transfection at 6, 18, 24, 48, 72, and 96 h. The results revealed that siTfR-1 transfected cells significantly decreased cell proliferation when compared with those of the control cells at 48, 72 and 96 h post transfection. Moreover, a clonogenic assay was performed to confirm growth ability after siTfR-1 transfection. Colonies were stained with 0.5% crystal violet. The results showed that the siTfR-1 transfected cells formed colonies significantly less than control media and control siRNA (Figure 4(f)). Furthermore, the suppression of TfR-1 expression significantly decreased cell migration compared with control media and control siRNA (p = 0.016 and 0.007, respectively; Figure 4(g) and (h)).
Discussion
Iron is necessary for cell growth and metabolism; however, an excess of free iron or LIP could induce oxidative stress via Fenton’s reaction. Normally, the cells are protected against LIP toxicity by controlling iron uptake and storage. When the intracellular demand for iron increases, the iron uptake is promoted by increasing the expression of TfR-1 and by decreasing the expression of iron storage protein ferritin.15–17 Several of these processes are altered in cancer development.
In this study, we found that there were high levels of iron in CCA tissues which was significantly correlated with poor prognosis. Moreover, we investigated the alteration of key proteins involved in iron metabolism (Tf, TfR-1, ferritin, ferroportin, hepcidin, and DMT-1) in CCA tissues using immunohistochemistry. Transferrin and TfR-1 were increased in CCA cells when compared with normal bile duct cells, indicating that cancer cells may increase iron uptake. However, most of the CCA tumor tissues reduced iron storage and iron efflux systems. Interestingly, a high expression of TfR-1 in CCA tissues was correlated with positive metastasis stage, suggesting that overexpression of TfR-1 plays a significant role in metastasis and CCA progression. Increased iron uptake has been identified in malignant cells. An increase in TfR-1, a cell surface receptor responsible for transferrin-mediated iron uptake, occurs in many cancers, such as breast cancer 11 and pancreatic cancer. 9 Therefore, our results together with previous studies suggest that TfR-1 is a possible candidate molecule for involvement in the increase of cellular iron uptake in CCA.
Inflammatory conditions are characterized by the intracellular deposition of LIP, which may promote tissue damage via Fenton chemistry resulting in oxidative stress. 18 Oxidative stress plays roles in inducing oxidative damage to biomolecules leading to mutation as well as in an altering gene expression through genetic instability and epigenetic changes. 19 Labile iron is an important determinant of ROS generation because this is a pool of iron which can undergo redox reaction and stimulate free radical formation. H2O2-mediated toxicity was found to be iron mediated. TfR-1 knockdown has been shown to reduce iron uptake by 80% in human hepatoma cell line. 20 TfR-1 has been reported to be a major molecule that responded to iron uptake mediated by insulin through hypoxia condition. 21 Andriopoulos et al. 22 found that exposure of cultured cells to sustained low levels of hydrogen peroxide that mimics its release by inflammatory cells leads to upregulation of TfR-1.
In addition, other studies showed that exposure to exogenous hydrogen peroxide upregulates IRP-1. IRP-1 is a bifunctional protein that functions as an iron-responsive element (IRE)-binding protein involved in the control of iron metabolism by binding messenger RNA (mRNA) to repress translation or degradation, in particular the mRNA encoding ferritin and TfR-1.23–25 Both in vitro and in vivo studies have shown that a number of conditions or agents known to increase cellular levels of ROS induce the reversible inactivation of IRP-1 aimed at decreasing TfR-1 levels while also increasing ferritin levels and hence diminishing the LIP.26–30 To define that TfR-1 is related with LIP accumulation in CCA, we detected TfR-1 expression and LIP accumulation in CCA cell lines compared with immortal cholangiocyte cell line. Our results showed that TfR-1 expression and LIP levels were significantly increased in CCA cell lines than in the cholangiocyte cell line, suggesting that TfR-1 may be associated with iron uptake and hence induce LIP in CCA.
We next elucidated the role of TfR-1 on CCA cell functions and LIP alteration using siRNA technique. Decrease in intracellular LIP level was determined upon knockdown of TfR-1, in parallel with a decrease in CCA cell migration and proliferation. Our finding was supported by previous studies which have demonstrated higher TfR-1 expression both in in vivo and in vitro, leading to sustained iron uptake in ovarian endometriosis. 31 Interest in the role of iron and proteins of iron metabolism in cell proliferation has been subsequently confirmed by studies of the regulation of TfR-1 expression. 32 Thus, cellular iron metabolism needs to be highly regulated, and abnormalities in any of its different steps may affect both the survival and the proliferative activity of the cells. 33
Conclusion
In conclusion, we confirmed that high accumulation of iron which may contribute to LIP is significantly correlated with a poor prognosis of CCA patients. Moreover, the aberrant expression of TfR-1 substantially contributed to the regulation of systemic iron levels and can play key roles in CCA development. Our present results suggest that TfR-1 is a key IRP in CCA and treatment of iron overload, which provide the basis to explore a new direction for CCA therapy.
Footnotes
Acknowledgements
The authors would like to thank Prof. Ross H Andrews for editing the manuscript through Publication Clinic KKU, Thailand.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Thailand Research Fund through Royal Golden Jubilee Ph.D. Program (to W.J. and P.Y.); the Khon Kaen University Research Fund; a Grant from Faculty of Medicine (Grant No. IN58246), Khon Kaen University, Thailand; and the Higher Education Research Promotion and National Research University Project of Thailand, Office of the Higher Education Commission, through the Center of Excellence in Specific Health Problems in Greater Mekong Sub-region Cluster (SHeP-GMS), Khon Kaen University (to P.Y.).
