Abstract
We evaluated autoantibodies against nine tumor-associated antigens, including p62, p16, Koc, p53, Cyclin B1, Cyclin E, Survivin, HCC1, and RalA as serological markers in lung cancer. Enzyme-linked immunosorbent assay (ELISA) was used to detect autoantibodies in sera from 50 lung cancer patients and 42 normal controls. Then, four tumor-associated antigens of higher values were selected and validated in sera from validation group. Western blot and serum absorption test were used to confirm positive findings from ELISA. When cutoff values were set as mean optical density values plus 3 standard deviation of normal controls, the positive rate of autoantibodies against four tumor-associated antigens (Survivin, Cyclin B1, HCC1, and p53) reached 32%, 20%, 22%, and 18%, with area under the curve values of 0.653, 0.767, 0.622, and 0.623 in sera from 50 lung cancer, respectively (all p < 0.05). Results from the validation group confirmed the results. When lung cancer patients were divided by their clinicopathological characteristics into different subgroups, we have found that serum anti-Cyclin B1 and anti-HCC1 autoantibodies increased in stages 1, 2, and 3 lung cancer; anti-Survivin autoantibodies increased in stages 2 and 3 lung cancer; and anti-p53 autoantibody only increased in stage 1 when compared with their corresponding levels in controls (all p < 0.05). Serum anti-Cyclin B1 and anti-Survivin autoantibodies increased with disease histological grade 2 and 3 (both p < 0.05). And higher serum level of anti-p53 autoantibodies is positively associated with tumor size. Parallel utilization of these four anti-tumor-associated antigens (any positive) can increase sensitivity to 65.0% at 100% specificity with area under the curve of 0.908 (p < 0.001) in lung cancer detection in validation group. Our results suggest that autoantibodies against these four tumor-associated antigens have higher values in lung cancer detection, and serum anti-Cyclin-B1 has the potential to serve as novel non-invasive biomarkers in early-stage lung cancer.
Introduction
Although the incidence of lung cancer has slightly declined in recent years, it remains the leading cause of cancer death in the United States because of its aggressive and heterogeneous nature. 1 During the past few decades, results from two major lung cancer cohort studies showed that chest X-ray was not an effective test in lung cancer screening (Shizuoka cohort study, the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial).2,3 The National Cancer Institute (NCI)-sponsored, National Lung Screening Trial (NLST) found that low-dose chest computed tomography (LDCT) screening could reduce lung cancer–specific mortality in high-risk subjects. However, LDCT showed not to be an ideal screening tool in average risk population due to high false positivity, cost, and radiation exposure. 3 Late-stage lung cancers are typically larger in size or/and have lymph node infiltration or have distant metastasis. Cells from higher grade of lung cancer are less differentiated, and they tend to grow quickly and are more likely to spread. Treatment options are greatly influenced by lung cancer stage and grade.
Many studies have shown that certain cellular proteins from cancer tissues can be shed into humoral system and trigger the immune system to generate autoantibodies long before the patients get diagnosed.4–7 The cellular proteins which can induce immune responses vary greatly in types, including tumor-suppressor gene proteins such as p53 8 and p16,9,10 messenger RNA (mRNA) binding proteins such as IGF2BP2/p62,5,11 cell-cycle regulatory proteins such as Cyclin B1,6,7 apoptosis inhibitor protein such as Survivin,12,13 and other cancer-related proteins. Although the mechanism underlying is not completely understood, researchers have been interested in the utilization of autoantibodies as serological markers for cancer diagnosis.14–21
Studies from our group as well as other groups have demonstrated that autoantibodies against these tumor-associated antigens (TAAs) can be used as biomarkers in cancer detection.6,22–31 Early in 1995, Lubin et al. 6 have indicated that sera p53 antibodies could be used as p53 alteration marker before clinical presence of lung cancer. After that, antinuclear antibodies were found in sera from lung cancer patients and the authors suggested that such markers might lead to the discovery of diagnostic and prognostic markers due to the autoimmune feature of lung cancer. 22 However, the diagnostic value of single marker is relatively low. Researchers suggest that panels of circulating autoantibodies and their corresponding antigens might be of use in cancer detection.23–26 Many efforts have been put trying to validate the diagnostic value and predictive value of such biomarkers.27–29,31 However, none of these biomarkers showed sufficient sensitivity and specificity to make them ideal serum biomarkers for early detection of lung cancer. It is well known that accumulation of somatic mutations may contribute to the oncogenesis of cancer. Thus, using a panel of biomarkers that cover multiple cellular process, better diagnostic performance may be achieved.
In this study, we evaluate the diagnostic performance of nine TAAs, including p62, p16, Koc, p53, Cyclin B1, Cyclin E, Survivin, HCC1, and RalA, which were previously studied in our research group, in the detection of lung cancer. And we further analyze which TAAs are more effective in the early detection of lung cancer and whether parallel utilization of these four biomarkers (any positive) can increase diagnostic value in lung cancer detection.
Materials and methods
Serum samples
Sera from 50 lung cancer patients were provided by Lung Cancer Biospecimen Resource Network (University of Virginia, USA) which includes patients with different stages and different grades. Patients’ clinical characteristics are summarized in the “Results” section (Table 1). Sera from 42 normal healthy controls (NHCs) were obtained from the sera bank of Cancer Autoimmunity and Epidemiology Research Laboratory at the University of Texas, El Paso (UTEP), which were collected during annual health examinations. Sera from another 60 lung cancer patients (without clinical characteristic information) and 41 normal healthy individuals were also obtained from the same sera bank to serve as validation group. All cancer sera were collected at the time of cancer diagnosis and all lung cancer patients were confirmed by histopathological diagnosis. Due to regulations concerning studies of human subjects, the patients’ names were blinded to investigators. This study was approved by the Institutional Review Board of UTEP.
Relationship between serum level of autoantibodies against four TAAs and clinicopathological factors in human lung cancer.
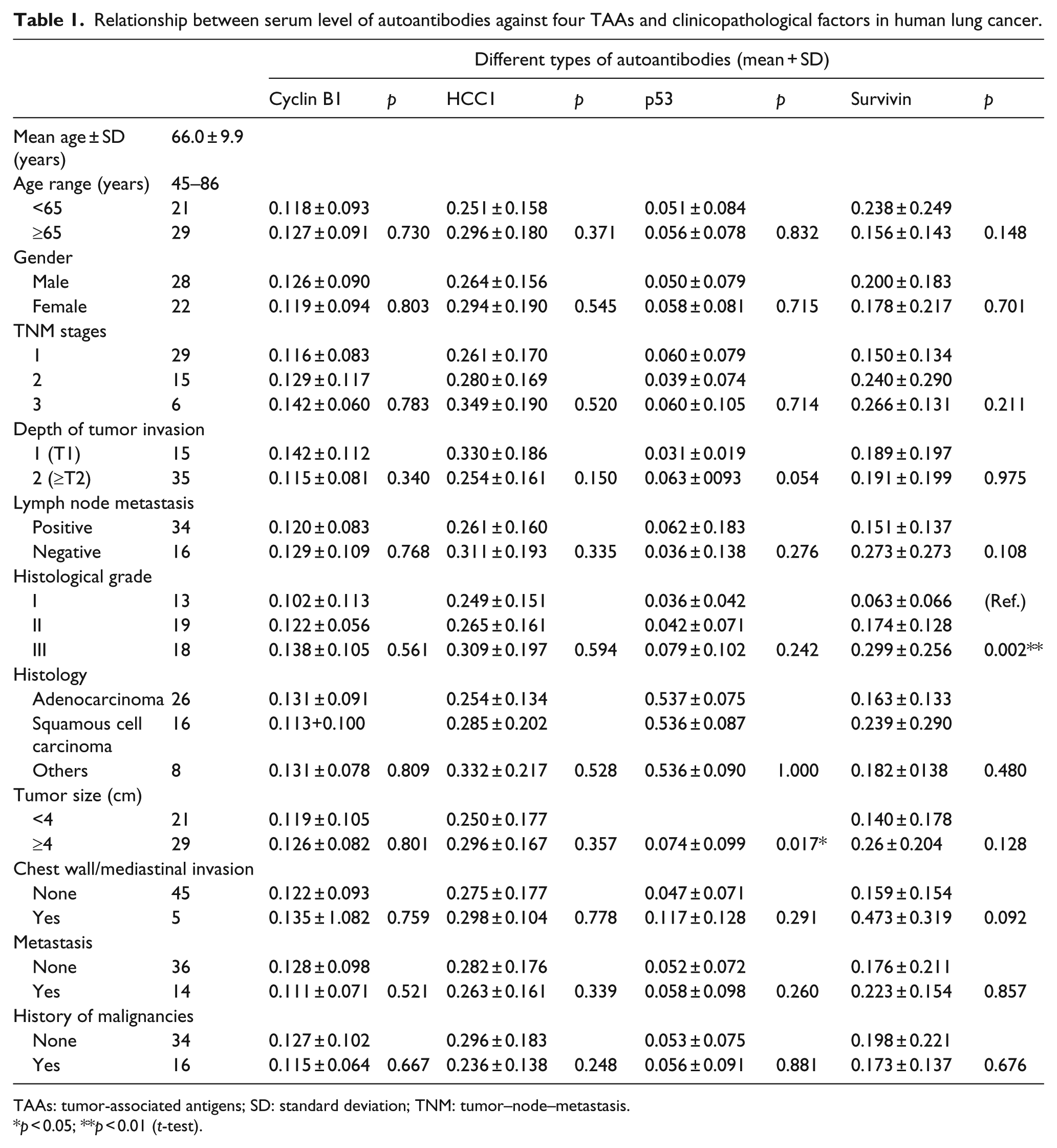
TAAs: tumor-associated antigens; SD: standard deviation; TNM: tumor–node–metastasis.
p < 0.05; **p < 0.01 (t-test).
Expression and purification of recombinant proteins
All TAAs used in this study, including Cyclin B1, Survivin, p53, HCC1, p62, p16, Koc, RalA, and Cyclin E, were constructed and purified as described in our previous studies.12,14
Enzyme-linked immunosorbent assay
Purified recombinant proteins were individually diluted in phosphate-buffered saline (PBS) to a final concentration of 1.0 µg/mL, and 50µL was pipetted into each well to coat onto 96-well enzyme-linked immunosorbent assay (ELISA) plates (Gibco, USA) overnight at 4°C. Human serum samples were diluted at 1:100, incubated with antigen-coated wells at 4°C overnight followed by washing with PBS containing 0.05% Tween-20 (PBST) three times, and then incubated at 37°C with horseradish peroxidase (HRP)-conjugated goat anti-human IgG or rabbit anti-human IgG (Caltag Laboratories, USA) as a secondary antibody diluted in anti-immunoglobulin diluent at 1:4000 for 1 h followed by washing with PBST. The solution of 2,2′-azino-bis(3-ethylbenzotjiazoline-6-sulfonic acid)diammonium salt (ABTS) was used as detecting agent. The optical density (OD) values of each well were read at 405 nm. Each sample was tested in duplicate well. The cutoff value for determining a positive reaction was designated as the mean OD value plus 3 standard deviations (SDs; (mean + 3 SD) of the control groups. In addition, all positive sera were confirmed with repeat testing, as were some negative sera. More detailed protocol of ELISA has been described previously.5,16
Western blot and serum absorption test
Positive serum samples that were determined to contain autoantibodies using ELISA were further tested by western blot to confirm the immunoreactivity to TAAs. In brief, 10 µg purified recombinant proteins (p62, p16, Koc, p53, Cyclin B1, Cyclin E, Survivin, HCC1, and RalA) were electrophoresed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a nitrocellulose membrane subsequently. After blocking in PBST with 5% nonfat milk for 2 h at room temperature, the NC membranes were cut into stripes and incubated overnight at 4°C with patient’s sera diluted at 1:100 and then incubated with HRP-conjugated goat anti-human IgG (Santa Cruz Biotechnology, Inc., USA) diluted at 1:10,000 for 60 min followed by washing with PBST solution. Positive signals were captured by autoradiography using chemiluminescence (Pierce Biotechnology, USA) per the manufacturer’s instructions. To confirm specificity of western blotting, sera at above dilutions were absorbed with recombinant proteins (final concentration: 0.03 µg/mL) overnight at 4°C, the mixture was centrifuged, and the supernatant was examined by western blot.
Statistical analysis
All data are presented as means ± SD, and unpaired two-tailed t-test or χ2 test was used to compare whether there was statistical difference between the two groups. Receiver operating characteristic (ROC) analysis was performed to illustrate the diagnostic performance of serum autoantibodies. The methods for calculating the positive and negative predictive values (NPVs) were based on the methodology provided in Epidemiology textbook. 17 Disease-free survival (DFS) curves and overall survival (OS) time were analyzed using the Kaplan–Meier method, and differences were examined using log-rank tests. All p values were two-sided, and those less than 0.05 were considered statistically significant. All statistical analyses were carried out using the IBM SPSS Statistics 21.0 (IBM Corp., USA).
Results
Frequency and area under the curves of autoantibodies against the panel of nine TAAs
Nine purified recombinant TAAs were used as coating antigens in ELISA to detect their corresponding autoantibodies in sera from lung cancer patients as well as normal controls in this study. As shown in Table 2, the frequency of autoantibodies against nine TAAs in sera from 50 patients with lung cancer ranges from 0% to 36%. The frequency of autoantibodies against Survivin, Cyclin B1, HCC1, and p53 ranks top four in the research group. Although the positive predictive values (PPVs) are relatively high for most TAAs, the NPVs are relatively low. The area under the curve (AUC) values were calculated to evaluate the diagnostic value of every TAA in an unbiased way, and the results are also shown in Table 2. The distribution of autoantibodies against these nine TAAs and their corresponding ROC curves are shown in Figure 1. Results from ROC curve analysis revealed that sera autoantibodies against Cyclin B1, Survivin, P53, and HCC1 were robust in discriminating lung cancer patients from normal controls with AUCs of 0.767, 0.653, 0.623, and 0.622, respectively (p < 0.05, Figure 1(f), (j), (i) and (b)). Since upregulated/overexpressed markers are easier to be detected and determined than downregulated markers, they are more suitable to serve as diagnostic markers. These data suggest that these four TAAs have better diagnostic value in lung cancer detection, and this needs to be further validated and evaluated. So, we choose these four TAAs to further validate and evaluate their diagnostic performance in a validation group.
Positive frequency and diagnostic values of nine anti-TAAs in research group.

Cutoff = mean + 3 SD; TAA: tumor-associated antigen; LC: lung cancer; NHC: normal health control; PPV: positive (+) predictive value = number of true positives/(number of true positive + number of false positives) × 100; NPV: negative (−) predictive value = number of true negative/(number of true negative + number of false negative) × 100%; YI: Youden index; *p < 0.05, **p < 0.01.
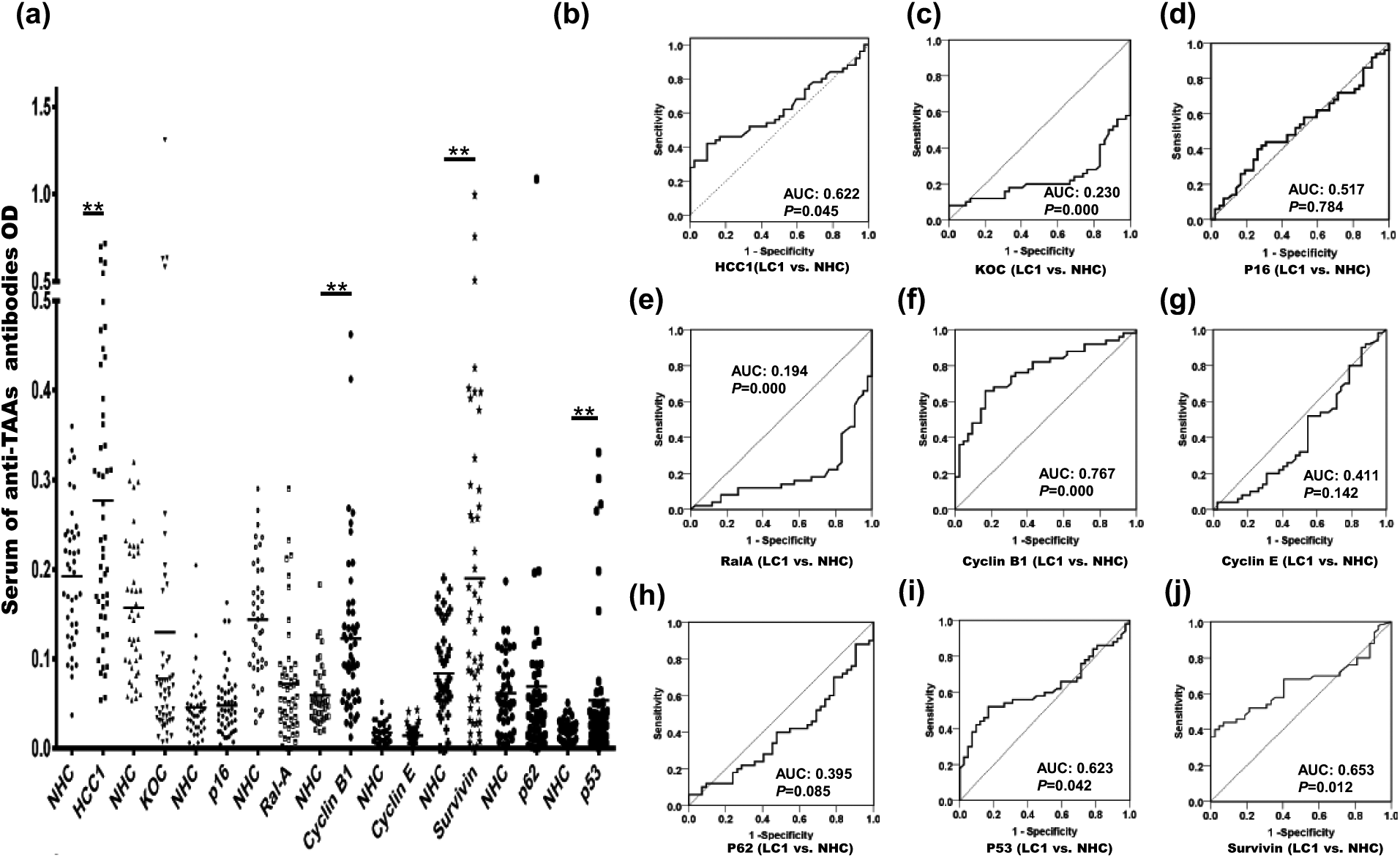
Distribution of nine anti-TAAs autoantibodies in research group and their corresponding ROC curves. (a) Different serum autoantibodies against TAAs distribution in NHC (n = 42) and lung cancer patients (n = 50); y-axis represents the OD value and dots represent serum samples (*p < 0.05; **p < 0.01). (b–j) ROC curve yielded by OD values of anti-TAAs autoantibodies in discriminating lung cancer patients (n = 50) from NHC (n = 42).
Reproducibility of serum anti-Cyclin B1, anti-Survivin, anti-p53, and anti-HCC1 autoantibodies as biomarkers in lung cancer
It is noteworthy that autoantibodies against these four TAAs are also significantly higher in lung cancer patients from validation group with positivity of 16.7% for anti-Cyclin B1, 26.7% for anti-Survivin, 41.7% for anti-p53, and 20% for anti-HCC1 (Table 3). Although the PPVs of autoantibodies against these TAAs have reached 100%, there is a slight drop for NPVs when these TAAs are used alone. ROC curves revealed that the AUC values of these four anti-TAAs are all higher than 0.7 (Figure 2(b)–(e); p < 0.001). When using these four anti-TAAs in conjunction, the AUC value has increased to 0.908, the NPV has increased to 59.6%, and the optimal sensitivity/specificity is 80%/90.3% (Table 3, Figure 2(f)).
Positive frequency and diagnostic values of four anti-TAAs in validation group.
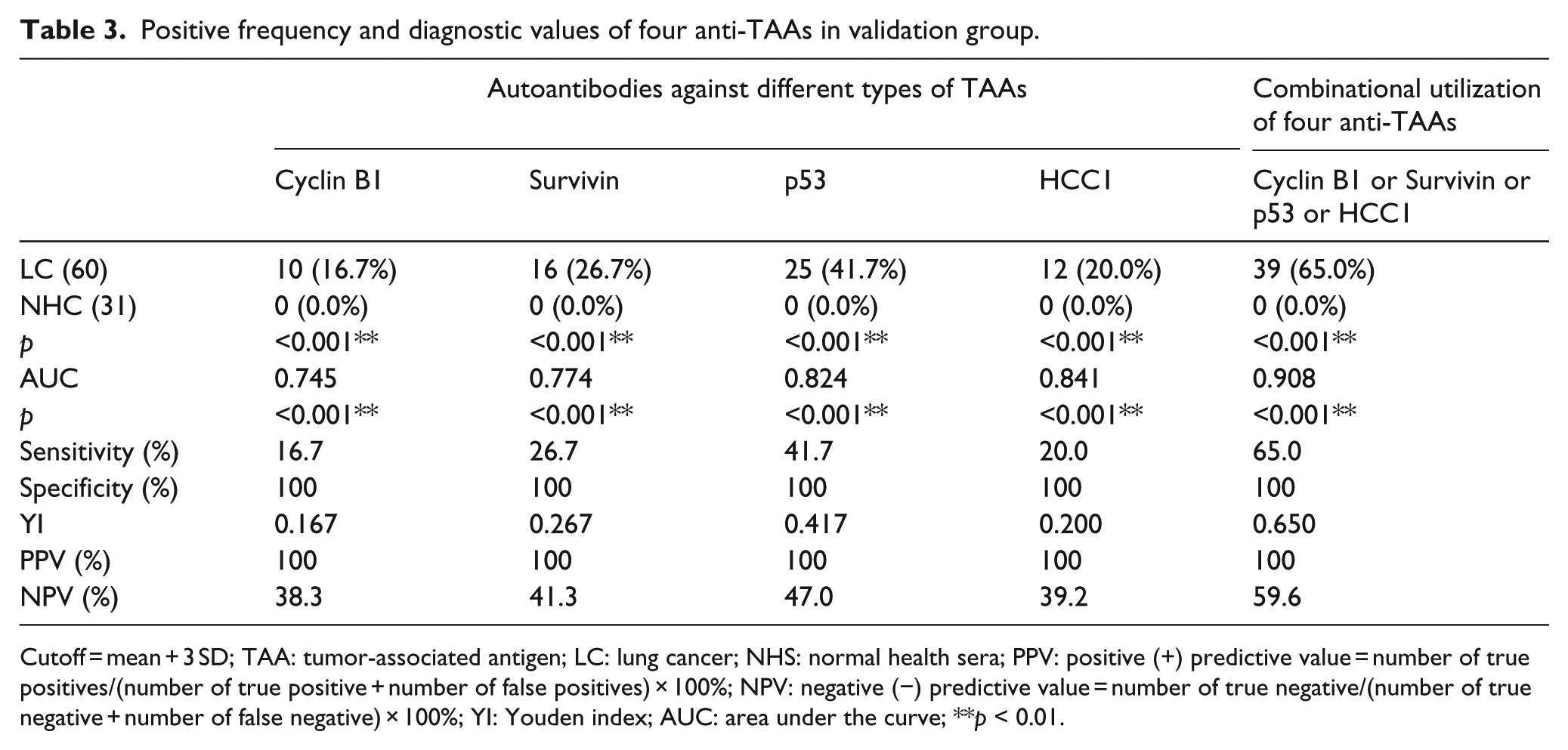
Cutoff = mean + 3 SD; TAA: tumor-associated antigen; LC: lung cancer; NHS: normal health sera; PPV: positive (+) predictive value = number of true positives/(number of true positive + number of false positives) × 100%; NPV: negative (−) predictive value = number of true negative/(number of true negative + number of false negative) × 100%; YI: Youden index; AUC: area under the curve; **p < 0.01.

Distribution of four anti-TAAs autoantibodies in validation group and their corresponding ROC curves. (a) Different serum anti-TAA autoantibody distribution in NHC (n = 31) and lung cancer patients (n = 60); y-axis represents the OD value and dots represent serum samples (*p < 0.05; **p < 0.01). (b–f) ROC curve yielded by OD values of anti-TAAs autoantibodies in discriminating lung cancer patients (n = 60) from NHC (n = 31).
Subgroup analysis of diagnostic value of serum anti-Cyclin B1, anti-Survivin, anti-p53, and anti-HCC1 autoantibodies in 50 lung cancer patients from research group
We have further divided 50 lung cancer cases from the research group into different subgroups according to different clinical parameters and tried to explore whether there are any correlations. For different stages, serum anti-Cyclin B1 and anti-HCC1 autoantibodies significantly increased in patients with stages 1, 2, and 3 lung cancer; Survivin autoantibodies significantly increased in patients with stages 2 and 3 lung cancer; and p53 autoantibody only increased in stage 1 lung cancer patients’ sera compared with their corresponding levels in NHCs (p < 0.05; Table 4). For different grades, sera anti-Cyclin B1 and anti-Survivin autoantibodies are significantly higher in patients with grades 2 and 3 lung cancer, and sera anti-p53 and anti-HCC1 autoantibodies showed only significant difference with grade 3 lung cancer (p < 0.05), compared with NHCs.
Subgroup comparisons of four serum anti-TAAs in NHC and lung cancer patients with different TNM stages and histological grades.
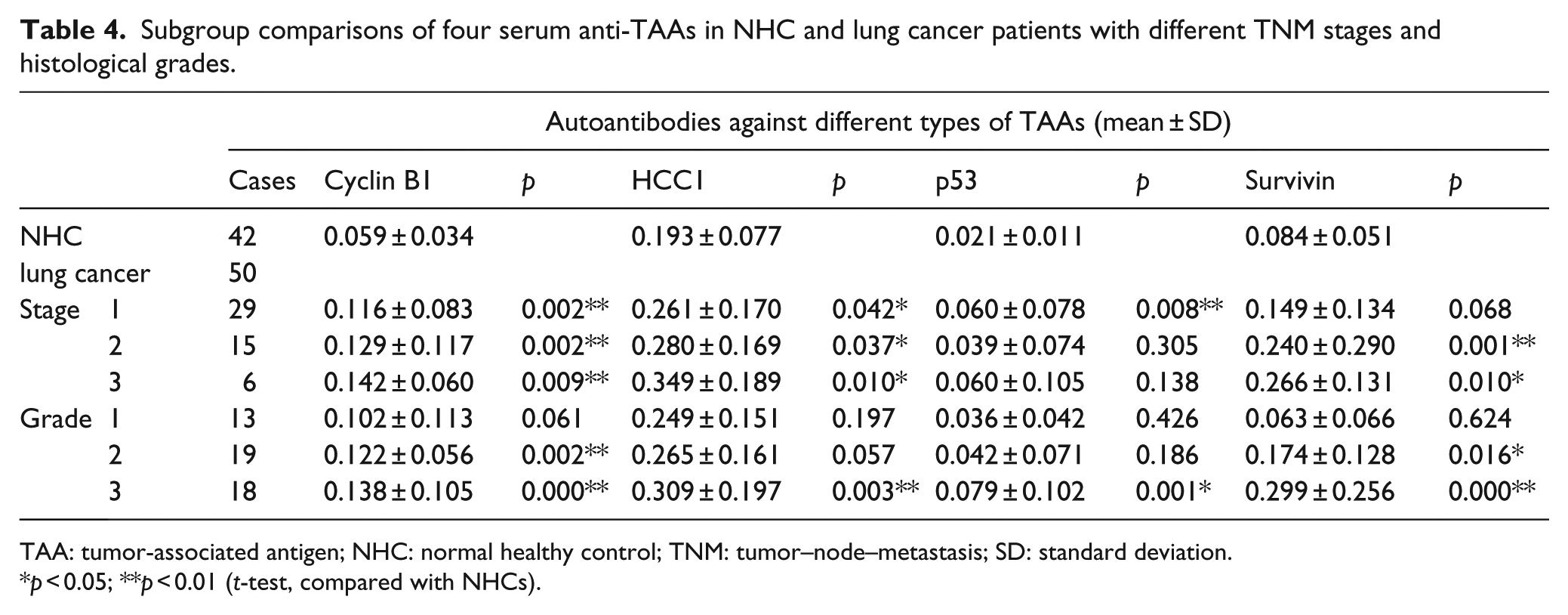
TAA: tumor-associated antigen; NHC: normal healthy control; TNM: tumor–node–metastasis; SD: standard deviation.
p < 0.05; **p < 0.01 (t-test, compared with NHCs).
We further generated ROC curves to check whether anti-Cyclin B1 or other autoantibodies could discriminate stage-specific lung cancer from NHCs. As shown in Figure 3, anti-Cyclin B1 and anti-p53 autoantibodies can discriminate stage 1 lung cancer from NHC with AUC values of 0.757 and 0.715, respectively (p < 0.01; Figure 3(e) and (g)), while anti-Survivin and anti-HCC1 autoantibodies could not discriminate stage 1 lung cancer from NHC (Figure 3(f) and (h)). Due to unavailability of the background information of lung cancer patients in the validation cohort, further analyses were not conducted.
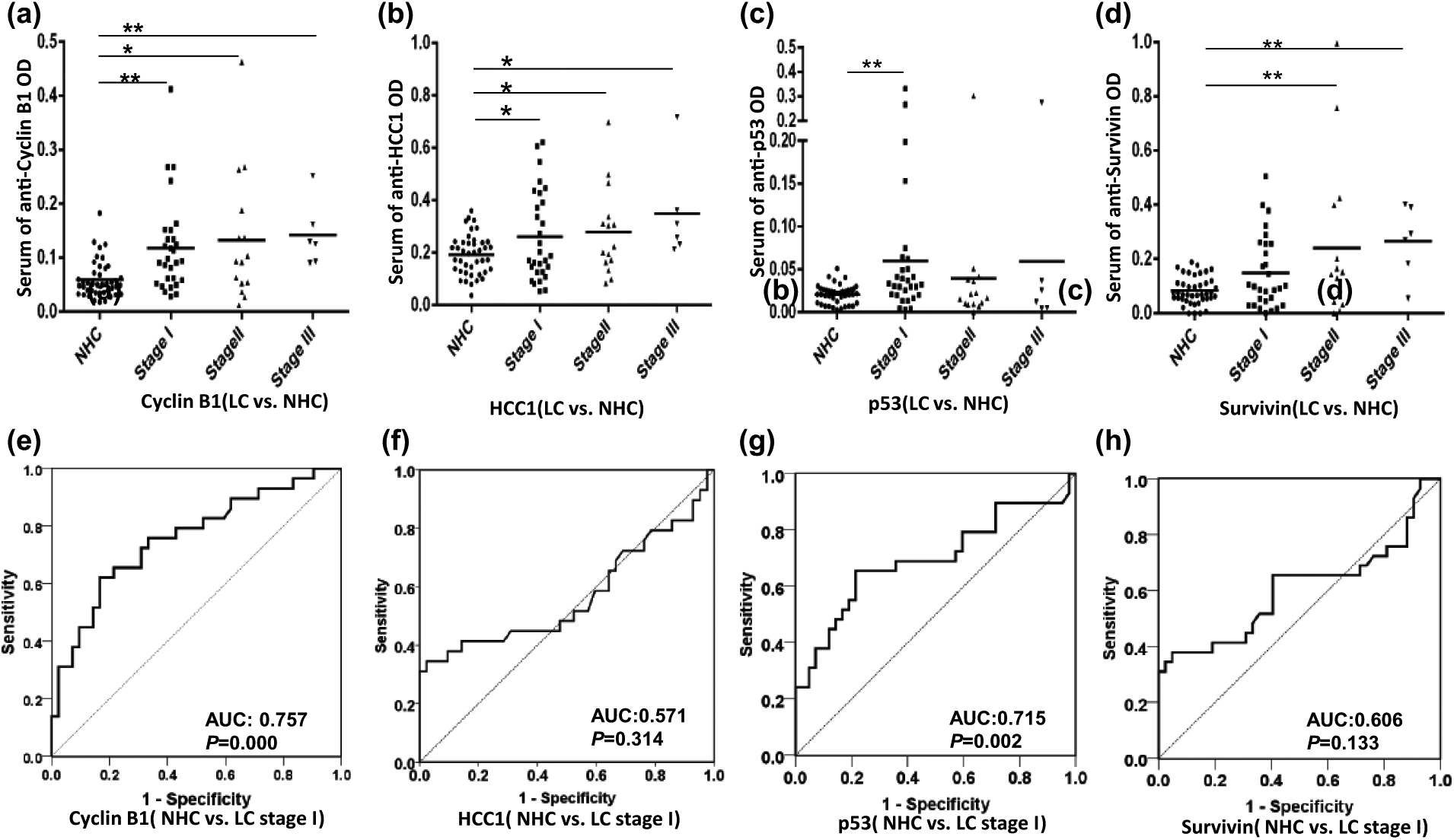
Distribution of four anti-TAAs autoantibodies in different stages of lung cancer and NHCs and their ROC curves distinguishing stage 1 lung cancer patients from NHCs; y-axis represents the OD value and the x-axis represents serum samples (*p < 0.05; **p < 0.01). (a–d) Different serum anti-TAA autoantibody distribution in NHC and patients with different TNM stages. (e–h) ROC curves yielded by OD values of different anti-TAAs autoantibodies in discriminating stage 1 lung cancer patients from NHC.
Next, we observed the relationship between autoantibodies’ response to Cyclin B1, HCC1, Survivin, and p53 with clinicopathological factors in human lung cancer. Table 1 illustrates that serum anti-p53 autoantibodies significantly increased in lung cancer patients with tumor size (p = 0.017). For other clinicopathological features, the results showed no significant differences. Kaplan–Meier survival analysis showed that there is no significant difference between serum level of four antibodies and DFS or OS.
Western blot and antibody absorption test confirmed the ELISA results
Representative serum samples were analyzed by western blotting to confirm the positive results from ELISA. Figure 4(a) shows that positive sera from ELISA results have strong reactivity with recombinant protein in western blot analysis. Figure 4(b) shows that after serum autoantibodies been neutralized by their corresponding TAAs, western blot reactions are significantly reduced. These results suggest that the positive findings from the ELISA are reliable.

Western blot and serum absorption test with representative sera from lung cancer patients and NHCs. (a) Lanes 1–10 are 10 representative lung cancer sera; lanes 11–15 are five representative normal human sera (NHS). (b) Antibody reactivity to Cyclin B1, HCC1, Survivin, and p53 before and after absorption with purified recombinant proteins. Lanes 1–2 are representative sera from LC patients.
Discussion
In the United States, lung cancer accounts for about 14% of all new cancer cases. Lung cancer is also the leading cause of cancer death both in men and women and contributes to about 25% of all cancer deaths. 32 During the past few decades, lung cancer incidence rate has slightly decreased due to the control of tobacco and smoking cessation program. 1 Intratumor diversity develops during the progression of cancer, so early diagnosis and treatment may improve the outcome of patients. 33 It is important that high specificity as well as high sensitivity are required for screening test to avoid false positivity and to improve accuracy.
As shown in our previous studies and studies from other groups, the frequency of autoantibodies against single TAA is still inadequate to make them as a good diagnostic biomarker. Previous studies have demonstrated that this drawback can be overcome by combinational utilization of several TAAs.34–36 And it is suggested that different type of cancer might have different profile of sera autoantibodies. In this study, we have measured serological autoantibodies against nine TAAs in two independent cohorts, which were previously confirmed with other types of cancers, including p62, p16, Koc, p53, Cyclin B1, Cyclin E, Survivin, HCC1, and RalA. These TAAs are involved in different cellular processes of cancer pathway, and therefore, they might hold the potential to pick up early-stage lung cancer. Cyclin B1 is a regulatory protein involved in mitosis and plays an important role in cell proliferation. 37 Higher level of Cyclin B1 in cancer tissues is associated with poorer prognosis of breast cancer and non-small-cell lung cancer.37,38 HCC1/RBM39 was identified as autoantigen with splicing factor motifs in hepatocellular carcinoma, 39 and studies have shown that this protein may promote breast cancer and lung cancer carcinogenesis.40,41 It plays a role in both steroid hormone receptor–mediated transcription and alternative splicing, and it is also a transcriptional coregulator of the viral oncoprotein v-Rel. 42 P53 acts as tumor-suppressor protein participating in cell-cycle arrest, apoptosis, senescence, and DNA repair (https://en.wikipedia.org/wiki/TP53). And p53 mutations are associated with poor prognosis with several types of human cancers. 43 Survivin plays a pivotal role in tumor cell survival due to its negative regulation of apoptosis or programmed cell death. 44 During the malignancy transformation of cancer cells, dysregulation or mutation of these proteins might happen and disrupt the normal cell cycle. In this study, we observed that serum level of anti-p53 autoantibodies significantly increased in lung cancer patients with larger tumor size, maybe due to the dysregulation of the cell-cycle arrest and apoptosis that resulted from the mutation and aberrant expression of p53 in cancer tissues.45–47
In this study, the frequency of autoantibodies against single TAAs ranges from to 0% to 32% in the research group when the cutoff value was set as mean OD values plus 3 SD in the control group. This is consistent with our previous findings that the positive frequency of autoantibodies against single TAA can vary greatly and usually are inadequate to make them diagnostic markers.14–19,21,48,49 AUC is independent of the choice of cutoff value, and AUC ranges from 0.194 to 0.764 in the research group. For diagnostic test, we usually chose markers that are upregulated because these types of markers are easier to be detected and determined. Therefore, we have chosen four TAAs, as were Cyclin B1, Survivin, p53, and HCC1 to further validate their diagnostic value in validation group. The frequency of autoantibodies against these four TAAs ranges from 16.7% to 41.7% and the AUC ranges from 0.747 to 0.841 in the validation cohort. The results further confirmed that anti-Cyclin B1, anti-Survivin, anti-p53, and anti-HCC1 autoantibodies have the potential to serve as biomarkers for lung cancer detection. To further check which of these four TAAs can detect early-stage lung cancer, we further generated ROC curves for stage-specific lung cancer versus normal controls in the research cohort. The results show that only anti-Cyclin B1 and anti-p53 autoantibodies have the potential to pick up stage 1 lung cancer, and anti-Cyclin B1 autoantibodies increase as lung cancer stage develops. This can be explained by the intratumor heterogeneity theory proposed by Gerlinger et al. 33 Cyclin B1 and p53 might locate in the trunk while the other seven TAAs might be located in the branch of the polygenetic tree. Therefore, autoantibodies against these two TAAs are more robust than the others in the immunodiagnosis of lung cancer. When autoantibodies against these four TAAs (Cyclin B1, Survivin, p53, and HCC1) were used in parallel, the sensitivity increased to 65.0% at 100% specificity with PPV of 100% and NPV of 59.6%. However, due to the stringent requirement of screening test, current panel is not fit to be as screening test for lung cancer since around 35% true lung cancer patients will not be picked up and more than 40% of individuals with negative results will need further tests to estimate their risk of lung cancer.
Based on above results, we suggest that a panel of TAAs that covers several different cellular biological processes might contribute to the detection of lung cancer, and sera anti-Cyclin-B1 antibody may have the potential to serve as novel non-invasive biomarkers in patients with lung cancer, especially in early-stage lung cancer. Although the results from this study are promising, further studies with larger sample size and clinical information for lung cancer patients as well as patients with other types of cancers are needed to validate the results reported in this study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant (SC1CA166016) from the National Institutes of Health (NIH); Program for New Century Excellent Talents in University, Ministry of Education, China (NCET-11-0949); and a grant from Greater East Cancer Center through MDHonors/WorldOne, London, UK. The authors would also like to thank the Border Biomedical Research Center (BBRC) Core facilities at The University of Texas at El Paso (UTEP) for their help, which were funded by NIH grant (5G12MD007592).
