Abstract
Circular RNAs are novel identified type of endogenous non-coding RNAs, which exert vital functions in human and animals. However, the in-depth role of circular RNAs in the progression of tumorigenesis, especially osteosarcoma, is still undefined. Our preliminary study had found that cir-GLI2 was significantly upregulated in osteosarcoma tissues compared to adjacent non-tumor tissue. Moreover, cir-GLI2 silencing could effectively suppress the proliferation, migration, and invasion capacity of osteosarcoma cells, indicating the tumor-promoting role. Besides, bioinformatics analysis and luciferase reporter assay predicted the direct binding to miR-125b-5p, which has been reported to function as a tumor suppressor in osteosarcoma. Furthermore, functional experiments validated that cir-GLI2 exerted the tumor-promoting effects on osteosarcoma cells via negatively targeting miR-125b-5p. In conclusion, our study demonstrated that cir-GLI2 acts as an oncogenic circular RNA in osteosarcoma genesis, providing a novel diagnostic and therapeutic target for osteosarcoma.
Introduction
Osteosarcoma (OS) is the most common primary bone tumor in children and adolescents with high morbidity rate and causes poor prognosis in teenager. 1 At present, the clinical commonly used treatments contain systemic chemotherapy and local control surgery. 2 Although many OS patients are cured by multi-agent chemotherapy and surgery, the overall survival rate of OS has not been observably improved and 5-year survival rate is still dissatisfied. 3 It is important to explore pivotal molecules or aberrant tumor-associated pathways to regulate the occurrence and development of OS and finally overcome it. On the basis of in-depth understanding of molecular mechanisms of OS tumorigenesis and progression, novel and more effective therapeutic targets are being discovered to improve the curative ratio and prognosis. 4
Circular RNAs (circRNAs) are a novel type of non-coding RNAs (ncRNAs) in biologic systems, which is generated from genomic transcripts by joining the 5′ and 3′ ends closed loops. 5 In human, circRNAs usually contain 1–5 exons and these circRNAs are generated from exons, which was called exonic circRNAs. Similarly, the circRNAs that generated from introns are named intronic circRNAs. 6 Presently, the circRNAs function remain unclear. One of the mainstream viewpoints is that circRNAs harbor multiple binding sites to sponge microRNAs and serve as competing endogenous RNA for microRNA. 7 For example, the Cdr1as and cir-ITCH contain binding sites for miR-7 and represses the activity.8,9
The circRNA GLI2 (circBase ID: hsa_circ_0056288; http://www.circbase.org) locates at chr2:121708818-121713006, and its corresponding gene symbol is GLI2. Cir-GLI2 is generated from chromosomal region 2q12.7 with spliced sequence length 389 derived from pre-mRNA of GLI2. In our previous microarray screening, we found cir-GLI2 is markedly over-expressed in OS tissue compared to adjacent normal tissue. The Gli transcription factors are primary transcriptional regulators that mediate the activation of Hedgehog (Hh) signaling. 10 In humans, the three Gli proteins, namely Gli1, Gli2, and Gli3, coordinate specific Hh responses in the cell by modulating gene expression. 11 Both Gli1 and Gli2 act as transcriptional activators in the response to Shh stimuli. 12 MiR-125b has been reported to suppress the proliferation and migration of OS cells and enhance chemosensitivity to cisplatin.13,14
In this study, we measured the expression of cir-GLI2 using quantitative reverse transcription polymerase chain reaction (qRT-PCR) and find that cir-GLI2 is upregulated in OS tissue and cell lines. Therefore, we hypothesize that cir-GLI2 participates in the mediation of OS tumorigenesis and progression and might act as a diagnostic value biomarker. Series of functional experiments are performed to validate our hypothesis and investigate the possible regulation mechanism.
Materials and methods
Tissue samples and ethics statement
A total of 23 cases of OS tissues and adjacent non-tumor tissues were collected from patients who underwent exairesis from March 2015 to November 2016 at Huaihe Hospital of Henan University. The age range is 11–35 years, and the average age is 21.6 years. After resection, the OS tumor and adjacent non-tumor tissues were collected within 10 min and immediately stored at −80 °C until use. The non-tumor tissues were more than 2 cm distant from the tumor edge. None of these patients received chemotherapy or radiotherapy before surgery. This study had got the approval from the Biomedical Ethics Committee of Huaihe Hospital of Henan University and written informed consent had obtained from each patient.
Cell lines and culture
OS cell lines (MG63, Saos-2, HOS, and U2-OS) were provided by the American Type Culture Collection (ATCC, Manassas, VA, USA) and cultured in RPMI 1640 medium (Gibco, USA) supplemented with 10% fetal bovine serum (FBS; Gibco Carlsbad, CA, USA). The normal human osteoplastic cell line (hFOB) and HEK293 were provided by the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China) and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) 1640 medium (Gibco) supplemented with 10% FBS (Gibco). The cultured environment was maintained at 37°C in 5% CO2.
RNA extraction and RNase R digestion
Total RNA was extracted from samples and cell lines using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s recommendations. Subsequently, the extracted RNA was quantified by a NanoDrop 2000 (Thermo Fisher Scientific, Waltham, MA, USA). Total RNA was incubated with RNase R (Epicentre, WI, Madison, USA) for 10 min at 37°C and the proportion was 1 mg RNA paired with three units of RNase. RNA was subsequently purified by phenol-chloroform extraction and reprecipitated in three volumes of ethanol.
Cell transfection
Cell transfection was performed in small interfering RNA sequence that was supplied and synthesized by GenePharma (Shanghai, China). The amplified cir-GLI2 flanking sequence was cloned into the lentiviral vector pLVX-IRES-neo (pLVX-cir-GLI2; Clontech Laboratories, Inc., San Francisco, CA, USA) according to the manufacturer’s instructions. The transfection was operated using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s procedure. The siRNA sequences were shown as follows: si-cir-GLI2-1, 5′-GUGGACACCAAAUUAACAAUU-3′; si-cir-GLI2-2, 5′-CCACCAAGCCAUCAGGAGAUU-3′; si-cir-GLI2-3, 5′-UACACCGCAGAGAUAGAAGUU-3′; and si-miR-125b-5p, 5′-AGCGGAUACGGACUGCGGGUUU-3′.
Real-time quantitative polymerase chain reaction (qRT-PCR)
Real-time quantitative PCR (qRT-PCR) was performed to detect the expression levels of circRNA and microRNAs. Total RNA was extracted from OS tissue and cells. The extracted RNA was quantified by a NanoDrop 2000 (Thermo Fisher Scientific). Then, complementary DNA (cDNA) was synthesized by RevertAid First-Strand cDNA Synthesis Kit (Thermo Fisher Scientific), and qRT-PCR was operated using SYBR Premix Ex Taq (TaKaRa, Dalian, China). Relative levels of gene expression were normalized to β-actin housekeeping genes and calculated using the 2−ΔΔCt method. All detections were performed in triplicate. The primers used in the study were displayed as follows: cir-GLI2, forward 5′-CAGGCGACGGTCAGCGTT-3′, reverse 5′-GGCAATGGCGACCGTTATAC-3′; miR-125b-5p, forward 5′-GCATCTACCAATCTGGTAGCGTG-3′, reverse 5′-GTGACGGGCAGTTGCGT-3′; and β-actin, forward 5′-GCATAAGGGGAGGTCTCATCT-3′, reverse 5′-TCGTTGTGTGAGCGCCCAAT-3′.
Luciferase reporter gene assay
The putative binding site of miR-125b-5p at 3′-untranslated region (UTR) of cir-GLI2 was subcloned into pGL3 luciferase promoter plasmid (Promega). Cells were transiently co-transfected with luciferase reporter plasmids (Promega) with Lipofectamine 2000 according to the manufacturer’s instructions. Whereafter, pGL3 vectors were co-transfected with miR-125b-5p mimics and negative control (NC) into HEK293 cells. After 36 h, Dual-Luciferase Reporter Gene Assay Kit (Promega) was used to detect the luciferase activities according to the manufacturer’s instructions. The relative firefly luciferase activities were normalized with the Renilla luciferase activities as a control.
Cell proliferation assay
Cell proliferation assay was performed and detected using Cell Counting Kit-8 (CCK-8; Dojindo). A total of 1 × 104 cells were cultured in 96-well plates. Subsequently, the cells were added with 10 mL CCK-8 solution at 37°C for 90 min, and the absorbance was assessed at 450 nm after incubation at 0, 24, 48, 72, and 96 h, respectively. All detections were performed in triplicate.
Colony formation assay
OS cell lines (MG63, Saos-2) were seeded in six-well plates at the density of 500 per well for 2 weeks. Subsequently, the colonies were fixed with 4% paraformaldehyde for 5 min and stained with 1% crystal violet for 10 min. Colonies were examined and counted under microscope. The assays were repeatedly performed in triplicate.
Flow cytometry and apoptosis
Apoptotic cells were detected by Annexin V/Dead Cell Apoptosis Kit (Invitrogen). Briefly, after centrifugation, the precipitate was washed by phosphate-buffered saline (PBS) three times. Then, cells were seeded in six-well plates at a density of 4 × 105 per well and starved in FBS-free medium for 12 h before treated with indicated agents for 48 h. Cells were incubated at room temperature for 15 min and then 400 µL of binding buffer was added. Finally, the apoptotic cells were performed using flow cytometric analyses (Attune, Darmstadt, Germany).
Transwell assay
The invasive and migratory cells were evaluated by Transwell chamber according to the manufacturer’s instructions. The 8-mm polycarbonate filters were coated with Matrigel Matrix (BD Biosciences, Franklin Lakes, NJ, USA). Then, 5 × 104 cells were seeded in the upper well of the migration chamber with 200 mL serum-free RPMI 1640 and 800 mL RPMI 1640 supplemented with 10% FBS were added to the lower chamber. After 24 h of incubation, the cells on the top were removed and the cells on the lower chamber were fixed in 4% paraformaldehyde for 30 min and stained with 0.1% crystal violet. The cell number was counted under microscope.
Statistical analysis
Statistical analyses were performed using SPSS version 19.0 (IBM SPSS Inc, Chicago, IL, USA) and GraphPad Prism version 6.0 (GraphPad Software, GraphPad Software Inc., La Jolla, CA, USA). Student’s t-test or one-way analysis of variance (ANOVA) test was used to evaluate the difference; p < 0.05 was considered to indicate a statistically significant result.
Results
Cir-GLI2 was upregulated in OS tissue and cells
In our previous study, we performed high-throughput sequencing and circRNA microarray assay to screen the circRNA expression profiles in OS tissue and discovered the distinctly upregulated cir-GLI2. In this study, we validated the expression of cir-GLI2 using qRT-PCR, indicating that cir-GLI2 expression was observably increased in OS tissue compared to adjacent non-cancerous tissue (Figure 1(a)). Besides, cir-GLI2 expression was upregulated in 82% (19/23) OS tissues compared with their adjacent non-tumor tissues (Figure 1(b)). In OS cell lines, the expression of cir-GLI2 was significantly increased in MG63 and Saos-2 cell lines among all OS cell lines (Figure 1(c)). The constructed plasmids expressing cir-GLI2 were transiently transfected into MG63 and Saos-2. Random and oligo-dT primers were used in the reverse transcription experiments. Because circRNAs were depleted in the poly-(A)-enriched samples, as shown in Figure 1(d), the relative expression level of linear GLI2 was significantly higher than that of circular GLI2 in MG63 and Saos-2 cell lines. RNase R assay showed that circRNA cir-GLI2 could powerfully resist RNase R treatment (Figure 1(e)).
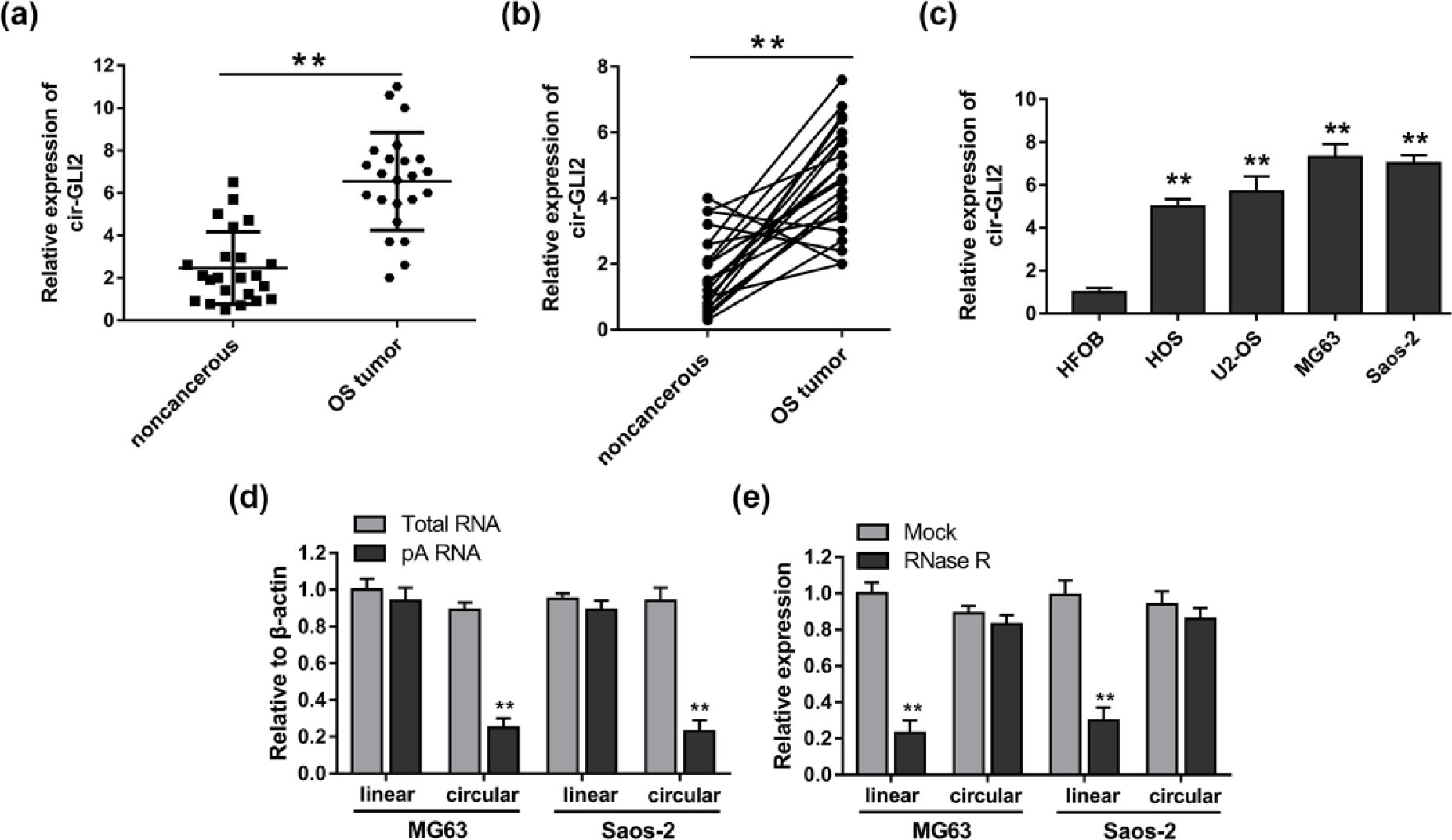
Expression of cir-GLI2 was upregulated in the OS tissues and cell lines. Besides, the covalent ring structure was verified. (a) Expression of cir-GLI2 was detected in the OS tissues and adjacent non-tumor tissues using qRT-PCR. (b) Expression of cir-GLI2 was upregulated in the 82% (19/23) OS tissues compared with their adjacent non-tumor tissues. (c) Overexpression of cir-GLI2 in OS cell lines (MG63, Saos-2, HOS, and U2-OS) compared to normal human osteoplastic cell line (HFOB). (d) Random primers and oligo-dT primers were applied in the reverse transcription experiments and the predicted circular RNA was absent in poly-A-enriched samples. (e) The predicted circular RNA was resistant to RNase R digestion. Data are presented as mean ± SD (*p < 0.05 and **p < 0.01 compared to HFOB or non-cancerous or mock or total RNA group).
Cir-GLI2 silencing suppressed the proliferation, migration, and invasion of OS cells
CircRNA GLI2 had been measured to be over-expressed in OS tumor tissue and cell lines, indicating the feasible oncogenic role in OS. Thus, we hypothesized that cir-GLI2 participated in the tumorigenesis of OS. To confirm the mediation of cir-GLI2 on the occurrence and progression of OS, cir-GLI2 silencing interfered by interfering RNAs was performed. Three siRNAs were designed and synthesized to knock down the expression of cir-GLI2, revealing different interference efficiency (Figure 2(a)). CCK-8 and colony formation assay illustrated that cir-GLI2 silencing suppressed the proliferation activity of OS cell lines (MG63 and Saos-2; Figure 2(b) and (c)). Flow cytometry showed that cir-GLI2 silencing observably increased the apoptotic cell rate (Figure 2(d)). Furthermore, Transwell assay demonstrated that the lower expression of cir-GLI2 inhibited the migratory and invasive capacity of OS cell lines (Figure 2(e) and (f)). Above results revealed that cir-GLI2 silencing could effectively suppress the proliferation, migration, and invasion of OS cells, indicating the definite tumor-promoting role in OS.

Cir-GLI2 silencing knocked with interference sequence significantly suppressed the proliferation, migration, and invasion of OS cells. (a) Three siRNAs (si-cir-GLI2-1, si-cir-GLI2-2, and si-cir-GLI2-3) were designed and synthesized to knock down the expression of cir-GLI2. (b) Proliferation activity of MG63 and Saos-2 measured by CCK-8. (c) Colony formation number counted after 48 h of transfection. (d) Apoptotic cell percentage of MG63 and Saos-2 using flow cytometry. (e) Representative images and statistical analysis of Transwell migration assay for MG63 and Saos-2 cells in vitro. (f) Representative images and statistical analysis of Transwell invasion assay. Data were calculated and presented as mean ± SD (*p < 0.05 and **p < 0.01 compared to si-NC group).
Cir-GLI2 sponged and interacted with miR-125b-5p
In the previous research, we had discovered the potential oncogenic function of upregulated cir-GLI2, which was validated by loss-of-function assays. Amount of reported researches had revealed that the major regulation pattern is competing endogenous RNA (ceRNA) and the mechanism had been widely verified within long non-coding RNA (lncRNA) and miRNAs. 15 We performed qRT-PCR to detect the differently expressed miRNAs in OS tissue to uncover the possible targets of cir-GLI2 (Figure 3(a)). Among the aberrated miRNAs, we predicted the specific miRNA targeting cir-GLI2 using bioinformatics analysis (starBase; http://starbase.sysu.edu.cn/panCancer.php). We focused on the obviously over-expressed miR-125b-5p, containing 10 complementary binding sites and exhibiting extremely high interaction score (Figure 3(b)). In MG63 and Saos-2 cells, dual-luciferase reporter assay showed that the co-transfection of pGL3-cir-GLI2 wild type and miR-125b-5p mimics distinctly decreases the luciferase activity compared to NC group (Figure 3(c)). Moreover, we assessed the expression of miR-125b-5p in 23 OS patients using qRT-PCR, indicating the remarkable lower expression of miR-125b-5p compared to adjacent non-tumorous tissue (Figure 3(d)). Besides, miR-125b-5p expression was downregulated in 78% (18/23) OS tissues compared to adjacent non-tumor tissues (Figure 3(e)). Above results powerfully suggested the negative targeting relationship between cir-GLI2 and miR-125b-5p.

Cir-GLI2 sponged and interacted with miR-125b-5p. (a) The variant expression of miRNAs in the OS tissue detected by qRT-PCR, containing miR-125b-5p. (b) Bioinformatics analysis predicted the putative complementary sites within miR-125b-5p and cir-GLI2. (c) Luciferase activity of wild-type or mutant-type cir-GLI2 3′-UTR reporter gene in MG63 and Saos-2 cells transfected with miR-125b-5p mimic. (d) qRT-PCR analysis of the expression of miR-125b-5p in 23 OS patients. (e) Expression of miR-125b-5p was downregulated in the 78% (18/23) OS tissues compared with their adjacent non-tumor tissues. Data were calculated and presented as mean ± SD (*p < 0.05 and **p < 0.01 compared with the corresponding control group).
The carcinogenic regulation of cir-GLI2 on OS cells targeting miR-125b-5p
To investigate and validate the ceRNA regulation of cir-GLI2 on OS cells via targeting miR-125b-5p, interference oligonucleotide and/or mimics sequences were transfected into MG63 cell line and functional experiments were performed. The expression of miR-125b-5p was significantly decreased in OS cell lines (Figure 4(a)). Besides, cir-GLI2 silencing could upregulate the expression of miR-125b-5p (Figure 4(b)). CCK-8 and colony formation assay showed that miR-125b-5p mimics inhibited the proliferation and colony formation capacity and miR-125b-5p inhibitor promoted it, while si-cir-GLI2 reversed the inhibition of miR-125b-5p mimics (Figure 4(c) and (d)). Afterwards, flow cytometry showed that miR-125b-5p inhibitor suppressed the apoptosis of MG63 cells, which was rescued by si-cir-GLI2 (Figure 4(e) and (f)). Similarly, migration and invasion assay showed that miR-125b-5p mimics inhibited the migration and invasion ability and miR-125b-5p inhibitor promoted them; however, si-cir-GLI2 reversed the inhibition of miR-125b-5p mimics (Figure 4(g) and (h)). Series of functional experiments demonstrated that cir-GLI2 acted as ceRNA and exerted oncogenic role on OS cells via negatively targeting miR-125b-5p.

Cir-GLI2 mediated OS cell proliferation, apoptosis, migration, and invasion by targeting miR-125b-5p in MG63 cell line in vitro. (a) Expression of miR-125b-5p in OS cell lines. (b) Expression of miR-125b-5p was detected in multiple treated groups. (c) Cell proliferation viability of OS cells detected by CCK-8 assay. (d) Cell colony formation assay. (e and f) Cell apoptosis was detected by flow cytometry. (g) Migration capacity was detected by Transwell assays. (h) Invasion ability was detected by Transwell assays. Data are presented as mean ± SD (*p < 0.05 and **p < 0.01 compared to control group).
Discussion
In this study, we uncover that circRNA cir-GLI2 is upregulated in the OS tissue, as well as OS cell lines, and exerts oncogenic role in OS tumorigenesis. Besides, we also illustrate one of the potential regulating mechanisms of cir-GLI2 is that cir-GLI2 negatively regulates the expression of miR-125b-5p, known as a tumor suppressor, to motivate the OS occurrence.
In other tumor research areas, circRNAs have been incrementally uncovered and reported depending on high-throughput sequencing and microarray analysis. 16 Moreover, the biological functions of huge quantity of newfound circRNAs need to be verified in subsequent studies. Our previous study has screened the circRNA expression profiles in OS tissue compared to adjacent normal tissues using circRNA microarray. A mass of aberrantly expressed circRNAs have been discovered in OS tissue, and one of observably over-expressed circRNA, cir-GLI2, was arisen after filtering. CircRNA cir-GLI2, derived from pre-mRNA of GLI2, locates at chr2:121708818-121713006, and its corresponding gene symbol is GLI2. RNase R assay showed that cir-GLI2 could powerfully resist RNase R treatment, indicating the covalently closed continuous loop construction. We constructed three interference sequences to knock down the expression of cir-GLI2 and found that cir-GLI2 silencing could effectively suppress the proliferation, migration, and invasion of OS cells, revealing the definite tumor-promoting role in OS.
CircRNA is a type of RNA characterized with covalently closed continuous loop, which has been detected in viruses, plant, and animals. 17 Recently, the unique characterizations and functions of circRNA have caught public attention again. More and more researches have focused on the potential functions of circRNAs, one of which was demonstrated to be miRNA sponges. 18 The best-known circRNA is Cdr1as, which acts as miR-7 sponge to regulate myocardial infarction, 9 hepatocellular carcinoma (HCC) cell proliferation and invasion, 19 and insulin secretion. 20 In colorectal cancer, hsa_circ_001569 is identified as a sponge of miR-145 to mediate the proliferation and invasion. 21
MiR-125b-5p has been predicted and validated as target of cir-GLI2 using bioinformatics analysis and luciferase reporter assay. Admittedly, similar to lncRNAs, circRNAs could function as miRNA sponge to modulate the transcription and post-transcription regulation. 18 To explore the in-depth regulatory mechanism within cir-GLI2 and miR-125b-5p, we performed functional experiments to test the interaction of them on OS cell line proliferation, apoptosis, and invasion, demonstrating that cir-GLI2 acted as ceRNA and exerted oncogenic role on OS cells via negatively targeting miR-125b-5p. Nevertheless, due to the specific configuration and ambiguous function, the more deep-going functional mechanism of cir-GLI2 involved in the OS tumorigenesis is still inconclusive.
Additionally, high stability, abundance, and tissue-specific expression make circRNA sponges very effective for clinical research. 18 Hsa_circ_002059 and hsa_circ_0000190 were found to be significantly dysregulated in gastric cancer tissues, being accordant with plasma level, providing a diagnostic biomarker for gastric cancer.22,23 The levels of hsa_circ_0001649 and hsa_circ_0005075 showed significantly different HCC tissues and could act as a potential HCC biomarker.24,25 Our study reveals that cir-GLI2 sponged miR-125b-5p to function as tumor promoter in OS tissue, indicating a valuable diagnostic biomarker and a novel insight for the occurrence and progression of OS.
In conclusion, our study preliminarily investigates the regulating role of cir-GLI2, acting as ceRNA of miR-125b-5p, in OS tumorigenesis. Obviously, the research prospect of circRNAs on OS is expansive and their in-depth biological regulation remains to be further elucidated.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Basic Medical Research Center and Department of Orthopaedics of Huaihe Hospital of Henan University.
