Abstract
Periostin is an extracellular matrix protein involved in tumorigenesis and metastasis. However, the role of serum periostin as a surrogate marker for treatment efficacy is still unknown. In 122 advanced non–small cell lung cancer cases, 37 patients with benign lung disease and 40 healthy controls, serum periostin was measured by enzyme-linked immunosorbent assays. The associations of serum periostin levels with the clinic-pathological parameters, chemotherapy response, and clinical outcomes of non–small cell lung cancer patients were analyzed. Serum periostin levels were significantly higher in non–small cell lung cancer patients, and it was related significantly to bone metastasis (p = 0.021). Serum periostin of 65 non–small cell lung cancer patients were detected before and after two cycles of chemotherapy. The patients with and without periostin response had significant difference in objective response to chemotherapy (p = 0.001). For the 122 non–small cell lung cancer patients, the median progression-free survival was 5 months. In a multivariate analysis, performance status (hazard ratio, 1.71; 95% confidence interval, 1.10–2.67), baseline periostin (hazard ratio, 1.01; 95% confidence interval, 1.00–1.01), and periostin response (hazard ratio, 0.50; 95% confidence interval, 0.29–0.86) were significantly correlated with prognosis. In conclusion, serum periostin was elevated in advanced non–small cell lung cancer patients. Baseline periostin and periostin responses appeared to be reliable surrogate markers to predict chemotherapy response and survival in patients with advanced non–small cell lung cancer.
Introduction
Non–small cell lung cancer (NSCLC) is the leading cause of cancer-related mortality worldwide. 1 The majority of patients present with advanced-stage disease because of the lack of early symptoms and specific clinical manifestations. In recent years, treatment options have improved greatly, especially for patients who carry sensitive mutations in the epidermal growth factor receptor or the echinoderm microtubule-associated protein-like 4–anaplastic lymphoma kinase (EML4-ALK) translocation. 2 Nonetheless, platinum-based doublet chemotherapy still remains the cornerstone of the treatment for advanced-stage patients, and the 5-year survival rate is still less than 15%. 3 It is very important to adjust the treatment regimen according to the patient’s response. Response Evaluation Criteria in Solid Tumors (RECIST) based on radiologic detections are widely used to assess objective response (OR) after systemic therapy. 4 However, potential and early metastases cannot be easily detected by imaging data. Furthermore, we still have some NSCLC patients who have no measurable lesions. Thus, more studies exploring the potential role of tumor markers in predicting chemotherapy response and survival in patients with NSCLC are in great need.
Periostin is an extracellular matrix (ECM) N-glycoprotein of 93 kDa. Over-expression of periostin protein and/or messenger RNA (mRNA) has been detected in different types of malignancies, including colorectal cancer (CRC), 5 ovarian cancer, 6 breast cancer, 7 prostate cancer, 8 pancreatic cancer, 9 and NSCLC. 10 Accumulating evidences indicate that over-expression of periostin in tissue or serum is closely correlated with tumorigenesis and metastasis. 11 Previous studies on the use of serum periostin levels as a biomarker in patients with malignant mainly put the emphasis on diagnosis and prognosis.10,12–14 There are few researches focused on the relationship between periostin level in serum and chemotherapy response.
In this study, we detected periostin levels in blood specimens from patients with advanced NSCLC before and after chemotherapy using the enzyme-linked immunosorbent method. In addition, we investigated the correlations between periostin expression levels and clinic-pathological parameters as well as subsequently evaluated whether it can be used as surrogate marker to predict tumor response and survival in patients with advanced NSCLC.
Materials and method
Patient enrollment
Serum samples from three different groups of patients were collected. The first group was a cohort of 122 advanced NSCLC patients. It was intended that these patients with previously untreated stage III-IV NSCLC should receive at least two cycles of standard first-line combination chemotherapy. They were enrolled prospectively into this study at a single institution (Jinling Hospital, Nanjing, China) between March 2011 and March 2012. To be eligible for first-line combination chemotherapy, patients were required to have cytologically or histologically proven NSCLC; unresectable stage III-IV; measurable lesions; an Eastern Cooperative Oncology Group (ECOG) performance status (PS) from 0 to 2; normal hepatic, renal, and hematologic function; and no serious concomitant disease. The second group consisted of 37 patients with benign lung disease who were hospitalized at the same period, involving pneumonia (n = 20), tuberculosis (n = 8), tuberculosis pleural effusion (n = 2), chronic obstructive pulmonary diseases (n = 2), tuberculous pleuritis (n = 2), acute bronchitis (n = 1), inflammatory pseudotumor (n = 1), and allergic alveolitis (n = 1). All cases were diagnosed as benign lung disease according to clinical symptom, chest computerized tomography scans, and histological examination. The third group comprised healthy adult volunteers who came to our hospital for health examination at the same period (n = 40), who had no history of any malignancies and were confirmed to have no lung disease by chest X-ray.
All procedures were approved by the Medical Ethical Committee of Jinling Hospital, and written informed consent was obtained from all participants before obtaining the serum samples.
Sample collection
Serum samples were obtained immediately before the start and the third chemotherapy course. A volume of 5 mL peripheral blood was collected from each patient on the day of admission. The blood samples were allowed to stand in 4°C refrigerator for 2 h. In the presence of clot activator, serum was isolated subsequently after centrifugation at 3,000 rpm for 10 min. All serum samples were divided into aliquots and stored at −80°C until use.
Enzyme-linked immunosorbent assay examination
The concentrations of periostin in the serum samples were measured with an enzyme-linked immunosorbent assay (ELISA) kit (USCN Life Science Inc, Wuhan, China) according to the manufacturer’s instructions. In brief, 100 µL of serum sample (1:40 diluted) was added into each well, coated with mouse anti-human periostin monoclonal antibody, and incubated for 2 h at 37°C. Excess serum was then washed off and the periostin conjugate was added into the wells, followed by coloring the subjects. The absorbance was read immediately by an Elx808 Ultra Microplate Reader (Bio-Tek Inc., Winooski, VT, USA) using 450 nm as the primary wavelength and optionally 620 nm as the reference wavelength.
Evaluation of chemotherapy response
Eligible patients were required to receive at least two cycles of platinum-based chemotherapy. Tumor response was assessed according to the RECIST criteria (Version 1.1). In this study, patients who achieved a complete response (CR) or partial response (PR) were classified with OR, and all remaining patients (stable disease (SD) and progressive disease (PD)) were considered as non-responders.
Patients follow-up
Progression-free survival (PFS) is defined as the time from diagnosis to documented progression or death. For patients without a PFS-reported event, the date of the last documented evaluation was censored.
Statistical analysis
The statistical tests used included Kolmogorov–Smirnov test, variance analysis test, independent-sample t test, paired-samples t test, chi-square test, Pearson correlation method, logistic regression models, Kaplan–Meier survival analysis, and Cox’s proportional hazards model. To find the cutoff value of periostin and to evaluate and compare the performance of periostin decrease in predicting response to therapy, receiver operating characteristic (ROC) curves were constructed.
All calculations were performed with SPSS software, version 17.0 (SPSS Inc., Chicago, IL, USA). All comparisons were two-tailed, and p values of <0.05 were considered to be significant.
Results
Measurement of baseline periostin levels
The characteristics of patients with advanced NSCLC are summarized in Table 1. There were significant differences between the overall mean of the three groups (one-way analysis of variance (ANOVA), p < 0.001). The baseline serum periostin levels were significantly higher in patients with advanced NSCLC (552.37 ± 135.67 ng/mL) than in those with benign lung diseases (428.45 ± 121.95 ng/mL, p < 0.001) and in healthy controls (299.29 ± 42.32 ng/mL, p < 0.001). Patients with benign lung diseases had higher periostin levels than healthy controls (p < 0.001; Table 2).
NSCLC patients characteristics.
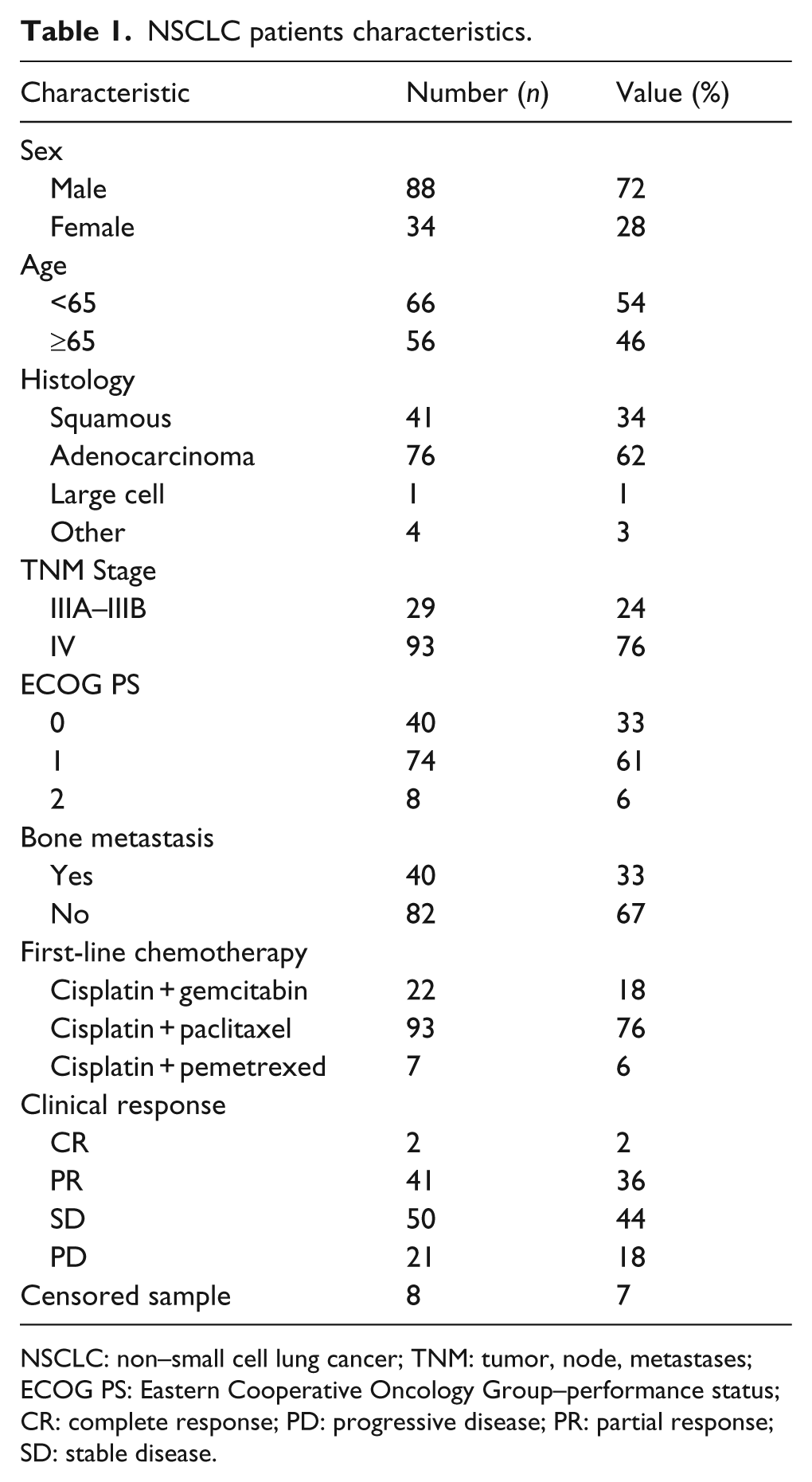
NSCLC: non–small cell lung cancer; TNM: tumor, node, metastases; ECOG PS: Eastern Cooperative Oncology Group–performance status; CR: complete response; PD: progressive disease; PR: partial response; SD: stable disease.
Serum periostin level in patients.

One-way analysis of variance (ANOVA).
Baseline serum levels of periostin were significantly related to bone metastasis (independent-samples t test, p = 0.021). Conversely, no association was observed between pretreatment serum periostin levels and gender (p = 0.151), age (p = 0.205), histology (p = 0.401), or stage of cancer (p = 0.499).
An ROC curve analysis was performed to determine the values to distinguish patients with malignancy from patients with benign lesions and normal controls. For baseline periostin (Figure 1), the area under the ROC curve (AUC) is 0.87 (95% confidence interval (CI), 0.81–0.92). The best efficacy (combination between sensitivity and specificity) was observed at 435.04 ng/mL, with a sensitivity of 84% and a specificity of 78% (positive predictive value (PPV), 86%; negative predictive value (NPV), 75%).

Receiver-operating characteristic curves for serum periostin level to distinguish malignant from benign and normal groups.
Association between periostin levels and response rate
Of 122 NSCLC patients, 65 could be evaluated both for serum markers response and for efficacy assessment after the two cycles of chemotherapy. The baseline serum periostin levels of this group were 550.27 ± 145.15 ng/mL. After treatment, serum periostin levels decreased significantly compared with baseline levels (p < 0.001). The mean serum concentrations of periostin decreased to 496.28 ± 142.74 ng/mL before the third course of chemotherapy. The median reduction in periostin was 15% (7% in non-responders and 18.5% in responders; p < 0.05).
An ROC curve analysis was also performed to assess whether a decrease of periostin would predict an OR to chemotherapy (Figure 2). The AUC is 0.74 (95% CI, 0.62–0.86). And the best efficacy was observed for a 14.5% reduction, with a sensitivity of 60% and a specificity of 80%. Based on the ROC curve analysis, a post-treatment 14.5% reduction in serum periostin was used as cutoff value for defining a response. Overall, a periostin response occurs in 25 patients (38%; Table 3). A periostin response was achieved in 18 of 30 patients (60%) who have an OR and in 7 of 35 patients (20%) without an OR (p = 0.001). When the analysis was focused only on patients who had pretreatment serum marker levels greater than the cutoff limit, an OR was observed in 16 of 25 patients (64%) who had a periostin response and in only 7 of 29 patients (24%) who did not have a marker response (p = 0.003). Logistic analysis confirmed the correlation between periostin response and efficacy of chemotherapy (odds ratios, 7.57; 95% CI, 1.98–28.89; p = 0.003; logistic regression analysis). Factors of age, gender, histology, disease stage, and smoking did not predict an OR to treatment.

Receiver-operating characteristic curves for the cutoff value of periostin deduction rate to predict the effect of chemotherapy.
Marker response and radiologic objective response.
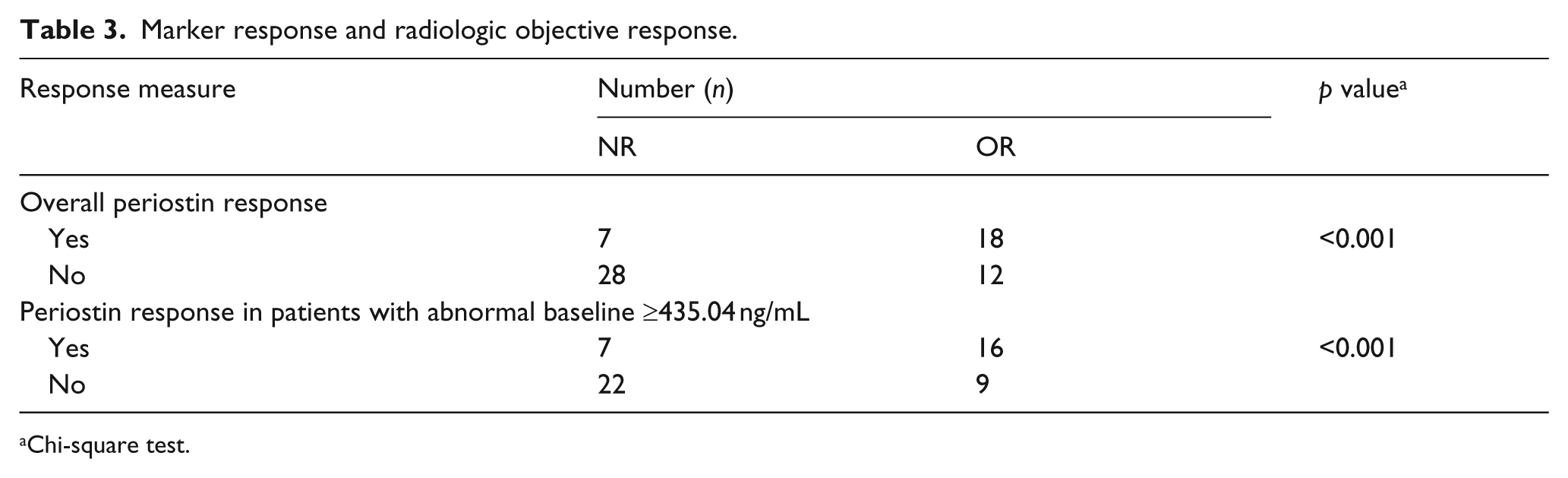
Chi-square test.
Survival analysis according to periostin levels
For the 122 NSCLC patients, 8 patients were lost to follow-up—the median PFS from the landmark date was 5 months. Patients with a baseline periostin level less than 435.04 ng/mL had better prognosis than patients with higher baseline levels (7 months vs 4 months, p = 0.001; log-rank test). Patients who experience a reduction of more than 14.5% in serum periostin levels had longer survival than patients without marker response (8 months vs 5 months, p = 0.044; log-rank test). The landmark survival curves for baseline periostin and periostin response were shown in Figure 3.

Survival of patients stratified according to (a) baseline periostin levels and (b) periostin response to chemotherapy.
Results from the Cox regression analysis were shown in Table 4. The independent prognostic roles of PS (hazard ratio (HR), 1.71; 95% CI, 1.10–2.67), baseline periostin (HR, 1.01; 95% CI, 1.00–1.01), and periostin response (HR, 0.50; 95% CI, 0.29–0.86) were confirmed.
Survival assessment of patients according to several variables.

Cox’s proportional hazards model.
p < 0.05.
Discussion
Recent studies have shown that tumor development was not only influenced by the epithelium or stroma but also impacted by the tumor-host interface micro-environment. 15 The ECM is an important component of tissue micro-environment. The cross talk between cancer cells and ECM proteins can create a supportive micro-environment for the initiation and growth of primary tumor and for the metastatic dissemination of tumors to distant secondary sites.
Periostin was originally found in mouse osteoblasts, which could regulate cell adhesion and the differentiation function of osteoblasts 16 and was considered as an ECM protein. This matricellular protein was involved in regulating intercellular adhesion via an interaction with itself and other ECM protein, such as collagen I, fibronectin, and tenascin C. 17 Periostin was involved in the pathobiology of various diseases, including formation and maintenance of dental tissues, 18 skin-related diseases, 19 valvular heart diseases, 20 and wound healing response. 21 An increasing number of studies have shown that periostin over-expression presents in various malignant tumors and closely relates with disease progression. In vitro and in vivo researches demonstrated that periostin participate in key processes in cancer metastasis such as cancer stem cell maintenance, 22 tumor cell survival, 5 protumoral macrophages recruitment, 23 pre-metastatic niche formation, 24 epithelial mesenchymal transition (EMT), 25 and angiogenesis. 26
Increased periostin levels have been detected in serum of patients with some malignant tumors. Ben et al. 27 found that the serum levels of periostin in CRC patients were significantly higher than that in healthy volunteers and benign colorectal polyps or adenomas. Elevated preoperative serum levels of periostin in CRC patients were correlated with distant metastasis and advanced-stage disease. In NSCLC patients, Sasaki et al. 28 found that serum periostin levels had decreased significantly 4 weeks after the resection of the NSCLC tumor. Our previous study have found that, compared with a normal control group, periostin levels were significantly higher in the serum of NSCLC cases. 10 Periostin is not just expressed in tumor tissues, it could also be found in a variety of acute and chronic inflammatory diseases, 29 even in normal tissues. Baril et al. 30 demonstrated serum periostin levels were significantly elevated in patients with pancreatic ductal adenocarcinoma (PDAC) and enable distinction between PDAC and control group that includes patients with various inflammatory diseases as well as healthy volunteers. However, limit information is available on the serum expression of periostin in NSCLC and benign lung diseases. In the present study, we expanded the sample size of NSCLC patients and supplemented with benign lung disease group. We found that the baseline serum periostin levels are significantly higher in patients with advanced NSCLC and enable distinction between NSCLC and other benign lung diseases and healthy controls. Baseline serum levels of periostin were significantly related to bone metastasis. These were in accordance with previous research, which suggested that over-expression of serum periostin may be a novel serodiagnostic marker for malignancies 31 and a sign of bone metastasis. 32
There were few studies focused on periostin expression and efficacy of chemotherapy. Ryner et al. 33 declared that high periostin expression level predicted shorter PFS following first-line chemotherapy in patients with epithelial ovarian cancer. While Sung et al. 6 demonstrated that patients with high level of stromal periostin tend to have higher percentage of cisplatin resistance than those with lower level of stromal periostin. The role of serum periostin as a surrogate marker for treatment efficacy has not been investigated before. In our study, we have shown that serum periostin levels decreased significantly after two cycles of chemotherapy compared with baseline levels. A strong point of our research is that we found obvious differences in chemotherapeutic efficacy between patients with or without periostin responses. As easy minimally invasive collection and detection of expression changes, serum periostin may be a potential biomarker that predicts the efficacy of chemotherapy. Based on further study of the molecular mechanisms, Hu et al. 34 demonstrated that periostin was significantly up-regulated in cisplatin-resistant A549 cells, compared with parental controls. In their study, over-expression of periostin rendered A549 cells more resistant to cisplatin-induced apoptosis, and knockdown of periostin re-sensitized cisplatin-resistant A549 cells to cisplatin. Sung et al. 6 found that periostin treatment can induce cisplatin resistant through activating AKT pathway in A2780 cells in vitro.
Our previous study had demonstrated that the 3-year survival rate of patients with high-level periostin expression in cancer tissue was much higher than patients with low-level periostin expression. 35 In this study, the PFS of patients with abnormal expression of serum periostin is significantly shorter than that of patients who have lower baseline levels. Cox multivariate analysis confirmed that the level of serum baseline periostin and periostin response are independent prognostic factors in advanced NSCLC patients. Our study further demonstrated that over-expression of periostin predict a poor prognosis.
There are several limitations in this study. First, early-stage NSCLC patients were not included. Therefore, the role of periostin in tumorigenesis and progression of lung cancer could not be fully investigated. Second, the number of study samples was relatively small, which limits the power of multivariate analyses. Finally, the underlying mechanism of serum periostin as a biomarker for treatment efficacy was not explored, which will be investigated in the further research.
Conclusion
In summary, our study suggests that periostin might become an effective biomarker for NSCLC which can aid diagnosis and predict survival. Moreover, we provide new insights into serum periostin as surrogate marker of chemotherapy response. In view of the lack of specific tumor markers and the poor prognosis of patients with NSCLC, periostin and its relationship with lung cancer worth further investigation.
Footnotes
Compliance with ethical standards
All procedures were approved by the Medical Ethical Committee of Jinling Hospital, and written informed consent was obtained from all participants before obtaining the serum samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
