Abstract
Several studies have revealed the potential of normalizing tumor vessels in anti-angiogenic treatment. Recombinant human endostatin is an anti-angiogenic agent which has been applied in clinical tumor treatment. Our previous research indicated that gold nanoparticles could be a nanoparticle carrier for recombinant human endostatin delivery. The recombinant human endostatin–gold nanoparticle conjugates normalized vessels, which improved chemotherapy. However, the mechanism of recombinant human endostatin–gold nanoparticle–induced vascular normalization has not been explored. Anterior gradient 2 has been reported to be over-expressed in many malignant tumors and involved in tumor angiogenesis. To date, the precise efficacy of recombinant human endostatin–gold nanoparticles on anterior gradient 2–mediated angiogenesis or anterior gradient 2–related signaling cohort remained unknown. In this study, we aimed to explore whether recombinant human endostatin–gold nanoparticles could normalize vessels in metastatic colorectal cancer xenografts, and we further elucidated whether recombinant human endostatin–gold nanoparticles could interrupt anterior gradient 2–induced angiogenesis. In vivo, it was indicated that recombinant human endostatin–gold nanoparticles increased pericyte expression while inhibit vascular endothelial growth factor receptor 2 and anterior gradient 2 expression in metastatic colorectal cancer xenografts. In vitro, we uncovered that recombinant human endostatin–gold nanoparticles reduced cell migration and tube formation induced by anterior gradient 2 in human umbilical vein endothelial cells. Treatment with recombinant human endostatin–gold nanoparticles attenuated anterior gradient 2–mediated activation of MMP2, cMyc, VE-cadherin, phosphorylation of p38, and extracellular signal–regulated protein kinases 1 and 2 (ERK1/2) in human umbilical vein endothelial cells. Our findings demonstrated recombinant human endostatin–gold nanoparticles might normalize vessels by interfering anterior gradient 2–mediated angiogenesis in metastatic colorectal cancer.
Keywords
Introduction
Tumor vessels are structurally and functionally abnormal, which formatted by disorganized and active endothelial cells. It is well established that in tumor vessels, pericytes are either lacking or attaching loosely to endothelial cells. In addition, vascular endothelial growth factors (VEGF), especially VEGFA, decreases adherent junctions of adjacent endothelial cells and responses for vascular leakage. Those immature vessels decrease blood flow and increase tumor hypoxia, causing tumor development and resistant to chemotherapy or radiotherapy.1–3
Tumor vascular normalization is an approach to improve vascular morphology and function. This therapy reduces tumor vascular density while offering vessels with improved pericyte coverage and more integrated basement membrane. Those tumor vessels ensure blood flow and decrease hypoxia in tumor. Therefore, normalized vessels improved drug delivery when vascular normalization therapy was applied in combination with chemotherapy.4,5 It has been reported that anti-angiogenic agents may result in transient tumor vascular normalization, which improve blood perfusion, tumor hypoxia, and antitumor therapy.6,7 Anti-angiogenic drugs such as sunitinib and bevacizumab have been shown to be potential vascular normalization agents.8–10 Gold nanoparticles (AuNPs) exert anti-angiogenic function via antagonizing the VEGF-induced proliferation, migration, and activation of VEGF/AKT signaling cohort in human umbilical vein endothelial cells (HUVECs).11,12 Recombinant human endostatin (rhES), the N-terminal modified human endostatin, is approved as the first-line anti-angiogenic drug for non–small cell lung cancer by the China Food and Drug Administration.13,14 rhES interrupts the VEGF-induced angiogenesis and obtains better curative effect when combined with chemotherapy decreasing tumor hypoxia in the process.15–17 Our previous work implicates that AuNPs, as a nanoparticle drug-delivery system, induce tumor vessel normalization by delivering rhES into solid tumor and accordingly enhance chemotherapy efficacy. It illustrates that AuNPs could serve as a rhES-carrier that assemble in tumor and normalize tumor vessels. 18 Nevertheless, the mechanism of vascular normalization induced by rhES-AuNPs remains indistinct.
Anterior gradient 2 (AGR2) have been widely researched and mostly related to the promotion of tumor cell proliferation, migration, drug resistance, and tumor transformation.19,20 AGR2 is now considered as one of the latent tumor angiogenesis factors (TAFs) and assumes to be a potential anti-angiogenic drug target in tumor. It has demonstrated that in vitro, secreted AGR2 could induce migration and tube formation in HUVECs. AGR2 is highly expressed in tumor cell and regulated by hypoxia-induced factor-1 (HIF-1) in hypoxic tumor microenvironment. 21 In addition, more evidence indicates that HIF-1 could be stabilized by AGR2. 22 Interestingly, it is believed that AGR2 plays dissimilar role in various kinds of tumor. As for colorectal cancer (CRC), the expression of AGR2 is deemed to be the independent prognostic factor which remains low levels. 23 However, it is assumed that the role of AGR2 is necessary in the metastatic progression in CRC, and AGR2 may also be an impactful target for metastatic colorectal cancer (mCRC) treatment. 24 Indeed, previous studies show that AGR2 over-expressed in SW620, which was one kind of mCRC cells. 25 Overall, tumor angiogenesis is an extremely complex process, which covers a multitude of underlying TAFs. 26 Given AGR2 is a novel angiogenic factor and over-expressed in mCRC, it may be responsible for abnormal tumor vascular growth in mCRC.
So far, the precise efficacy of rhES-AuNPs conjugates on AGR2-induced angiogenesis or AGR2-mediated activation of signal pathways remained unknown. In this study, we aim to elucidate whether rhES-AuNPs can normalize tumor vessels by interrupting AGR2-mediated angiogenesis in mCRC xenografts. Furthermore, to clarify the mechanism of vascular normalization, we explore AGR2-activated signal pathways interfered by rhES-AuNPs. In this study, our results show that rhES-AuNPs can normalize vessels in mCRC xenografts via its interrupting the AGR2-mediated tumor angiogenesis.
Materials and methods
Preparation of the rhES-AuNPs
rhES was obtained from Simcere-Medgenn Bio-pharmaceutical Co., Ltd (Shandong, China) and stored at 4°C. AuNPs was purchased from Shanghai Jie Ning Biotech Co. Ltd. The AuNPs had an average concentration of 0.16 mg/mL and stored in light-resistant containers at 4°C. rhES-AuNPs was prepared as our previously described report. 18 Briefly, a calculated amount of rhES was added to the dispersion of AuNPs (the volume fractions between AuNPs and rhES is 3:1), and the reaction system was incubated in the dark at 4°C for 12 h. The preparation was oscillated gently every 2 h. Subsequently, mPEG-Thiol was introduced and incubated for another 4 h for in vivo and in vitro treatment.
Cell culture
mCRC cell lines SW620 were purchased from SHANGHAI ZZBIO CO., LTD (Shanghai, China). Human umbilical vessel endothelial cells (HUVECs) were digested with 0.25% trypsin (Gibco, NY, USA) for 10 min at 37°C from human umbilical veins and then centrifuged for 10 min at 1000 r/min before harvested. All cells were grown at 37°C with 5% CO2 and 95% humidity. Both SW620 and HUVECs were cultured in DMEM (Gibco, Life Technologies, USA) containing 10% fetal bovine serum (Gibco, Life Technologies) and 1% penicillin/streptomycin (Hyclone). HUVECs were harvested at 85% confluence for in vitro functional assays.
In vivo tumor grow assay
Female BALB/c nude mice (15–25 g weight, 6–8 weeks) were purchased from the Medical Laboratory Animal Center (Guangdong, China) and maintained under pathogen-free conditions. All animal study protocols were approved and conducted in accordance with the guidelines of the Laboratory Animal Ethics Committee of Jinan University. mCRC cell lines SW620 (4 × 106/100 µL/mouse) suspend in saline were inoculated into the flank of mice subcutaneously. rhES (5 mg/kg) was intraperitoneally injected into mice, while AuNPs (5 mg/kg), rhES-AuNPs (5 mg/kg), and saline (5 mg/kg) were subcutaneously injected into mice, respectively, when the tumor volume reached 175–200 mm3 and were repeated daily from days 0 to 4. After the treatment for 4 days, mice were weighed and sacrificed. The tumors were harvested and examined (n = 3 each group).
Plasma AGR2 ELISA
Blood samples were collected and centrifuged at 3000g for 15 min and store at −40°C (n = 6 each group). Level of AGR2 in plasma were detected by ELISA Kit for Anterior Gradient Protein 2 (AGR2) (MEXN USA), according to the manufacturer’s instruction.
Wound healing assay and tube formation assay
HUVECs were grown and seeded into six-well plate at a density of 1 × 105 cells/well. Three straight lines were drawn in the bottom of each well in one direction. Then the wells were washed twice with medium to remove the detached cells after scratching. HUVECs were seeded to 24-well plates (1 × 105 cells/well), which were coated with Matrigel (BD Biosciences, Bradford, MA, USA). HUVECs were supplied with 1 µg AGR and treated with rhES (100 µg), AuNPs (100 µg), rhES-AuNPs (100 µg), and saline (5 mM), respectively. The width of wound was evaluated at 0 and 18 h by using ImageJ software. The enclosed tubes were obtained at 24 h with an inverted phase contrast microscope (Olympus, Tokyo, Japan). All the experiments were repeated three times independently.
Immunofluorescence
Tumor tissue was fixed (4% paraformaldehyde, 24 h), paraffin-embedded, sectioned, dewaxed (Xylene), rehydrated (graded alcohol), and blocked (2% normal goat serum, 1 h). Then the sections were stained with α–smooth muscle actin (SMA) antibody (1:100; Proteintech, Chicago, USA), anti–vascular endothelial growth factor receptor 2 (VEGFR2), antibody (1:100; Abcam, Cambridge, UK), anti-AGR2 antibody (1:500; Abcam, Cambridge, UK), and anti-CD31 antibody (1:500; Abcam, Cambridge, UK). The sections were washed and incubated with rhodamine-conjugated goat anti-rat IgG (H+L) (1:50; Proteintech, Chicago, USA) or goat anti-rabbit IgG-FITC (1:200; Santa Cruz, Dallas, USA) for 40 min at room temperature. Tissue was visualized using a fluorescence microscope (Leica DM6000B).
Histology and immunohistochemistry
Depending upon the procedure, tumors were fixed in 10% formalin solution, embedded in paraffin, and cryopreserved in isopentane. Hematoxylin and eosin (H&E) staining was performed according to standard procedures on 3 mm paraffin sections. Pimonidazole staining was conducted with Hypoxyprobe™-1 Plus kit (HPI Inc., Burlington, MA, USA). Pimonidazole was applied intraperitoneally at 60 mg/kg and tumors were harvested 60 min later. Hypoxyprobe-1 adducts were detected using an affinity-purified rabbit IgG polyclonal antibody conjugated with horseradish peroxides following the manufacturer’s instructions. More than six photos were taken randomly from each slice on a microscope.
Protein extraction and western-blotting assay
Tumors were harvested. Radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime, Shanghai, China) was used to lyse tumor tissue. Bicinchoninic acid (BCA) protein assay kits (TaKaRa, Dalian, China) were used to detect protein concentrations. Western-blotting assay was conducted for detection of HIF-1α and AGR2 by using HIF-1α antibody (1:100; Proteintech, Chicago, USA) and anti-AGR2 antibody (1:500; Abcam, Cambridge, UK) as primary antibody. Well-grown HUVECs were cultured with 1 µg AGR2. Then HUVECs were treated with rhES (100 µg), AuNPs (100 µg), rhES-AuNPs (100 µg), and saline (5 mM) for 24 h, respectively. Lysates of HUVECs were collected and applied for detection of MMP2, cMyc, VE-cadherin (VE-ca), and phosphorylation of p38 and ERK1/2, by using rabbit monoclonal p44/p42 (ERK1/2) antibody, rabbit monoclonal phosphop44/42 (ERK1/2) antibody, rabbit monoclonal p38 antibody, mouse monoclonal phosphop38 (Thr180/Tyr182) antibody, and rabbit polyclonal anti-β-actin antibody (Cell Signaling, Boston, USA). The internal control protein was β-actin. It was detected by anti-beta actin antibody (1:1000; Abcam, Cambridge, UK). Detection was performed with the ECL kit (GE Healthcare, Pittsburgh, USA).
Statistical analysis
All data are displayed as the mean ± standard error from three independent experiments. Statistics for different groups were compared by one-way analysis of variance (ANOVA). Statistics between two groups were analyzed by sample t-test. And statistics from any groups were measured by Student–Newman–Keuls (SNK). All statistical analyses were exhibited by GraphPad Prism (version 5.0; GraphPad Software, La Jolla, CA, USA). It is considered statistically significant when p values are <0.05 or 0.01 (*p < 0.05 and **p < 0.01).
Results
Treatment of rhES-AuNPs modulated vascular morphology in mCRC xenografts
CD31 was measured as endothelium marker. Tumor vessels appeared to be tortuous and distensible after treated with saline, while vessels became organized and continuous after treated with rhES-AuNPs. There was no significant difference in vascular density among tumors treated with saline, rhES, AuNPs, or rhES-AuNPs (Figure 1(a)). Our data suggested that short-term rhES, AuNPs, or rhES-AuNPs treatment did not reduce vascular density but modulated vascular morphology. Next, we used α-SMA as pericyte marker. Pericyte expression was increased in rhES-AuNPs-treated tumors (p = 0.022; Figure 1(b)). VEGFR2 regulated vascular permeability in tumor. 27 There was a significant reduction of VEGFR2 expression in rhES-AuNPs-treated tumors, suggesting rhES-AuNPs treatment resulted in vascular permeability improvement (p = 0.041; Figure 1(c)).
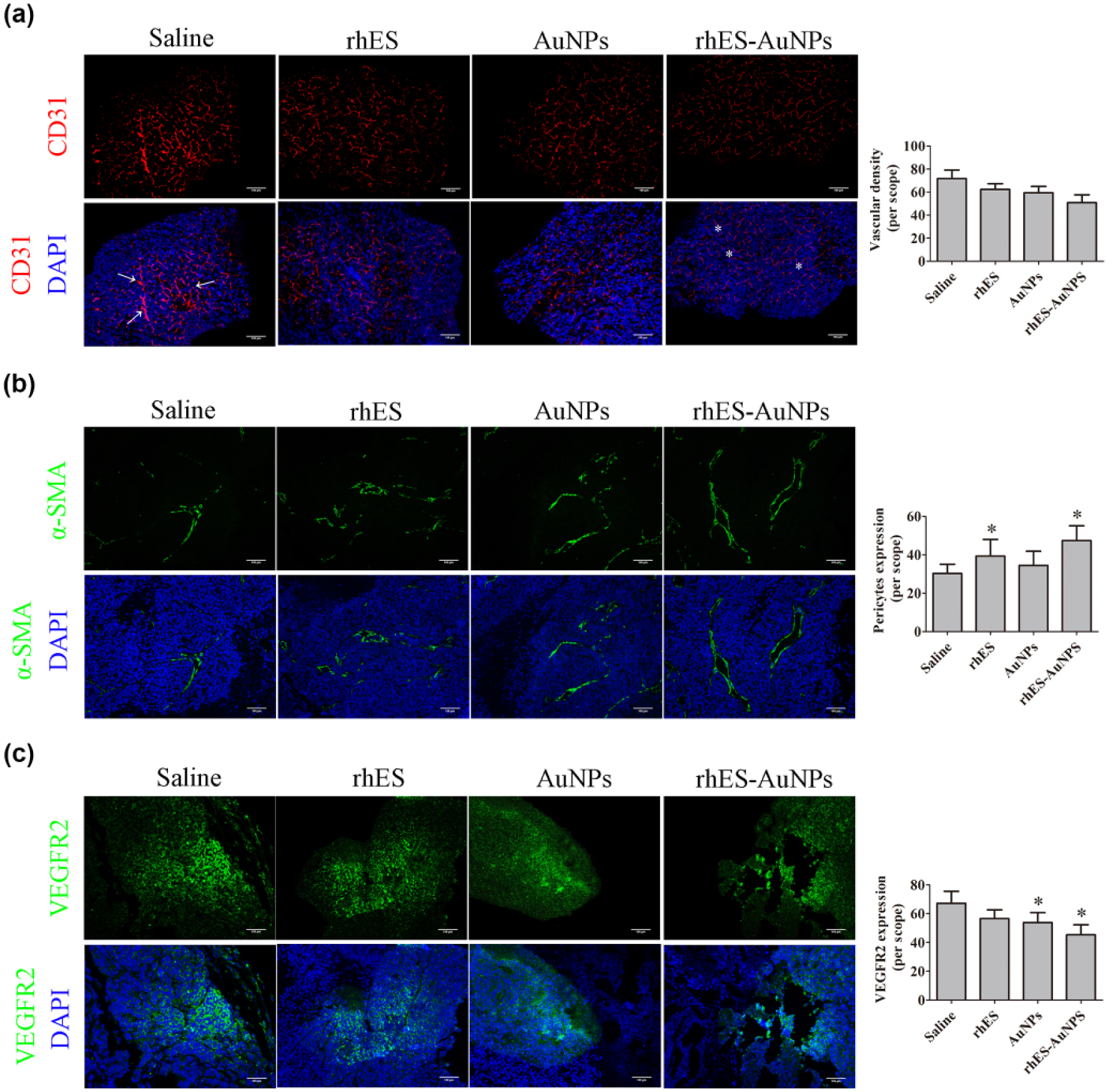
Treatment of rhES-AuNPs modulated vascular morphology in mCRC xenografts. (a) CD31 (red) staining for tumors that were treated with saline, rhES, AuNPs, and rhES-AuNPs. Vascular density of each group had no obvious difference after treatment for 4 days. (b) α-SMA (green) staining for tumors that were treated with saline, rhES, AuNPs, and rhES-AuNPs. α-SMA positive areas in rhES-AuNPs-treated tumor were significantly higher (p < 0.05). (c) VEGFR2 (green) staining for tumors that were treated with saline, rhES, AuNPs, and rhES-AuNPs. VEGFR2 positive areas in rhES-AuNPs-treated tumor were significantly lower (p < 0.05). Scale bars: 100 µm.
Treatment of rhES-AuNPs alleviated tumor hypoxia and preserved tissue viability
There was significantly lower level of pimonidazole staining areas in tumor tissue that were treated with rhES-AuNPs as compared with mice treated with saline, rhES, or AuNPs (rhES, p = 0.021; AuNPs, p = 0.004; rhES-AuNPs, p = 0.026; Figure 2(a)). Next, we identified HIF-1α expression was lower in rhES-AuNPs-treated tumor tissue by western-blotting assay (p = 0.047; Figure 2(a)). These data indicated that treatment of rhES-AuNPs improved tumor hypoxia. Also, H&E staining revealed that tissue viability remained indistinctive after treatment with rhES-AuNPs.

Treatment of rhES-AuNPs alleviated tumor hypoxia and preserved tissue viability. (a) Pimonidazole (brown) staining. Lower pimonidazole positive staining was observed in tumor tissue from mice that were treated with rhES-AuNPs. (b) Western-blotting assay revealed lower HIF-1α expression in rhES-AuNPs-treated tumor tissue. (c) H&E images. Scale bars: 200 µm. There are no variations among the mice that were treated with saline, rhES, AuNPs, or rhES-AuNPs, respectively.
Treatment of rhES-AuNPs decreased AGR2 expression in tumor
To investigate AGR2 expression in rhES-AuNPs treated tumor, we conducted AGR2 staining in tumor tissues. Tumor treated with rhES-AuNPs had less AGR2 expression than that treated with saline, rhES, or AuNPs (rhES, p = 0.026; AuNPs, p = 0.032; rhES-AuNPs, p = 0.043; Figure 3(a)). Moreover, western-blotting assay further confirmed that AGR2 expression in rhES-AuNPs-treated tumor was lower than in saline-, rhES- or AuNPs-treated tumor (rhES, p = 0.011; AuNPs, p = 0.019; rhES-AuNPs, p = 0.022; Figure 3(b)). Meanwhile, previous report indentified that secreted AGR2 was responsible to tumor abnormal vessels growth. 21 Therefore, plasma AGR2 in mice was detected and the result indicated that rhES-AuNPs reduced plasma AGR2 in mice.

Treatment of rhES-AuNPs decreased AGR2 expression in tumor. (a) AGR2 (green) staining areas. Scale bars: 200 µm. Lower AGR2 expression was observed in tumor tissue from mice that were treated with rhES-AuNPs. (b) Western-blotting assay confirm that rhES-AuNPs-treated tumor had lower AGR2 expression. (c) Treatment with rhES-AuNPs resulted in lower level of plasma AGR2.
Treatment of rhES-AuNPs suppressed AGR2-mediated cell migration, tube formation, and AGR2-induced activation of signaling pathway in HUVECs
We observed that migration of HUVECs stimulated by AGR2 had significantly lesser relative width of scratched gap than saline-treated HUVECs (p = 0.037; Figure 4(a)), and HUVECs treated with rhES-AuNPs had less migration distance than those treated with rhES or AuNPs (Figure 4(a)). For tube formation assay, HUVECs stimulated by AGR2 had more tube formation units than those with saline-treated HUVECs (p = 0.016; Figure 4(b)). MMP2, cMyc, VE-ca, and mitogen-activated protein kinase (MAPK) signaling pathway plays vital role in regulation of cell migration, proliferation, and adhesion in the process of tumor angiogenesis. Based on the finding that rhES-AuNPs suppressed the AGR2-mediated proliferation, migration, and tube formation in HUVECs, the significance of rhES-AuNPs on AGR2-mediated activation of intracellular MMP2, cMyc, VE-ca, and MAPK signaling pathway in HUVECs was further investigated (Figure 4(c)). The results indicated that AGR2 could stimulate the phosphorylation of p38 (p = 0.030) and ERK1/2 (p = 0.021) and expression of MMP2 (p = 0.037), cMyc (p = 0.011), and VE-ca (p = 0.040), as compared to saline-treated group. But the protein expression levels of p38 and ERK1/2 remained stable in HUVECs.

Treatment of rhES-AuNPs suppressed AGR2-mediated cell migration, tube formation, and AGR2-induced activation of signaling pathway in HUVECs. (a) Wound healing assay of HUVECs. Cells cultured with AGR2 had longer migration distance than those treated with saline. Treatment with rhES-AuNPs had significantly less migration distance. (b) Tube formation assay of HUVECs. Cells cultured with AGR2 had more numbers of tubes formed than those treated with saline. Treatment with rhES-AuNPs had significantly less tube formation. (c) Western-blotting assay of HUVECs. The relative levels of phosphorylated p38, phosphorylated ERK1/2, MMP2, cMyc, and VE-ca were significantly higher in HUVECs that treated with AGR2, but reduced after treated with rhES-AuNPs.
Discussion
AuNPs are identified as nano-drug carriers and delivered drugs without impacting the efficacy of them. 28 It is known that rhES is an endogenous anti-angiogenesis factor that inhibits the growth of solid tumor and has been approved in the clinical treatment. 29 Our previous research showed that the treatment of rhES-AuNPs had promising efficacy in tumor vascular normalization and improved chemotherapy as compared to rhES or AuNPs monotherapy. 18 Few revealed molecular basis of the anti-angiogenic efficacy of AuNPs and rhES was their participation in the VEGF/VEGFR signaling pathway.12,30 However, the exact molecular mechanism through which rhES-AuNPs exerted its vascular normalization effect was still indistinct.
As a new antitumor strategy, vascular normalization induced by antiangiogenic therapy was widely studied in the treatment of tumor, but for AGR2 over-expressed malignant tumors, the influence of rhES-AuNPs on the expression or the function of AGR2 was not elucidated.21–25,31
In this study, the effect of rhES-AuNPs on mCRC tumor growth in vivo was firstly evaluated. The results suggested that rhES-AuNPs experimental group could modulate vascular morphology in mCRC tumor and improve tumor hypoxia. Treatment with rhES-AuNPs inhibited AGR2 expression in tumor tissue, accompanied by lower plasma AGR2 levels in mice. Based on the theory, AGR2 contributed to tumor angiogenesis and poor survival of clinical mCRC patients.21,25 Our finding indicated that rhES-AuNPs could normalize tumor vessels by inhibiting angiogenesis mediated by high secretion of AGR2, and the vascular normalization effect of rhES-AuNPs was more remarkable than rhES or AuNPs monotherapy. In addition, previous studies found that suppressing secreted AGR2 could restrain tumor metastasis and tumor angiogenesis, and reduced intracellular expression of AGR2 could reverse chemotherapy resistance. Actually, anti-AGR2 monoclonal antibodies exhibited antitumor effects, and AGR2 might be a new target for the anti-angiogenesis treatment. 32 Our findings, coincided with those previous studies mentioned above, indicated that AGR2 might be a promising drug target for the anti-angiogenesis and vascular normalization therapy in the treatment of mCRC.
Hypoxia, as an activate stimulator for tumor angiogenesis, responses for the attraction and cooperation of multiple cells in tumor environment, including vascular endothelial cells. 33 Previous studies demonstrate that both rhES and AuNPs could inhibit the proliferation of HUVECs induced by VEGF.12,30 To further investigate the influence of rhES-AuNPs on AGR2-mediated angiogenesis, the HUVECs was applied as cell model in vitro. Our results indicated that treatment with rhES-AuNPs could obviously suppress AGR2-mediated migration and tube formation of HUVECs. These findings directly proved that rhES-AuNPs could antagonize AGR2-mediated angiogenesis in vitro.
Ultimately, we explored the possible molecular mechanism of anti-angiogenesis mediated by rhES-AuNPs. Given that AGR2 contributed to migration and tube formation of HUVECs, western-blotting was conducted to evaluate the effect of rhES-AuNPs on multiple intracellular protein include MMP2, cMyc, VE-ca, phosphorylation of p38, and ERK1/2, which played important role in pro-angiogenic signaling pathways in HUVECs. Our result showed that treatment with rhES-AuNPs in HUVECs noticeably suppressed AGR2-induced activation of MMP2, cMyc, VE-ca, phosphorylation of p38, and ERK1/2. These findings, for the first time, suggested that rhES-AuNPs suppressed AGR2-activated signaling pathway in HUVECs. Our present findings were also consistent with the previous reports that treatment with VEGF could promote angiogenesis by activating p38 and ERK1/2 in HUVECs, and the sensitization of MMP2, cMyc could regulate the VEGF expression, while VE-ca accounted for the integrity and abnormal function of new vessel.30,34–36
In summary, our findings demonstrated that rhES-AuNPs might modulate vascular normalization and interrupted AGR2 expression in mCRC xenografts. Furthermore, rhES-AuNPs interrupted AGR2-induced vascular formation in HUVECs. Those finding suggested that rhES-AuNPs might normalize vessels by interfering AGR2-mediated angiogenesis in mCRC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81472849), the Guangdong Natural Science Research (2014A030313383), and the Guangdong High-level University Construction Fund for Jinan University (88016013034).
